- LOGIN
- MemberShip
- 2026-03-17 11:28:03
- The key elements of government’s drug pricing reform plan
- by Jung, Heung-Jun | translator Alice Kang | 2026-03-17 09:22:15
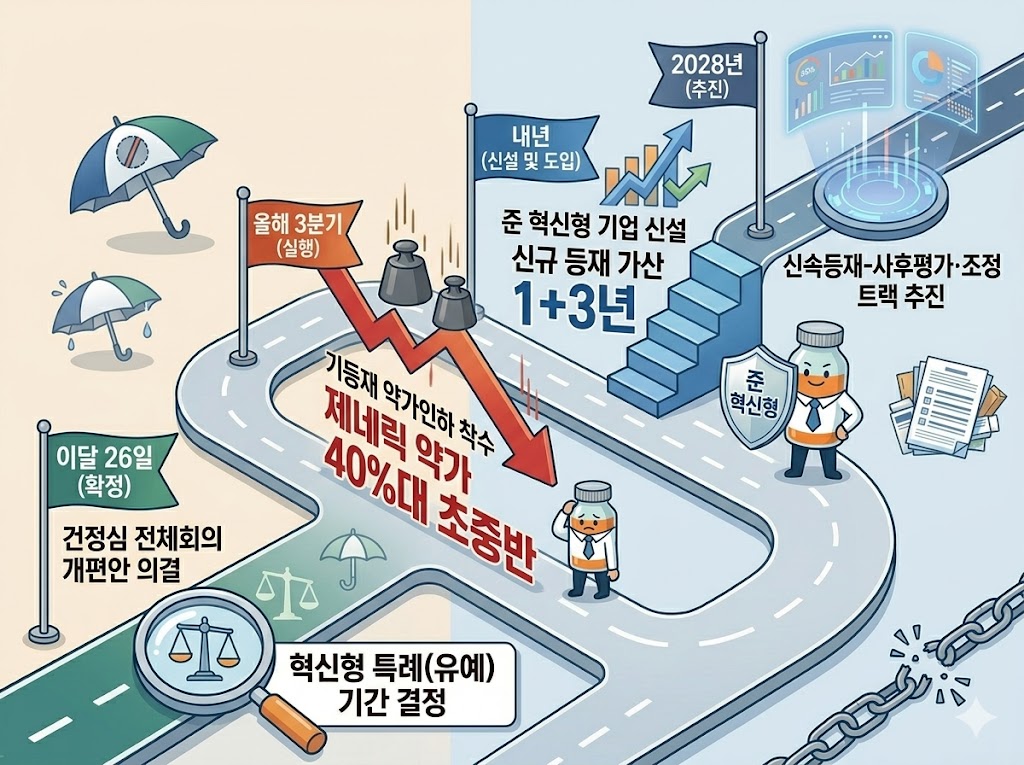
Under the government’s new drug pricing reform, price cuts for already listed drugs are expected to begin in the third quarter, with preferential treatment for innovative companies’ new listings expected to begin next year.
Innovative companies are set to receive a temporary exception from price cuts for existing drugs. However, the duration of these exceptions is scheduled to be finalized at the Health Insurance Policy Deliberation Committee meeting on the 26th.
In addition, the fast-track listing–post-evaluation and adjustment track is planned for 2028, and its criteria will be established to select which products are eligible.
According to industry sources on the 16th, the Ministry of Health and Welfare plans to finalize the drug pricing reform at the HIPDC plenary meeting on the 26th. Based on the discussions at the subcommittee, the direction of the reform will be fixed after some further adjustments.
◆ Generic pricing ratio in low-to-mid 40% range…failure to meet required criteria: 85% → 80% = The pricing ratio, which had been described as “in the 40% range” last November, has recently narrowed to the low-to-mid 40% range at the HIPDC subcommittee.
The industry expects it to fall somewhere between 43% and 45%. Since many had hoped for the high 40% range, the industry had expressed disappointment. However, because time remains before the HIPDC meeting, the final rate still remains to be seen.
If the required conditions for generic pricing are not met, the pricing level is expected to be lowered from 85% to 80%. If a company does not submit its own bioequivalence data or fails to use APIs registered with the MFDS, the reduction rate will become greater.
◆ Price cuts for already listed drugs to begin in Q3…cuts will be deferred for innovative companies, but duration undecided=The government plans to begin price cuts for already listed products in Q3. It intends to divide companies into groups based on their listing dates and implement sequential adjustments using varying calculation rates.
Innovative pharmaceutical companies will be granted a deferral period under a separate pricing calculation rule. However, an exception is being discussed for ingredients with 21 or more listed products. A deferral period of 4 to 5 years has been mentioned, but it will be finalized at the HIPDC meeting on the 26th.
For innovative companies, their reduction ratio under the price-volume agreement will be raised from 30% to 50%, with application expected to start at the end of this year.
◆ New “innovation-equivalent” category to be established next year…50% preferential pricing=When newly listing drugs, innovative companies will receive a 60% pricing add-on. The period for this premium will be 1+3 years. If the product is manufactured domestically, an additional 3 years will be granted.
In addition, the government is considering adding a category of companies equivalent to innovative firms, with 50% preferential pricing. These “quasi-innovative” companies would be defined based on annual sales above or below KRW 100 billion, with R&D investment ratios of at least 5% or 7% of sales, respectively.
However, companies that have received administrative sanctions for rebates within the past 5 years will be excluded. The preferential period for quasi-innovative companies is expected to be the same as that for certified innovative companies.
The preferential measures for innovative and quasi-innovative companies are expected to take effect starting next year.
◆ Stepwise price reduction from the 13th product onward…15% reduction rate maintained=To manage multiple generic listings, the government plans to strengthen the stepwise reduction system. The stepwise reduction, which currently begins from the 21st product, is expected to shift to the 13th listed product.
The reduction rate itself is expected to remain unchanged, with the price set at 85% of the previous lowest price. However, if multiple products are listed at the point when the 13th product is entered, the price of those products will be adjusted to the 85% level one year later.
The strengthened stepwise reduction and multi-product listing management are expected to take effect next year.
◆ 100-day fast-track listing to be institutionalized next year…selection criteria for ‘innovative new drugs’ to be established=The government plans to institutionalize fast-track listing for innovative new drugs next year. It also plans to establish post-evaluation and price-adjustment mechanisms.
Selection criteria will also be established for determining which innovative new drugs will qualify for fast-track listing. The operation of a grading system is also under review.
The government plans to operate a performance-based evaluation model incorporating digital healthcare elements such as hospital EMR systems and AI data, and is also reviewing the creation of a specialized agency.
It appears that decisions to delist drugs will be made through post-market evaluation. The plan is to establish an evaluation and adjustment system to determine selective reimbursement coverage and appropriate drug prices.
◆ Flexible drug pricing contract system to expand in Q2…will prepare measures to prevent inconvenience to patients =The expansion of the so-called ‘flexible drug pricing contract system,’ which allows for contract prices different from the listed price, is expected to be implemented in Q2.
The expanded scope is expected to include listed new drugs, already listed off-patent originals, new drugs whose RSA refund period has ended, incrementally modified new drugs, and biosimilars.
As the number of products subject to dual pricing increases, the government is preparing measures to prevent inconvenience to patients. One approach under review is to charge patients based on the separately contracted price rather than the listed price, thereby preventing the need for refunds.
◆ Price evaluation and adjustment every 3–5 years…targeting ingredients 5 years after first generic entry=The government is establishing a system for evaluating and adjusting drug prices every 3–5 years. This involves comparing, by ingredient, ▲the number of products, ▲market structure, and ▲drug prices in major countries.
The target appears to be ingredients for which 5 years have passed since the first generic entered the market. The specific operational model will be established and implemented after gathering industry feedback.
The Ministry of Health and Welfare is reportedly expected to revise and supplement the reform plan containing these elements and approve it at the HIPDC meeting on the 26th.
-

- 0
댓글 운영방식은
댓글은 실명게재와 익명게재 방식이 있으며, 실명은 이름과 아이디가 노출됩니다. 익명은 필명으로 등록 가능하며, 대댓글은 익명으로 등록 가능합니다.
댓글 노출방식은
댓글 명예자문위원(팜-코니언-필기모양 아이콘)으로 위촉된 데일리팜 회원의 댓글은 ‘게시판형 보기’와 ’펼쳐보기형’ 리스트에서 항상 최상단에 노출됩니다. 새로운 댓글을 올리는 일반회원은 ‘게시판형’과 ‘펼쳐보기형’ 모두 팜코니언 회원이 쓴 댓글의 하단에 실시간 노출됩니다.
댓글의 삭제 기준은
다음의 경우 사전 통보없이 삭제하고 아이디 이용정지 또는 영구 가입제한이 될 수도 있습니다.
-
저작권·인격권 등 타인의 권리를 침해하는 경우
상용 프로그램의 등록과 게재, 배포를 안내하는 게시물
타인 또는 제3자의 저작권 및 기타 권리를 침해한 내용을 담은 게시물
-
근거 없는 비방·명예를 훼손하는 게시물
특정 이용자 및 개인에 대한 인신 공격적인 내용의 글 및 직접적인 욕설이 사용된 경우
특정 지역 및 종교간의 감정대립을 조장하는 내용
사실 확인이 안된 소문을 유포 시키는 경우
욕설과 비어, 속어를 담은 내용
정당법 및 공직선거법, 관계 법령에 저촉되는 경우(선관위 요청 시 즉시 삭제)
특정 지역이나 단체를 비하하는 경우
특정인의 명예를 훼손하여 해당인이 삭제를 요청하는 경우
특정인의 개인정보(주민등록번호, 전화, 상세주소 등)를 무단으로 게시하는 경우
타인의 ID 혹은 닉네임을 도용하는 경우
-
게시판 특성상 제한되는 내용
서비스 주제와 맞지 않는 내용의 글을 게재한 경우
동일 내용의 연속 게재 및 여러 기사에 중복 게재한 경우
부분적으로 변경하여 반복 게재하는 경우도 포함
제목과 관련 없는 내용의 게시물, 제목과 본문이 무관한 경우
돈벌기 및 직·간접 상업적 목적의 내용이 포함된 게시물
게시물 읽기 유도 등을 위해 내용과 무관한 제목을 사용한 경우
-
수사기관 등의 공식적인 요청이 있는 경우
-
기타사항
각 서비스의 필요성에 따라 미리 공지한 경우
기타 법률에 저촉되는 정보 게재를 목적으로 할 경우
기타 원만한 운영을 위해 운영자가 필요하다고 판단되는 내용
-
사실 관계 확인 후 삭제
저작권자로부터 허락받지 않은 내용을 무단 게재, 복제, 배포하는 경우
타인의 초상권을 침해하거나 개인정보를 유출하는 경우
당사에 제공한 이용자의 정보가 허위인 경우 (타인의 ID, 비밀번호 도용 등)
※이상의 내용중 일부 사항에 적용될 경우 이용약관 및 관련 법률에 의해 제재를 받으실 수도 있으며, 민·형사상 처벌을 받을 수도 있습니다.
※위에 명시되지 않은 내용이더라도 불법적인 내용으로 판단되거나 데일리팜 서비스에 바람직하지 않다고 판단되는 경우는 선 조치 이후 본 관리 기준을 수정 공시하겠습니다.
※기타 문의 사항은 데일리팜 운영자에게 연락주십시오. 메일 주소는 dailypharm@dailypharm.com입니다.







