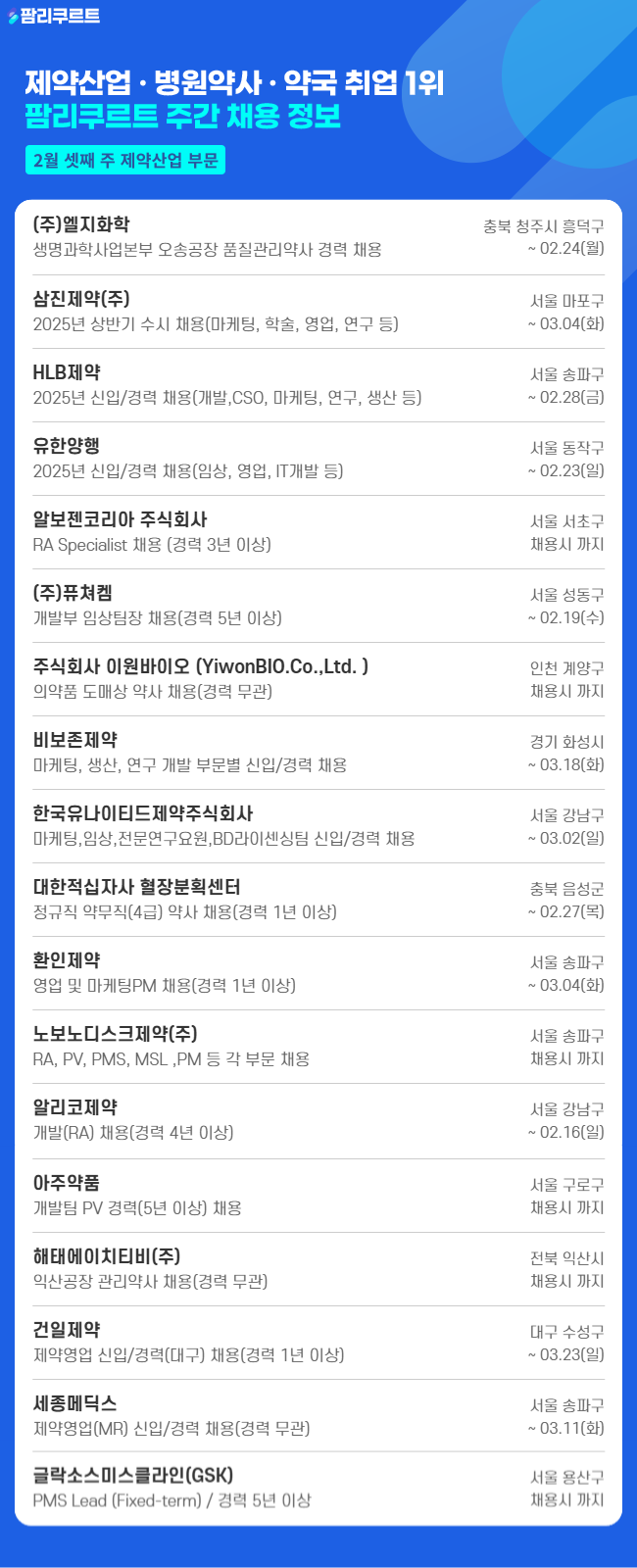
[팜리쿠르트] 유한양행·삼진제약·노보노디스크 등 채용
- 손형민
- 2025-02-18 06:15:27
-
가
- 가
- 가
- 가
- 가
- 가
- AD
- 4월 3주차 지역별 매출 트렌드 분석이 필요하다면? 제약산업을 읽는 데이터 플랫폼
- BRPInsight
손형민(shm@dailypharm.com)
Copyright ⓒ 데일리팜. All rights reserved. 무단 전재 및 재배포 금지.
- 익명 댓글
- 실명 댓글
- 댓글 0
- 최신순
- 찬성순
- 반대순
운영규칙
오늘의 TOP 10
- 1표제기 이부프로펜 감기약 속속 등장…종근당 모드콜도 가세
- 2건보 적자 늪 탈출구는 '지불제도' 개혁…사회적 대타협 필요
- 3이노엔·대웅·제일, P-CAB 적응증 강화…후발주자 견제
- 4씨투스 제네릭 발매 1년만에 점유율 30% 돌파
- 5"바이오시밀러 선택한 환자 인센티브"…처방 활성화 추진
- 6보령, 내달 카나브젯 급여 등판...복합제 라인업 강화
- 7[기자의 눈] 무색해진 판결…실리마린에 꽂힌 정부의 집요함
- 8바이오헬스, 수천억 CB 발행…주가 훈풍에 자금조달 숨통
- 9항체치료제 '누칼라 오토인젝터', 약가협상 최종 타결
- 10㉖ 최초 원발성 lgA 신병증 항체치료제 '시베프렌리맙'



















































![[SK케미칼] 속편한정 복합소화제](https://cdn.platpharm.co.kr/2025/12/2512040916400005920.webp)
![[리쥬올]리쥬올 PDRN 약국 1위 PDRN](https://cdn.platpharm.co.kr/2025/09/2509260220180000170.webp)
![[신신] 새사래 상처연고 습윤밴드](https://cdn.platpharm.co.kr/2025/10/2510210339570001784.webp)
![[유한양행] 콘택콜드 걸렸구나 생각되면](https://cdn.platpharm.co.kr/2025/10/2510282252420008436.webp)
![[유한양행] 미녹펜겔 탈모스팟 집중케어](https://cdn.platpharm.co.kr/2025/09/2509220824180004563.webp)
![[리쥬올]레티노 멜라세럼 저자극 레티놀](https://cdn.platpharm.co.kr/2025/09/2509260219360000145.webp)
![[종근당] 벤포벨에스 어른들의 피로회복제](https://cdn.platpharm.co.kr/2025/07/2507290841210004645.webp)
![[셀로맥스] 베베락스 온가족 안심 관장약](https://cdn.platpharm.co.kr/2025/09/2509171131320018843.webp)
![[SK케미칼] 트라스트패취 피록시캄 성분](https://cdn.platpharm.co.kr/2025/10/2510020656150002375.webp)
![[더본메디칼] ATC인쇄리본 특가](https://cdn.platpharm.co.kr/2025/04/2504100527360001454.jpg)

![[신신] 아렉스 두번효과로 강력한](https://cdn.platpharm.co.kr/2025/10/2510230254510000664.webp)
![[아워팜] 에너지 바로 충전, 바로콤](https://i.baropharm.com/products/202512/1764922282624.png)
![[레킷코리아] 목 아플 때, 스트렙실 허니&레몬 트로키 12정](https://i.baropharm.com/products/202502/1739520767049.png?label=PLAN_01)
![[레비온] PDRN+EGF, 레비온RX PDRN EGF 크림](https://i.baropharm.com/products/202512/1765949426601.png)
![[알엑스미] 알엑스미 리쥬영 울트라 PDRN 10000 딥리페어 크림](https://i.baropharm.com/partner/products/70c72dd0-cfd3-4d80-87e4-dc4f8de6658b.png?label=바뷰페로고)
![[아워팜] 우리아이 맞춤설계, 바로타민 kids 엘더베리맛](https://i.baropharm.com/partner/products/3f39593e-6318-4dd9-a778-c008c868b5c8.png)
![[켄뷰] 오리지널 폼타입, 로게인5%폼에어로졸60g](https://i.baropharm.com/products/dc84d96e-d0b4-46bc-bcc8-d62016406fe4.png)
![[켄뷰] 다양한 통증에, 타이레놀정 500mg 10정](https://i.baropharm.com/products/6c6ea4f4-7ab2-44f2-a165-f062d80f525b.png)
![[휴온스 ] 비듬을 한번에, 니조랄 2%액](https://i.baropharm.com/products/478a284d-4361-4b4a-8a00-8bab80f34319.png?label=PLAN_01)
![[한독] 붙이는 통증 전문가, 케토톱 액티브 플라스타(쿨) 40매](https://i.baropharm.com/products/202503/1741829602305.png)
![[아워팜] CJ웰케어, 바이오코어 1000억 유산균](https://i.baropharm.com/products/202512/1765955416559.png)
![[오펠라] 부드럽고 편안한, 둘코락스에스장용정 20정](https://i.baropharm.com/products/202511/1762260404625.png)
![[리쥬올] 닥터 리쥬올 어드밴스드 PDRN 리쥬비네이팅 크림 30ml](https://i.baropharm.com/partner/products/a201d2b4-f21e-4b13-957c-846d286b3d21.jpg?label=바뷰페로고)



