- LOGIN
- MemberShip
- 2026-04-17 04:57:35
- Study: Is convalescent plasma actually effective on COVID-19
- by choi, sun | translator Byun Kyung A | 2020-04-09 06:27:45
Following the case reported in China, Korea has published a case study of effective plasma therapy on COVID-19 patients.
As the Korean infectious disease control authority has announced its plan to establish a plasma therapy guideline for the COVID-19 patients, the public’s interest on the therapeutic mechanism and effect has heightened.
Following is a discussion over the issues regarding the plasma therapy’s mechanism of action and efficacy.
▲Plasma therapy using recovered patient’s antibody to fight against the virus A vaccine has a preventive effect but does not treat the disease after the viral infection.
Antiviral remdesivir, currently used to treat COVID-19 patients, relies on inhibition of viral replication and symptomatic therapy relieving the symptoms.
On the other hand, plasma therapy uses immune system of a patient who has completely recovered from the viral infection.
In the recovered patient’s blood, plasma containing antibody is generated during the process of post-viral infection recovery.
And if the convalescent plasma is extracted from the donor and transfused to another patient with severe symptoms, the antibody from the plasma fights against the virus in the patient.
In such case, the time to alleviation of symptoms and treatment against the virus could be shortened.
This is usually the reason why the plasma therapy is a last resort for patients in a critical state of a novel infectious disease without a treatment or vaccine.
The U.S.
Food and Drug Administration (FDA) on Mar.
24 and Korean health authority on Apr.
1 have respectively approved of using blood product extracted from people who have cured from COVID-19 for treatment purpose.
After China has published a clinical study result confirming the therapeutic benefit on five people, Korea has also reported cases cured from blood product.
▲Effect of plasma therapy?
“All cured” On Apr.
6, Journal of Korean Medical Science (doi.org/10.3346/jkms.2020.35.e149) has published a study on the use of convalescent plasma therapy in two COVID-19 patients led by Professor Choi Jun Yong (Department of Internal Medicine, Severance Hospital, Seoul) and his research team.
This is a first report of using convalescent plasma extracted from recovered patients to treat COVID-19 cases in Korea.
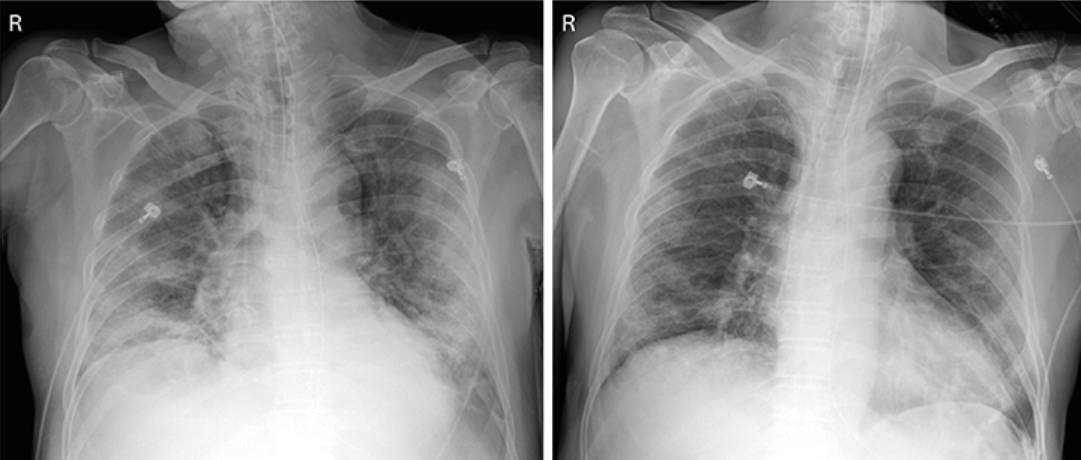
The first case, a 71-year-old man, has been treated with antimalarial and HIV drug, but his symptoms worsened.
The respiratory rate was over 30 times per minute (normally under 20 times per minute) and chest radiographs showed critical pneumonic symptoms in both of his lungs.
C-reactive protein (CRP) was up at 172.6 mg/ L (normally less than 8 mg/ L).
When the second case, a 67-year-old woman, was hospitalized, her respiratory rate was at 24 times per minute and oxygen saturation was at 93 percent (normally over 95 percent), while her CRP elevated up to 314 mg/ L with signs of lymphopenia.
Regardless of using corticosteroid on top of administering antimalarial and HIV drugs, the female patient sustained lymphopenia and the viral titer was increased.
The researchers administered 500 ml of convalescent plasma, obtained from a recovered donor, twice in 12-hour interval and initiated corticosteroid treatment simultaneously.
The patients were discharged from hospital after seeing lymphopenia recovery, reduction in viral titer, and recovery of CRP level.
A plasma therapy study on critically ill patient with COVID-19 in China, led by Professor Chenguang Shen (National Clinical Research Center for Infectious Disease), has also demonstrated similar outcome.
Five patients with COVID-19 and acute respiratory distress syndrome (ARDS) have recovered normal body temperature in three days after receiving convalescent plasma, and showed increase in oxygenation capacity and decrease in viral titer.
Three out of five patients were discharged (hospitalized for 53, 51 and 55 days, respectively) and two patients stabilized on day 37 after the plasma therapy.
▲Plasma therapy with positive outcomes, but why the negative issue?
The problem is the effect.
One says the effect has been confirmed, but the others say it has not been fully confirmed.
So why are there contrasting claims?
The dispute has sparked due to the limitations in the clinical environment the plasma therapy was conducted.
As the studies were conducted in critically ill and desperate patients with novel infectious disease, the variables were not properly controlled in all cases.
To appropriately prove the efficacy of the plasma therapy, the infected patients should be divided into three groups.
A large-scale long-term clinical study with patient groups receiving plasma therapy only, plasma therapy plus antiviral agent (symptomatic therapy), and placebo should be conducted to test the clinical efficacy.
Such study is leaves limitations in confirming the effect of each therapy type as various antibodies were administered to the patient due to their severity.” “However, the study in Korea has witnessed the drop in viral load, which could be assessed as an effect of transfusing convalescent plasma, as using systemic corticosteroids can only relieve inflammatory response but does not block viral replication,” the professor emphasized.
▲Korean disease control authority setting convalescent plasma use guideline, what to look out for?
The limitations still exist because plasma therapy is not the only option and is definitely not the fully proven option.
The government guideline should take account of limited convalescent plasma from a donor, prioritization of administrated subject and different level of effect depending on the donor’s condition.
But also the authority should keep in mind that the therapy lacks sufficient medical evidences.
Medical experts stress that confusion in clinical scene and waste in resources would be minimized if only the health authority clarifies detailed guideline of convalescent plasma use.
Professor Choi Jun Yong highlighted, “As for the guideline, the Korean Society for Laboratory Medicine has a lot to contribute.
More than anything, it would have to recommend standard of prioritizing the subject for limited convalescent plasma transfusion considering the efficiency and benefit.” And he added, “Depending on the donor’s health condition, the convalescent plasma’s effect and outcome could differ.
Future studies should follow to evaluate adequacy of donor and recipient.” The professor also suggested, “The level of neutralizing antibody related to the plasma therapy needs more research.
The guideline should consider testing the donor’s neutralizing antibody titer prior to the transfusion, as the young age of a donor does not guarantee high level of the antibody.”
-

- 0
댓글 운영방식은
댓글은 실명게재와 익명게재 방식이 있으며, 실명은 이름과 아이디가 노출됩니다. 익명은 필명으로 등록 가능하며, 대댓글은 익명으로 등록 가능합니다.
댓글 노출방식은
댓글 명예자문위원(팜-코니언-필기모양 아이콘)으로 위촉된 데일리팜 회원의 댓글은 ‘게시판형 보기’와 ’펼쳐보기형’ 리스트에서 항상 최상단에 노출됩니다. 새로운 댓글을 올리는 일반회원은 ‘게시판형’과 ‘펼쳐보기형’ 모두 팜코니언 회원이 쓴 댓글의 하단에 실시간 노출됩니다.
댓글의 삭제 기준은
다음의 경우 사전 통보없이 삭제하고 아이디 이용정지 또는 영구 가입제한이 될 수도 있습니다.
-
저작권·인격권 등 타인의 권리를 침해하는 경우
상용 프로그램의 등록과 게재, 배포를 안내하는 게시물
타인 또는 제3자의 저작권 및 기타 권리를 침해한 내용을 담은 게시물
-
근거 없는 비방·명예를 훼손하는 게시물
특정 이용자 및 개인에 대한 인신 공격적인 내용의 글 및 직접적인 욕설이 사용된 경우
특정 지역 및 종교간의 감정대립을 조장하는 내용
사실 확인이 안된 소문을 유포 시키는 경우
욕설과 비어, 속어를 담은 내용
정당법 및 공직선거법, 관계 법령에 저촉되는 경우(선관위 요청 시 즉시 삭제)
특정 지역이나 단체를 비하하는 경우
특정인의 명예를 훼손하여 해당인이 삭제를 요청하는 경우
특정인의 개인정보(주민등록번호, 전화, 상세주소 등)를 무단으로 게시하는 경우
타인의 ID 혹은 닉네임을 도용하는 경우
-
게시판 특성상 제한되는 내용
서비스 주제와 맞지 않는 내용의 글을 게재한 경우
동일 내용의 연속 게재 및 여러 기사에 중복 게재한 경우
부분적으로 변경하여 반복 게재하는 경우도 포함
제목과 관련 없는 내용의 게시물, 제목과 본문이 무관한 경우
돈벌기 및 직·간접 상업적 목적의 내용이 포함된 게시물
게시물 읽기 유도 등을 위해 내용과 무관한 제목을 사용한 경우
-
수사기관 등의 공식적인 요청이 있는 경우
-
기타사항
각 서비스의 필요성에 따라 미리 공지한 경우
기타 법률에 저촉되는 정보 게재를 목적으로 할 경우
기타 원만한 운영을 위해 운영자가 필요하다고 판단되는 내용
-
사실 관계 확인 후 삭제
저작권자로부터 허락받지 않은 내용을 무단 게재, 복제, 배포하는 경우
타인의 초상권을 침해하거나 개인정보를 유출하는 경우
당사에 제공한 이용자의 정보가 허위인 경우 (타인의 ID, 비밀번호 도용 등)
※이상의 내용중 일부 사항에 적용될 경우 이용약관 및 관련 법률에 의해 제재를 받으실 수도 있으며, 민·형사상 처벌을 받을 수도 있습니다.
※위에 명시되지 않은 내용이더라도 불법적인 내용으로 판단되거나 데일리팜 서비스에 바람직하지 않다고 판단되는 경우는 선 조치 이후 본 관리 기준을 수정 공시하겠습니다.
※기타 문의 사항은 데일리팜 운영자에게 연락주십시오. 메일 주소는 dailypharm@dailypharm.com입니다.





