- LOGIN
- MemberShip
- 2026-05-01 13:10:17
- Policy
- MFDS reviews emergency import of 'anakinra'
- by Lee, Hye-Kyung Jul 07, 2025 06:08am
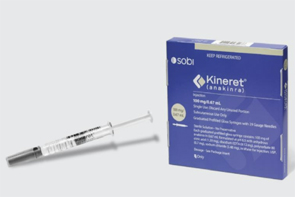
- Product photo of Kineret (anakinra) The Ministry of Food and Drug Safety (MFDS) is reportedly conducting a review of whether to urgently import anakinra (brand name Kineret) for the treatment of severe adverse reactions associated with CAR-T cell therapy. On June 2nd, in response to an official inquiry from specialized journalists, MFDS stated, "MFDS is currently reviewing the necessity of emergency import based on the safety, efficacy, and overseas usage status of this product and its indication." The current MFDS review is a follow-up action to the emergency import request for anakinra submitted by the Korean Society of Hematology. The Pharmaceutical Safety Bureau, which received the request, transferred the item to the MFDS Biopharmaceuticals Policy Division for an official review process. CAR-T cell therapy is an advanced, precision treatment that genetically modifies a patient's immune cells to target and attack cancer cells. It has garnered significant attention as a treatment offering different effects compared to conventional anti-cancer drugs, particularly for relapsed and refractory acute lymphoblastic leukemia, diffuse large B-cell lymphoma, and multiple myeloma. In South Korea, the treatment costs KRW 400-500 million. With national health insurance reimbursement applied, the patient's out-of-pocket burden has significantly decreased. Yet, while this high-cost therapy is supported with reimbursement, some adjuvant medications used to manage severe immune adverse reactions that can occur after treatment remain unreimbursed. 'Tocilizumab' or 'high-dose steroids' are primarily used as first-line agents. However, for patients who are refractory to these, anakinra, an Interleukin-1 (IL-1) inhibitor, serves as a crucial therapeutic alternative. However, anakinra's general use in Korea has been blocked since its withdrawal from the domestic market based on the manufacturer's decision. Anakinra is an interleukin-1 receptor antagonist manufactured by Sobi of Sweden. It's approved in countries such as the United States (FDA), Europe (EMA), and Japan (PMDA) for treating conditions including rheumatoid arthritis, CAPS (Cryopyrin-Associated Periodic Syndromes), FMF (Familial Mediterranean Fever), and Still's Disease. The 2025 European CAR T Handbook by the GoCART Coalition recommends high-dose anakinra administration for Cytokine Release Syndrome (CRS) and Immune Effector Cell-Associated Neurotoxicity Syndrome (ICANS), and its guidelines include co-administration with corticosteroids for patients with IEC-HS (Immune Effector Cell-Associated Hemophagocytic Lymphohistiocytosis/Macrophage Activation Syndrome). Specifically, the guidelines suggest anakinra as an essential treatment option for high-risk patients who do not respond to steroid and tocilizumab therapies. Furthermore, a study by Gazeau N et al. (Transplant Cell Ther. 2023) showed that among 43 patients who developed Grade 2 or higher complications after CAR-T therapy and were administered anakinra, the treatment-related mortality rate within 28 days was 0%, and the overall response rate was 77%. Meanwhile, in South Korea, anakinra is currently supplied only in a limited manner through the Korea Orphan & Essential Drug Center (KOEDC) as an urgently imported drug. Yet, it is not permitted for treating adverse reactions of CAR-T. Consequently, some hospitals are attempting to offer limited prescriptions through their own Institutional Review Board (IRB) approvals. However, the treatment is neither reimbursed nor guaranteed legal protection, making nationwide application impossible. The Korean Society of Hematology stated, "Currently, medical professionals are forced to off-label use anakinra, even risking illegal practice, to save patients' lives," and added, "This does not align with the fundamental principles of national public health systems and the right to life, and urgent institutional reform is desperately needed." The Korean Society of Hematology, which has submitted requests to the Ministry of Health and Welfare (MOHW) and the HIRA for several years following the reimbursement of CAR-T cell therapy, submitted a request via the 'Anti-Corruption & Civil Rights Commission (e-People)' portal in April regarding the allowance of off-label use of anakinra and institutional improvement. In response, MFDS replied that "upon request from a central administrative agency or professional organization, we can review the expansion of the reimbursement scope based on the necessity for patient treatment, availability of alternatives, and overseas approval status." In response, the Korean Society of Hematology formally submitted an emergency import request for anakinra to the MFDS Pharmaceutical Safety Bureau on May 19. The request included information such as product details, approval status in major overseas countries, usage cases in the U.S. and Europe, the necessity of its use in life-threatening situations, domestic demand prediction (less than 100 cases annually), and the lack of drug alternatives. The Korean Society of Hematology stated, "Rapid immune modulation is essential in life-threatening severe complications, and currently, there are no drugs with a similar mechanism to anakinra available in South Korea." They emphasized, "This emergency import request is an essential measure to protect patient lives and ensure access to treatment, and prompt institutional reform is required."
- Policy
- National Office of Investigation announces rebate crackdown
- by Lee, Jeong-Hwan Jul 04, 2025 06:06am
- Amidst the National Police Agency's announcement of a special crackdown on illegal drug rebates following the launch of the new administration, the Ministry of Health and Welfare is also busy assessing the situation. The MOHW plans to actively consult with the National Police Agency if it receives a specific request for cooperation to crack down on illegal rebates. On the 2nd, an official from the MOHW explained, “The issue of illegal rebates is understood to be part of a national policy agenda related to the National Police Agency's special crackdown, aimed at eradicating corruption and promoting integrity among public officials.” However, the official said that no separate request for cooperation has been made by the National Police Agency to MOHW for the special crackdown on rebates so far. The Ministry of Health and Welfare is currently maintaining its routine of monitoring cases and referring them to investigative agencies such as the police, in accordance with the guidelines for handling illegal pharmaceutical rebates. The ministry also plans to respond to future requests for cooperation. The official said, “The MOHW requests investigations by the police or the prosecution when it receives external reports or complaints related to rebates. This is in accordance with our guidelines for handling rebates. The police or the prosecution launches an investigation, then reports the results to the MOHW.” He added, “There are cases where the police request an authoritative interpretation to the MOHW when legal interpretation is necessary, such as whether there has been a violation of the Pharmaceutical Affairs Act, and some investigative agencies request the submission of expenditure reports. In response to some requests earlier this year, we provided (submitted expenditure reports) within the scope that could be disclosed.” He added, “There are no immediate plans or policies regarding the media reports, but we are cooperating with the prosecution and the police and are requesting investigations if necessary.”
- Policy
- Pressures to reform Korea's drug pricing system
- by Lee, Tak-Sun Jul 02, 2025 06:10am
- President Trump's signing of the executive order on May 12 to lower prescription drug prices in the United States to the same level as other countries has deepened the concerns of Korea’s authorities as well. This is because the U.S. Pharmaceutical Research and Manufacturers of America and others are demanding that Korea raise drug prices and improve its system. If the U.S. government directly demands improvements to Korea's drug pricing system during tariff negotiations, this may increase the new government's concerns as there will be no clear solutions to resolving the issue, On the 27th (local time), the Pharmaceutical Research and Manufacturers of America (phRMA) submitted a statement to the US Trade Representative (USTR) urging the improvement of unfair drug pricing policies by foreign governments, including South Korea, as leverage in trade negotiations. The USTR claimed that Korea imposes difficult review requirements on foreign pharmaceutical companies, delays market entry, and suppresses drug prices below fair market value. This statement gained attention because it was released after President Trump signed an executive order on May 12, known as the most-favored-nation (MFN) policy. The MFN policy aims to lower the price of prescription drugs in the US to the same level as other countries. Under the executive order, the US Secretary of Health and Human Services must establish a program that allows US patients to purchase drugs directly from pharmaceutical companies at MFN prices. In its statement, the phRMA identified South Korea, Australia, Canada, France, Germany, Italy, Japan, Spain, the United Kingdom, and the European Union as countries with unfair drug pricing policies. These countries are likely to become MFN countries, which will serve as a reference for U.S. drug price reductions. It is analyzed that the Trump administration is likely to demand that MFN countries, with which the US pharmaceutical industry is currently engaged in tariff negotiations, improve their drug pricing systems in order to push through the reduction of prescription drug prices in the US. When President Trump signed the MFN policy executive order, he instructed the USTR and the Department of Commerce to take action to prevent other countries from unfairly lowering their drug prices below market prices. The Korean government is concerned that the US MFN policy will result in ‘Korea Passing,’ where companies bypass Korea when making new drug releases and withdraw from the market. However, during Trump's first term, the US drug price reduction policy was abandoned due to opposition from US pharmaceutical companies, so the situation requires continued monitoring. However, if the US actually demands improvements to the drug pricing system during tariff negotiations, there are no clear measures that South Korea can take in response, which is expected to increase the South Korean government's concerns. The measures the South Korean government can take include significantly adjusting the ICER (Incremental Cost-Effectiveness Ratio) threshold, and expanding risk-sharing agreement programs to adjust prices. However, given the high proportion of drug expenditures in the health insurance budget, there are limits to regulatory relaxation. Flexible application of the ICER threshold or expansion of RSA programs has already been implemented last year as part of efforts to compensate for the value of innovative new drugs. If the U.S. government pushes for significant improvements to the drug pricing system under the guise of trade pressure, the new government's health authorities would have few cards to play, leaving them with even more challenges ahead. A government official stated, “We are currently monitoring and closely watching the U.S. government's implementation of the MFN policy. We are preparing for the worst-case scenario, such as the withdrawal of U.S. pharmaceutical companies' products from the market.”
- Policy
- More generic 'Vimovo' drugs with naproxen+PPI enters the mkt
- by Lee, Tak-Sun Jun 30, 2025 06:05am
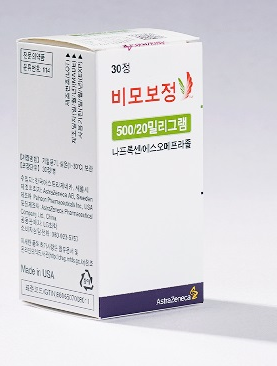
- Product photo of the original drug Generic drugs containing the same active ingredients as the 'Vimovo' (naproxen+esomeprazole magnesium trihydrate), a combination of an anti-inflammatory drug and an anti-ulcer agent, are set to be released. A generic has not been approved since Chong Kun Dang's 'Naxen S Tab' was approved in 2024. Considering the characteristics of a generic containing two types of active ingredients, proving a pharmaceutical equivalence test on each active ingredient may have been challenging. Furthermore, analysis suggests new product entry to this market poses a challenge due to 'Naxozole,' a salt-changed product that was launched at a relatively lower price, with a strong presence. According to the industry on June 27, four generic Vimovo drugs, including KyungDong Pharm's 'Nasopra Tab 500/20 mg,' will be listed with reimbursement next month. KyungDong Pharm is the organizing company. The company's Nasopra Tab 500/20 mg met two types of requirements. Thus, the price was assessed at KRW 715, the same as the original Vimovo. In contrast, the drug price of the generics meeting only one requirement was set as KRW 608. These include Genuonesciences' 'Gemovo Tab 500/20 mg,' Mother's Pharmaceutical's 'Vimo M Tab. 500/20 mg,' and Dongkook Pharmaceutical's 'Exoraxen Tab 500/20 mg.' These drugs treat symptoms of osteoarthritis, rheumatoid arthritis, and ankylosing spondylitis in patients at risk of gastric or duodenal ulcers associated with NSAIDs, such as naproxen, or in those who are not adequately treated with low-dose naproxen or other NSAIDs. Joined later by four companies, products containing the same active ingredients as Vimovo have now increase from two to six. Previously, LG Chem's 'Vimovo Tab 500/20 mg' and Chong Kun Dang's 'Naxen S Tab' were the only generics available. Furthermore, Naxen S Tab has been on the market for over 10 years since its approval in 2014. Even without re-examination periods or patent barriers, generic versions have not been made available, likely due to the significant difficulty of conducting bioequivalence tests for this combination therapies. To prove bioequivalence for generic Vimovo drugs, it's necessary to compare the human absorption rates of each component, naproxen and esomeprazole. This process is complex, and achieving favorable results is challenging. Korean pharmaceutical companies have shifted their development efforts towards modified drugs that use a different salt of esomeprazole. Currently, there are five such salt-changed products available, including Hanmi Pharm's 'Naxozole Tab. 500/20 mg. Naxozole recorded KRW 25.8 billion in outpatient prescription sales last year, according to UBIST data, surpassing the original Vimovo (KRW 21.7 billion). The issue, however, is that Naxozole is relatively low-priced, at KRW 445 per tablet, which is the cheapest among competing products. Naxen S, a generic version of Vimovo, is relatively inexpensive at KRW 490 and recorded KRW 4.2 billion in outpatient prescription sales last year. In contrast, these newly reimbursed generic drugs are priced KRW 100-200 higher than Naxen S, based on the estimation criteria. Therefore, it's uncertain whether they will exhibit drug price competitiveness against Naxozole and Naxen S in the market. However, an analysis suggests that if they enter the market with high CSO (Contract Sales Organization) fees based on their higher drug prices, they could increase their market share. Amid other generic companies also preparing for market entry, attention is now focused on the market performance of the products from KyungDong Pharm and the other three companies that have recently obtained reimbursement.
- Policy
- Polivy granted partial reimbursement after 5 years
- by Lee, Tak-Sun Jun 30, 2025 06:05am
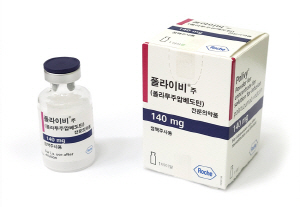
- Roche’s Polivy (polatuzumab vedotin), a treatment for diffuse large B-cell lymphoma (DLBCL) that is currently non-reimbursed in Korea, has been added to the reimbursement list as a part of combination therapy. With the listing, the other drugs used in the combination, excluding Polivy, will be reimbursed. This measure is in accordance with the partial reimbursement policy for combination cancer therapies that was implemented in May. On the 26th, the Health Insurance Review and Assessment Service announced the reimbursement criteria for anticancer drugs and announced 2 new partial reimbursements for combination anticancer therapies that included Polivy. Partial reimbursement for combination therapy using anticancer drugs grants reimbursement to already covered drugs that are included as part of combination therapy. Previously, combination therapies were not covered by reimbursement unless they were officially added as a whole to the reimbursement criteria. The Ministry of Health and Welfare implemented this policy in May, and earlier this month, HIRA announced 35 therapies to clarify the general principles. HIRA plans to review applications for partial reimbursement for combination therapy submitted by academic societies and make additional revisions as necessary through its Cancer Disease Review Committee. As a result, two combination therapies will be added to the partial reimbursement list in July. Polivy is the first first-line treatment for DLBLC in 20 years, but it has faced difficulties in obtaining reimbursement since its approval in 2020. As a result, it remains non-reimbursed to date. The combination therapy included in the partial reimbursement criteria this time is " polatuzumab Vedotin(non-reimbursed)+rituximab, cyclophosphamide, doxorubicin, and prednisone/prednisolone (R-CHP)" for treatment-naïve adult patients with Diffuse Large B-Cell Lymphoma (DLBCL). HIRA plans to apply reimbursement for all drugs except Polivy. A combination therapy for cervical cancer that uses the immuno-oncology drug Keytruda (pembrolizumab) has also been added. As with the previous case, partial reimbursement will be provided, so all other drugs will be reimbursed excluding Keytruda and Avastin (bevacizumab). The treatments granted partial reimbursement are: pembrolizumab + paclitaxel + carboplatin ± bevacizumab and pembrolizumab + paclitaxel + cisplatin ± bevacizumab. Reimbursement will be applied to patients with recurrent, metastatic (stage IV) cervical cancer or stage IB2 or higher that meet one or more of the following conditions: ▲positive pelvic lymph nodes (pelvic LN) after surgery, ▲positive para-aortic lymph nodes (para-aortic LN) after surgery, or ▲positive parametrium after surgery (stage I or higher, palliative treatment). The reimbursement criteria also include revisions to Tier 1 and 2 anticancer drugs. HIRA explained, “When the criteria were first established in 2006, in the details regarding the application criteria and methods for reimbursement of drugs prescribed and administered to cancer patients, drugs subject to re-evaluation, orphan drugs, or drugs with potential for abuse were classified as Tier 2 drugs and was granted use within the scope of its reimbursement criteria. Tier 1 drugs were to be administered at the discretion of the doctors within the scope of the indication and general principles for anticancer therapy." "However, since the initial enactment, new clinical evidence is being updated every year due to drug price fluctuations, the listing of generic drugs and many new high-priced anticancer drugs, and the development of new drugs with new mechanisms of actions, rendering it necessary to review the old treatment guidelines (mainly the deletion of Tier 1 anticancer drugs that are highly toxic and ineffective and the reclassification of Tier 2 anticancer drugs) and recommend the use of anticancer treatments that are more clinically proven and safer for the public. In this regard, we gathered opinions from academic societies and held TFT meetings on improving the reimbursement criteria for anticancer drugs to establish a reimbursement criteria (draft) for anticancer drugs. After final discussions by the Cancer Disease Deliberation Committee, we established new reimbursement criteria for anticancer therapies that removed the classification of anticancer drugs into Tiers 1 and 2.” However still, for cancers that are difficult to establish clinical evidence due to the small number of drug options and patient population, the current system will be maintained. Detailed information on the revisions can be found on the HIRA website (System/Policy → Drug Criteria Information → Drugs and Therapies Used for Cancer).
- Policy
- Ensuring stable supply of drugs in short supply
- by Lee, Jeong-Hwan Jun 27, 2025 06:03am
- Following President Lee Jae-myung's pledge to establish a stable supply system for drugs with supply shortages, attention is drawn to the Ministry of Health and Welfare's (MOHW) opinion that a social consensus on the criteria and scope of 'supply shortage' is first needed. It is anticipated that legislative review in the National Assembly to resolve the issue of drug shortages will progress if criteria for drug shortages are established, including whether the causes of the shortages are isolated or chronic. On June 26, an MOHW official met with the Korea Special Press Association and explained, "The government also agrees on the need to reform national governance to respond to drugs with supply shortages." The official proposed the necessity of establishing a definition and criteria for drugs with supply shortages to find solutions to the drug shortage problem properly. Establishing the criteria first is necessary because drug shortage issues arise from various types and cases, and finding a consensus on how to quantify them is essential for laying the groundwork to establish policies and proceed with legislation. The MOHW stated that while national essential medicines are designated through various criteria and procedures, defining criteria for drugs with supply shortages will be challenging due to their often variable nature, such as differing shortage durations for each case. An MOHW official explained, "It's crucial how we measure supply shortages. We monitor the situation through supply history reports, but it's difficult to confirm why shortages occur at the final distribution stage," and added, "Since we do not have any information on the inventory levels so there are data limitations in determining whether a shortage is due to an absolute lack of supply or a hoarding problem." The official further explained, "It takes about 2-3 months for data on actual drug prescriptions and usage to accumulate. Hospitals and pharmacies don't report every time a drug is used, and it's even harder to secure data for non-reimbursable drugs," and added, "We need to establish some level of supply shortage criteria to come up with countermeasures. Even during public-private meetings, we discussed whether a drug with supply shortage needed an intervention or not." Meanwhile, four amendments to the Pharmaceutical Affairs Act aimed at resolving the issue of drugs with supply shortages are currently pending in the National Assembly (proposed by Rep. Han Jeoung-ae, Rep. Kim Yoon, Rep. Kim Sunmin, and Rep. Seo Mi-hwa).
- Policy
- Yungjin, Ildong’s Ofev generics enter market at half price
- by Lee, Tak-Sun Jun 26, 2025 06:07am
- A generic version of Ofev (nintedanib), a treatment for chronic fibrotic interstitial lung disease), will enter the market at half the price of the original drug. With the entry of generic drugs, the Ofev market now faces competition, just two months after the original drug was listed for reimbursement. According to industry sources on the 25th, Yungjin and Ildong Pharmacuetical will list the generic version of Ofev with reimbursement at a price much lower than the calculated price, differentiating it from the original product. Also, the original product is a capsule formulation, while the generic version is a tablet formulation. When reimbursement was approved in May, the maximum insurance price for Boehringer Ingelheim Korea's Ofev Soft Capsules 100 mg was set at KRW 29,600, and Ofev Soft Capsules 150 mg at KRW 26,220. The generic versions are much cheaper than this. Three generic drugs will be listed for reimbursement next month: Yungjin Pharmaceutical's Nintebro Tab 150 mg and Nintebro 100 Tab mg, and Ildong Pharmaceutical's Cuninta Tab 150 mg. Among them, Nintebro 100 mg has a maximum price of KRW 9,000, less than half the original Ofev Soft Capsule 100 mg (KRW 20,960). Nintebro 150 mg is KRW 15,000, and Cuninta 150mg is KRW 13,500. When considering how Ofev Soft Cap 150mg costs KRW 26,220, the generic versions cost half the original price. Being rare disease treatments, the generic versions were eligible to receive the same price as the original Ofev, but it is believed that the companies significantly lowered the price in consideration of market competitiveness. As a result, patients will now be able to purchase the same ingredient drug at a lower price. In May, after nine years since being approved in Korea, Ofev was listed for reimbursement as a treatment for chronic fibrotic interstitial lung disease. Due to the delay in reimbursement listing, the substance patent expired on January 25. This is how generic versions were able to enter only 2 months after the original drug was listed for reimbursement. Meanwhile, Yungjin and Ildong, which are launching the generic versions, are competing fiercely in the pirfenidone market, a representative treatment for idiopathic pulmonary fibrosis, which is another indication for Ofev.
- Policy
- Daewoong's high-dose generic 'Xeljanz' wins nod
- by Lee, Hye-Kyung Jun 26, 2025 06:06am
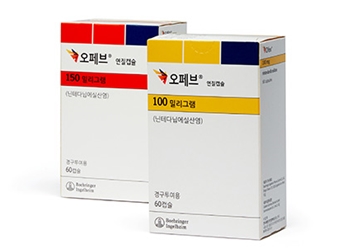
- Product photo of Daewoong Pharmaceutical's oral generic version of 'Xeljanz (tofacitinib),' which is used to treat rheumatoid arthritis, received approval. On June 23, the Ministry of Food and Drug Safety (MFDS) approved Daewoong Pharmaceutical's two dosages of 'Xeltofa Tab': 5 mg and 10 mg. The latest approval grabs attention, particularly because Daewoong Pharmaceutical has developed high-dose formulation of generic Xeljanz. Over 65 generic Xeljanz have been approved to date. However, only five items with a 10 mg high-dose formulation, including Pfizer Korea's 'Xeljanz,' Chong Kun Dang's 'Tofacell Tab,' Jeil Pharm's 'Topazan Tab,' Jeil Pharm's 'Topazan Tab,' and Alvogen Korea's 'Gencinib Tab,' are available. High-dose formulations have not been popular because the approved indications were fewer than those of low-dose ones. While the low-dose formulation holds five indications, including rheumatoid arthritis, psoriatic arthritis, ankylosing spondylitis, ulcerative colitis, polyarticular juvenile idiopathic arthritis (pJIA), and juvenile psoriatic arthritis, the high-dose formulation is only approved for ulcerative colitis, thereby limiting its therapeutic scope. Safety concerns and fewer indications have hindered high-dose generic Xeljanz approvals. However, an increasing number of pharmaceutical companies are now securing approvals for both low-dose and high-dose products. With the substance patent for Xeljanz expiring in November, Korean companies are expected to expedite getting approvals for their generics. Xeljanz holds a substance patent expiring in November 2025 and a crystal form patent expiring in November 2027. Twenty companies, initially by Chong Kun Dang, attempted to overcome these patents by filing invalidation trials. Additionally, 16 companies, including Boryung Pharmaceutical, filed passive rights scope confirmation trials to circumvent them. In January 2018, generic companies successfully circumvented the Xeljanz patent. Following another victory in an invalidation trial in November 2019, the Xeljanz crystal form patent was removed in March 2020. Generic Xeljanz items that have obtained marketing authorization can be launched after November 22, 2025, when the substance patent expires.
- Policy
- Samsung Bioepis joins Prolia market with Obodence
- by Lee, Tak-Sun Jun 25, 2025 06:02am
- With Samsung Bioepis entering the biosimilar market for Prolia (denosumab), competition is expected to intensify among co-promoter companies Daewoong Pharmaceutical (Stoboclo, Celltrion), Hanmi Pharmaceutical (Obodence, Samsung Bioepis), and Chong Kun Dang (original Prolia, Amgen). Chong Kun Dang signed a co-promotion agreement with the original manufacturer, Amgen; Daewoong Pharmaceutical with Celltrion; and Hanmi Pharmaceutical with Samsung Bioepis, respectively. According to industry sources on the 24th, Samsung Bioepis' Prolia biosimilar Obodence Prefilled Syringe (denosumab, 60mg/1mL) will be listed for reimbursement in July. As a result, Celltrion's ‘Stoboclo Pre-filled Syringe’ and Samsung Bioepis' ‘Obodence Pre-filled Syringe’ are expected to compete fiercely in the domestic market. Park Jae-Hyun, President of Hanmi Pharmaceutical (right), and Kim Kyung-Ah, CEO of Samsung Bioepis (left), sign a copromotion agreement in March and pose for a commemorative photo. Celltrion’s Stoboclo was listed for reimbursement in March. The maximum reimbursement ceiling price for Stoboclo and Obodence is the same. Obodence’s price was set the same as the lowest price of the same ingredient drug already listed when it was added to the reimbursement list. As there are between 2 and 19 drugs of the same type, and the price of the drugs have already been adjusted to 70%, Obodence’s price was calculated at the same price as the lowest price of the same drug already listed in accordance with the calculation rules for biological drugs. The lowest price among the same-ingredient drugs was Stoboclo. Stoboclo’s price was voluntarily reduced by Celltrion from KRW 111,384 to KRW 108,290 starting in July, and Obodence’s price was also set accordingly at KRW 108,290. The original Prolia’s insurance ceiling price is KRW 123,760. Prolia is the absolute leader in the osteoporosis treatment market. According to IQVIA, Prolia recorded sales of KRW 174.9 billion in the domestic market last year. Prolia suppresses the activity of osteoclasts responsible for bone degradation, thereby preventing bone resorption while promoting an increase in bone mineral density. The drug is regarded to have improved the convenience of administration for patients, as its efficacy can be maintained for 6 months with a single injection. Daewoong Pharmaceutical and Celltrion Pharmaceutical announced in October last year that they signed an agreement for the copromotion of a Prolia biosimilar With a domestic market size worth KRW 170 billion for that single drug, large pharmaceutical companies have actively joined in the competition to become the distributors of denosumab and its biosimilars. The original Prolia has been sold jointly by Chong Kun Dang and Amgen since 2017. Daewoong Pharmaceutical stepped in as a relief pitcher for Celltrion's Stoboclo. Chang-Jae Lee, CEO of Daewoong Pharmaceutical, said after the copartnership agreement signing ceremony, “We expect that the agreement will leverage the strong sales and marketing capabilities of Daewoong Pharmaceutical and the outstanding biosimilar technology of Celltrion to dominate the osteoporosis treatment market. Furthermore, we will strive to reduce the risk of fractures and improve the quality of life for osteoporosis patients in Korea with CT-P41, in a prescription market worth KRW 170 billion.” Hanmi Pharmaceutical will co-market Samsung Bioepis' Obodence. Jae-Hyun Park, President of Hanmi Pharmaceutical, said, “Hanmi Pharmaceutical is a well-prepared partner with an established foothold in the musculoskeletal treatment market. Through the partnership, we will closely collaborate with our partner to create innovative results that promote mutual growth.”
- Policy
- Inspections target faulty packaging·labeling of drugs
- by Lee, Jeong-Hwan Jun 24, 2025 06:00am
- The Korean government and local governments will launch inspections targeting pharmaceutical companies with a history of recalls due to faulty packaging or labeling of medicinal products and illegal advertising of GLP-1 obesity drugs, such as Wegovy. For GLP-1 obesity drugs, to prevent misuse and abuse, inspections will focus on handling medical clinics and pharmacies to check for direct-to-consumer advertising of specialized medicines and whether information exceeding the scope of approved indications is being exaggerated or falsely advertised to the general public. Following the inspections, pharmaceutical companies, medical institutions, pharmacies, and wholesalers found to violate these terms will face measures such as administrative guidance or administrative penalties. On June 23, the Ministry of Food and Drug Safety (MFDS) announced that it would conduct planned joint inspection with local MFDS agencies and local governments to strengthen the safety management of medicinal products· biopharmaceutical products·Korean traditional medicines·quasi-drugs·medical devices. This second-quarter joint inspection of medical products will run until June 27. For medicinal products, manufacturers with a history of recalls due to packaging·labeling defects will be inspected, given the continuous occurrence of such recalls. Key inspection points include, ▲Whether the company has implemented follow-up measures and recurrence prevention plans submitted during previous drug recalls ▲Whether follow-up measures from self-inspections related to packaging·abeling processes have been carried out. If follow-up measures are not implemented or violations are confirmed during the inspection, necessary actions, including administrative penalties, will be taken. During the summer, concerns about misuse and abuse are rising due to illegal advertising of specialized medicines, including online posts featuring post-treatment reviews that describe GLP-1 obesity biopharmaceuticals as 'weight loss drugs' or promotional materials placed in patient waiting areas. To prevent the misuse and abuse of obesity treatments, MFDS will focus on handling medical clinics and pharmacies to inspect ▲Whether prescription drugs are being advertised to the general public through various advertising media ▲Whether exaggerated·false information exceeding the scope of approved indications is being released to an unspecified public. If violations are confirmed during the inspection, necessary actions such as administrative guidance or administrative penalties will be taken against the medical clinics, pharmacies, and, if necessary, wholesalers or pharmaceutical manufacturers. For Korean traditional medicines (Hanyak), rigorous quality control is required for high-value herbal ingredients, such as 'Nokyong' and 'Uhwang'. Therefore, manufacturers of these herbal ingredients and manufacturers of hanyak ingredients containing 'Uhwang,' such as 'Uhwangcheongsimwon,' will be inspected. Key inspection points will include ▲Appropriateness of quality control for raw materials and finished products ▲Appropriateness of raw material storage management and finished product manufacturing management. For quasi-drugs, to ensure a safe distribution environment for anti-snoring products used to reduce or suppress snoring noise, manufacturing (importing) companies will be inspected for ▲Performance of quality inspections for raw materials and finished products ▲Microbiological quality control ▲Appropriateness of hygiene management of manufacturing facilities. Additionally, inspections for false·exaggerated advertising will be conducted on sanitary product vendors frequently found with identical advertising violations on numerous online sales sites. If violations are confirmed in advertisements provided by manufacturing (importing) companies, those manufacturers (importers) will also be inspected. As instances of unapproved illegal medical devices being imported through direct overseas purchases or purchasing agencies continue to occur, companies frequently caught importing illegal medical devices at the import·customs clearance stage will be inspected. Key inspection points will include ▲Whether illegally imported medical devices are being illegally distributed ▲Whether return or disposal measures have been taken ▲Other violations of the Medical Devices Act. If inspections confirm violations, such as storing or distributing illegally imported medical devices without returning or disposing of them, necessary actions, including administrative penalties, will be taken. Meanwhile, to enhance the effectiveness of this planned joint inspection, the MFDS conducted pre-training on inspection methods for the '2025 Q2 Medical Products Sector Inspection Officer Training' (6.19.~20.), involving local MFDS agencies and local governments (cities·provinces, cities·counties·provinces).