- LOGIN
- MemberShip
- 2026-05-04 19:44:18
- Company
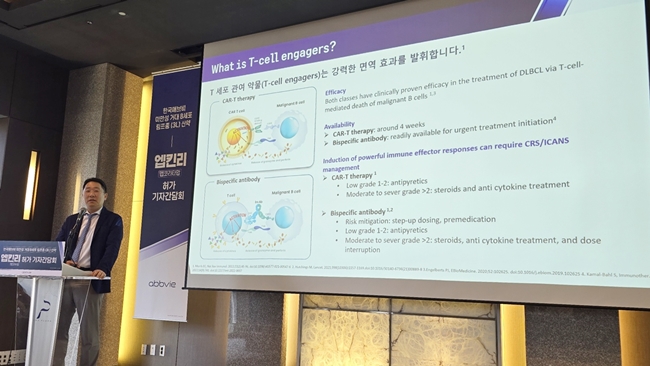
- Will Epkinly become a game-changer in the blood cancer mkt?
- by Hwang, Byung-woo Jul 12, 2024 05:47am
- The arrival of bispecific antibody-based therapies in Korea is expected to shift the blood cancer treatment market paradigm. The industry is welcoming the introduction of a new treatment option, as an unmet need exists in diffuse large B-cell lymphoma (DLBCL), which has a poor prognosis even after three or later lines of treatments. AbbVie hosted a press conference on the 10th to highlight the significance of Epkinly’s approval. Professor Jin Seok Kim. Department of Hematology-Oncology at Severance Hospital, is presenting data related to blood cancer.AbbVie hosted a press conference on the 10th to celebrate the launch of 'Epkinly (epcoritamab)’ as a third-line treatment for DLBCL and highlight the implications of the approval. Epkinly was approved by the Ministry of Food and Drug Safety in late June for the treatment of adult patients with relapsed or refractory DLBCL who have received 2 or more prior systemic therapies. Epkinly is a humanized bispecific antibody (IgG1) that binds to CD20 on B cells and CD3’s extracellular specific epitopes on T cells. It has a mechanism of action that induces specific T-cell activation and T-cell-mediated CD20-expressing cell death by simultaneously acting on CD20-expressing cancer cells and CD3-expressing endogenous T cells. Epkinly is the first subcutaneous bispecific antibody approved in Korea for the treatment of DLBCL. It has the advantage of being administered in less than one minute, allowing for a relatively short hospital stay and rapid treatment. The duration of the treatment is not limited and can be administered until disease progression or unacceptable toxicity Epkinly’s approval is noteworthy because it provides a new option in the treatment of DLBCL, an area with much unmet need. Professor Deok Hwan Yang, Department of Hematology-Oncology at Chonnam National University Hwasun Hospital, said, "Even after receiving first-line standard therapy for DLBCL, 30 to 40% of patients relapse or become refractory to treatment and move on to the next line of treatment. Patients who relapse after receiving autologous stem cell transplantation as a second-line treatment have poor prognosis, and patients who relapse after receiving CAR-T as a third-line treatment also have poor outcomes." This means that patients who relapse after receiving three or more lines of treatment generally have a poor prognosis, with lower overall response rates and worse survival outcomes. "Currently, there are limited options in the third and later lines of treatment, with no set standard of care. In this way, there is a large unmet need for new options.” (From the left) Professor Deok Hwan Yang, Department of Hematology-Oncology at Chonnam National University Hwasun Hospital; Professor Jin Seok Kim. Department of Hematology-Oncology at Severance HospitalThe study that became the basis of Epkinly’s approval is the EPCORE NHL-1 study, a non-randomized, single-arm trial. Its efficacy analysis showed an overall response rate (ORR) of 62%, with 39% achieving complete response (CR). Professor Jin Seok Kim. Department of Hematology-Oncology at Severance Hospital, said, “We found Epkinly was well tolerated in heavily pretreated third-line patients, and most adverse events were manageable and predictable. At 30 months of follow-up, median overall survival (mOS) was 19.4 months, confirming that it is a viable option for prolonging survival in this patient population." The advent of such bispecific antibody-based therapies sheds light on the question of how they will compare to CAR-T therapies that are being reimbursed through national health insurance. Experts predict that bispecific antibody therapies such as Epkinly will become complementary options in the future, assuming that they also be reimbursed. Professor Kim explained, "If you look at the average cost, I think they will be very similar to CAR-T therapies, and I don't think one can be a substitute for the other. I think it's going to be a patient-specific choice because they have different targets, but I think there will be cost concerns."
- Company
- "Enhertu gets expanded indication to treat lung cancer"
- by Son, Hyung-Min Jul 11, 2024 06:12am
- Ahn Myung Ju, Professor of the Division of Hematology-oncology in the Department of Medicine at Samsung Medical Center “Previously, patients could only use Enhertu by participating in clinical trials conducted in tertiary general hospitals. Current approval in South Korea will provide a new treatment option for patients with HER2 mutant metastatic non-small cell lung cancer (NSCLC). Brain metastasis is common in lung cancer patients. Because Enhertu demonstrated a response rate of 50% in patients with brain metastasis, it is clinically significant.” During a recent meeting with Daily Pharm, Ahn Myung Ju, Professor of the Division of Hematology-oncology in the Department of Medicine at Samsung Medical Center, highly regarded the clinical value of Enhertu (ingredient: trastuzumab deruxtecan). Enhertu, an antibody-drug conjugate (ADC) jointly developed by Daiichi Sankyo and AstraZeneca, has been approved for the treatment of HER2-positive breast cancer and gastric cancer. As Enhertu demonstrated to be effective in HER2 mutant NSCLC and HER2-low breast cancer, its indication was expanded in May. HER2 mutant NSCLC is a rare cancer that occurs in about 2-4% of patients with NSCLC. Previously, the treatment of this cancer with chemotherapy and immunotherapy for cancer was limited, and HER2-targeted therapy showed inconsistent results. Therefore, there has been an unmet need for an effective treatment option to increase the survival in patients. Previously, several targeted treatment options, such as Hanmi Pharm’s poziotinib, were tested for the treatment of HER2 mutant NSCLC but failed to demonstrate efficacy. Therefore, Enhertu is likely to provide new hope to patients with HER2 NSCLC, which is categorized as a rare cancer. Ahn said HER 2 mutation gained attention in NSCLC after the availability of next-generation sequencing (NGS) testing and targeted therapies like Enhertu. It will provide a new treatment opportunity to patients with NSCLC. Enhertu is approved in South Korea based on phase 2 trials Enhertu is an ADC that targets HER2 mutations by linking an antibody to a payload that destroys tumor cells. This treatment works by binding antibodies to specific antigens, triggering an Antibody-Dependent Cellular Cytotoxicity (ADCC) reaction, and allowing the payload to enter tumor cells and exit through their membranes. This mechanism results in a bystander effect, enabling the destruction of cancer cells, including those without HER2 mutations, demonstrating its anti-tumor efficacy. Enhertu is effective in various diseases, including breast cancer, gastric cancer, colorectal cancer, and NSCLC. In DESTINY-Lung02 study, Enhertu demonstrated antitumor response to the second-line treatment of HER2 mutant metastatic NSCLC. This study evaluated the efficacy and safety in patients with advanced, unresectable, or metastatic NSCLC who have previously been treated with systemic therapy, including platinum-based chemotherapy, more than once. The clinical results demonstrated that Enhertu had confirmed ORR of 49%, complete response (CR) of 1%, and partial response (PR) of 48%. The mean duration of response (DOR) was 16.8 months. “Previously, the second-line treatment, docetaxel, showed a response rate of only 10-15% and a median progression-free survival (PFS) of just 5-6 months. In contrast, Enhertu showed a response rate of 50% and a durable response period lasting up to 16 months. Moreover, brain metastasis is commonly found in lung cancer, and Enhertu has demonstrated an intracranial objective response rate (IC-cORR) of over 50%, even in patients with brain metastasis,“ Ahn said. “Kadcyla, previously used for HER2-positive breast cancer, showed a response rate of over 50% in non-small cell lung cancer (NSCLC) but had limitations due to the short duration of response. In this context, Enhertu has recently gained attention for demonstrating efficacy in HER2-mutated metastatic NSCLC patients,“ Ahn said. ”Although it’s a phase 2 single-arm study, Enhertu demonstrated better outcomes than data from existing medications, warranting consideration for reimbursement,“ Ahn added. "Enhertu use will be greater" in HER2 NSCLC In the case of metastatic non-small cell lung cancer (NSCLC) with HER2 mutation, it represents about 2-4% of all NSCLC cases.This cancer type is characterized by adenocarcinoma histology, prevalence in non-smokers and females, and a higher incidence among East Asian populations, including Japan and Korea. Additionally, it exhibits a higher rate of brain metastases. HER2 abnormalities are categorized into ‘gene mutations,‘ ‘gene amplifications,‘ and ‘protein overexpression.‘ In most cancers, such as gastric cancer and breast cancer, HER2 abnormalities mainly manifest as an overexpression of the HER2 protein. Previously, HER2 overexpression gene abnormalities were generally considered to not play a significant role in NSCLC. First-generation platinum-based chemotherapy has traditionally been the standard treatment for HER2 mutant non-small cell lung cancer. Recently, combination therapies of platinum-based chemotherapy and immunotherapy for cancer have also been employed. However, non-smokers are known to have a poorer response to immunotherapy for cancer, leading to worse prognosis. “So far, in HER2-mutated metastatic non-small cell lung cancer (NSCLC), drugs like Giotrif (afatinib), which is developed to target Epidermal Growth Factor Receptor (EGFR) mutations, have been used, but their response rates have been low, around 10%,“ Ahn said. “Additionally, poziotinib showed a better response rate of approximately 27%, but its use has been challenging due to significant toxicity and side effects,“ Ahn added. Ahn said that Enhertu will bring many changes to the treatment of NSCLC. “In the United States, Enhertu is indicated for the treatment of HER2 mutant metastatic NSCLC with the U.S. FDA approval, and it is widely used with reimbursement coverage. Even in Europe with stringent standards, non-small cell lung cancer patients are strongly recommended to undergo HER2 mutation testing, and those with HER2 mutations are advised to use Enhertu,“ Ahn said. “It is unfortunate that patients cannot afford the monthly cost of approximately 7 to 8 million won for Enhertu, which allows them to maintain their daily lives for over a year. Therefore, it would be beneficial if the government explores various measures, such as selective reimbursement, to alleviate the financial burden on patients, even if it means slightly increasing the portion they pay out of pocket,“ Ahn emphasized.
- Company
- Will Trodelvy be deliberated for reimb by DREC in August?
- by Eo, Yun-Ho Jul 11, 2024 05:47am
- The road to reimbursement for the new ADC breast cancer drug Troldelvy remains a bumpy one. The agenda remains pending for 8 months now. Gilead Sciences' triple-negative breast cancer (TNBC) drug Troldelvy, whose reimbursement request received 100,000 consents in a public petition, was not presented for deliberation to the Health Insurance Review and Assessment Service's Drug Reimbursement Evaluation Committee in July. The drug’s application has remained without progress since its reimbursement standard was set by the Cancer Disease Review Committee in November last year. Therefore, attention is now focused on whether the drug will be submitted to DREC in August. Trodelvy is already listed in about 30 countries around the world. Taiwan, which has a single-payer healthcare system similar to South Korea's, began reimbursing Trodelvy in February this year. The global rush to improve patient access to Trodelvy has been driven by the poor treatment environment for metastatic triple-negative breast cancer and the clinical value of Trodelvy. Triple-negative breast cancer is an aggressive form of breast cancer that recurs and metastasizes rapidly. Patients with metastatic triple-negative breast cancer who have metastasized despite treatment have a life expectancy of only a few months even with chemotherapy. However, chemotherapy has long been the standard of care due to the lack of targets that can effectively kill cancer cells. Trodelvy, the first Trop-2-targeted antibody-drug conjugate (ADC), is the only treatment for metastatic triple-negative breast cancer in the second-line or higher setting that has been shown to prolong survival compared to chemotherapy and has settled as the global standard of care since its introduction. Currently, major guidelines in the U.S. and Europe specify Troldelvy as the preferred agent for patients with previously treated metastatic triple-negative breast cancer. In a Phase III study, the overall survival of the chemotherapy arm was 6.9 months, compared to a nearly one-year survival. (11.8 months) in the Troldelvy arm, In addition, Troldelvy demonstrated an effect in controlling symptoms and pain caused by cancer and an improvement in patients' quality of life by improving their overall health status. Trodelvy was awarded the highest possible score of 5 points on ESMO-MCBS, the European Society for Medical Oncology's (ESMO) scale used to rate the value of anticancer drugs. A score of 5 indicates that a drug is effective not only in prolonging patient survival but also in improving quality of life, and Troldelviy is the only treatment for metastatic triple-negative breast cancer to receive a score of 5 on ESMO-MCBS. In fact, the U.K. has detailed the rationale behind its assessment, stating that the state’s reimbursement decision was based on the severity of metastatic triple-negative breast cancer and the survival benefit of Troldelvy. Similar to Korea, the U.K. uses the incremental cost-effectiveness ratio (ICER) to evaluate new drugs for health insurance coverage. While the UK has one of the highest reimbursement barriers for new drugs, it applies flexible pharmacoeconomic evaluation criteria for innovative drugs used for serious conditions to improve patient access. In the UK, Troldelvy was granted preferential economic evaluation because it prolonged survival in terminally ill patients with less than 2 years of life expectancy, whose population is even smaller than those of rare diseases. As a result, Trodelvy gained access with an ICER threshold that was approximately twice higher than that of general drugs. Meanwhile, Troldelvy has been the subject of a series of petitions this year, gathering more than 100,000 consents online. Since the petition system’s inception, Trodelvy is the only drug to be referred to the National Assembly for garnering more than 50,000 signatures. The Korean Alliance of Patients' Organizations also responded to the desperate pleas of patients and their caregivers when the petition was abandoned due to the expiration of the 21st National Assembly's term. In May, the organization submitted a letter directly to the Ministry of Health and Welfare requesting a prompt review of the reimbursement of drugs with high patient demand, including Trodelvy.
- Company
- From obesity to brain diseases...expansion of GLP-1 agonists
- by Son, Hyung-Min Jul 11, 2024 05:46am
- In-Young Choi, Head of R&D at Hanmi Pharmaceutical The domestic pharmaceutical bio industry is looking into the possibility of developing various new drugs with GLP-1 agents. As Novo Nordisk and Lilly's GLP-1-based obesity drugs have become global blockbusters, latecomers are also avidly developing jumping in to develop the next blockbuster. Major domestic companies are conducting clinical studies on the use of GLP-1 agents not only for obesity, focusing on the mode of administration and quality of weight loss effects, but also in the field of degenerative brain diseases such as metabolism-associated steatohepatitis (MASH) and Alzheimer's disease. On July 10, the Korea Biotechnology Industry Association and RX Korea hosted the BIOPLUS-INTERPHEX KOREA 2024 (BIX 2024). At the event, major Korean pharma and biotech companies introduced their GLP-1 drug candidates. GLP-1 drugs can be effective in weight loss by increasing satiety and improving insulin secretion and sensitivity, resulting in favorable glycemic control. Recently, GLP-1 agents have been reported to be effective against cardiovascular diseases and kidney diseases, and various clinical studies are underway on their use in patients with alcoholism and dementia. At the event, Hanmi Pharmaceutical introduced its new drug candidate for obesity which has a novel mode of action. The company is developing HM15275, an obesity drug candidate that simultaneously acts on glucagon-like peptide-1 (GLP-1), glucose-dependent insulinotropic peptide (GIP), and glucagon. HM15275 recently entered a Phase 1 clinical trial in the U.S. To date, a dual GLP-1-GIP receptor agonist Zepbound has been released, but there is no commercialized triple agonist that also includes glucagon. Hanmi Pharmaceutical is aiming to launch a first-in-class drug in this category. In preclinical trials, HM15275 showed less muscle mass loss and higher weight loss than Lilly's obesity drug tirzepatide (Zepbound). In-Young Choi, Head of R&D at Hanmi Pharmaceutical, said, "There are three strategies to developing a third-generation obesity drug following Wegovy and Zepbound. The strategies include increasing the dose, targeting a new mode of action (MOA), or adding substances such as glucagon that can increase the weight loss effect. We are exploring all possibilities through our various pipelines.” Choi added, "There is still an unmet demand for the weight loss effects of GLP-1 obesity drugs such as Zepbound and Wegovy, and the quality of weight loss and reduction of the weight rebound will be the indicators of competitiveness." Mi-Kyung Kim, Vice President and Head of Research HQ at Dong-A ST Also at the event, Mi-Kyung Kim, Vice President and Head of Research HQ at Dong-A ST, introduced its company’s new obesity drug DA-1726. According to Kim, The company completed preclinical studies and is in Phase I clinical trials for DA-1726., with the first patient dosing in April of this year. DA-1726 is a new drug candidate that is being developed as a long-acting oxyntomodulin peptide analog for the treatment of obesity. It acts simultaneously on both GLP-1 and glucagon receptors to suppress appetite, stimulate insulin secretion, and increase basal metabolism in the periphery, ultimately leading to weight loss and glycemic control. In preclinical trials, DA-1726 demonstrated similar weight loss efficacy despite higher food intake compared to tirzepatide, with less rebound after weight loss compared to tizetapide. Kim explained, "The U.S. Food and Drug Administration (FDA) requires a weight loss effect of 5% or more for an obesity drug, but the market is looking for more. We are focusing on the quality of weight loss." GLP-1 agents show promise in MASH·brain disorders GLP-1 drugs are also showing promise in the field of brain diseases. Recently, the mechanism of action of GLP-1 drugs that block neuroinflammatory responses by targeting microglia has been discovered, and active clinical studies are underway on their use in Parkinson's disease and Alzheimer's disease. There is already evidence that GLP-1s are associated with a reduced risk of developing dementia. A study of 88,381 patients with type 2 diabetes aged 65 years and older who were treated with liraglutide-based Victoza showed that those taking Victoza had a lower risk of dementia than those in the other treatment groups. Seulki Lee, CEO of D&D Pharmatech Among domestic companies, D&D Pharmatech is conducting clinical trials of GLP-1 agents in various areas, including Parkinson's disease and dementia. However, in 2020, D&D Pharmatech failed to demonstrate efficacy in a Phase II clinical trial involving 255 patients with Parkinson's disease. The primary endpoint, symptom improvement after a total of 36 weeks of treatment, did not show a statistically significant effect compared to placebo. A closer look revealed a significant difference between the NLY01 and placebo groups at 24 weeks post-dose. However, between weeks 24 and 36, the placebo group showed more improvement than NLY01. D&D Pharmatech is also conducting clinical studies on GLP-1 agents in MASH as well as neurodegenerative diseases. Recently, the company received the U.S. Food and Drug Administration's (FDA) IND approval to initiate a global Phase II trial for DD01, a new drug candidate for MASH. The Phase II trial will enroll 68 overweight and obese patients with MASH at 10 clinical sites in the U.S. D&D Pharmatech’s DD01 is a GLP-1-glucagon dual receptor agonist that has demonstrated weight loss and fatty liver reduction in preclinical studies. In a Phase I trial, DD01 reduced fatty liver by more than 50% at 4 weeks of treatment. Seulki Lee, CEO of D&D Pharmatech, said, "Although we did not meet the primary endpoint while developing a new drug for Parkinson's disease, we have seen potential. Our current focus is on metabolic diseases. We are looking forward to further clinical results based on the promise we have seen with GLP-1 agents in MASH."
- Company
- GC Biopharma ships first batch of ‘Alyglo’ to the U.S.
- by Son, Hyung-Min Jul 10, 2024 05:48am
- GC Biopharma announced on the 8th that it has completed the shipment of the first batch of its blood product ‘Alyglo’, which was approved by the U.S. Food and Drug Administration (FDA) late last year. The shipment will be delivered to specialized pharmacies through warehouses and distributors in the U.S. and is expected to be available for prescription from the middle of this month. Alyglo is a 10% liquid intravenous immunoglobulin treatment used to treat Primary Humoral Immunodeficiency. It received marketing authorization from the FDA in December last year, After approval, GC Biopharma has been carrying out activities to commercialize the drug around its U.S. subsidiary (GC Biopharma USA, Inc.), including signing PBM contracts formulary listings, as well as securing specialty pharmacies. On the 1st of this month, GC Biopharma signed a contract with a large Pharmacy Benefit Manager (PBM) in the U.S. to include Alyglo in its formulary and has also completed contracts with well-known specialty pharmacies and distributors that it targeted as core distribution channels. The company plans to actively secure additional vertically integrated channels, such as PBMs, specialty pharmacies, and distributors, to tap into the U.S. market. The U.S. immunoglobulin market is the world's largest at approximately KRW 16 trillion (USD 11.6 billion) and has been growing at a CAGR of 10.9% over the past 10 years (2013-2023). GC Biopharma’s strategy is to rapidly expand its market share by generating $50 million in sales this year and recording a growth rate of more than 50% every year thereafter.
- Company
- Tecentriq reapplies for reimb in early stage NSCLC
- by Eo, Yun-Ho Jul 10, 2024 05:48am
- The immuno-oncology drug Tecentriq is reattempting to expand insurance reimbursement to early-stage lung cancer. According to Dailypharm coverage, Roche Korea's PD-L1 inhibitor Tecentriq (atezolizumab) will be presented to the Health Insurance Review and Assessment Service's Cancer Disease Deliberation Committee today (July 10). The specific indication is “postoperative adjuvant therapy after resection and platinum-based chemotherapy for the treatment of patients with PD-L1 TC ≥50% Stage II-IIIA NSCLC. NSCLC is the leading type of lung cancer, accounting for about 85-90% of lung cancer deaths in Korea. Most patients are diagnosed at locally advanced or metastatic stages and about half of the NSCLC patients who undergo complete resection still experience relapse after surgery, which poses a significant burden on the patients’ part. Tecentriq was deliberated as an agenda by the CDDC in May last year but failed to receive reimbursement standards at the time. This time, however, the company is equipped with a new weapon. At the recent American Society of Clinical Oncology (ASCO) meeting, the company added data on Tecentriq’s overall survival (OS) improvement. The data was from the 5-year follow-up of Tecentriq’s Phase III trial, IMpower010. Results showed that in patients with stage II-IIIA NSCLC with PD-L1 expression greater than 50% following complete resection and platinum-based chemotherapy, the OS was 82.7% with Tecentriq adjuvant therapy, significantly higher than that found in the optimal supportive care arm (65.3%). It remains to be seen if Tecentriq can achieve a different outcome by the CDDC this time. Tecentriq is indicated in multiple types of lung cancer and is the first immuno-oncology drug to be approved as a first-line treatment for patients with extensive stage small cell lung cancer in combination with carboplatin and etoposide (chemotherapy). The company is also conducting multiple clinical studies to address unmet medical needs in advanced or metastatic NSCLC as a monotherapy or in combination with other targeted therapies, chemotherapies, and immuno-oncology drugs.
- Company
- Industry struggles to develop new atopic dermatitis drugs
- by Son, Hyung-Min Jul 09, 2024 05:51am
- The domestic pharmaceutical bio-industry is facing difficulties in developing new drugs for atopic dermatitis. Recently, Kangstem Biotech's stem cell therapy drug completed Phase III clinical trials, but the results fell short of expectations. JW Pharmaceutical's atopic dermatitis drug candidate failed to prove its efficacy in a Phase II clinical trial. Nevertheless, the companies’ challenge to develop atopic dermatitis drugs continues. Enzychem Lifesciences submitted an IND for Phase II trial earlier this month, and Shapreon is conducting Phase I/IIa clinical trials in Korea and the United States. SCM Life Science, Daewoong Pharmaceutical, and Novacell Technology are also developing new drugs with new mechanisms of action for atopic dermatitis. #iOn the 19th, according to industry sources, Kangstem Biotech announced on the 4th that its atopic dermatitis drug candidate ‘Furestem-AD inj' failed to meet the primary endpoint in Phase III clinical trials. This is the second time in 2019 that Kangstem Biotech has failed to achieve statistical significance with Furestem-AD. In October 2019, the company failed to achieve statistical significance in the Eczema Severity Index (EASI)-50 endpoint with Furestem-AD and discontinued trials. Furestem-AD is being developed for patients with moderate-to-severe atopic dermatitis eczema who have not responded to existing treatments, including topical and systemic steroids. The Phase III trial was conducted from September 2021 through August 2023 and enrolled a total of 315 adult patients with moderate-to-severe chronic atopic dermatitis who had not responded to existing treatments. The primary endpoint was the percentage of patients achieving Eczema Severity Index (EASI)-50, a measure of atopic severity, at 12 weeks. EASI-50 is defined as a 50% improvement in atopic dermatitis symptoms after treatment. Clinical results showed that 33.0% of patients achieved EASI-50 in the Furestem-AD treatment arm compared to 29.3% of patients in the placebo arm. The percentage of patients achieving EASI-50, which is a measure of atopic severity, was 58% at 1 year, 66% at 2 years, and 75% at 3 years with Furestem-AD. While Furestem-AD was associated with a higher rate of EASI-50 achievement than placebo, the difference was not statistically significant (P=0.6111). However, Kangstem Biotech plans to proceed with the license application due to the significant effect of Furestem-AD in long-term administration. Furestem-AD increased the EASI-50 achievement rate to 48.1% at week 16 and 58.1% at week 24. In terms of safety, Furestem-AD was associated with fewer adverse events than placebo after week 12. JW Pharmaceutical's innovative new drug candidate also failed to meet the primary endpoint. The Danish pharmaceutical company LEO Pharma returned the rights to JW1601, an atopic dermatitis drug, and terminated the technology transfer agreement with JW Pharmaceutical. JW1601 has a dual mechanism of action that selectively acts on the histamine H4 receptor to block the activity and migration of immune cells that cause atopic dermatitis and inhibit histamine signal transduction. Histamine is a neurotransmitter involved in allergic reactions. However, the global Phase II trial reportedly failed to meet its primary endpoint. As there are no atopic dermatitis drugs with this mechanism of action, it could have become a first-in-class drug if developed but did not reach commercialization. The company plans to review future development directions, including the possibility of securing new indications. Development of new atopic dermatitis drugs continue Despite the failure experienced by some companies in proving the efficacy of their drug candidates in clinical trials, the challenge of developing new drugs for atopic dermatitis continues in the domestic pharmaceutical and biotech industry, SCM-AGH, a stem cell therapy for atopic dermatitis being developed by SCM Life Science, also secured positive results in Phase II clinical trials. The company held an IR meeting last month to explain the detailed clinical results of SCM-AGH and future development plans. In a study comparing the efficacy and safety of SCM-AGH versus placebo, SCM-AGH met its primary endpoint. The drug also showed significant results in the secondary endpoint, ESAI-90 at 24 weeks. Also, no adverse events were reported among the 55 subjects who received SCM-AGH. The no adverse events were deemed positive as some inflammation treatments have been associated with safety concerns. SCM Life Science plans to conduct a Phase III clinical trial in Korea with Handok. On the 3rd, Enzychem Lifesciences filed an IND to initiate a Phase II clinical trial for atopic dermatitis to the Ministry of Food and Drug Safety. Based on its immunomodulatory function, EC-18’s mechanism quickly eliminates the hypersensitive immune response caused by allergens, which is the main cause of atopic dermatitis. The trial will evaluate the efficacy and safety of EC-18 in 120 patients with moderate or severe atopic dermatitis. Shapreon’s atopic dermatitis treatment, NuGel, is in clinical trials in the U.S. After receiving global Phase II IND approval for NuGel from the U.S. Food and Drug Administration (FDA) in September last year, Shapreon successfully enrolled its first patient in March of this year. NuGel works by activating GPCR19, which blocks the inflammasome pathway to inhibit cytokine secretion. In addition, Daewoong Pharmaceutical is conducting 2 clinical trials in the United States. DWP212525, which targets JAK3 and TEC family kinase (TFK), is in the preclinical trial, and DWP213388, a BTK-ITK inhibitor, is in its Phase I clinical trial. Novacell, an affiliate of DongKoo Bio&Pharma, is developing NCP112 for the treatment of mild-to-moderate atopic dermatitis targeting formyl peptide receptor 2 (FPR2), a G protein-coupled receptor (GPCR) involved in inflammation.
- Company
- Takeda seeks top-line growth to KRW 300 billion
- by Hwang, Byung-woo Jul 09, 2024 05:51am
- Takeda Pharmaceuticals Korea achieved a double win last year, seeing improvements in sales and operating income. In particular, thanks to the growth of its anti-cancer drug portfolio, the company's sales exceeded the KRW 250 billion mark for the first time in 3 years since 2020, increasing sales growth for the second consecutive year. Takeda Pharmaceuticals Korea logoIn the long run, the key question will be whether the growth of products such as Obizur, which will be listed for reimbursement this year, can offset the decline in sales of Dexilant and Pantoloc. Japanese pharmaceutical companies count the period from April 1 to March 31 as the first fiscal year, and the 16th fiscal year audited report, which includes last year's sales, records performance from April 1, 2023, to March 31, 2024. Sales rebound since 2021... second consecutive year of growth According to the audit reports disclosed in the Data Analysis, Retrieval and Transfer System (DART), Takeda Pharmaceuticals Korea’s (Takeda Korea) 16th fiscal period sales amounted to KRW 253.9 billion, surpassing the KRW 250 billion mark for the first time in 3 years. Takeda had posted sales of KRW 252.6 billion in 2020, and the sales fell to KRW 231.5 billion in 2021. It then rebounded to KRW 249.5 billion in 2022 and continued its upward trend. In line with the revenue growth, operating profit also rose, reaching KRW 9 billion. This is the highest in the last 5 years, with operating profit in the last 4 years being KRW 8.1 billion in 2020, KRW 8 billion in 2021, and KRW 7.5 billion in 2022. Net income also grew from KRW 6.8 billion to KRW 7.9 billion during the same period. This revenue growth indicates that Takeda Korea improved externally and internally last year. This was influenced by the company’s decrease in selling, general, and administrative expenses (SG&A). Commissions paid decreased from KRW 19.5 billion in the 15th period to KRW 5.1 billion in the 16th period, offsetting the increase in other SG&A expenses. In the footnote, it was indicated that the amount of commission paid to Baxalta GmbH of KRW 25.8 billion in the 15th period was eliminated in the 16th period with the company being merged and acquired by Takeda Pharmaceutical International AG. (from the left)Adcetris, ZejulaOncology products lead sales growth...sees sales growth in all major items Takeda Korea's sales recovery to KRW 250 billion was driven by growth in its oncology portfolio. Sales of Alunbrig and Adcetris grew more than 20% year-on-year, and Zejula grew 19%. According to the drug research institution IQVIA, Luprin recorded the highest sales of KRW 30.6 billion. After reaching KRW 32.2 billion in 2019, sales dropped to KRW 27.6 billion in 2022 but surpassed the KRW 30 billion mark again in 2023. In addition, Zejula, which is expanding its influence every year, had the second highest sales among anti-cancer drugs, with sales of KRW 22.6 billion in 2023, up from KRW 19 billion in 2022, and Alunbrig's sales increased from KRW 11 billion to 13.6 billion won during the same period. Sales of Adcetris showed the highest growth. Its sales increased by 29%, from KRW 7.6 billion in 2022 to KRW 9.8 billion in 2023. Taken together, the sales growth of major anticancer drugs last year was about KRW 13 billion. As a result, the company’s oncology portfolio will continue to drive sales growth this year. However, there is the possibility that Adcetris' sales growth may be limited hereon due to its lowered drug price cap on July 1. GI drug sales will inevitably decline...Obizur’s reimbursement is soon expected In the GI space, Mezavant's sales growth was notable. It crossed the KRW 10 billion mark for the first time with sales of KRW 10.1 billion in 2023, up 26% from the KRW 8 billion in 2022. In addition, sales of Kinteles grew 14% to KRW 16.5 billion in 2023 (from KRW 14.5 billion in 2022), and orphan drug Replagal grew 17% to KRW 16 billion (from KRW 13.6 billion in 2022). However, there were also some items that took a turn for the worse. Sales of Dexilant and Pantoloc decreased 8% (from KRW 16.7 billion to KRW 15.3 billion) and 7% (KRW 13.3 billion to KRW 12.4 billion), respectively, compared to 2022, and sales of Adynovate decreased by nearly 20%, from KRW 6.8 billion in 2022 to KRW 5.4 billion in 2023. Considering this, there are voices that it is necessary to prepare a portfolio to expand sales in the long term. The most anticipated item is the reimbursement of Obizur, a treatment for adults with acquired hemophilia A, which was reimbursed in February. The drug for the rare blood disorder has a limited patient population but is expected to contribute to sales with its differentiated sales.
- Company
- HCV testing added to the National Health Screening Program
- by Hwang, Byung-woo Jul 08, 2024 05:46am
- The government's decision to introduce the long-discussed hepatitis C antibody testing to the national screening program is expected to have a positive impact on its treatment market. According to industry sources, the decision is encouraging as it enables the first step of identifying hepatitis C patients. However, how to link diagnosis and treatment will be a challenge in the future, as the treatment rate has been dropping despite patient identification. Recently, the Ministry of Health and Welfare held the 2nd National Health Screening Committee Meeting in 2024 and confirmed the introduction of new hepatitis C testing and the expansion of osteoporosis screening subjects in Korea’s general health screening program. As a result of the decision, people aged 56 and older will be able to receive an antibody test for hepatitis C during national health screening from 2025. According to the Korean Association for the Study of the Liver (KASL), hepatitis C is responsible for about 10% to 15% of liver cancer cases in Korea. Between 54% and 86% of hepatitis C patients progress to chronic hepatitis, and 15% to 51% of which progress to cirrhosis. The risk of liver cancer in cirrhosis is 1-5% per year, with the risk increasing with age. However, hepatitis C can be cured by taking oral antiviral drugs for 8 to 16 weeks, so screening and treating infected people early is the best way to prevent the spread of the disease. This is why the pharmaceutical industry’s hopes are rising on how the introduction of hepatitis C testing will lead to increased patient identification and treatment. An industry official said, "There had been a large unmet need in hepatitis C, but due to a lack of policy and awareness, many patients were unaware of their infection and were diagnosed late, often after the disease had progressed to liver cancer. The industry is pleased that a meaningful policy has been established." Will sales of AbbVie and Gilead’s treatments also rebound? Currently, the leading hepatitis C treatments are AbbVie’s Mavyret (glecaprevir/pibrentasvir) and Gilead Sciences' Epclusa (sofosbuvir/velpatasvir) and Vosevi (velpatasvir/sofosbuvir/voxilaprevir). In terms of sales, Mavyret sold KRW 57.3 billion in 2019, KRW 46.9 billion in 2020, KRW 46.6 billion in 2021, KRW 39.3 billion in 2022, and KRW 24.3 billion in 2023, showing a yearly decline. Epclusa and Bosebi Vosevi to generate sales in earnest after being granted reimbursement in November 2022, generating KRW 16.1 billion and KRW 3.5 billion respectively last year. In general, Mavyret’s sales have declined with Epclusa and Bosebi taking its place. However, the overall market for hepatitis C drugs has been on a decline or drifting sideways. Since 2020, the combined sales of the 3 products have been in the low to mid KRW 40 billion range. This is not unrelated to the decline in the number of hepatitis C patients in Korea. According to statistics from the Korea Disease Control and Prevention Agency’s Infectious Disease Portal, the number of hepatitis C patients decreased from ▲11,849 in 2020 to ▲11,115 in 2021, ▲8,308 in 2022, and ▲7,225 in 2023 Considering this situation, the introduction of hepatitis C antibody testing in the national health screening program is expected to lead to an increase in the use of hepatitis C treatment as well. An official from the KASL said, "Our society has been pondering how to combat hepatitis C for a long time, and although it is late, we are glad that screening for hepatitis C became possible through the national health screening program. The increase in screening will help us identify and treat patients at an earlier stage." However, there is also a view that additional efforts would need to be made to actively treat patients even after the national screening. According to KASL’s Fact Sheet which was published in 2023, the cure rate for hepatitis C patients was only 58.1% in 2019. This means that 4 out of 10 people diagnosed with hepatitis C do not actually receive treatment. It's also unclear how well the national screening program will be able to identify patients, given that the target age for screening is 56. For this reason, the KDCA is working on a plan to subsidize the cost of confirmatory tests for those who test positive for hepatitis C antibodies in the national health screening program so that they can be confirmed faster. A doctor at A General Hospital said, "The problem in Korea has been the low screening rate itself. Various studies have been conducted on how to treat the confirmed patients afterward. I think the introduction of hepatitis C testing in the national health program will increase patient treatment as well as the use of treatment."
- Company
- New PNH drugs introduced into AstraZeneca’s reign
- by Son, Hyung-Min Jul 08, 2024 05:45am
- Competition is in full swing in the paroxysmal nocturnal hemoglobinuria (PNH) market, which is currently dominated by AstraZeneca's treatments such as Soliris and Ultomiris, with the introduction of new drugs that have new mechanisms of action soon to emerge in the market. Recently, Handok's Empaveli is nearing reimbursement in Korea, and Novartis has filed for domestic marketing authorization of its new drug Fabhalta. Amid the introduction of such new drugs, AstraZeneca is defending its market by receiving approval for Voydeya, which is used as an adjuvant therapy with Soliris and Ultomiris. According to industry sources on the 6th, Handok's Empaveli (pegcetacoplan) was approved adequate for reimbursement by the Health Insurance Review and Assessment Service's Drug Reimbursement Evaluation Committee. Empaveli passed the committee 2 months after being approved in May, ahead of drug pricing negotiations with the National Health Insurance Service. New PNH drug introduced by Handok in Korea Empaveli is a treatment for paroxysmal nocturnal hemoglobinuria (PNH) developed by the U.S. company Apellis. The drug’s license in countries other than the U.S. is owned by the Swedish Orphan Biovitrum (Sobi). Handok entered into a strategic partnership with Sobi in October last year to introduce Empaveli in Korea. PNH is a rare and life-threatening disease caused by the destruction of red blood cells in the blood vessels, leading to symptoms of bloody urine and complications such as acute renal failure. Empaveli is the first PNH treatment to target the C3 protein. By blocking C3 cleavage, the drug prevents intravascular and extravascular hemolysis. Empaveli’s efficacy was confirmed over Soliris (eculizumab) in the Phase III PEGASUS trial. In the study, the level of lactate dehydrogenase (LDH), a marker of intravascular hemolysis, remained below 1.5 times the upper limit of normal for 48 weeks in the Empaveli treatment arm. The percentage of patients who remained transfusion-free for 16 weeks was also higher in the Empaveli treatment arm (85%) compared with the Soliris arm (15%). The efficacy of Empaveli was also confirmed in the PRINCE trial in treatment-naïve PNH patients. After 26 weeks of follow-up, 85.7% of patients in the Empaveli arm showed stable hemoglobin levels, and LDH levels were well controlled to normal levels in the Empaveli arm. In addition to Empaveli, other new drugs from multinational pharmaceutical companies are also preparing to enter Korea. Novartis recently submitted a new drug application (NDA) for its PNH drug Fabhalta (iptacopan) in Korea. Novartis Fabhalta is a factor B inhibitor that acts proximally in the immune system's alternative complement pathway, providing comprehensive control of red blood cell destruction. The advantage of Fabhalta is its formulation. As an oral agent, Fabhalta offers dosing convenience compared to the existing intravenous formulations like Soliris and Ultomiris (ravulizumab). Fabhalta’s efficacy was confirmed in patients who failed complement C5 inhibitor treatment or had received no prior therapy. Study results showed that 82% of patients in the Fabhalta arm achieved a 2g/dL increase in hemoglobin at week 24 without red blood cell transfusions. In addition, Roche's C5 complement inhibitor Piasky (crovalimab) also showed clinical benefit. Piasky demonstrated non-inferiority to Soliris across multiple endpoints, including the rate of abrupt hemolysis and the rate of hemoglobin level stabilization. AZ Voydeya approved in Korea as add-on therapy to Soliris and Ultomiris The PNH market has been dominated by AstraZeneca, which owns new C5 complement inhibitor class drugs, including Soliris and Ultomiris. However, the introduction of other new drugs is expected to increase competition in the area. AstraZeneca will defend the market with its oral factor D inhibitor, Voydeya add-on therapy. Voydeya was approved in Korea on March 28 as an add-on therapy for PNH patients with symptoms or signs of extravascular hemolysis (EVH) who are using Soliris and Ultomiris. Complement C5 inhibitors are expected to complement the development of EVH and anemia experienced by some patients with PNH. Voydeya has received the Breakthrough Therapy designation in the U.S. and PRIority MEdicines (PRIME) status in Europe. Voydeya's domestic approval was based on the Phase III ALPHA trial. In the trial, Voydeya met its primary endpoint of change in hemoglobin level in patients on C5 complement inhibitors that experience EVH. Specifically, Voydeya demonstrated an improvement compared with placebo in the primary endpoint of change in hemoglobin concentration from baseline to week 12. Voydeya also achieved a 60% increase in hemoglobin of 2g/dL or greater at Week 12 without transfusion compared to 0% with placebo. In terms of safety, the most common treatment-emergent adverse events were headache and diarrhea.