- LOGIN
- MemberShip
- 2026-05-01 04:34:34
- Company
- Competitiveness of K-anticancer drugs reaffirmed
- by Son, Hyung Min Sep 08, 2025 06:17am
- Korean pharmaceutical companies gathered in one place to show off their anti-cancer pipelines, including both in-house developed and imported new drugs. At the Korean Society of Medical Oncology 2025 International Conference (KSMO 2025), which was held over three days from September 3 at Walkerhill Hotel in Seoul, major domestic pharmaceutical companies showcased their oncology pipelines, including both self-developed and in-licensed drugs. Companies such as Yuhan, Boryung, GC Cell, Jeil Pharmaceutical, and Celltrion highlighted their therapies, including Lazertinib, Zepzelca, Immuncell-LC, Lonsurf, and Vegzelma, covering a wide range of solid and hematologic cancers. Domestic companies focus on promoting self-developed drugs and biosimilars Yuhan spotlighted its non-small cell lung cancer (NSCLC) therapy, Lazertinib. Lazertinib was approved as Korea’s 31st novel homegrown drug approved in in January 2021. The technology of the drug, which was originally developed by Genosco (a subsidiary of Oscotec) was transferred to Yuhan in 2015. Yuhan later signed a USD 1.4 billion out-licensing deal with Janssen in November 2018. Janssen confirmed Lazertinib’s efficacy compared with Tagrisso monotherapy, the current standard of care. Through the MARIPOSA Phase III trial, In August 2023, the U.S. FDA approved Lazertinib as a combination therapy as a first-line treatment for EGFR-positive NSCLC. The combo regimen is expected to extend overall survival (OS) by more than a year versus existing therapies. With over half of the patient group still alive, the trial is expected to reveal further significant improvements as new data emerges. (from the left) GC Cell and Celltrion GC Cell promoted Immuncell-LC, an autologous cell therapy originally developed by InnoCell (the predecessor of GC Cell). The therapy uses mononuclear cells extracted from a cancer patient’s blood, cultured for over two weeks with anti-CD3 and IL-2 stimulation. The activated immune cells target and eliminate cancer cells in the body. Immuncell-LC functions by inducing activated T-lymphocytes and autologous cytokine-induced killer cells (CIK) to seek out and eliminate cancer cells within the body. In a nine-year extended follow-up study of a Phase III trial in hepatocellular carcinoma, Immuncell-LC showed recurrence-free survival (RFS) of 43.5 months, compared with 27.4 months for the control group, with cancer-specific survival (CSS) not yet reached in either group. Hanmi Pharmaceutical showcased Rolontis, a neutropenia drug that was approved in Korea in 2021 as the 33rd novel homegrown drug. Marketed as Rolvedon in the U.S., it generates over KRW 20 billion in quarterly revenue. Hanmi has been working to strengthen Rolontis’s competitiveness through a same-day administration trial. Unlike existing drugs such as Neulasta, which require administration 24 hours post-chemotherapy, Rolontis allows for same-day dosing, reducing hospital stays and improving convenience. Celltrion highlighted Vegzelma, a biosimilar to Roche’s Avastin (bevacizumab). Now launched across Korea, the U.S., and Europe, Vegzelma has become the top-selling bevacizumab biosimilar in Europe. (clockwise from the upper left) Jeil Pharmaceutica, Yuhan Corp, Boryung, and Hanmi PharmaceuticalIntense Competition in In-Licensed Oncology Drugs Boryung emphasized its portfolio of in-licensed drugs, including Gemzar and Alimta. Boryung acquired Korean rights to the cytotoxic anticancer drug for NSCLC and pancreatic cancer, Gemzar (originally by Eli Lilly), in October 2020, later adding the NSCLC drug Alimta’s rights in 2022 and transitioning from imports to in-house production. Boryung also pinned hopes on the SCLC drug Zepzelca (developed by Spanish company PharmaMar), for which it holds exclusive sales rights in Korea. A symposium on the Phase III IMforte study on its use in combination with the immunotherapy drug Tecentriq was also being held, focusing efforts on establishing Zepzelca as the standard of care. Zepzelca is a novel drug developed by the Spanish pharmaceutical company PharmaMar, and is already being marketed in North America by Jazz Pharmaceuticals. Boryung holds exlclusive right to its sales and distribution in Korea. Zepzelca inhibits DNA transcription in cancer cells and reduces tumor-associated macrophage (TAM) activity, thereby blocking cancer cell proliferation, immune evasion, and angiogenesis. Zepzelca is being tested in the IMforte Phase III trial as a first-line option for small-cell lung cancer in combination with the immunotherapy Tecentriq (atezolizumab). Early results show survival benefits compared with Tecentriq alone. Jeil Pharmaceutical presented Lonsurf, which the company licensed from Japan’s Taiho. Approved in the U.S. in 2015 and now in 75 countries, Lonsurf is indicated for metastatic colorectal cancer and, since 2021, for metastatic gastric cancer.
- Company
- Kerendia+Jardiance combo demonstrates early benefit
- by Hwang, byoung woo Sep 08, 2025 06:16am
- Kerendia (finerenone), a treatment for chronic kidney disease, has strengthened its clinical presence by demonstrating efficacy in combination with the SGLT-2 inhibitor Jardiance (empagliflozin). Pic of Kerendia Kerendia is the first non-steroidal mineralocorticoid receptor antagonist (MRA), with a novel mechanism of action that directly suppresses inflammation and fibrosis in the kidney. In June, results from the CONFIDENCE study drew attention by confirming the benefit of early combination therapy with SGLT-2 inhibitors. The trial included 818 patients with type 2 diabetes and CKD (eGFR 30–90 mL/min/1.73m², urine albumin-to-creatinine ratio [UACR] ≥100–
- Company
- Zynyz receives orphan drug designation in Korea
- by Eo, Yun-Ho Sep 05, 2025 06:20am
- The immuno-oncology drug Zynyz that Handok has decided to market in Korea has been designated as an orphan drug in Korea. The Ministry of Food and Drug Safety (MFDS) recently announced the designation through a public notice. Specifically, the drug is indicated for ▲Merkel cell carcinoma and ▲ squamous cell carcinoma of the anal canal (SCAC). Among these, the Merkel cell carcinoma indication had already been granted orphan drug status earlier in June. Zynyz (retifanlimab), which was developed by the US company Incyte, was also designated in July as a subject for the “Global Innovative Products on Fast Track (GIFT)” program. Since receiving approval from the U.S. FDA in May, the PD-1 inhibitor Zynyz has been gaining attention as the first first-line treatment for adult patients with unresectable locally recurrent or metastatic anal cancer, in combination with platinum-based chemotherapy (carboplatin, paclitaxel). The efficacy of Zynyz in anal cancer was demonstrated in the Phase III POD1UM-303 study. This study evaluated 308 patients with unresectable locally recurrent or metastatic squamous cell carcinoma of the anal canal (SCAC) by comparing outcomes between the Zynyz + carboplatin + paclitaxel combination arm and the standard-of-care arm. Results showed that the median progression-free survival (PFS) was 9.3 months in the Zynyz arm, which is a 37% reduction in the risk of disease progression or death compared with the 7.4 months in the standard-of-care arm. In the interim analysis, the median overall survival (OS) was 29.2 months in the Zynyz arm and 23 months in the standard-of-care arm. The objective response rate (ORR) for the Zynyz arm was 56%, with 22% achieving complete response and 33% partial response—higher than the 44% observed in the standard-of-care arm. The median duration of response (DoR) was 14 months in the Zynyz arm, compared with 7.2 months in the standard-of-care arm. SCAC accounts for about 85% of all anal cancers and is classified as a rare cancer. The majority of cases are caused by human papillomavirus (HPV) infection, and HIV-positive individuals face a 25–35 times higher risk. Early symptoms often resemble hemorrhoids, leading to delayed diagnosis, with many patients visiting hospitals at advanced stages.
- Company
- MenQuadfi’s indication includes infants as young as 6 weeks
- by Hwang, byoung woo Sep 05, 2025 06:19am
- On the 4th, Sanofi Kore announced that its fully liquid quadrivalent meningococcal vaccine, MenQuadfi, received expanded indication approval from the Ministry of Food and Drug Safety (MFDS) on August 26. The new approval allows MenQuadfi’s vaccination for infants from 6 weeks to under 2 years of age. With the indication extension, MenQuadfi has become the first quadrivalent meningococcal conjugate vaccine in Korea that can be administered to infants as young as 6 weeks. This approval allows a broader age range to have preventive options against invasive meningococcal disease. MenQuadfi contains 10μg of antigen each for the four meningococcal serogroups (A, C, W, and Y) and is formulated as a fully liquid formulation that requires no reconstitution or mixing, enhancing ease of use. It utilizes tetanus toxoid (TT) as a protein carrier to induce a strong T cell–mediated immune response. Following the indication extension, MenQuadfi now offers flexible vaccination schedules to those 6 weeks to 55 years of age. Infants aged 6 weeks to under 6 months may receive a 4-dose series, with the initial three doses given at minimum 8-week intervals, and a fourth booster at least 6 months after the third dose, administered at ≥12 months of age. For infants aged 6 to less than 24 months with no prior meningococcal vaccination, a 2-dose schedule is recommended, with a minimum 3-month interval between doses and the second dose administered after 12 months of age. For individuals aged 2 to 55 years, a single dose is sufficient for protection. MenQuadfi’s indication extension was based on results from the MET42 and MET61 studies. The Phase III MET42 trial enrolled about 2,627 infants and toddlers aged 2 to 18 months to evaluate the immunogenicity and safety of MenQuadfi versus an existing quadrivalent meningococcal vaccine. After just three doses starting at 2 months of age, MenQuadfi demonstrated strong immune responses across all 4 meningococcal serogroups: A (64.4%), C (96.4%), W (92.8%), and Y (88.7%). These results were higher compared to the comparator vaccine (A: 50.6%, C: 82.8%, W: 85.6%, Y: 81.8%). The MET61 study assessed infants aged 6 to 23 months with a 2-dose schedule, comparing the immunogenicity and safety of MenQuadfi against control vaccines, including Sanofi’s own Menactra. The results confirmed that MenQuadfi induced non-inferior immune responses across all serogroups (A, C, W, Y) compared with control vaccines, with a safety profile comparable with existing vaccines. Hee-kyung Park, Director of Sanofi’s Vaccines Business Unit, said, “With the indication extension of MenQuadfi in Korea, we are pleased to be able to protect infants as early as 6 weeks of age from invasive meningococcal disease. Sanofi will continue to work closely with health authorities to strengthen preventive strategies and secure protection against meningococcal infections across various age groups.”
- Company
- HK Inno.N and Pfizer partner to distribute COVID-19 vaccine
- by Lee, Seok-Jun Sep 05, 2025 06:18am
- HK inno.N (CEO Dal-Won Kwak) announced on the 4th that it has signed a co-promotion agreement with Pfizer Korea (CEO Dong-Wook Oh) for the 2025–2026 season distribution of the new COVID-19 variant vaccine, Comirnaty LP.8.1 Prefilled Syringe (SARS-CoV-2 mRNA Vaccine), as part of the National Immunization Program (NIP). Last year, HK inno.N was responsible for the private distribution of Comirnaty JN1 Injection (single-dose, bretobameran, SARS-CoV-2 mRNA vaccine). More recently, the company has also been in charge of distribution for the government-funded NIP program, which targets high-risk groups such as seniors aged 65 and older and immunocompromised individuals, a market valued at approximately KRW 200 billion. Through this co-promotion agreement, HK inno.N and Pfizer Korea have expanded their partnership to include promotional activities for those included in the National Immunization Program. Comirnaty® LP.8.1 Prefilled Syringe received approval from the MFDS on January 29 for the prevention of COVID-19 caused by SARS-CoV-2 in individuals aged 12 years and older. The vaccine is administered as a single 0.3 mL intramuscular injection, regardless of previous COVID-19 vaccination history. For individuals who have received prior COVID-19 vaccination, the new shot should be given at least three months after the last dose. HK inno.N CEO Dal-Won Kwak said, “Based on our sales and distribution capabilities, we will contribute to the stable supply of Comirnaty® LP.8.1 Prefilled Syringe and to the improvement of public health in Korea.
- Company
- Fruzaqla may be prescribed at general hospitals in KOR
- by Eo, Yun-Ho Sep 04, 2025 06:12am
- ] The new colon cancer drug Fruzaqla is may now be prescribed at general hospitals in Korea. According to industry sources, Fruzaqla (fruquintinib), a colorectal cancer therapy from Takeda Korea that selectively inhibits vascular endothelial growth factor receptors (VEGFR)-1, 2, and 3, has passed the drug committees (DC) of 41 major medical institutions nationwide, including Samsung Medical Center, Seoul National University Hospital, and Severance Hospital. Prior to its approval in Korea this June, Fruzaqla had been designated as an orphan drug in February and as a “Global Innovative products on Fast Track” (GIFT) in November of last year. Specifically, Fruzaqla is indicated for the treatment of adult patients with metastatic colorectal cancer (mCRC) who have previously received fluoropyrimidine-, oxaliplatin-, and irinotecan-based chemotherapy; an anti-VEGF therapy; an anti-EGFR therapy (for RAS wild-type); and trifluridine/tipiracil and/or regorafenib, but have progressed on or are intolerant to these therapies. Fruzaqla demonstrated efficacy in the Phase III FRESCO-2 trial. Results showed that the median overall survival (mOS) for the Fruzaqla arm was 7.4 months compared with 4.8 months in the placebo arm, reducing the risk of death by 34%. In addition, the median progression-free survival (mPFS) was 3.7 months (95% CI: 3.5–3.8) with Fruzaqla, more than doubling the placebo group’s 1.8 months, and reducing the risk of disease progression or death by 68%. Moreover, Fruzaqla is an oral therapy that can be taken once daily without complicated dietary restrictions, the convenience of which is expected to improve not only treatment outcomes but also patients’ quality of life. Professor Sang-Cheul Oh, Department of Oncology, Korea University Guro Hospital (Chair of the Colorectal Cancer Subcommittee, Korean Cancer Study Group) said, “Fruzaqla’s mechanism of action that selectively inhibits VEGFR-1, 2, and 3 provides strong efficacy with lower toxicity, making it a very meaningful option for later-line patients in the 4th line or beyond who have already received long-term treatment.”
- Company
- Novo Nordisk, Kakao Health sign digital healthcare MOU
- by Cha, Jihyun Sep 04, 2025 06:09am
- Kakao Healthcare (CEO Hee Hwang) announced on the 3rd that it has signed a memorandum of understanding with the multinational pharmaceutical company, Novo Nordisk Korea (General Manager Kasper Roseeuw Poulsen) to build a digital healthcare ecosystem for obesity and diabetes patients. The signing ceremony, held on September 2 at Novo Nordisk Korea’s headquarters in Songpa-gu, Seoul, was attended by Kakao Healthcare CEO Hee Hwang and Novo Nordisk Korea CEO Kasper Rosseeu Poulsen. At the event, the two companies agreed to collaborate by utilizing various digital technologies to improve the treatment journey of obesity and diabetes patients, ultimately creating a sustainable treatment environment that can deliver better outcomes. This is the second collaboration between the two companies, following their first partnership in 2023. Under the agreement, they linked Kakao Healthcare’s AI-based health management app ‘Pasta’ with Novo Nordisk’s insulin FlexTouch pen and its smart cap ‘Mallya,’ providing a medication management solution for diabetes patients. The new partnership is significant in that it expands the scope of collaboration from diabetes to obesity, creating a comprehensive alliance. The companies plan to address the unmet medical needs of Korea’s rapidly growing obesity and diabetes populations by offering patient-centered digital solutions. Specifically, in the field of obesity, the partnership will focus on ▲developing a personalized digital support program, ▲providing solutions to improve treatment effectiveness and quality of life. and In particular, the partnership will integrate Novo Nordisk’s patient support program Novo Fit Care, which is offered to patients prescribed its obesity treatments, into the Pasta app. Through this, patients will be able to manage weight and other long-term health aspects in a comprehensive way. In the field of diabetes, the companies will jointly develop digital solutions aimed at improving disease awareness. Kasper Roseeuw Poulsen, General Manager of Novo Nordisk Korea, said, "Obesity and diabetes are chronic conditions which, if left untreated, can lead to serious comorbidities and impose a tremendous burden on individuals and society. With Novo Nordisk’s more than 100 years of dedication to diabetes and obesity, and Kakao Healthcare’s leadership in Korea’s digital healthcare sector, the partnership will further accelerate integrated innovation for patient support in Korea." Hee Hwang, CEO of Kakao Healthcare, said, "Kakao Healthcare has leveraged AI, big data, and other technologies to bring positive changes to patients’ health journeys. By combining this experience and technology with Novo Nordisk, a global leader in obesity and diabetes care, we aim to play a major role in expanding patient-centered digital healthcare on a global scale."
- Company
- Moderna’s latest variant-targeted COVID-19 vaccine approved
- by Whang, byung-woo Sep 03, 2025 06:08am
- Moderna Korea announced on the 1st that its LP.8.1 variant-targeted COVID-19 vaccine, ‘Spikevax LP Inj’, has been approved by the Ministry of Food and Drug Safety (MFDS). Spikevax LP has been confirmed to induce broad cross-immune responses against currently circulating variants, including the LP.8.1 strain, and it is authorized for use in adolescents aged 12 and above as well as adults. Moderna plans to supply the newly approved vaccine in time for the government’s 2025–2026 seasonal immunization program, which begins in October. The company noted that its COVID-19 vaccines have demonstrated strong immune effect and safety in large-scale Phase III clinical trials and extensive real-world evidence (RWE). A key feature is that elderly individuals aged 65 and above showed immune responses comparable to those in younger adults. Also, regardless of which vaccine type had been administered previously, the Moderna vaccine showed high immunogenicity when used as a subsequent dose. A domestic study conducted by the Korea Disease Control and Prevention Agency (KDCA) also confirmed that Moderna’s vaccine recorded the lowest breakthrough infection rate among the vaccines used in the early stages of the pandemic. According to the National Immunization Program guidelines, Spikevax LP will be provided free of charge to high-risk groups, including seniors aged 65 and over and residents of long-term care facilities. The Korean Society of Infectious Diseases has stressed that immunity acquired through infection or vaccination wanes over time, and with the emergence of new variants, periodic updated COVID-19 vaccination for high-risk groups is recommended. The LP.8.1 series vaccines supplied this season have already been recommended for use by the World Health Organization (WHO), the European Medicines Agency (EMA), and the U.S. Food and Drug Administration (FDA). Based on recommendations from its Vaccination Expert Committee, the KDCA decided to adopt the LP.8.1 vaccine, which demonstrated stronger neutralizing antibody responses compared to last season’s JN.1-based vaccines. Moderna is the only company manufacturing mRNA COVID-19 vaccines in Korea, through its partnership with Samsung Biologics, and continues to ensure a stable vaccine supply via ongoing cooperation with Boryung Biopharma. Sang Pyo Kim, General Manager of Moderna Korea, said, “COVID-19 remains a threat to high-risk groups, with hospitalizations on the rise for 7 consecutive weeks. Moderna is committed to delivering updated COVID-19 vaccines that address the latest variants in a timely manner, ensuring that people can be vaccinated safely.”
- Company
- 'Prevenar 20' now available at general hospitals
- by Eo, Yun-Ho Sep 02, 2025 06:11am
- Product photo of Prevenar 20 PFS The pneumococcal conjugate vaccine 'Prevenar 20,' which will soon be included in the National Immunization Program (NIP), is becoming available for prescription at general hospitals. According to industry sources, Pfizer Korea's Prevenar 20 has passed the drug committees (DC) of tertiary general hospitals, including Samsung Medical Center, Seoul National University Hospital, Asan Medical Center in Seoul, and Sinchon Severance Hospital, and medical institutes, including Kangnam Sacred Heart Hospital, Kyung Hee University Hospital at Gangdong, Pusan National University Hospital, Seoul National University Bundang Hospital, Ajou University Hospital, Pusan National University Yangsan Hospital, and Chungnam National University Hospital. Several of these hospitals, including Seoul National University Hospital, have approved codes for adult patients only. However, as Prevenar 20 will be included in the NIP effective October, more hospitals are expected to have pediatric vaccines. Prevenar 20 is a pneumococcal conjugate vaccine that was approved by the Ministry of Food and Drug Safety (MFDS) on October 31, 2024. Compared to the 13-valent vaccine, Prevenar 20 has added seven additional pneumococcal serotypes. Among domestically approved pneumococcal conjugate vaccines, it contains the most serotypes. In addition to existing serotype components in the 13-valent vaccine, Prevenar 20 contains seven additional serotypes (serotypes 1, 3, 4, 5, 6A, 6B, 7F, 8, 9V, 10A, 11A, 12F, 14, 15B, 18C, 19A, 19F, 22F, 23F, 33F). It can be used to prevent invasive diseases and pneumonia in all ages, including infants aged six weeks and above. Pneumococcus is a major bacterial pathogen that causes various diseases in infants and young children, including otitis media, pneumonia, and meningitis. Vaccination is crucial, especially as it can cause life-threatening invasive pneumococcal disease (IPD) in immunocompromised children. Despite South Korea's NIP supporting the 23-valent pneumococcal polysaccharide vaccine (PPSV23) for adults aged 65 and older, this age group has the highest incidence of invasive pneumococcal disease (IPD). From September 2014 to mid-November 2023, a total of 3,734 cases of IPD were reported. The incidence rate for adults aged 65 and over was 32.1 cases per 100,000 people, accounting for 54.8% of all cases. This rate is the highest among all age groups under 65 years old. The inclusion of Prevenar 20 in the NIP was decided after a comprehensive review by the Korea Expert Committee on Immunization Practices (KECIP) on the vaccine's safety, immunogenicity, and cost-effectiveness. Meanwhile, Korea Vaccine is responsible for the domestic promotion of pediatric Prevenar 20, while Chong Kun Dang handles the promotion of the adult vaccine.
- Company
- Patent suits active to release Rinvoq generics
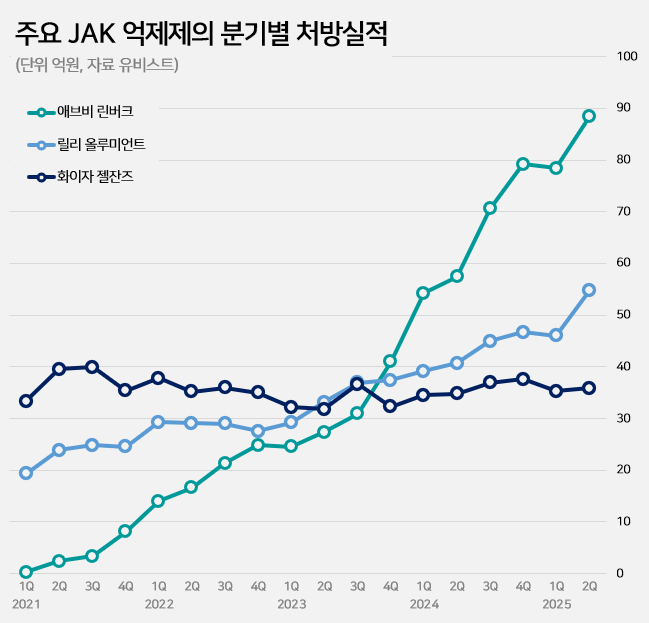
- by Kim, Jin-Gu Sep 02, 2025 06:10am
- Pic of Rinvoq The race to launch generics of AbbVie’s Rinvoq (upadacitinib), an oral treatment for autoimmune diseases, is showing signs of intensifying patent disputes. With the JAK inhibitor market expanding rapidly and Rinvoq solidifying its dominance, multiple companies are expected to mount patent challenges. According to industry sources on the 1st, Daewoong Pharmaceutical recently filed a passive scope confirmation trial against AbbVie, challenging the crystalline form patent of Rinvoq. Currently, two patents related to Rinvoq are registered: a substance patent expiring in May 2032 and a crystalline form patent expiring in October 2036. Daewoong's strategy is to first circumvent the crystalline form patent and then launch a generic version early, timed for the expiration of the substance patent. Chong Kun Dang opened the door to this dispute by challenging Rinvoq’s patents before Daewoong Pharmaceutical. Chong Kun Dang filed a related trial request on the 19th of last month. The pharmaceutical industry expects more companies to challenge Rinvoq’s patents from here on. One of the key requirements for obtaining first generic exclusivity is filing the initial invalidation trial. However, if another company files the same trial within 14 business days of the first filing date, it is also deemed to meet this requirement. This is why additional trial filings are expected in the coming weeks as companies move to secure first generic exclusivity for Rinvoq. According to UBIST, a pharmaceutical market research institution, outpatient prescriptions of JAK inhibitors in the first half of this year reached KRW 38.6 billion, up 40% from the KRW 27.5 billion in the same period last year. JAK inhibitors are experiencing rapid growth in the autoimmune disease treatment market, driven by the convenience of being oral formulations. The JAK inhibitor market, which was KRW 18.7 billion in 2020, grew by 36% to KRW 25.5 billion the following year. It continued to grow significantly each year: KRW 35.5 billion in 2022, KRW 40 billion in 2023, and KRW 62.2 billion in 2024. The expanded reimbursement granted for key products drove this market growth. Growth accelerated further last October when switching between JAK inhibitors for rheumatoid arthritis was allowed reimbursement. Major JAKi Prescriptions (AbbVie During this market expansion, Rinvoq has tightened its grip as the dominant player. In the first half of this year, Rinvoq recorded KRW 16.7 billion in prescriptions, a 49% increase from KRW 11.2 billion in the same period last year. Its growth has outpaced competitors such as Xeljanz (tofacitinib) and Olumiant (baricitinib). Having claimed the number one position in Q4 2023, Rinvoq has since expanded its market share, which now stands at 43% in the first half of this year. Rinvoq is a JAK inhibitor used for autoimmune diseases such as rheumatoid arthritis and atopic dermatitis. Its mechanism involves inhibiting inflammatory cytokines, thereby suppressing inflammation, pain, and cell activation.