- LOGIN
- MemberShip
- 2026-04-02 07:36:13
- Global pharmas cautiously welcome pricing reform
- by Son, Hyung Min | translator Alice Kang | 2026-04-01 08:15:51
“Domestic firms are crying, multinational firms are smiling.” This is the prevailing assessment of Korea’s latest drug pricing reform.
With the reform centered on lowering prices of generic drugs now finalized, Korea’s drug pricing structure is approaching a turning point. While the government has proposed reallocating savings toward rewarding innovative drugs and improving patient access, the industry is also voicing cautious views that outcomes will depend on the system’s effectiveness and execution.
According to industry sources, the Ministry of Health and Welfare finalized the drug pricing reform plan on the 26th through the Health Insurance Policy Deliberation Committee (HIPDC). The reform focuses on lowering the pricing benchmark for generic and off-patent drugs from the current 53.55% to around 45%.
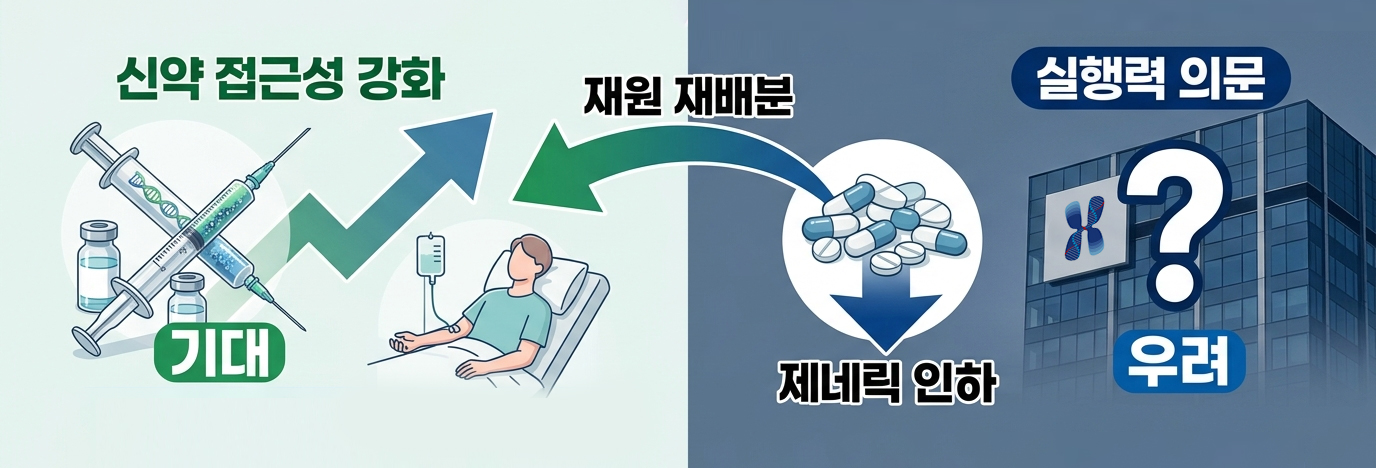
This reform is regarded as significant not simply as a price cut, but as a structural reorganization aimed at strengthening incentives for innovative drugs using the savings generated.
The key lies in resource reallocation. The strategy is to use the funds secured by adjusting the generic-centric pricing structure to lower barriers to reimbursement for new drugs through measures such as ▲faster reimbursement listing of treatments for rare and severe diseases, ▲introduction of flexible drug pricing contracts, and ▲raising the ICER (incremental cost-effectiveness ratio) threshold.
This is interpreted as a response to long-standing criticism that Korea’s pricing system has focused excessively on cost containment, limiting access to innovative therapies.
The Ministry of Health and Welfare also defined the reform as a structural transformation of the pricing system.
The Ministry stated, “By advancing the drug pricing system to the level of major countries, we can enhance public access to treatment and coverage while r reducing drug expenditure burdens. Establishing a compensation system for research and development and efforts to ensure the stable supply of essential medicines will serve as a momentum for the pharmaceutical and biotech industries to take the leap forward.”
Global pharmas express “cautious optimism”…System design is key
Global pharmaceutical companies are generally welcoming the reform. Given that patient access and reimbursement rates for new drugs in Korea have lagged behind major countries, there is an expectation that savings from generic price cuts could improve access to innovative therapies.
The Korea Research-based Pharmaceutical Industry Association (KRPIA), which is primarily composed of multinational pharmaceutical companies, also offered a positive assessment.
The association stated, “he policy reflects a commitment to reflecting the value of innovative new drugs and enhancing patient access. It is crucial whether the reform’s intent is actually realized through system design and implementation,” thereby emphasizing the importance of policy implementation.
This expectation is also supported by data. According to PhRMA’s ‘2023 Global Access to New Medicines Report,’ among the 460 new drugs covered by health insurance worldwide from 2012 to 2021, South Korea’s coverage rate was 22%, falling below the G20 (28%) and OECD (29%) averages.
For innovative cancer drugs, the rate was 23%, and for rare disease treatments, it was just 12%, both significantly lower than the G20 and OECD averages, respectively.
However, global pharmaceutical companies are also expressing conditional caution. While the entry environment may improve, there are concerns that requirements for demonstrating value during reimbursement listing have become more stringent.
An official from a global pharmaceutical company noted, “It is positive in terms of improving access to new drugs and strengthening clinical value-based evaluation. However, the strengthened post-listing price control raises concerns about predictability.”
Another official from a global pharmaceutical company agreed with the direction but raised questions about its implementation.
The official said, “The changes in government perception toward rare and severe diseases are significant. Even drugs under the pilot approval-evaluation-negotiation linkage program are facing reimbursement delays, so concrete execution plans are urgently needed.”
Regarding the increase in the ICER threshold, the official emphasized, “The magnitude of the increase is more important than the direction itself. If implementation is delayed under the pretext of policy research, it will just become another waiting period for patients.”
Institutional issues surrounding global pharmaceutical companies also remain unresolved. Industry feedback regarding the revision of certification criteria for innovative pharmaceutical companies was, “only the name has changed, with limited substantive improvement.”
Apart from the fact that bonus points are awarded for certain factors such as attracting foreign capital, joint research, and open innovation, the assessment is that the industry’s long-standing demand for the inclusion of achievements in attracting headquarter-level R&D investment has not been sufficiently reflected. Furthermore, the fact that indicators, such as the scale of pharmaceutical exports, which are difficult for multinational companies to meet, remain unchanged, poses disadvantages.
While some companies are already expanding cooperation with the government and joint research with domestic firms with certification in mind, the extent to which these efforts will be reflected in evaluations remains uncertain.
An industry official stated, “To foster the domestic biotech ecosystem, collaboration with global pharma is essential. Since the role of domestic branches is crucial in attracting headquarter-level R&D investment, these characteristics need to be reflected in the design of the system.”
Another industry official noted, “The new system is already affecting future pipeline processes. Multinational companies have been strengthening preparations to obtain the Innovative Pharmaceutical Company certification,” suggesting that this reform is bringing about changes in actual business strategies in practice.
-

- 0
댓글 운영방식은
댓글은 실명게재와 익명게재 방식이 있으며, 실명은 이름과 아이디가 노출됩니다. 익명은 필명으로 등록 가능하며, 대댓글은 익명으로 등록 가능합니다.
댓글 노출방식은
댓글 명예자문위원(팜-코니언-필기모양 아이콘)으로 위촉된 데일리팜 회원의 댓글은 ‘게시판형 보기’와 ’펼쳐보기형’ 리스트에서 항상 최상단에 노출됩니다. 새로운 댓글을 올리는 일반회원은 ‘게시판형’과 ‘펼쳐보기형’ 모두 팜코니언 회원이 쓴 댓글의 하단에 실시간 노출됩니다.
댓글의 삭제 기준은
다음의 경우 사전 통보없이 삭제하고 아이디 이용정지 또는 영구 가입제한이 될 수도 있습니다.
-
저작권·인격권 등 타인의 권리를 침해하는 경우
상용 프로그램의 등록과 게재, 배포를 안내하는 게시물
타인 또는 제3자의 저작권 및 기타 권리를 침해한 내용을 담은 게시물
-
근거 없는 비방·명예를 훼손하는 게시물
특정 이용자 및 개인에 대한 인신 공격적인 내용의 글 및 직접적인 욕설이 사용된 경우
특정 지역 및 종교간의 감정대립을 조장하는 내용
사실 확인이 안된 소문을 유포 시키는 경우
욕설과 비어, 속어를 담은 내용
정당법 및 공직선거법, 관계 법령에 저촉되는 경우(선관위 요청 시 즉시 삭제)
특정 지역이나 단체를 비하하는 경우
특정인의 명예를 훼손하여 해당인이 삭제를 요청하는 경우
특정인의 개인정보(주민등록번호, 전화, 상세주소 등)를 무단으로 게시하는 경우
타인의 ID 혹은 닉네임을 도용하는 경우
-
게시판 특성상 제한되는 내용
서비스 주제와 맞지 않는 내용의 글을 게재한 경우
동일 내용의 연속 게재 및 여러 기사에 중복 게재한 경우
부분적으로 변경하여 반복 게재하는 경우도 포함
제목과 관련 없는 내용의 게시물, 제목과 본문이 무관한 경우
돈벌기 및 직·간접 상업적 목적의 내용이 포함된 게시물
게시물 읽기 유도 등을 위해 내용과 무관한 제목을 사용한 경우
-
수사기관 등의 공식적인 요청이 있는 경우
-
기타사항
각 서비스의 필요성에 따라 미리 공지한 경우
기타 법률에 저촉되는 정보 게재를 목적으로 할 경우
기타 원만한 운영을 위해 운영자가 필요하다고 판단되는 내용
-
사실 관계 확인 후 삭제
저작권자로부터 허락받지 않은 내용을 무단 게재, 복제, 배포하는 경우
타인의 초상권을 침해하거나 개인정보를 유출하는 경우
당사에 제공한 이용자의 정보가 허위인 경우 (타인의 ID, 비밀번호 도용 등)
※이상의 내용중 일부 사항에 적용될 경우 이용약관 및 관련 법률에 의해 제재를 받으실 수도 있으며, 민·형사상 처벌을 받을 수도 있습니다.
※위에 명시되지 않은 내용이더라도 불법적인 내용으로 판단되거나 데일리팜 서비스에 바람직하지 않다고 판단되는 경우는 선 조치 이후 본 관리 기준을 수정 공시하겠습니다.
※기타 문의 사항은 데일리팜 운영자에게 연락주십시오. 메일 주소는 dailypharm@dailypharm.com입니다.
- "Joint bioequivalence licensing system is driving abundant generics"
- Reporter's view | Lee, Jeong-Hwan
- ‘Revisiting Xeljanz safety concerns based on accumulated long-term data’
- Reporter's view | Son, Hyung Min







