- LOGIN
- MemberShip
- 2026-04-30 19:05:35
- Policy
- MFDS allocates KRW 812.2 billion for next year's budget plan
- by Lee, Hye-Kyung Sep 04, 2025 06:09am
- The Ministry of Food and Drug Safety (MFDS; Minister, Yu-Kyoung Oh) has announced that its budget for 2026 has been set at a total of KRW 812.2 billion, an increase of KRW 63.3 billion (8.4%) from this year's budget of KRW 748.9 billion. This budget plan was focused on facilitating the smooth execution of the new government's national tasks and restructuring expenditure to ensure efficient financial management. The 2026 MFDS budget plan includes four areas: enhancing safety and expanding the foundation for innovative growth in the pharmaceutical and bio-health sectors, strengthening customized safety support for food and medicine, considering the regulatory environment, creating a safe food and healthy dietary environment, and building a proactive food and drug safety management system for the future. A total of KRW 170.4 billion has been allocated to enhance safety and expand the foundation for innovative growth in the pharmaceutical and biotech sectors. The Fund for the Korea Orphan & Essential Drug Center will increase from KRW 4.5 billion this year to KRW 6.7 billion next year. To resolve the unstable supply of rare and essential medicines, MFDS will strengthen the stable supply foundation by expanding contract manufacturing of discontinued products and the emergency import of self-administered medicines with minimal demand. For supporting and building a management system, such as innovative medical devices, KRW 2 billion will be invested. A newly allocated budget of KRW 15 billion will be invested next year to fund the rapid commercialization of AI-driven products. MFDS will assist in the swift commercialization of promising AI-based products in the food and medical device sectors, thereby shortening Korean companies' development timelines and accelerating their market entry. The budget for the Korean Association Against Drug Abuse has been increased from KRW 16.5 billion this year to KRW 17.1 billion next year. A total of KRW 105.4 billion has been allocated for strengthening customized safety support for food and medicine, taking into account the regulatory environment. With the biohealth industry continuing to grow and the demand for systematic regulatory support from industries with limited experience and expertise increasing, MFDS plans to expand regulatory support by building an integrated consultation platform and securing customized consultation personnel for advanced and next-generation biopharmaceuticals. The budget for this will be significantly increased from KRW 500 million this year to KRW 11.4 billion next year. To lead the development of the pharmaceutical industry, MFDS will create review guidelines for new technologies and concepts, such as AI-driven products, and establish review standards that consider the characteristics of advanced and next-generation biopharmaceuticals. This will secure approval and review capabilities at the level of developed countries. To counter non-tariff barriers, such as country-specific approval regulations that hinder the export of Korean pharmaceuticals, MFDS will operate export approval support hubs. These hubs will analyze and provide case studies of approvals by product and offer regulatory consultations for export countries, thereby supporting the swift acquisition of overseas drug approvals. A new budget of KRW 5.5 billion has been allocated for operating a regulatory science talent development program in which universities, industries, and research institutes participate. The goal is to cultivate regulatory science professionals who can scientifically evaluate the safety of advanced bio-health products. A total of KRW 187.1 billion has been allocated for creating a safe food and healthy dietary environment, and a total of KRW 146.9 billion has been allocated for building a food and drug safety management system for the future. Next year, MFDS plans to build an automated drug approval and review system. This system will verify submitted document requirements, handle repetitive and routine tasks, and generate data summaries and reports. The system is intended to address the shortage of review personnel and expand patients' treatment opportunities through a faster drug approval process. The automation system will be expanded from generic drugs next year to active pharmaceutical ingredients (APIs) in 2027 and new drugs in 2028. To respond to the changing environment, such as online food distribution and the development of artificial intelligence, MFDS will establish an Information Strategy Plan (ISP) to build an "Integrated Food Safety Information Network." This network will consolidate 15 food-related information systems and automate public service and administrative tasks. In addition, as the scope of narcotics investigations expands, MFDS will secure digital forensics personnel and equipment dedicated to investigating medicinal narcotics. It will also broaden the synthesis of standard materials for new psychoactive substances and the evaluation of dependence on temporary narcotics. MFDS stated, "Once the 2026 budget plan is finalized through the National Assembly's deliberation process, we will do our best to execute the new government's national tasks and key projects without delay and protect the health and safety of the public."
- Policy
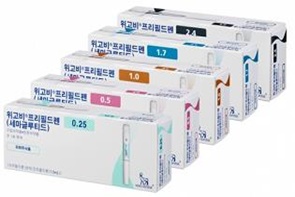
- HIRA "Reimb application of Wegovy has not been filed"
- by Lee, Jeong-Hwan Sep 04, 2025 06:08am
- Product photo of Wegovy The Health Insurance Review & Assessment Service (HIRA) announced that it will conduct a fair and swift evaluation if an application for reimbursement is submitted for Novo Nordisk's popular obesity drug, Wegovy (semaglutide). HIRA clarified that since Wegovy's company has not yet applied for reimbursement, HIRA is yet at the stage of determining Wegovy's National Health Insurance reimbursement. HIRA stated recently after a recent inquiry from Rep. Kim Seon-min of the Cho Kuk Innovation Party regarding the reimbursement status of Wegovy. Rep. Kim had asked HIRA for its stance on a plan to transition the non-reimbursed prescription drug Wegovy to National Health Insurance coverage and management. Rep. Kim likely inquired about the management plan related to the significant prescription volume of Wegovy, which has garnered immense popularity since its launch in Korea. According to data from the Drug Utilization Review (DUR) system, approximately 400,000 prescriptions for Wegovy have been issued in the roughly eight months since its launch in October 2024, which translates to about 80,000 prescriptions per month. The non-reimbursed prescription price for Wegovy ranges from KRW 200,000 to 300,000 for the 0.25mg, 0.5mg, and 1.00mg doses, and over KRW 400,000 for the 1.70mg and 2.40mg doses. Related to this, Rep. Kim has raised the need for managing the side effects that arise from people who are not overweight or obese using these drugs for cosmetic purposes. The medical community has also pointed out that some doctors are prescribing Wegovy and other obesity drugs for cosmetic, rather than therapeutic, purposes. This practice, they argue, can lead to repeated prescriptions and illegal trading of excessively prescribed medicines on online platforms. To address these issues, the medical community is proposing that obesity treatments be brought under the National Health Insurance system, subjecting them to a public monitoring and management system. In response to these concerns, Rep. Kim asked HIRA for its plan to manage the side effects of Wegovy through reimbursement. However, HIRA's response was general and procedural. HIRA explained, "For a new drug to be listed for reimbursement, the pharmaceutical company must first apply for reimbursement to the Minister of Health and Welfare and the head of HIRA, along with the necessary documentation," and added, "Then, this is followed by HIRA will evaluate the drug's clinical utility and cost-effectiveness, followed by drug price negotiations with the National Health Insurance Service. Finally, a notification is issued by the Ministry of Health and Welfare." HIRA said, "Wegovy's manufacturer had not yet submitted a reimbursement application after its approval by the Ministry of Food and Drug Safety," and added, "If an application for this drug is submitted in the future, we will ensure that it is evaluated fairly and swiftly." Meanwhile, the decision on National Health Insurance reimbursement for new obesity drugs, such as Wegovy, will be based on South Korea's health insurance finances, reimbursement equity, and the cost-effectiveness of these drugs.
- Policy
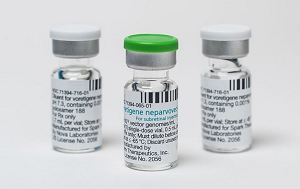
- Luxturna shows signif improvement in 3 out of 6 patients
- by Lee, Tak-Sun Sep 03, 2025 06:09am
- Luxturna (voretigene neparvovec, Novartis), a one-shot gene therapy that costs approximately KRW 300 million, showed clinically significant changes set by reimbursement criteria in only half of the patients. As indicated in the performance evaluation results disclosed last year, the effectiveness was only 50%. The Health Insurance Review & Assessment Service (HIRA) disclosed the latest performance evaluation results for Luxturna on August 29. Luxturna is a gene therapy for patients with inherited retinal disease, administered as a single subretinal injection in each eye. The ceiling price for one vial is KRW 325.8 million, with a patient co-payment of approximately KRW 10.5 million per person. Due to its high cost, health authorities have a risk-sharing agreement (RSA) with three types of contracts (refund, expenditure cap, and performance-based refund) to manage the drug expenditure. The performance-based refund contract, in particular, requires a post-administration performance evaluation to adjust the refund rate. The detailed reimbursement criteria for this drug are as follows. 1. A clinical evaluation (light sensitivity, vision, visual field, etc.) must be performed before administration (within 90 days before the first eye's injection) and at 1-3 months, 12 months, and annually for up to 4 years after administration (after the second eye's injection if both eyes are treated). Objective records, such as medical charts, must be submitted. 2. Light sensitivity must be evaluated using a full-field light threshold test with white light. 3. A clinically significant change is defined as an improvement of 1 log unit (average value for both eyes) or more from the baseline in the full-field light threshold test. The first performance evaluation result was disclosed on October 31 of last year, following the reimbursement listing in February of that year. The first evaluation followed up on four patients 1-3 months after administration. The result showed that two patients had a significant improvement, while two did not, indicating a 50% success rate. The latest evaluation results are based on a total of six patient cases. The evaluation was conducted 1-3 months after administration for two patients and 12 months after for four patients. The results showed that one patient at 1-3 months and two patients at 12 months had a significant improvement. In contrast, it was evaluated that one patient at 1-3 months and two patients at 12 months did not meet the criteria for significant improvement. It means that half of the patients were successful, while the other half failed. Since Luxturna's performance evaluation will continue for up to 4 years, the efficacy is expected to be more accurately verified as more data accumulates. By adjusting the refund rate accordingly, the high cost of the drug can be controlled. HIRA is currently conducting performance evaluations for other high-cost drugs, including Kymriah, Zolgensma, and Qarziba.
- Policy
- Bill to mandate generic substitutions gains momentum
- by Lee, Jeong-Hwan Sep 03, 2025 06:09am
- On September 2, the ruling party introduced a bill to mandate generic (ingredient-based) prescriptions for medicines with unstable supply. The bill establishes the legal basis for “shortage drugs” and allows prescribing by ingredient name instead of brand name. The bill holds significance in that it amends the Pharmaceutical Affairs Act to define drugs frequently in shortage and sets out legal procedures to designate such through public-private consultations, and amends the Medical Service Act to legislate the legitimacy and standards for mandatory ingredient prescriptions. In other words, the bill enforces a law that stipulates that “drugs in such unstable supply that generic prescribing must be enforced” through deliberations by a public-private consultative body. Representative Jong-tae Jang of the Democratic Party of Korea, who spearheaded the amendments, has stated he will push for their swift passage. The participation of fellow Democratic Party representative Yoon Kim (a physician) and Rebuilding Korea Party Rep Sun-min Kim (also a physician) is expected to reduce obstacles in upcoming committee reviews. Specifically, the bill requires the Ministry of Health and Welfare (MOHW) to establish a Supply Management Committee for Shortage Drugs. After deliberation and resolution by this committee, the Minister of Health and Welfare will designate shortage drugs. The committee will have up to 30 members, including the Vice Minister of Health and Welfare as Chair, Deputy Commissioner of the Ministry of Food and Drug Safety (MFDS) as Vice Chair. The remaining 28 will consist of : ▲Senior officials from relevant central government agencies (appointed by Presidential Decree), ▲ representatives recommended by the Chair of the Korean Pharmaceutical Association, ▲ representatives recommended by the Korean Medical Association under Article 28 of the Medical Service Act, ▲ representatives recommended by the Korea Pharmaceutical and Bio-Pharma Manufacturers Association and other relevant industry groups, as well as ▲ experts with sufficient knowledge and experience. Thus, government officials, pharmacists, physicians, manufacturers, and academia will collectively decide which drugs require mandatory generic prescribing due to unstable supply. The Minister of Health and Welfare will also have dedicated staff and budget authority to designate and de-designate shortage drugs, monitor supply conditions, and implement distribution improvement measures. The bill also legalizes distribution interventions alongside generic prescribing. If supply is deemed significantly disrupted, or upon request from another central administrative agency head, the Minister may—after committee review—order measures to improve distribution regarding sales outlets, procedures, volumes, and conditions. Pharmacies, medical institutions, wholesalers, and other designated entities will be legally obligated to comply with these measures. However, the Minister must first consult with the Minister of Strategy and Finance and the Chair of the Fair Trade Commission before issuing such orders. The MOHW Minister will also build and operate a shortage drug management system. This will allow requesting and collecting information necessary for distribution control, which includes production, shipments, sales, prescriptions, and dispensing data, from manufacturers, importers, wholesalers, pharmacies, and medical institutions. This effectively grants the MOHW authority over the entire supply chain, but also increases the Ministry’s accountability when shortages occur. The Minister may also designate certain shortage drugs as “emergency production/import drugs”, subject to committee review, which empowers the Minister to order manufacturers to produce or import the drugs. The Medical Service Act amendment stipulates that physicians and dentists must prescribe designated shortage drugs by generic name, not brand name. Violation will result in up to 5 years imprisonment or a fine up to KRW 50 million. This significant level of criminal penalty is expected to draw strong opposition from the Korean Medical Association and the broader medical community.
- Policy
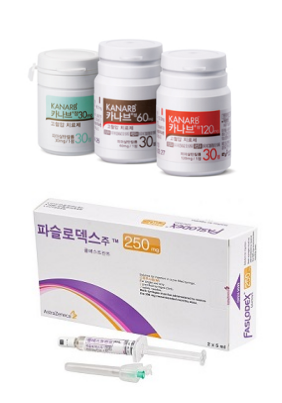
- Kanarb and Faslodex to keep their price until ruling
- by Lee, Tak-Sun Sep 02, 2025 06:11am
- The drug prices for Boryung's hypertension treatment ‘Kanarb’ and AstraZeneca's anticancer drug ‘Faslodex,’ which were determined to be reduced by the MOHW due to the entry of generics, will be maintained at their previous levels for now. The court has decided to suspend the execution of the price reduction until the first-instance ruling. As a result, the industry’s eyes are on whether these pharmaceutical companies will be able to halt the price reduction disposition through the main lawsuit. According to industry sources on the 1st, the Seoul Administrative Court accepted an application on the 27th of last month to suspend the execution of the price reduction order for 11 items, including Kanarb Tab. Consequently, the previous insurance price ceiling for the drugs will be maintained until two months after the date of the final judgment of the main trial. The affected items are Boryung's Kanarb Tab (3 dosages), Kanarb Plus Tab (2 dosages), Dukarb Tab (4 dosages), and Dongwha Pharmaceutical's LaCor Tab (2 dosages). At the end of June, the MOHW announced an ex officio adjustment and termination of the premium for these items following the entry of generic drugs containing the active ingredient, the single-ingredient fimasartan. Implementation was scheduled for July 1st. Four pharmaceutical companies' Kanarb generic products were listed for reimbursement last May. Consequently, health authorities proceeded with the ex officio price reduction process for Kanarb. Despite Boryung's objection, the price adjustment was ultimately finalized. The price caps for Kanarb Plus Tab and LaCor Tab, which are combination drugs that contain fimasartan, were also administratively reduced due to the price adjustment. For Dukarb, the premium granted for incrementally modified new drug combinations ended as two or more fimasartan-based single-ingredient drugs became available. Kanarb’s price was reduced by 30%, Dukarb by 21%, and Kanarb Plus and LaCor by 47%. Given that Kanarb and Dukarb each generate sales in the KRW 60 billion range, the price adjustment is expected to lead to a significant decrease in Boryung's overall sales performance. Consequently, many believed Boryung would seek to provisionally maintain its drug prices through litigation. The first trial for the main suit will commence in earnest with the first hearing scheduled for November 13. Kim & Chang is representing Boryung. The drug price for AstraZeneca's anticancer drug Faslodex (fulvestrant) will also be maintained until 30 days after the ruling date of the main case. With more than three companies producing generic versions, Faslodex's price adjustment period was scheduled to end last July. As a result, its insurance price cap was set to decrease from KRW 376,724 to KRW 288,194 starting August 1. AstraZeneca was reportedly considering withdrawing from the domestic market due to the Faslodex price cut disposition. This issue also surfaced during the confirmation hearing for Minister of MOHW Eun-Kyung Jeong. Domestic pharmaceutical companies have also expressed the view that if the original anticancer drug withdraws, it would be difficult for patients to find a substitute, given the market characteristics. Fortunately, the company did not withdraw its product but opted for litigation. On the 29th of last month, the Seoul Administrative Court granted its application for a stay of execution of the price reduction order. The main lawsuit was filed on July 25th, and no hearing date has been set yet. AstraZeneca's legal representative is the law firm Sejong. Although both pharmaceutical companies chose litigation to maintain drug prices, the risk is not entirely absent. This is because the Act on Drug Price Litigation Recovery and Refund that was implemented last year allows the National Health Insurance Service to refund the claimed amount for the trial period if the company loses the case. Nevertheless, analysis suggests the companies ultimately chose litigation due to the significant immediate financial loss and the potential for dispute over the price reduction decision.
- Policy
- "MFDS' data-based drug shortage response"
- by Lee, Jeong-Hwan Sep 02, 2025 06:09am
- Jeong Eun Keong, Minister of Health and Welfare The Ministry of Health and Welfare (MOHW) announced that it will address drug shortages by utilizing information from the "Pharmaceutical Production, Supply, and Prescription Data-based Supply Risk Prediction System," which is currently being established by the Ministry of Food and Drug Safety (MFDS). The MOHW has also proposed drug shortage response strategies, including domestically producing the raw materials and sub-materials for medicines and vaccines, as well as expanding a production support program for drugs with unstable supply to include up to four pharmaceutical companies. The MOHW also stated that the introduction of abortion drugs could only be expedited after the scope of legal abortion is defined through amendments to the Mother and Child Health Act and the Criminal Act. The following Q&A is based on the MOHW's responses to written inquiries from People Power Party Representative Han Ji-a and Democratic Party of Korea Representative Nam In-soon of the National Assembly's Health and Welfare Committee on August 27. "Responding to unstable supply by linking production, supply, and prescription systems" The MOHW's Division of Pharmaceutical Policy and Division of Health Industry Promotion responded to Rep. Han Ji-ah's inquiries about policy plans to strengthen the drug supply chain. First, the MOHW stated that "securing a stable drug supply chain is essential for protecting and maintaining public life and health, and is an essential task directly related to health security." The Ministry explained that it is "monitoring drugs with supply interruptions or shortages in cooperation with the MFDS, and taking customized measures through a public-private consultative body when necessary." The MOHW also stated that it would create countermeasures by linking its system with the one being developed by the MFDS for drugs with unstable supply. The MFDS is currently building a data-based drug supply risk prediction support system to enhance the prediction of unstable supply situations. The system is designed to comprehensively analyze drug production, supply, and prescription information. In particular, the National Assembly has a pending amendment to the Pharmaceutical Affairs Act that would strengthen the linkage of information on drugs with unstable supply between the MOHW and the MFDS. The Ministry expressed its commitment to support this legislation to address drug shortages actively. The National Assembly also promised to make efforts to produce drug and vaccine raw materials and sub-materials domestically, support the production of drugs with unstable supply, and stockpile essential medicines. The plan is to expand the project that provides budget support for pharmaceutical companies producing drugs with unstable supply from one to four companies. To respond to human infections of avian influenza and bioterrorism, the Ministry plans to prioritize the introduction of the latest overseas vaccines and stockpile them with domestically developed vaccines after their development is complete. It will also focus on national cooperation to diversify the sources of imports for active pharmaceutical ingredients (APIs) from China and India. "Introducing abortion drug requires an amendment to both Mother and Child Health Act and the Criminal Act" The MOHW states that amendments to the Mother and Child Health Act and the Criminal Act are essential for the domestic approval of abortion drugs like Mifepristone. It stated that to create a safe abortion environment, the scope of abortion must be established through amendments to the Criminal Act. While the Constitutional Court's ruling in April 2019 that the abortion ban was unconstitutional effectively nullified the relevant penal provisions, a legislative void has persisted for over six years due to the failure to pass supplementary legislation. As a result, the MOHW's stance is that it is difficult to carry out abortion-related administrative tasks. The MOHW plans to work closely with relevant ministries, including the Ministry of Justice and the MFDS, to support future legal and institutional improvements. Minister Jeong Eun Keong gave the same response during a questioning session with People Power Party Representative Cho Bae-sook at the National Assembly's Special Committee on Budget and Accounts on August 27. Minister Jeong explained, "As the Criminal Act and Mother and Child Health Act have not been amended since the unconstitutional ruling, there are safety issues." She added, "While there are no approved drugs in Korea, most countries in the world use approved drugs." And added, "The World Health Organization (WHO) also provides recommendations based on gestational age." Minister Jeong said, "I will consult with the MFDS on safe usage methods. We will consider both the fetus's right to life and the woman's right to health in a balanced manner."
- Policy
- Industry requests incentives for rare and orphan drug supply
- by Lee, Hye-Kyung Aug 29, 2025 06:07am
- The pharmaceutical industry has voiced the need for incentives to expand supply support, such as official certification, for companies supplying rare and essential drugs. The Ministry of Food and Drug Safety (MFDS, Minister Yu-kyoung Oh) visited the Korea Orphan & Essential Drug Center on the 28th to review the center's current projects supporting the supply of rare and essential medicines. It then held an ‘On-site discussion forum to support drug supply’ with the Korean Organization for Rare Diseases, the Korean Society of Health-System Pharmacists, the Korean Society of Cardiology, Huons, Korea United Pharm, and Sinwon Chemical. Pharmaceutical companies attending the meeting reportedly proposed measures such as incentive schemes and ways to shorten the emergency introduction period for rare and essential medicines. Patient groups requested guidance on the supply plans for medicines that had recently experienced supply disruptions, resulting in treatment difficulties. MFDS Minister Yu-Kyoung Oh said, “We will actively support and continuously communicate with the industry and patient groups to ensure the stable and prompt supply of rare and essential medicines needed for treating patients with rare and intractable diseases.” Young-Rim Kim, Director of the Korea Orphan & Essential Drug Center, said, “We will work with the MFDS to reflect the opinions provided by patient groups and pharmaceutical companies in the operation of the rare and essential medicines supply support project and ensure these medicines are safely supplied to patients.” The Ministry of Food and Drug Safety (MFDS), together with the Korea Orphan & Essential Drug Center, is working to supply rare disease medicines in a timely manner and reduce patient burden by rapidly introducing orphan drugs and operating patient support programs. The center is also pursuing self-sufficiency and reducing reliance on foreign countries by utilizing the technological development of raw and finished drugs for essential national medicines that are taken daily through consignment production with domestic pharmaceutical companies. The MFDS plans to continue communicating with patient groups and industry stakeholders to identify and implement various policies ensuring the stable supply of medicines in need. Meanwhile, this forum was arranged to thoroughly explore government support measures for the stable supply of medicines to patients with rare and intractable diseases, an issue raised during the ‘Food and Drug Policy Connection Open Forum (Medical Products Sector)’ held on July 22. Opinions presented at the meeting will be actively reflected in future policies for the stable supply of medicines.
- Policy
- Next year’s reimb reevaluations to be discussed further
- by Lee, Tak-Sun Aug 29, 2025 06:07am
- The government has decided to continue discussions on the 2026 reimbursement adequacy reevaluation plan. While the reevaluation was initially expected to be approved this month through the Health Insurance Policy Deliberation Committee review, it is reported that disagreements remain unresolved regarding selection criteria, procedural improvements, and the ingredients targeted for 2026. Some suggest the findings from an ongoing post-listing drug control study commissioned by the MOHW may be incorporated. According to industry sources on the 28th, the ‘Plan to Promote Drug Reimbursement Adequacy Reevaluations’ reported to the HIPDC subcommittee was not placed on the agenda for the HIPDC plenary session on the 28th. The government stated it would continue discussions. The plan reported to the subcommittee shows that the selection criteria for evaluation will change from the current threshold of an increase of 0.1% or more of the average claim amount over three years (approximately KRW 20 billion) to claims of KRW 10 billion or more. Furthermore, the condition for “'listed reference countries' will be expanded from the current requirement of fewer than 2 countries to fewer than 3 countries. The seven drug ingredients subject to reevaluation in 2026 are: Ginkgo biloba extract that was approved in 1989, Calcium dobesilate hydrate, Calladinogenase, Meglumine gadoterate, Diacerein, Afloqualone, and Octilonium bromide. The level of reimbursement will also be differentiated based on evaluation results. For drugs with unclear clinical utility but high social necessity, a 50% selective reimbursement rate will apply; for those with low social necessity, an 80% selective reimbursement rate will apply. Additionally, if the drug cost is high compared to its alternatives, an additional price cut will be applied. Previously, decisions only excluded drugs from reimbursement or maintained reimbursement, with drug prices adjusted through voluntary reductions by manufacturers to demonstrate cost-effectiveness. The pharmaceutical industry has clearly stated its opposition to this plan. Specifically, the argument is that the late selection of next year's reevaluation targets has rendered insufficient time to prepare data, making the 2026 reevaluation unfeasible. This is because it takes about a year for drug efficacy to be included in textbooks. Additionally, opinions have emerged that the selection of ginkgo biloba extract preparations as next year's target ingredients is unreasonable. So the pharmaceutical industry has expressed temporary relief as the government decided to further discuss the reevaluation plan. It is known that the government authorities were not in agreement regarding this reevaluation plan at the Drug Reimbursement Evaluation Committee and the Health Insurance Review and Assessment Service subcommittee level. Particularly, it is reported that further discussion would be needed regarding changes to the criteria. Some predict that the reimbursement reevaluation plan will be revisited based on the ‘Study on Integrated Post-listing Control of Drug Prices’ currently underway as an MOHW research project. This study is scheduled to be conducted by the Catholic University of Daegu Industry-Academic Cooperation Foundation (Director Hyeop-sang Yoon) until November. It is anticipated that the study conducted last year by the Korea Institute for Health and Social Affairs will include implementation plans related to integrated post-listing control measures. An industry official explained, “Given that the post-listing control study is currently underway, it appears the new administration will comprehensively review the post-listing control system. Consequently, there are projections that the finalization of the reevaluation plan may extend beyond this year.”
- Policy
- Bill banning reverse payment agreements passes committee
- by Lee, Jeong-Hwan Aug 28, 2025 06:10am
- The amendment to the National Health Insurance Act, which regulates "reverse payment agreements" where original drug companies and generic drug companies collude by exchanging money to delay or not release generics, thereby avoiding a price reduction for the original drug, passed the National Assembly Health and Welfare Committee review on the morning of the 27th. The illegal collusion among pharmaceutical companies to withhold generic drug launches has forced patients to pay unfairly high prices for medications. This practice must be eradicated to eliminate such disadvantages and prevent unnecessary leakage of health insurance funds. The bill on revising the Pharmaceutical Affairs Act, which expands the post-notification system for generic substitution at pharmacies to the information system established and operated by the Ministry of Health and Welfare and the Health Insurance Review and Assessment Service, and improves the definition of essential medicines and expands support to include drugs with no substitutes or facing supply instability, has also cleared the Health and Welfare Committee. These bills will take effect according to the implementation date specified in the supplementary provisions once they pass the plenary session after review by the Legislation and Judiciary Committee and are promulgated by the government. Original-Generic Pharma Company Collusion Prohibition…Price cuts or reimbursement suspensions applied for offenses The core provision of the National Health Insurance Act amendment bill proposed by Democratic Party of Korea lawmaker Young-seok Seo stipulates that if the Fair Trade Commission uncovers collusion where a generic drug company agrees not to launch a generic in exchange for exclusive domestic distribution rights from an original drug company, the price of the unfairly traded drug will be reduced or its reimbursement suspended. The bill passed by the Health and Welfare Committee amends Article 41-2 (Reduction of Upper Limit Amount for Reimbursement Costs for Drugs, etc.) of the National Health Insurance Act. Specifically, the bill stipulates that for cases violating Article 40(1) or Article 45(1) of the Monopoly Regulation and Fair Trade Act, where the violation was committed “for the purpose of increasing or maintaining the reimbursement price ceiling for drugs,” the insurance price of the drug can be reduced or reimbursement suspended. The amendment allows for a maximum 20% reduction in drug prices when a reverse payment agreement violation is first detected. If another reverse payment agreement is confirmed within 5 years of the price reduction, the price can be cut by up to 40%. If a second violation of the reverse payment agreement is detected within five years after the second price reduction, the application for reimbursement for the drug can be suspended for up to one year. The effective date for the provisions preventing reverse payment agreements is ‘the day six months after the government's promulgation’. Consequently, collusive reverse payment agreements will now face the same level of penalties as illegal pharmaceutical rebates, including drug price cuts and reimbursement suspensions. The Health and Welfare Committee explained, “This bill enables the reduction of the maximum reimbursement amount for drugs related to unfair joint conduct or unfair trade practices, and the suspension of reimbursement coverage. It will prevent pharmaceutical companies and others from profiting through unfair joint conduct or unfair trade practices and establish a fair drug sales order.” Post generic substitution notification system expanded to HIRA A revision to the Pharmaceutical Affairs Act, proposed by Democratic Party of Korea lawmakers Young-seok Seo, Su-jin Lee, and Byung-duk Min, was passed as an alternative bill by the Health and Welfare Committee. It expands the pharmacists’ post-dispensing notification system to the information system operated by the Health Insurance Review and Assessment Service (HIRA). The bill incorporates the existing regulation requiring pharmacists to inform patients when substituting a drug listed on a prescription with an item recognized by the Ministry of Food and Drug Safety as bioequivalent (generic substitution) and to notify the prescribing physician or dentist within one day (or three days if unavoidable circumstances exist). The key provision approved by the Health and Welfare Committee is the establishment of a new Article 27-2 (Establishment and Operation of a Substitution Dispensing Information System) in the Pharmaceutical Affairs Act, enabling the Minister of Health and Welfare to establish and operate an information system to support post-notification of generic substitution.. Notably, the bill allows the Minister of Health and Welfare to delegate this task to the Health Insurance Review and Assessment Service (HIRA), with the necessary details to be stipulated under the Ministry of Health and Welfare ordinance. The effective date for the simplified generic substitution regulations is set for five months after the government's promulgation. This establishes the legal basis for the Ministry of Health and Welfare and HIRA to build and operate a generic substitution information system to support post-notification of generic substitution under the Pharmaceutical Affairs Act. It is expected that this will enable more efficient communication of generic substitutions while clarifying the accuracy of such notifications, thereby enhancing information sharing between physicians and pharmacists. Expansion of the Definition of Essential Medicines & Legal Basis for the National Essential Medicines Stable Supply Council The Committee also passed a revision to the Pharmaceutical Affairs Act that improves and expands the definition of essential medicines to include those without substitutes or facing supply instability, and elevates the legal basis for the National Essential Medicines Stable Supply Council from a presidential decree to a law. The bill provides a detailed definition of essential medicines. These include medicines essential for maintaining the national health system, such as disease control and radiation disaster prevention (Item a), and medicines that are essential for healthcare but lack substitutes or face supply instability despite market mechanisms (Item b). Furthermore, a new Article 83-5 (National Essential Medicines Stable Supply Council) was added to the Pharmaceutical Affairs Act. This mandates the establishment of a National Essential Medicines Stable Supply Council within the Ministry of Food and Drug Safety (MFDS) to develop countermeasures for both national essential medicines and medicines that, while not designated as such, require stable supply due to temporary increases in demand. Notably, the composition of the National Essential Medicines Supply Council, previously chaired solely by the MFDS Vice Minister, was expanded to include ‘one senior official from the Ministry of Health and Welfare designated by the Minister of Health and Welfare,’ bringing the total to two chairs. This appears aimed at strengthening national management capabilities by adding the Ministry of Health and Welfare to the MFDS as a government ministry with authority over national essential medicines. Furthermore, the legal basis for the composition of the council members was clarified. The effective date of the bill was set as one year after its promulgation. While the definition of essential medicines is being expanded, the government's basis for supporting medicines without substitutes and medicines with unstable supply requiring stable supply due to temporary increases in demand is expected to be strengthened.
- Policy
- Reimb listing approved for the first generic of 'Angeliq'
- by Lee, Tak-Sun Aug 27, 2025 06:07am
- Bayer The first generic of Angeliq Tab (drospirenone·estradiol, Bayer), a hormone-based medicine for use in postmenopausal women, will be included in the reimbursement listing. Analysis suggests that the commercialization of domestically produced generic is significant, considering that there had been supply issues related to Angeliq, which is an imported medicine. According to industry sources on August 26, Dalim BioTech's 'Anzeno Tab' will be added to the reimbursement list on September 1 with a ceiling price of KRW 5,565. The product is manufactured directly by Dalim BioTech. Anzeno Tab is a generic drug with the same active ingredients as Bayer's Angeliq Tab. Angeliq is approved for ▲Hormone replacement therapy for estrogen deficiency in women who are at least one year post-menopause, and for the ▲Prevention of osteoporosis in post-menopausal women who are intolerant of or have contraindications to other approved drugs and have an increased risk of fracture. Based on 2024 UBIST data, Angeliq's outpatient prescription sales amounted to KRW 12 billion. Because it is a hormonal drug requiring a separate manufacturing facility had previously prevented generic products from entering the market. Consequently, when supply issues arose with the imported original drug, Angeliq, pharmacies had no identical alternatives, leading to significant difficulties. Alternative prescriptions with similar drugs were the only option, resulting in a continuous demand for generic development. In 2021, the persistent shortage of Angeliq due to production delays at Bayer's Berlin plant caused difficulties for pharmacists. With the launch of this in-house-manufactured generic, the issue of supply instability is expected to be largely resolved. The price of Anzeno Tab was set at KRW 5,565, which is 53.55% of Angeliq's price, without any additional premium, as it met all the required criteria. Angeliq Tab's current ceiling price is KRW 10,393.