- LOGIN
- MemberShip
- 2026-04-30 04:35:45
- Policy
- Will incentives for low-price purchasing be expanded?
- by Jung, Heung-Jun Nov 27, 2025 06:14am
- Concerns are emerging that expanding low-price purchase incentives without safeguards could lead to a market collapse, ridden with ultra-low-price bidding wars, eroding the entire pharmaceutical ecosystem. The criticism is that a system focused solely on price competition directly contradicts the government’s stated goals of encouraging pharmaceutical and biotech R&D. According to industry sources on the 25th, the government’s upcoming drug-pricing reform package is expected to include a substantial expansion of low-price purchasing incentives. The low-price purchase incentive is a system where, if a healthcare institution purchases a drug at an actual transaction price lower than the reimbursement ceiling, a portion of the price reduction is provided as an incentive payment. Approximately 20-30% of the price difference is paid to the healthcare institution based on the incentive payment standards announced by the Ministry of Health and Welfare. Although the specific details remain undisclosed, one option being discussed is maintaining the current rule of not lowering drug prices based on actual transaction prices, while increasing the incentive rate to up to 50%. However, there are concerns that even without lowering drug prices based on actual transaction prices, a system that encourages ultra-low-price competition will ultimately lead to a steep decline in profits for manufacturers and distributors. Mr. A from a domestic pharmaceutical company expressed concern, “They are essentially fueling price competition. While I understand the intent to create an ecosystem where competition enables cheaper transactions, it will ultimately lead to a resurgence of 1-won and 2-won bids. Everyone will flock to what is effectively legalized rebates, and the manufacturing ecosystem will collapse.” Critics point out that in a situation where small and medium-sized distributors, including CSOs, proliferate and the lack of transparency in distribution channels is a constant issue, a policy solely focused on encouraging price competition will likely cause more harm than good. The same representative added, “Many injectable drugs have already been through 5 rounds of price cuts based on actual transaction prices, with reductions of around 20% each time. If price competition intensifies further, the damage to injectables will be greater. It’s not enough to simply expand incentives; the government must implement safeguards.” Advice was also given that setting an appropriate minimum bid price would be necessary. Another domestic company representative, Mr. B, stated, “It's hard to predict the outcome, but without setting an appropriate minimum bid price, profits will plummet dramatically. At the very least, it should guarantee cost recovery. Considering the relationship between large hospitals and pharmaceutical companies, they might all rush in with a ‘let's all go down together’ attitude.” Mr. B added, "Furthermore, if incentives rise to 50%, even institutions that have been quiet until now may start making more demands. Currently, payments aren't high enough to warrant claiming the maximum price, but if the reimbursement rate hits 50%, that changes. It may essentially turn into a legalized rebate structure." Some have proposed specific restrictions similar to the minimum-price guarantee applied to Essential Exit-Prevention Drugs, arguing that price-protection mechanisms must accompany any incentive expansion. Mr. A added, “It's necessary to put brakes in place. For injectables with high in-hospital usage rates, we could cap prices at 91% of the drug price, similar to exit prevention drugs. Simply trying to intensify low-price competition is armchair theorizing. Once the ecosystem collapses, it's difficult to recover. The government needs to listen more to real-world concerns.”
- Policy
- Legalizing order for manufacturer to supply essential drugs
- by Lee, Jeong-Hwan Nov 25, 2025 06:13am
- A bill has been proposed in the Korean National Assembly to stipulate the Government Supply System in the Pharmaceutical Affairs Act to ensure the stable supply of National Essential Medicines, which are frequently in short supply due to low profitability. The bill also includes an article expanding treatment opportunities for patients using essential medicines by entrusting the practical work of ensuring a stable supply to the Korea Orphan & Essential Drug Center. On November 24, Democratic Party of Korea Rep. Young-seok Seo announced that he had officially proposed this amendment to the Pharmaceutical Affairs Act. Rep. Seo stated that the number of discontinued products has recently been increasing, primarily in essential medicines with low commercial viability. He noted that the government is currently operating a Government Supply System, such as supporting drug production through agreements with pharmaceutical companies or directly importing overseas pharmaceuticals, when supply is halted due to a company's product withdrawal. However, Rep. Seo expressed concern that the current law does not explicitly provide a legal basis for the Government Supply System, limiting its application and expansion. He further pointed out that, concerning this issue, the legal basis for supporting the resumption of production of medicines not supplied domestically is also inadequate. Therefore, Rep. Seo introduced a bill that legally mandates the Government Supply System for essential medicines and entrusts its practical implementation to the Korea Orphan & Essential Drug Center. In detail, the bill allows the Minister of Food and Drug Safety (MFDS) to mandate a pharmaceutical manufacturer to manufacture National Essential Medicines for products not currently manufactured or imported into Korea when a stable domestic supply is necessary. This bill is to be newly established under Article 41-2, titled 'Manufacture and Supply of National Essential Medicine,' of the Pharmaceutical Affairs Act. In such cases, the MFDS Minister may prepay all or part of the manufacturing costs to the relevant pharmaceutical company. Furthermore, the MFDS Minister can provide administrative and technical support to ensure a swift review process when the company applies for a product license. The MFDS Minister is also mandated to entrust the duties of manufacturing, storage, and supply of National Essential Medicines to the Korea Orphan & Essential Drug Center. The proposed bill also introduces a new article, 'Article 43-2 Emergency Import of Medicines,' into the Pharmaceutical Affairs Act. This new bill allows the MFDS Minister to supply medicines domestically through methods such as importation and provide related information to patients when an urgent domestic supply is deemed necessary or when the head of a relevant central administrative agency or related organizations and groups requests the supply. Rep. Seo explained, "This bill legally specifies the government system for the stable supply of essential medicines and entrusts the practical execution to the Korea Orphan & Essential Drug Center," and added, "It will guarantee treatment opportunities for patients requiring essential medications."
- Policy
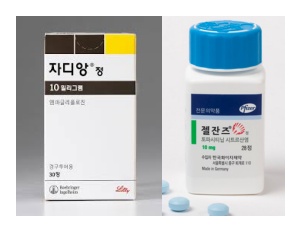
- Price of Jardiance, Xeljanz cut 30% with entry of generics
- by Lee, Tak-Sun Nov 24, 2025 06:19am
- The diabetes treatment Jardiance (empagliflozin, Boehringer Ingelheim) and the rheumatoid arthritis treatment Xeljanz (tofacitinib citrate, Pfizer) will have their maximum insurance prices officially reduced due to the listing of generic versions. When a therapeutically equivalent product is newly listed, the price ceiling is adjusted to 53.55%, and a one-year 70% add-on premium is applied. As a result, starting in December, the previous prices will drop by 30 percentage points. According to industry sources on the 21st, starting December, the price of Xeljanz 5mg Tab will decrease from KRW 10,996 to KRW 7,697, and Xeljanz 10mg Tab will decrease from KRW 18,250 to KRW 12,775. Additionally, the price of Jardiance 10mg Tab will be adjusted from KRW 582 to KRW 408, and Jardiance 25mg Tab from KRW 762 to KRW 533. This price adjustment is due to the inclusion of generic drugs in the reimbursement list. With the substance patent for Xeljanz expiring on the 22nd of this month, 17 products from 12 companies, including those from Daewoong Pharmaceutical and Chong Kun Dang, will be reimbursed starting on the 23rd. Generic versions of Jardiance also entered the market en masse in October. Following the expiration of the substance patent, 235 products from 37 companies were listed for reimbursement on October 24. Both products are achieving high sales in the market. Jardiance recorded KRW 66.3 billion last year based on UBIST data, while the combination product Jardiance Duo recorded KRW 41.8 billion, boasting a combined market size exceeding KRW 100 billion. Xeljanz also secured market presence with KRW 14.3 billion in outpatient prescriptions. The ex officio price reductions made for the extended-release and combination formulations are expected to cause significant concern for Pfizer and Boehringer. Starting December, the price ceiling of Xeljanz XR 11mg will drop from KRW 22,169 to KRW 17,071, and Xeljanz Syrup 1mg/mL will decrease from KRW 527,808 to KRW 369,443. Additionally, the price ceiling of Jardiance Duo Tab 5/1000mg will decrease from KRW 301 won to KRW 284, Jardiance Duo Tab 5/500mg from KRW 301 to KRW 278, and Jardiance Duo Tab 5/850mg from KRW 301 to KRW 278. Among the items subject to this ex officio adjustment, Xeljanz 5mg Tab, Xeljanz 10mg Tab, Xeljanz XR 11mg Tab, and Xeljanz syrup will see an additional price reduction on November 23 next year, once the one-year add-on premium ends, dropping to 53.55%, equal to the generic price. Jardiance 10 mg and 25 mg will undergo the same adjustment on October 24 next year, one year after generic entry. For both original manufacturers, generic competition and reimbursement price cuts will inevitably impact sales, prompting intense efforts to defend market share. Pharmacies are advised to take note of returns and settlement procedures following price changes for these frequently prescribed original products. Meanwhile, drug prices will also be adjusted for the menopausal hormone therapy drug Angelic Tab (drospirenone/estradiol, Bayer) and the antifungal agent Ambisome Inj (liposomal amphotericin B, Gilead) with the entry of their respective generics. The price of Angelic Tab will be adjusted from KRW 10,393 to KRW 5,565 starting in December. The price of Ambisome Inj will be reduced by 30% from KRW 129,392 to KRW 113,218, and after the add-on premium ends on October 1 next year, the price will drop to KRW 86,612.
- Policy
- Scope of use for Kisqali, Imfinzi expanded for combo use
- by Jung, Heung-Jun Nov 21, 2025 06:12am
- Use of combination therapies for breast cancer, endometrial cancer, urothelial carcinoma, and prostate cancer has been newly approved, expanding the expected treatment scope for drugs such as Kisqali and Imfinzi. Starting December, 5 new combination therapies for breast cancer, 2 for endometrial cancer, and 1 each for urothelial and prostate cancer will be established. The Health Insurance Review and Assessment Service (HIRA) is collecting industry opinion on the revision of the notification regarding drugs prescribed and administered to cancer patients until the 24th. For breast cancer, four new combination therapies using Novartis Korea's breast cancer treatment Kisqali (ribociclib) tablets will be added. Newly established methods include ribociclib + anastrozole, ribociclib + letrozole, and adding leuprorelin (LHRH agonist). Additionally, the non-reimbursed triple combination of inavolisib + palbociclib + fulvestrant—will also be added.. This option is used when cancer progresses despite treatment with other targeted therapies. Combination therapies using AstraZeneca's immunotherapy Imfinzi (durvalumab) for endometrial cancer and urothelial carcinoma will also be expanded. For endometrial cancer, two new regimens are introduced: either durvalumab monotherapy following combination therapy with durvalumab + carboplatin + paclitaxel, or durvalumab + Olaparib combination therapy. For urothelial carcinoma, the approach involves combination therapy with durvalumab + gemcitabine + cisplatin followed by radical cystectomy, with durvalumab used as adjuvant therapy. For both endometrial cancer and urothelial carcinoma, the addition of durvalumab to existing reimbursed regimens (carboplatin + paclitaxel, gemcitabine + cisplatin) will be added. For prostate cancer, a new combination therapy combining the non-reimbursed talazoparib with the reimbursed enzalutamide has been newly established. The newly established combination therapies expand treatment options using targeted therapies and immunotherapies based on genetic mutations and DNA repair capabilities. HIRA also revised reimbursement criteria for 19 items among Groups 1-2 anticancer drugs. This aims to clarify reimbursement standards for therapies that are not considered standard treatments. Changes, such as adding or deleting phrases, were made to the reimbursement criteria for anticancer drugs used in malignant melanoma, rhabdomyosarcoma, neuroblastoma, Wilms tumor, and retinoblastoma. HIRA stated, “We have announced additional combination therapies requested by academic societies that were deemed to comply with the general principles of the official notice and announcement.”
- Policy
- Ineffective dual pricing system to be revised
- by Jung, Heung-Jun Nov 20, 2025 06:16am
- The government has acknowledged the failure of the 'Separate Contracting System: Dual Pricing System', implemented last March to support the export of domestically developed new drugs. It will soon introduce a revised plan under a new name. The revised plan is expected to be submitted as an agenda item to the Health Insurance Policy Review Committee meeting on November 28 of this month, with the option to allow dual pricing for drugs other than those for severe and rare diseases being strongly considered. According to industry sources November 19, the government plans to significantly relax requirements for the dual pricing system, including conditions for ▲new drugs developed by companies that received Innovative Pharmaceutical Companies designation ▲eligibility for expedited review ▲domestic clinical trial requirements. Currently, no product meets all three requirements to use the dual pricing system. The system, though well-intentioned to support the development of domestic new drugs, has essentially remained a regulation without practical use. An official from pharmaceutical company A explained, "There were many restrictions. The number of new drug developments by domestic companies is at a low point, and few multinational pharmaceutical companies have received innovativeness designation," and added, "Moreover, the target for expedited review is often narrowed to severe and rare disease drugs, meaning ultimately, there are no drugs utilizing the dual pricing system." Currently, 45 domestic pharmaceutical and biotechnology companies of South Korea and four multinational pharmaceutical companies have received the innovativeness designation. Only drugs held by these companies that are also eligible for expedited review and have undergone domestic clinical trials could apply for the dual pricing system. Multinational pharmaceutical companies also assessed the original dual pricing system as insufficiently incentivizing. Since many multinational companies already utilize the refund-type Risk-Sharing Agreement (RSA), they were not interested in the dual pricing system, which offered no additional benefit. An official from multinational pharmaceutical company B assessed, "When the policy direction came out, we thought it would be widely used, but when we actually looked into it, there was no major advantage," and added, "There was many opportunity to utilize the RSA refund type, and since (applying the dual pricing system) did not grant an exemption from health-economic evaluation, it was difficult to see any added value." Following criticism in the National Assembly that the system lacked practical effectiveness due to zero uptake, the government is revising the regulation and renaming it the 'Flexible Drug Pricing Contracting System'. To avoid repeating the mistake of merely changing the name without addressing the lack of efficacy, incorporating field feedback is critical. The National Health Insurance Service (NHIS) is currently conducting internal reviews and gathering industry opinions for the introduction of the Flexible Drug Pricing Contracting System. Prospective expansion for eligible drugs for the dual pricing...will be gradually introduced to RSA drugs Even before the government announced the finalized plan, there was already discussion about the prospective expansion of the drugs eligible for the dual pricing system. Some predict that market-entry hurdles, such as the requirement for innovativeness designation and eligibility for expedited review, will be entirely removed. A multinational company official said, "The direction will likely be to allow it even for companies outside of the innovativeness designation. We anticipate that a wide range of products from pharmaceutical companies that show commitment will be permitted." The official also predicted, "We expect that RSA drugs will switch to the dual pricing system when their cost-sharing period ends or when the listing of a subsequent drug mandates the disclosure of their official listed price." Domestic pharmaceutical companies preparing for overseas export are also awaiting the government's detailed plan. They are, however, optimistic about the decision to expand eligibility. A domestic company official stated, "Since there are countries that reference our drug prices, it is beneficial if the official external price is higher. If implemented, we plan to enter the flexible contract system," and added, "Although the number of items for domestic companies is limited, it is much better than nothing. We will have to wait and see exactly how much they relax the rules, but I understand they are going to expand the scope of application proactively." This official estimated, "They seem to be aiming for implementation around the second half of next year, after consulting with the industry for necessary adjustments." Another industry official commented, "Given the external issues, the Lee Jae Myung administration is also interested in the dual pricing system, so I think it will be discussed as a priority item in the drug pricing reform." The commenter strongly suggested that the plan be submitted to the committee on the 28th of this month. The Ministry of Health and Welfare (MOHW) is being cautious ahead of the official announcement. A MOHW official said, "We are preparing improvements, and because the announcement will be made soon, it is difficult to disclose the details," and added, "We are discussing with the personnel in charge at the executive agency (NHIS) and are in the process of gathering opinions from stakeholders regarding the system improvement."
- Policy
- Temporary relaxation of reimb criteria for Dupixent ends
- by Jung, Heung-Jun Nov 20, 2025 06:15am
- The temporary relaxation of reimbursement requirements for certain drugs, which was implemented since April last year due to the medical staffing crisis, will end this year. Drugs like Dupixent, MabThera, and Soliris must resume the patient response assessments, which the drugs were granted exemption from, starting January next year. According to industry sources on the 19th, the Ministry of Health and Welfare (MOHW) will end the temporary relaxation of drug reimbursement requirements as a follow-up measure after lifting the severe-level health crisis alert. When a shortage of medical staff occurred due to the resident physician walkout, medications requiring pre-administration tests and evaluations were omitted at the discretion of treating physicians. Frontline clinicians had requested relief from strict reevaluation requirements that mandated reassessment before renewing prescriptions. In recognition of the practical difficulties in adequately conducting patient tests and evaluations, the reimbursement criteria were temporarily relaxed as an unavoidable measure. The alert was lifted on October 20th as the medical staffing shortage was resolved. However, to prevent confusion in clinical settings, a grace period of approximately 2 months was granted before ending the reimbursement relaxation. A wide range of medications are affected, including anticancer drugs, epilepsy treatments, dementia drugs, stimulants, and atopic dermatitis therapies. High-profile injectables such as Dupixent, MabThera, Soliris, Eylea, Humira, and Spinraza will once again require documented patient evaluations. Oral agents such as Xeljanz for autoimmune disease, Concerta OROS for ADHD, and Opsumit for pulmonary arterial hypertension are also included. These medications are covered under insurance only if administered based on test results. Periodic evaluations, such as every 3 to 6 months, must be conducted to qualify for coverage. For example, initial and maintenance treatment criteria often require submission of objective evidence such as prior treatment history, EASI score calculation, and lesion photographs. Beginning January, reimbursement will once again be contingent upon meeting such evaluation requirements, and providers are advised to exercise particular caution.
- Policy
- 'Photos' to be excluded from files for negotiated drugs
- by Jung, Heung-Jun Nov 19, 2025 06:08am
- "Some companies submitted product photos and test certificates, but their production records were not found the following year. This must not happen." The National Health Insurance Service (NHIS) made this statement during a recent drug price consultation session with pharmaceutical industry stakeholders, announcing future institutional changes. According to industry sources on November 18, the NHIS is gathering opinions on changes to the documentation required for negotiated drugs and the scope of disclosure for Risk-Sharing Agreement (RSA) drugs, until November 21. The main point of the changes to the negotiated drug documentation is to replace product photos with the Manufacturing Instruction Record. The goal is to prevent instances such as the submission of a semi-finished product test certificate or the forgery of a finished product test certificate when no finished product inventory is actually available for shipment. Consequently, product photos will not be required unless requested. The primary objective is to substitute these with the Manufacturing Instruction Record to secure credibility. The NHIS plans to conduct an internal review in December and apply these changes starting with the main negotiations for negotiated drugs in January next year. The NHIS is also pursuing the disclosure of information regarding RSA drugs. The NHIS is also seeking opinions on the scope of information to be disclosed, including the drug name and type, within the same deadline. Some in the National Assembly have argued that related information must be disclosed to ensure the transparent operation of the RSA system. The NHIS is seeking opinions on two options: Option 1, which discloses only the drug name, and Option 2, which discloses both the drug name and the RSA type. The NHIS is also gathering general opinions on the overall disclosure of RSA drug information. The NHIS plans to disclose drug name and the contractually set refund rate to subsequent reimbursement applicants. An NHIS official previously stated, "We believe it is right to disclose the refund rate information for RSA drugs. We will ensure that this information is provided upon request when a reimbursement application is submitted to HIRA. However, the requesting applicant must sign a non-disclosure agreement."
- Policy
- World’s first dutasteride+tadalafil combo to launch in Dec
- by Lee, Tak-Sun Nov 19, 2025 06:07am
- The world’s first dutasteride and tadalafil combination approved for benign prostatic hyperplasia (BPH) is expected to be released next month as a non-reimbursed product. This effectively confirms that the companies failed to secure reimbursement for their respective products. According to industry sources on the 17th, the dutasteride+tadalafil fixed-dose combination will begin sales next month at an expected price of KRW 1,300. Four products from four companies received approval: Dong-A ST's Dutana Tab 0.5/5mg, Shinpoong Pharm’s Avocial Tab 0.5/5mg, Dongkook Pharmaceutical's Uresco Tab 0.5/5mg, and DongKoo Bio&Pharma's Uroguard Tab 0.5/5mg. The four drugs were approved on January 23rd through joint development. The approved indication is the treatment of moderate to severe symptoms of benign prostatic hyperplasia. Dongkook Pharmaceutical is the manufacturer. These companies received approval in January and have been pursuing reimbursement listing. However, because tadalafil is a non-reimbursed ingredient, the combination could not be priced under standard calculations and would have had to enter the new drug reimbursement track, which requires substantial data submission and a prolonged evaluation period. Ultimately, manufacturers agreed to proceed with a non-reimbursed market launch. Some companies are informing distributors and retailers of their new product launch plans for December. The KRW 1,300 price is viewed as reasonable. Currently, the maximum price for dutasteride tablets 0.5mg is KRW 709. Given that the lowest price for tadalafil 5mg is around KRW 700, the combination drug’s price is lower than the sum of the two components. Still, since most BPH treatments in Korea are reimbursed, it remains uncertain how much demand there will be for a non-reimbursed option. Industry observers note that manufacturers may lean toward highlighting tadalafil’s erectile dysfunction benefits as a marketing angle.
- Policy
- First batch of drugs gain reimb through AEN pilot project
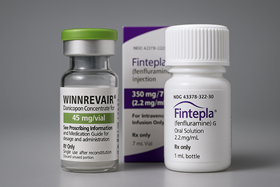
- by Jung, Heung-Jun Nov 17, 2025 06:11am
- With the first drugs under the Approval-Evaluation-Negotiation pilot program all securing reimbursement, the timing for the second batch of drugs currently under review to enter reimbursment listing is also approaching. Expectations are building for the possibility of their sequential insurance coverage as early as the first half of next year. With Bylvay Cap (odevixibat) gaining reimbursement coverage last month, all first-phase drugs under the Approval-Evaluation-Negotiation (AEN) project have now entered Korea’s reimbursement system, joining the previously covered Qarziba Inj (dinutuximab beta). The second batch of products under review are Winrevair (sotatercept) by MSD Korea for pulmonary arterial hypertension, Fintepla (fenfluramine) by UCB Korea for Dravet syndrome, and Limcato (anbal-cel) by the domestic company Curocell for large B-cell lymphoma. According to industry sources on the 16th, approximately 9 to 10 months have passed since the companies applied for reimbursement of these second-batch drugs. They are currently receiving cost-effectiveness reviews. Except for Winrevair, which received marketing authorization in July, the other two are also near approval. Among the three, Limcato was the first to apply for reimbursement in January, followed by Winrevair and Fintepla in February. Winrevair and Fintepla are being reviewed for cost-effectiveness, while Limcato is being reviewed for its clinical usefulness. Looking at the precedent set by the first batch of drugs that went through the reimbursement listing process, reimbursement for the second batch is expected early next year. As Qarziba was listed about 6 months after approval, if Winrevair, the only second batch drug approved as of now, follows a similar process, its reimbursement listing could occur as early as late this year or early next year. However, both Qarziba and Bylvay encountered hurdles at the Drug Reimbursement Evaluation Committee (DREC) stage, facing twists and turns before achieving reimbursement. Qarziba, which was approved by the Ministry of Food and Drug Safety (MFDS) in June last year, was initially deemed non-reimbursable at its first DREC review. It was subsequently granted for reimbursement in December last year after reapplying. It took approximately two years from Bylvay’s first reimbursement application in October 2023 to listing. Even at the point of its re-application last August, it had already been 14 months. This underscores the need to pass the DREC review without rejection and secure recognition of its reimbursement appropriateness, to achieve swift reimbursement listing. If a re-review process is required, reimbursement may be delayed beyond expectations. Meanwhile, the AEN linkage project is a pilot initiative aimed at enhancing access to high-priced treatments for severe diseases by shortening the period to listing of new drugs—which previously exceeded 300 days, including 120 days for MFDS (Ministry of Food and Drug Safety) approval, 150 days for HIRA (Health Insurance Review and Assessment Service) reimbursement evaluation, and 60 days for NHIS (National Health Insurance Service) drug price negotiations. During this year's National Assembly audit, the Ministry of Health and Welfare stated it would review plans to institutionalize the expedited listing system based on an analysis of the AEN pilot project's outcomes and feedback from the field.
- Policy
- Pricing nego for Erleada underway…last hurdle to reimb
- by Jung, Heung-Jun Nov 14, 2025 06:12am
- Janssen Korea's prostate cancer treatment Erleada (apalutamide) has entered price negotiations, the final hurdle for its reimbursement expansion in Korea. Erleada passed the Drug Reimbursement Evaluation Committee review last October and was deemed appropriate to expand reimbursement coverage to ‘high-risk non-metastatic castration-resistant prostate cancer’. According to industry sources on the 13th, the National Health Insurance Service (NHIS) is in pricing negotiations for Erleada’s reimbursement expansion. It being a drug subject to reimbursement under the risk-sharing agreement, the contract will be finalized based on the refund rate determined during negotiations. Once the price negotiations are complete, Erleada will be covered for ‘high-risk non-metastatic castration-resistant prostate cancer (nmCRPC)’ in addition to its existing coverage for ‘metastatic hormone-sensitive prostate cancer (mHSPC)’. The hormone-sensitive metastatic prostate cancer (mHSPC) indication was granted reimbursement in April 2023 as a refund-type risk-sharing agreement drug at KRW 20,045. At that time, Janssen focused its efforts on securing reimbursement for Erleada, even voluntarily lowering the price of its own prostate cancer treatment, Zytiga. It also became the first androgen receptor targeted agent (ARTA) to receive essential reimbursement at a 5% coinsurance rate. About six months later, competition intensified when Astellas Pharma Korea's Xtandi Soft Cap (enzalutamide) was added to the list as an essential reimbursement drug. Another competitor is Bayer Korea's Newbeqa, which also indicates treating ‘high-risk non-metastatic castration-resistant prostate cancer’. The company submitted a reimbursement application for Newbeqa in June this year, but the drug’s clinical benefit is still under review. With Erleada poised for reimbursement expansion, its prescription scope is also expected to broaden. Consequently, attempts to expand reimbursement for competing prostate cancer treatments are likely to follow. According to the pharmaceutical market research institution UBIST, Erleada's sales last year reached KRW 31.2 billion, a 368% increase from the previous year's KRW 6.6 billion.