- LOGIN
- MemberShip
- 2026-04-08 15:26:04
- K-biosimilars gain influence in domestic market
- by Chon, Seung-Hyun | translator Alice Kang | 2026-04-08 07:46:56
Domestically developed biosimilars have gradually expanded their influence in the domestic market. Celltrion’s Remsima and Samsung Bioepis’s Onbevezi competed for the top spot by a narrow margin, both recording sales in the KRW 40 billion range. Celltrion’s Prolia biosimilar, joined by Daewoong Pharmaceutical, surpassed KRW 10 billion in sales in its first year of release. This trend reflects the increasing involvement of traditional pharmaceutical companies in biosimilar sales, which has strengthened market penetration.
According to the Financial Supervisory Service on the 7th, Celltrion’s Remsima recorded KRW 45.5 billion in sales last year, up 3.4% year-on-year, securing the top spot among domestically developed biosimilars. In 2024, Samsung Bioepis’s Onbevzi took the lead with KRW 45.2 billion, but last year Remsima overtook it by KRW 5.5 billion. The figures are based on sales disclosed by Celltrion Pharm and Boryung, which handle domestic sales for Celltrion and Samsung Bioepis biosimilars, respectively.
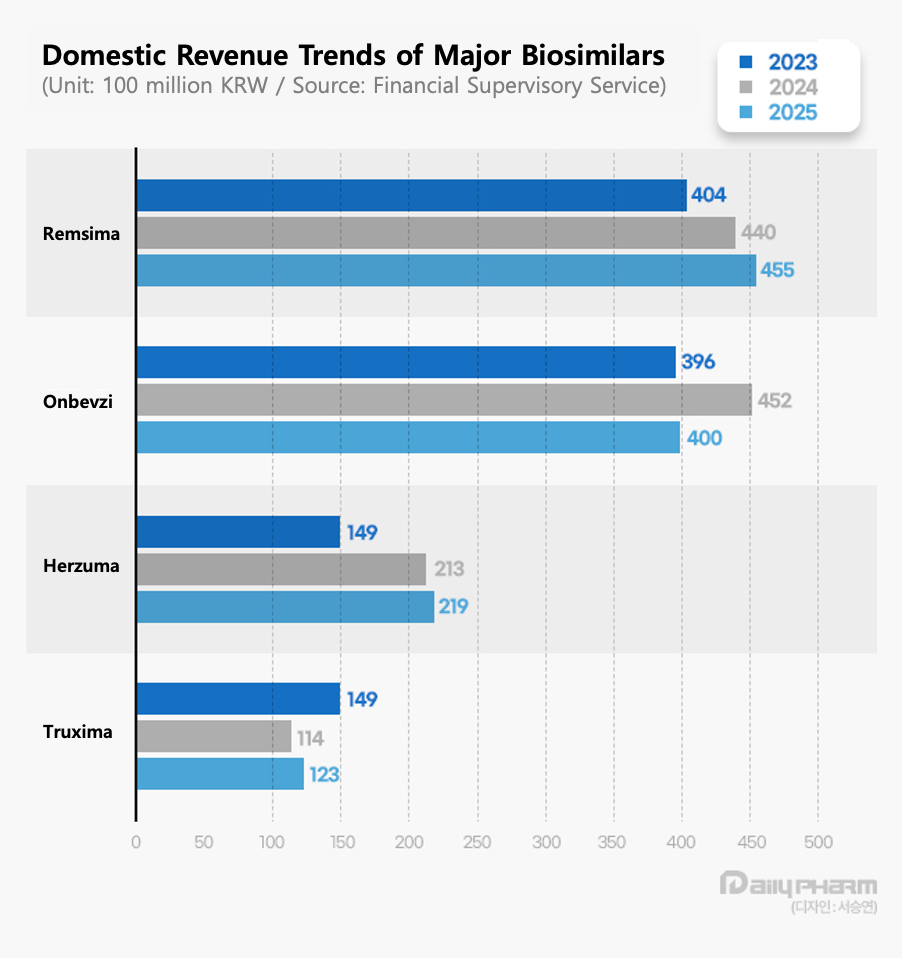
Remsima is a biosimilar of the autoimmune treatment Remicade. It was approved in 2012 as Korea’s first domestically developed antibody biosimilar. It is used to treat Crohn’s disease, ankylosing spondylitis, ulcerative colitis, and rheumatoid arthritis.
Onbevzi is a biosimilar version of the oncology drug Avastin. It is an anticancer drug used to treat metastatic colorectal cancer, metastatic breast cancer, non-small cell lung cancer, advanced or metastatic renal cell carcinoma, glioblastoma, epithelial ovarian cancer, fallopian tube cancer, primary peritoneal cancer, and cervical cancer.
Being the first biosimilar product introduced to the domestic market, Remsima had long led sales, but the rapid growth of Onbevzi since its introduction shifted the market into a duopoly structure.
In 2023, Remsima maintained a narrow lead of KRW 0.8 billion, but in 2024, Onbevzi overtook it for the first time by KRW 1.2 billion. However, Onbevzi’s sales declined 11.5% year-on-year to KRW 40 billion last year, allowing Remsima to regain the top position.
The domestic biosimilar market is drawing attention as a battleground driven by traditional pharmaceutical companies’ sales capabilities.
In the Avastin market, Samsung Bioepis launched Onbevzi in September 2021, followed by entries from Celltrion and Alvogen Korea. As the first entrant, Onbevzi maximized its advantage by leveraging tailored sales strategies. Samsung Bioepis also secured an exclusive domestic sales agreement with Boryung immediately after approval. Boryung is known for its strong presence in oncology sales.
Celltrion’s biosimilars are marketed domestically through Celltrion Pharm. Celltrion’s Herzuma, a biosimilar version of Herceptin, recorded KRW 21.9 billion in sales last year, up 2.8%, ranking behind Onbevzi and Remsima. Truxima, a biosimilar version of MabThera, recorded KRW 12.3 billion in sales, up 7.9%.
While domestically developed biosimilars are still far from achieving commercial success in the global market, they appear to be gradually expanding their influence with the support of traditional pharmaceutical companies.
Last year, 8 of Celltrion’s 12 biosimilar product lines sold in the global market exceeded KRW 100 billion in sales. Remsima recorded sales of KRW 1.0495 trillion last year, surpassing the KRW 1 trillion mark for the second consecutive year.
Remsima SC generated KRW 717.2 billion in sales, up 27.1% year-on-year. Zymfentra grew from KRW 36.6 billion in 2024 to KRW 122.2 billion last year, more than tripling and surpassing KRW 100 billion. Remsima SC is a subcutaneous formulation developed from the original intravenous Remsima, and it has been approved in the U.S. as a new drug under the name Zymfentra. Truxima and Herzuma generated sales of KRW 526.3 billion and KRW 217.1 billion, respectively, in the global market last year.
Recently, sales competition has been intensifying with traditional pharmaceutical companies joining the biosimilar market en masse.
Samsung Bioepis has established disease-specific sales capabilities tailored to biosimilars.
It initially partnered with Daewoong Pharmaceutical for its Herceptin biosimilar in 2017 but switched to Boryung in 2021. Immediately after receiving domestic approval for Onbevzi, a biosimilar of Avastin, in 2021, the company signed an exclusive domestic sales agreement with Boryung. Samsung Bioepis selected Samil Pharmaceutical as the sales partner for biosimilars of Lucentis and Eylea, treatments for ophthalmic diseases.
Last year, Samsung Bioepis selected Hanmi Pharmaceutical as its domestic sales partner for its Prolia biosimilar Obodence. Samsung Bioepis handles production and supply, while both companies jointly manage marketing and sales. Its original version, Prolia, which was developed by Amgen, works by inhibiting the activity of osteoclasts that break down bone, thereby preventing bone resorption and increasing bone density. It prevents bone loss in postmenopausal women and reduces the risk of fractures, while in cancer patients, it inhibits bone metastasis and protects bone structure to reduce complications.
Daewoong Pharmaceutical signed a joint sales and distribution agreement with Celltrion Pharm last year and began domestic sales of Celltrion’s Prolia biosimilar, Stoboclo. Daewoong Pharmaceutical is conducting joint sales of Stoboclo with Celltrion Pharm at general hospitals and clinics nationwide. Celltrion has sold biosimilars in the domestic market through its affiliate, Celltrion Pharm. Stoboclo is the first Celltrion biosimilar to be sold by a pharmaceutical company other than Celltrion Pharm. Stoboclo generated sales of KRW 11.8 billion last year.
Daewoong Pharm’s strategy is to expand prescriptions at major general and university hospitals nationwide to grow Stoboclo into a ‘mega blockbuster’ with annual sales exceeding KRW 100 billion. Currently, Stoboclo has been introduced to more than 50 major general and university hospitals nationwide, rapidly expanding its prescription base. Daewoong has also joined the sales efforts for LG Chem’s Humira biosimilar Xelenka.
-

- 0
댓글 운영방식은
댓글은 실명게재와 익명게재 방식이 있으며, 실명은 이름과 아이디가 노출됩니다. 익명은 필명으로 등록 가능하며, 대댓글은 익명으로 등록 가능합니다.
댓글 노출방식은
댓글 명예자문위원(팜-코니언-필기모양 아이콘)으로 위촉된 데일리팜 회원의 댓글은 ‘게시판형 보기’와 ’펼쳐보기형’ 리스트에서 항상 최상단에 노출됩니다. 새로운 댓글을 올리는 일반회원은 ‘게시판형’과 ‘펼쳐보기형’ 모두 팜코니언 회원이 쓴 댓글의 하단에 실시간 노출됩니다.
댓글의 삭제 기준은
다음의 경우 사전 통보없이 삭제하고 아이디 이용정지 또는 영구 가입제한이 될 수도 있습니다.
-
저작권·인격권 등 타인의 권리를 침해하는 경우
상용 프로그램의 등록과 게재, 배포를 안내하는 게시물
타인 또는 제3자의 저작권 및 기타 권리를 침해한 내용을 담은 게시물
-
근거 없는 비방·명예를 훼손하는 게시물
특정 이용자 및 개인에 대한 인신 공격적인 내용의 글 및 직접적인 욕설이 사용된 경우
특정 지역 및 종교간의 감정대립을 조장하는 내용
사실 확인이 안된 소문을 유포 시키는 경우
욕설과 비어, 속어를 담은 내용
정당법 및 공직선거법, 관계 법령에 저촉되는 경우(선관위 요청 시 즉시 삭제)
특정 지역이나 단체를 비하하는 경우
특정인의 명예를 훼손하여 해당인이 삭제를 요청하는 경우
특정인의 개인정보(주민등록번호, 전화, 상세주소 등)를 무단으로 게시하는 경우
타인의 ID 혹은 닉네임을 도용하는 경우
-
게시판 특성상 제한되는 내용
서비스 주제와 맞지 않는 내용의 글을 게재한 경우
동일 내용의 연속 게재 및 여러 기사에 중복 게재한 경우
부분적으로 변경하여 반복 게재하는 경우도 포함
제목과 관련 없는 내용의 게시물, 제목과 본문이 무관한 경우
돈벌기 및 직·간접 상업적 목적의 내용이 포함된 게시물
게시물 읽기 유도 등을 위해 내용과 무관한 제목을 사용한 경우
-
수사기관 등의 공식적인 요청이 있는 경우
-
기타사항
각 서비스의 필요성에 따라 미리 공지한 경우
기타 법률에 저촉되는 정보 게재를 목적으로 할 경우
기타 원만한 운영을 위해 운영자가 필요하다고 판단되는 내용
-
사실 관계 확인 후 삭제
저작권자로부터 허락받지 않은 내용을 무단 게재, 복제, 배포하는 경우
타인의 초상권을 침해하거나 개인정보를 유출하는 경우
당사에 제공한 이용자의 정보가 허위인 경우 (타인의 ID, 비밀번호 도용 등)
※이상의 내용중 일부 사항에 적용될 경우 이용약관 및 관련 법률에 의해 제재를 받으실 수도 있으며, 민·형사상 처벌을 받을 수도 있습니다.
※위에 명시되지 않은 내용이더라도 불법적인 내용으로 판단되거나 데일리팜 서비스에 바람직하지 않다고 판단되는 경우는 선 조치 이후 본 관리 기준을 수정 공시하겠습니다.
※기타 문의 사항은 데일리팜 운영자에게 연락주십시오. 메일 주소는 dailypharm@dailypharm.com입니다.
- "Joint bioequivalence licensing system is driving abundant generics"
- Reporter's view | Lee, Jeong-Hwan
- ‘Revisiting Xeljanz safety concerns based on accumulated long-term data’
- Reporter's view | Son, Hyung Min





