- LOGIN
- MemberShip
- 2026-03-16 20:51:35
- Evolving HIV treatment strategies… new treatment options emerge
- by Son, Hyung Min | translator Alice Kang | 2026-03-16 09:25:24
The paradigm of HIV treatment is expanding beyond simple viral suppression toward a “lifecycle management” strategy that considers the entire lifespan of people living with HIV.
Within the current HIV treatment landscape, a variety of options have emerged, ranging from oral dual-drug regimens that reduce the medication burden during the initial treatment phase to long-acting injectables that prioritize quality of life during long-term treatment. Consequently, treatment strategies are becoming increasingly multifaceted.
HIV had long been perceived as a fatal infectious disease characterized by rapid disease progression and high mortality. However, with advances in antiretroviral therapy (ART), the life expectancy of people living with HIV has increased to approximately 78 years, reaching a level not significantly different from that of the general population.
At the same time, the U=U (Undetectable = Untransmittable) concept—which states that the virus cannot be transmitted to others once the viral load in the blood becomes undetectable— has positioned HIV treatment as a core pillar of public health strategy, moving beyond individual care.
Accordingly, HIV treatment is shifting from a question of survival to ‘how to manage the disease for a long time and in good health.’ In actual clinical practice, the number of patients newly diagnosed in their 20s and 30s is increasing, alongside those in their 60s and 70s who have been on treatment for decades, underscoring the growing importance of long-term treatment management strategies.
Amid these changes, there is an emphasis on an approach that considers the entire treatment journey, from the initial treatment phase through long-term management. Among the leading treatment options are GSK’s oral two-drug regimen ‘Dovato (dolutegravir and lamivudine)’ and the combination therapy of the long-acting injectable combination therapy ‘Vocabria (cabotegravir) + Rekambys (rilpivirine).’
Two-drug regimen ‘Dovato’ emerges as an initial treatment strategy…effective viral suppression with minimal drug burden
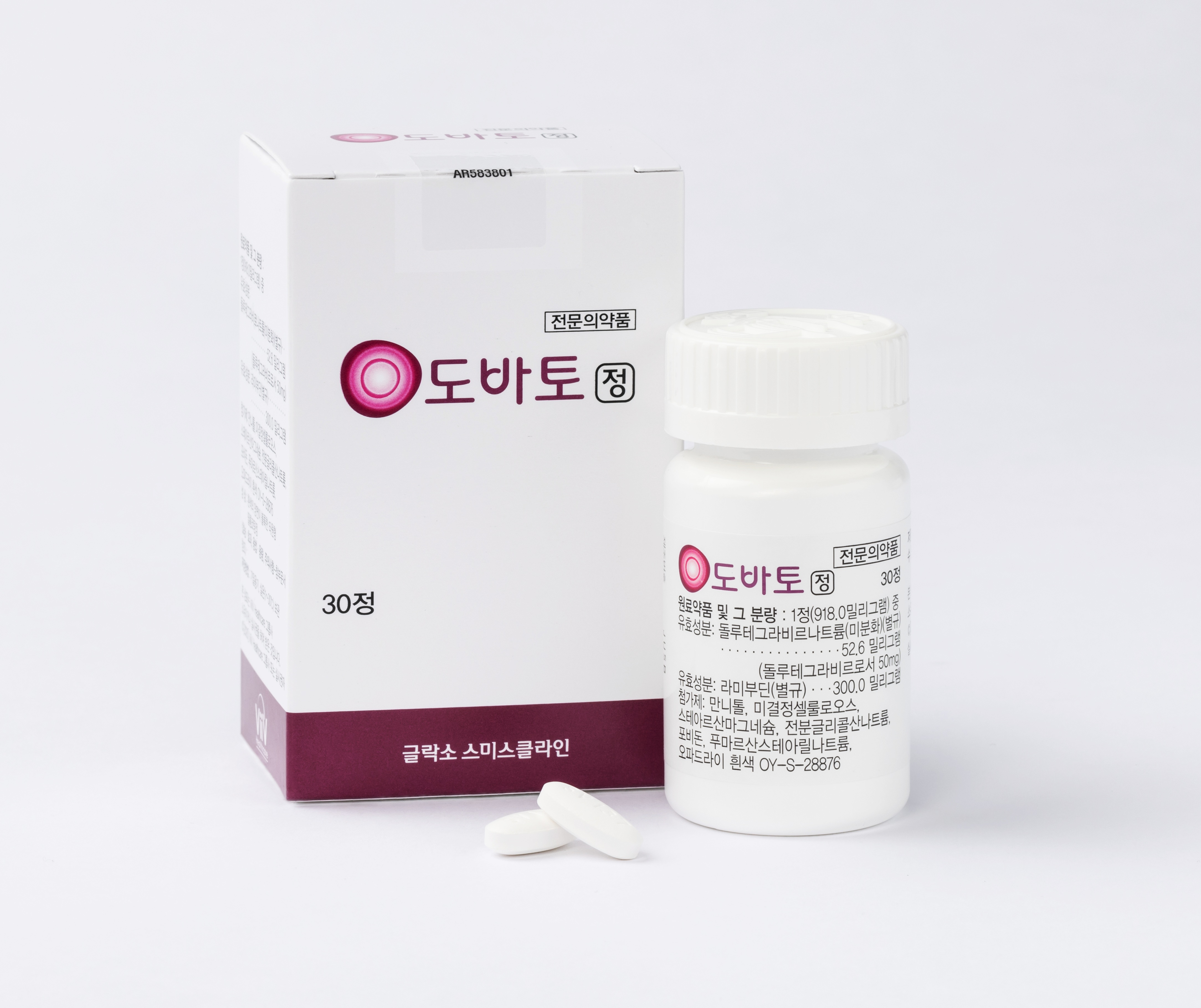
More than half (approximately 66.7%) of newly diagnosed HIV infections in Korea are diagnosed in people in their 20s and 30s. Since lifelong treatment is required from the time of diagnosis, the importance of strategies to minimize the medication burden during the initial treatment stage is increasing.
Dovato, a two-drug oral regimen, is characterized by its ability to reduce drug exposure while demonstrating viral suppression efficacy non-inferior to existing three-drug regimens.
In the GEMINI I and II studies, Dovato demonstrated non-inferiority in viral suppression compared with the dolutegravir (DTG) + tenofovir disoproxil fumarate/emtricitabine (TDF/FTC)-based three-drug regimen, and the TANGO study reported positive results regarding bone and renal biomarkers and lipid changes compared to TAF-based therapy. The risk of weight gain was also found to be relatively lower.
Thermore, the PASO-DOBLE study, which directly compared the regimen to the existing three-drug regimen of bictegravir (BIC), FTC, and TAF, confirmed non-inferiority in viral suppression rates at week 48, and reported relatively lower risks of weight gain and metabolic side effects.
The DOLCE study, conducted in a high-risk group of treatment-naïve HIV-infected individuals with low CD4 counts and high viral loads, and the ATTEND study, which focused on late-diagnosed individuals, also confirmed similar viral suppression effects compared to triple therapy.
Based on these study results, guidelines from the Spanish AIDS Research Group GeSIDA (Grupo de Estudio de SIDA), as well as those in Norway, Sweden, and other countries, recommend Dovato as an initial treatment option regardless of baseline viral load.
Every 2 month injectable better utilized for HIV in the long term…offers greater utilization in long-term treatment stages
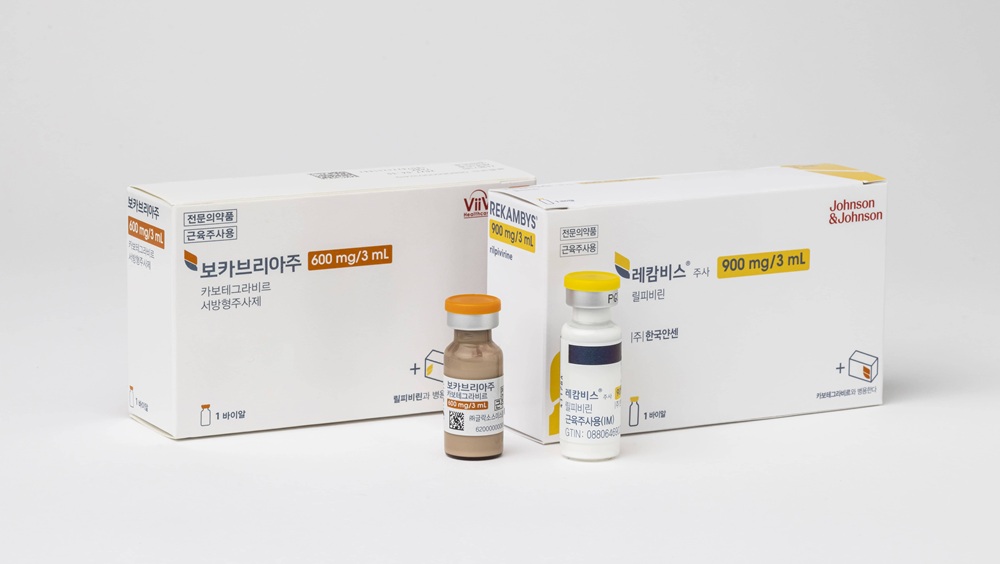
As HIV treatment enters the long-term management phase, the focus of treatment has naturally shifted toward quality-of-life management.
Amid this trend, the long-acting injectable combination therapy ‘Vocabria + Rekambys,’ administered once every two months, is also establishing itself as a major treatment option.
In the Phase III SOLAR study, the therapy demonstrated non-inferiority in both virologic failure rates and viral suppression rates compared with the standard three-drug oral regimen (BIC/FTC/TAF).
Treatment satisfaction was also high. The study found that approximately 90% of patients who switched to the long-acting injectable regimen preferred it over the existing oral regimen, citing the following main reasons: ▲ the absence of the need to take medication daily, ▲ treatment convenience, and ▲ reduced psychological burden from being repeatedly reminded of their HIV status through daily medication.
The U.S. Department of Health and Human Services (DHHS) guidelines also state that switching to long-acting injectable therapies can improve treatment convenience and reduce medication-related fatigue and the burden of social stigma.
In the CARES study, which reflects real-world clinical practice, the virologic success rate of the long-acting injectable therapy reached 96.9%, showing similar efficacy to the oral medication group. Treatment adherence was also found to be high, with 96% of scheduled doses administered within ±7 days.
Based on these study results, the 2025 European AIDS Clinical Society (EACS) removed HIV Subtype A1, which had previously been identified as a risk factor for virologic failure and resistance in certain ethnic groups when switching to the Vocabria + Rekambys combination therapy.
Currently, the Vocabria + Rekambys combination therapy is the only long-acting HIV injectable treatment available in Korea. It is being used as a switch therapy option to improve treatment convenience and quality of life in adult patients whose viral load has been stably suppressed through existing treatments.
This treatment has reportedly accumulated more than 1,000 treatment cases within about one year since its launch in Korea last April.
-

- 0
댓글 운영방식은
댓글은 실명게재와 익명게재 방식이 있으며, 실명은 이름과 아이디가 노출됩니다. 익명은 필명으로 등록 가능하며, 대댓글은 익명으로 등록 가능합니다.
댓글 노출방식은
댓글 명예자문위원(팜-코니언-필기모양 아이콘)으로 위촉된 데일리팜 회원의 댓글은 ‘게시판형 보기’와 ’펼쳐보기형’ 리스트에서 항상 최상단에 노출됩니다. 새로운 댓글을 올리는 일반회원은 ‘게시판형’과 ‘펼쳐보기형’ 모두 팜코니언 회원이 쓴 댓글의 하단에 실시간 노출됩니다.
댓글의 삭제 기준은
다음의 경우 사전 통보없이 삭제하고 아이디 이용정지 또는 영구 가입제한이 될 수도 있습니다.
-
저작권·인격권 등 타인의 권리를 침해하는 경우
상용 프로그램의 등록과 게재, 배포를 안내하는 게시물
타인 또는 제3자의 저작권 및 기타 권리를 침해한 내용을 담은 게시물
-
근거 없는 비방·명예를 훼손하는 게시물
특정 이용자 및 개인에 대한 인신 공격적인 내용의 글 및 직접적인 욕설이 사용된 경우
특정 지역 및 종교간의 감정대립을 조장하는 내용
사실 확인이 안된 소문을 유포 시키는 경우
욕설과 비어, 속어를 담은 내용
정당법 및 공직선거법, 관계 법령에 저촉되는 경우(선관위 요청 시 즉시 삭제)
특정 지역이나 단체를 비하하는 경우
특정인의 명예를 훼손하여 해당인이 삭제를 요청하는 경우
특정인의 개인정보(주민등록번호, 전화, 상세주소 등)를 무단으로 게시하는 경우
타인의 ID 혹은 닉네임을 도용하는 경우
-
게시판 특성상 제한되는 내용
서비스 주제와 맞지 않는 내용의 글을 게재한 경우
동일 내용의 연속 게재 및 여러 기사에 중복 게재한 경우
부분적으로 변경하여 반복 게재하는 경우도 포함
제목과 관련 없는 내용의 게시물, 제목과 본문이 무관한 경우
돈벌기 및 직·간접 상업적 목적의 내용이 포함된 게시물
게시물 읽기 유도 등을 위해 내용과 무관한 제목을 사용한 경우
-
수사기관 등의 공식적인 요청이 있는 경우
-
기타사항
각 서비스의 필요성에 따라 미리 공지한 경우
기타 법률에 저촉되는 정보 게재를 목적으로 할 경우
기타 원만한 운영을 위해 운영자가 필요하다고 판단되는 내용
-
사실 관계 확인 후 삭제
저작권자로부터 허락받지 않은 내용을 무단 게재, 복제, 배포하는 경우
타인의 초상권을 침해하거나 개인정보를 유출하는 경우
당사에 제공한 이용자의 정보가 허위인 경우 (타인의 ID, 비밀번호 도용 등)
※이상의 내용중 일부 사항에 적용될 경우 이용약관 및 관련 법률에 의해 제재를 받으실 수도 있으며, 민·형사상 처벌을 받을 수도 있습니다.
※위에 명시되지 않은 내용이더라도 불법적인 내용으로 판단되거나 데일리팜 서비스에 바람직하지 않다고 판단되는 경우는 선 조치 이후 본 관리 기준을 수정 공시하겠습니다.
※기타 문의 사항은 데일리팜 운영자에게 연락주십시오. 메일 주소는 dailypharm@dailypharm.com입니다.







