- LOGIN
- MemberShip
- 2026-05-01 23:44:47
- Policy
- Gov’t works to stop the shortage of essential drugs
- by Lee, Jeong-Hwan Apr 07, 2025 05:50am
- The government will come up with improvement measures within this year to eradicate and alleviate the instability in the supply of national essential drugs, which has been occurring frequently since the COVID-19 pandemic. The government plans to begin the groundwork for overhauling the entire system, including the method for designating national essential drugs, the operation of the council of relevant government ministries and agencies, and government policy support and preferential measures for stable supply. This is done by examining the current status of the essential drug systems in major countries overseas and seeking ways to improve the stability of supply based on an analysis of the current status of Korea’s operation of the essential medicine system. On the 6th, the Ministry of Food and Drug Safety announced that it would begin research on ways to collaborate on the classification and stable supply of essential medicines. This is a response to the growing risk of health security crises and the increasing frequency of shortages of essential drugs such as cold medicines that arose as countries around the world have adopted a national drug policy centered on their own countries since the COVID-19 pandemic. Since 2017, the government has designated and managed drugs that are essential for medical sites such as disease control and radiation disaster prevention, and therefore must be supplied stably as national essential medicines. The Minister of Health and Welfare and the Minister of Food and Drug Safety designate the essential medicines in consultation with the heads of relevant central administrative agencies. However, as front-line pharmacies are constantly experiencing difficulties in obtaining essential medicines, citizens are repeatedly unable to purchase essential drugs such as cold medicine and fever reducers when they need them. The Ministry of Food and Drug Safety examines the purpose of the essential medicine system in advanced pharmaceutical countries and investigates the classification method by type, such as the nature of the product and its intended use. The plan is to look into the current state of the system, including the selection criteria, procedures, and collection of advisory opinions, and analyze the current use of policy support and preferential measures for the stable supply of essential medicines, as well as the current state of operation of organizations such as selection and advisory councils and committees. Based on this, the research team will seek direction for the advancement of Korea's national essential drug policy. As the number of national essential medicines that were first designated around government stockpiles has gradually increased to include items that require a stable supply in the private medical field, the overall goal is to find improvement measures that reflect the characteristics of each national essential medicine. Afterward, the government will prepare a plan to improve the criteria for designating national essential medicines, review the classification plan by use, and come up with a plan to reorganize the list, as well as an operational plan to maintain its suitability in the field. The research will comprehensively review the necessity of each product in the product group when designating and re-evaluating national essential medicines. In addition, it will consider the need to distinguish the system from similar systems and review the scope of designation and exclusion of national essential medicines. Ultimately, it will establish a plan for reorganizing and operating the Stable Supply Council and Sub-Council to facilitate discussions on the designation of national essential medicines and stable supply. The MFDS explained, “We will identify the government policy tools and the collaboration needs with relevant ministries and agencies that can be used to enable the council to determine and implement policies for the stable supply of essential medicines. We will also identify the improvements needed to strengthen the capacity to support the stable supply of national essential medicines, and find ways to strengthen the human and material infrastructure for this.”
- Policy
- COVID-19 Vaccine Damage Compensation Act passes NA
- by Lee, Jeong-Hwan Apr 04, 2025 05:58am
- A special law that compensates and supports patients who have suffered damage after COVID-19 vaccination was passed at the plenary session of the National Assembly on the afternoon of the 2nd. Also, a partial amendment to the Framework Act on Health and Medical Services, which includes the establishment of a Health and Medical Manforce Planning Estimation Committee under the direct control of the Minister of Health and Welfare, passed the plenary session along with the special act. The National Assembly passed the 'Special Act on Compensation for Damage Caused by COVID-19 Vaccination' with 263 in favor and 2 abstentions out of 265 members present. The Act on the Medical Manpower Planning Committee was passed at the plenary session with 247 in favor, 11 against, and 8 abstentions out of 266 members present. The Special Act expands the scope of vaccine damage compensation, such as by presuming that there is a causal relationship if the temporal correlation between COVID-19 vaccination and the disease occurrence is proven. The government has been administering vaccinations and providing state compensation in accordance with the current Infectious Disease Control and Prevention Act. However, the need for the Special Act was raised as there were indications that the damage caused by COVID-19 vaccination was not being properly compensated, due to limited causality of the damage being accepted. The Special Act, which passed the plenary session, was passed by the National Assembly Health and Welfare Committee in January with consensus from the ruling and opposition parties. The Medical Manpower Planning Committee Act, which will deliberate the number of medical school enrollees from 2027, has also passed the National Assembly, but it is unlikely to serve as a clue to resolve the conflict between the government and the medical community in Korea. The amendment proposes the establishment of the Medical Manpower Planning Committee, an independent deliberation body under the direct control of the MOHW, to deliberate on the estimation of medical personnel by job type. The committee members will consist of no more than 15 experts recommended by medical provider representative organizations, consumer representative organizations, and relevant academic societies, with the majority of the members recommended by providers, such as the Korean Medical Association (KMA), and the chairman of the committee will be elected from among the members recommended by the academic community. In addition, the revised act ensures the independence of the committee and ensures transparency by disclosing the minutes and reference materials, and the appointment of a Medical Manpower Planning center to ensure the professionalism of the planning work. When the committee estimates the size of the medical personnel required, the Health and Medical Services Policy Deliberation Committee, chaired by the Minister of MOHW, will then determine the number of medical school students.
- Policy
- First RSV vaccine Arexvy to soon be released in Korea
- by Lee, Hye-Kyung Apr 01, 2025 05:52am
- With the release date of the first RSV (respiratory syncytial virus) vaccine in Korea confirmed the MFDS has also set a fee for its national lot release approval. GSK Korea announced that it will launch the RSV-LRTD vaccine ‘Arexvy’ in May. Once the pharmaceutical company releases the vaccine, full-scale vaccination is expected to become possible from June. Arexvy is the first RSV vaccine in Korea approved by the Ministry of Food and Drug Safety in December last year for the purpose of preventing lower respiratory tract disease (LRTD) caused by RSV in adults aged 60 and over. According to industry sources on the 1st, the MFDS also set a national lot release approval fee for the first supply of RSV vaccine in Korea. According to the “Partial Amendment to the Notification of Fees for Permits for Medicines, etc.,” the MFDS has set a national lot release approval fee upon the approval of a new type of biological drug. The new fees have been set at KRW 895,000 for electronic civil services and 995,000 won for in-person and mail civil services for respiratory syncytial virus vaccines (recombinant). National Lot Release is a process in which the MFDS comprehensively evaluates the results of the MFDS's inspection and the manufacturer's manufacturing and testing results for each manufacturing lot of a biological drug to once more ensure that the quality of the biological drug is verified before it is released on the market. Last year, the government agreed on the need to consider the introduction of RSV vaccines into the NIP (National Immunization Program). In a written inquiry during the National Assembly's audit last year, the Korea Disease Control and Prevention Agency said, “Currently, there are no RSV vaccines approved in Korea,” and “We will closely monitor the situation, including the MFDS's approval, related research, and overseas trends, and consider the need to introduce it into the NIP ahead of its domestic approval through expert consultations.” According to the medical community, the number of patients hospitalized for LRTD due to RSV in Korea last year was about 11,000, which is not a small number, and the condition is even more fatal for the elderly and infants. When adults are infected with RSV, they may have no symptoms or recover after experiencing cold-like symptoms, but in the case of the elderly with weak immunity, it can lead to pneumonia, chronic obstructive pulmonary disease, and congestive heart failure. Although Arexvy is the first RSV vaccine to be commercialized in Korea, Pfizer's Abrysvo and Moderna's mRESVIA are awaiting MFDS approval.
- Policy
- KPBMA proposes 'drug shortages·API·AI new drug' budget
- by Lee, Jeong-Hwan Apr 01, 2025 05:52am
- Amid both ruling and opposition parties discussing the allocation of a supplementary budget plan, the Korean pharmaceutical industry has begun requesting a supplementary budget for two key initiatives: the "Production Support Business for Drugs with Supply Instability" and the "Direct Support Business for the Production of Active Pharmaceutical Ingredients." In particular, the industry requested increased funding to train professionals and build infrastructure for AI-driven new drug development. The industry proposes a supplementary allocation of approximately KRW 32.05 billion for this fiscal year alone. On March 31, the Korea Pharmaceutical and Bio-Pharma Manufacturers Association (KPBMA) submitted a supplementary budget request to the National Assembly, which includes the budget for stabilizing the drug supply chain and supporting AI-developed new drugs. Specifically, the Korean pharmaceutical industry is seeking KRW 900 million to support production companies managing supply instability and KRW 2 billion for direct support to companies producing active pharmaceutical ingredients. For AI-developed new drugs, the proposal outlines KRW 150 million for education and promotional activities, KRW 17 billion for establishing an automated research facility aimed at discovering innovative new compounds, and KRW 12 billion for establishing a virtual AI new drug research center. ◆Reinstating a previously reduced budget of KRW 900 million for drugs in short supply=an allocated budget of KRW 900 million for production support of pharmaceuticals of supply instability is intended to support one additional pharmaceutical company to abolish the issue of drugs in short supply. Although a large-scale pharmaceutical shortage has been resolved, the requested budget aims to prevent sporadic events of individual pharmaceutical shortages. The budget is intended to overcome shortages of essential medications, including compounding acetaminophen, general cold remedies, as well as drugs for motion sickness, constipation, hypertension, and pediatric cold medications, to prevent further disruptions in patient care. Initially, the budget for supporting supply was set at KRW 1.8 billion but was later cut by KRW 900 million. The final allocation is set as KRW 900 million. The KPBMA maintains that reinstating the reduced KRW by 900 million and potentially increasing it is necessary to add one designated pharmaceutical company and support facilities·equipment. ◆API Production Support=a KRW 2 billion budget increase has been requested to boost the domestic production of active pharmaceutical ingredients (APIs). Requests have been made to allocate a budget to improve the self-sufficiency rate of domestically produced APIs. Following the COVID-19 pandemic, the reliance on imported raw materials has risen significantly, raising concerns about potential disruptions in domestic drug production and supply. The KPBMA insists that although the government has implemented preferential pricing for drugs using domestic raw materials, API manufacturers still report that the policy’s benefits have not been fully realized. The KPBMA calls for budget allocations to support the expansion of core production facilities, maintenance of equipment, and workforce training. Furthermore, a supplementary budget of KRW 2 billion is being proposed to establish a monitoring system for the stockpiling·statistical tracking of frequently used APIs. ◆Requesting supplementary budget of an additional KRW of 29.15 billion for AI new drug discovery=A supplementary budget proposal requests an additional 150 million AI new drug discovery related to AI-based new drug development education·promotion. This budget aims to train interdisciplinary professionals for utilizing AI-based digital technology for new drug development. This budget aims to train field-ready professionals who can be immediately deployed to new drug development using AI-based digital technology. It is expected to support establishing and operating Korea’s only AI new drug development education platform (LAIDD), coupled with project-based problem-solving training. Through top-tier project mentoring from leading experts across academia, industry, and research, the program aims to nurture professionals capable of addressing complex challenges in drug discovery. Amid rapid digital transformation of the bio-health sector and accelerated progress in AI new drug development, demand for advanced IT-BT interdisciplinary talent has surged. Because this area lacks comprehensive training programs, the KPBMA has proposed addressing this issue. ◆Building AI-automated Autonomous Data Lab (ADL)=The KPBMA has proposed a supplementary budget request of KRW 12 billion to establish and deploy an AI-automated new drug research center, Autonomous Data Lab (ADL), for this year. The lab will be built to utilize AI agents with specialized expertise and proactive thinking in various new drug development fields, aiming to boost the efficiency of domestic new drug development. The total project cost is set at KRW 51 billion, to be allocated KRW 17 billion for stage 1 this year, KRW 17 billion for stage 2 in 2026, and KRW 17 billion for stage 3 in 2027. The budget reflects on the synthesis process for candidate compounds with enhanced pharmacological properties in early-stage drug development. Synthesis process is a high-cost and high-value-added area. Recent advances in AI have enabled the development of synthesis design and optimization technologies, which have further evolved into autonomous laboratory systems when combined with robotics. The proposal aims to promote the domestic adoption of these technologies. ◆A virtual AI new drug research center=The KPBMA has proposed a supplementary budget request of KRW 12 billion to establish and deploy a virtual AI new drug research center. This center will use AI agents with specialized expertise and proactive thinking in various new drug development fields, aiming to boost the efficiency of domestic new drug development. The total project cost is set at KRW 36 billion, to be allocated over three years from this year through 2027. The KPBMA proposed that while adopting AI technology is essential for enhancing Korea’s new drug development competitiveness, there remains a shortage of AI experts with the specialized knowledge required for drug research. The KPBMA states that technological advancement allows AI to conduct new drug research independently without additional staffing. Thus, establishing a virtual AI new drug research center that can be integrated throughout the entire drug development process may be needed.
- Policy
- Advanced biodrugs subject to reimb eval before approval
- by Lee, Jeong-Hwan Mar 31, 2025 05:59am
- 'Advanced biopharmaceuticals' will be added to the group of drugs that can receive drug reimbursement evaluations before receiving the government's official marketing authorization. The revision comes into effect from April 1st. On the 28th, the MOHW announced this through a notice of the product groups subject to the approval - insurance drug price evaluation linkage system. The current laws and regulations only allow the linkage between approval and evaluation of insurance drug prices for new drugs and orphan drugs in accordance with the Regulations on Approval and Review of Pharmaceutical Products, the Regulations on Approval and Review of Biological Products, and the Regulations on Approval and Review of Herbal Medicinal Preparations (Raw Drugs). The MOHW has decided to apply the approval-insurance drug price evaluation linkage system to advanced biopharmaceuticals based on the ‘Act on the Safety and Support of Advanced Regenerative Medicine and Advanced Biopharmaceuticals’ starting next month. As a result, companies with advanced biopharmaceuticals can now apply for drug reimbursement evaluations for their respective products before marketing authorization by submitting a copy of the manufacturing (import) item license as the safety and efficacy review report notified by the Ministry of Food and Safety. The approval-insurance drug price evaluation linkage system is one of the fast-track reimbursement systems for medicines implemented by the government to improve patient access to new drugs.
- Policy
- "Will maintain stability of essential·short supply drugs"
- by Lee, Jeong-Hwan Mar 28, 2025 06:39am
- The government will reportedly provide necessary administrative support for a stable supply of essential drugs and medications and improve the system so that innovative new drugs and new medical devices can quickly introduced into medical practices. Preferential pricing for national essential drugs that use domestically sourced raw materials will be implemented in the first half of this year. In contrast, the government will continuously increase the prices of drugs that are in supply instability. For innovative new drugs, specific innovation criteria will be applied during cost-effectiveness evaluations, and the policy of offering preferential pricing for drugs developed by companies with a high proportion of R&D investment will be maintained. Additionally, detailed timelines have been established for initiatives aimed at ensuring fair compensation for essential medical services and for innovating non-reimbursable and private insurance to safeguard the sustainability of the National Health Insurance budget. On May 27, the Ministry of Health and Welfare (MOHW) convened the 6th Health Insurance Policy Review Committee for 2025. It reviewed and approved this year’s implementation plan under the '2nd National Health Insurance Comprehensive Plan (2024–2028).' Maintaining stability of essential drugs·providing preferential pricing of innovative new drugs The government will continue to ensure a stable supply of essential medicines and therapeutic materials while improving the regulatory system to enable the rapid market entry of innovative new drugs and medical devices. To secure supply stability, the government will implement preferential pricing to nationally essential drugs that use domestically sourced raw materials, and drugs with supply instability will have their prices promptly increased continuously. In addition, a system for monitoring, analyzing, and addressing shortages of therapeutic materials will be established. Establishment of monitoring·analyzing materials of supply instability and response measures For innovative new drugs, cost-effectiveness evaluations will incorporate specific innovation criteria (revised on August 2024). Drugs developed by pharmaceutical companies with a high R&D investment ratio will receive preferential pricing. Innovative medical devices that have undergone an extended deferment period and rigorous clinical evaluation and have subsequently received MFDS approval will be allowed immediate market entry. The government will also expand the access to and use of National Health Insurance data for public interest research, scientific studies, and self-directed health management while supporting international cooperation through organizations such as the WHO and OECD in matters related to health insurance systems and initiatives. Enhancing the Supply of Essential Medical Care·Ensuring Fair Compensation To eliminate low-reimbursement structures and address overall imbalances in health insurance fees, more than 1,000 fee items for surgeries, procedures, and anesthesia will receive targeted increases by the first half of this year. In particular, fees in high-difficulty areas and resource-intensive, such as pediatric and emergency services, will be significantly raised, with rapid adjustments planned for over 2,000 low-reimbursement items by 2027. Furthermore, the cost survey framework will be strengthened by establishing a fee-determination system linked to conversion indices and relative value scores, developing standardized cost-calculation guidelines, and expanding the panel hospital network. Reimbursement for high-difficulty medical procedures, such as additional age-based fees for pediatric surgery, will be reinforced, and public policy fee support for maintaining maternity care infrastructure will continue. The performance of pilot projects for alternative payment systems, which offer differential reimbursement based on the quality and outcomes of care rather than volume, will be evaluated, with ongoing efforts to sustain their implementation. Closing medical care access gaps and ensuring a healthy life To provide uninterrupted healthcare, long-term care, and support services across each region's acute, recovery, and chronic phases, the government will strengthen local medical institutions and expand integrated healthcare and care support. To establish a regionally comprehensive essential healthcare system, the government will provide support to key regional hospitals—such as national university hospitals and general hospitals—through funding for faculty salaries (KRW 26 billion) and for facilities and equipment (KRW 81.5 billion), as well as low-interest loans of KRW 120 billion for any additional necessary resources. These measures aim to enhance institutional capacity, expand the infrastructure for recovery-phase healthcare institutions, and reinforce long-term care support systems (including nursing and caregiving services) in preparation for an aging society. Efforts to promote routine health management to prevent complex and chronic diseases will be strengthened, with additional support provided in high-demand areas such as mental health, women's and pediatric care, and end-of-life care. Moreover, ongoing measures will continue to strengthen the healthcare safety net to address underserved areas. The government will strengthen the healthcare safety net to close access gaps. This includes the ongoing promotion of pilot projects for primary care for individuals with disabilities and in dental care, as well as the expansion of reimbursement for treatments targeting severe and rare diseases (with 20 new items expected to be added and the reimbursement scope broadened for 10 additional items), all designed to enhance access to healthcare for vulnerable populations and alleviate their financial burdens. Enhancing the financial sustainability of National Health Insurance The National Health Insurance system will be fostered sustainably by managing medical supply by ensuring adequate hospital beds, installing·operating high-quality medical equipment, and encouraging appropriate healthcare utilization through demand management. Non-reimbursable services and indemnity insurance management will also be strengthened to promote appropriate healthcare usage. For non-reimbursable services prone to overuse, measures such as applying managed reimbursement and requiring pre-service explanations and informed consent will be enforced. Reimbursement for non-reimbursable services related to cosmetic or plastic surgery will be limited, especially if provided alongside reimbursable services. Furthermore, to rationalize the co-payment coverage under private insurance and prevent distortions in the healthcare system, non-reimbursable services will be appropriately covered. At the same time, review processes are strengthened and transparency is enhanced. Moreover, financial management transparency will be boosted by expanding the disclosure of financial indicators, such as fund operation status (March) and financial settlement status (May), publishing annual five-year financial forecasts, and enhancing the accuracy of short-term forecasts, all aimed at improving the overall management framework. The MOHW stated, "Through the '2nd National Health Insurance Comprehensive Plan (2024–2028), we plan to reach a goal for strengthening essential medical care and establishing sustainable National Health Insurance," adding, "We will also integrate these initiatives with the '2nd Healthcare Reform Implementation Plan' and other reform projects and effectively implement."
- Policy
- "Regulatory hurdle eliminated to simply refund-type RSA"
- by Lee, Jeong-Hwan Mar 28, 2025 06:37am
- The government saw eliminating regulatory hurdle as a success in exempting the efficacy·cost-effectiveness evaluation procedure during the 'third-contract termination evaluation' for pharmaceuticals on the basic refund-type RSA for over 10 years. Even drugs that have been subject to fines or other administrative sanctions can still have their prices increased through negotiations with the National Health Insurance Service (NHIS) if supply shortages disrupt patient care. For example, if a pharmaceutical company that produces plasma fractionation products can prove production cost increases based on additional research into pricing models for raw plasma and related inputs, the government covers not only past losses but also future cost escalations, a policy to raise prices accordingly. This measure is regarded as a notable case of eliminating regulatory hurdle. On March 25, the government convened the 6th Bio-Health Innovation Commission at the ARPA-H Promotion Team conference room on the 16th floor of City Tower in Jung-gu, Seoul, chaired by Director Kim Young-tae, Vice Chairman from the private sector and Director of Seoul National University Hospital, to discuss improvements to so-called "killer regulations." The Ministry of Health and Welfare (MOHW) suggested several regulatory improvement initiatives, including enhancements to the repeated re-evaluation process for risk-sharing agreements, revisions to the evaluation criteria for drug price caps, and establishment of a cost-calculation methodology for plasma fractionation products. Under revised guidelines, drugs subject to a basic refund-type RSA contract that has been used for more than 10 years can exempted from the utility and cost-effectiveness assessment during the 'third-contract termination evaluation.' The MOHW believes these changes will help resolve issues such as the devaluation of new drugs due to repeated re-evaluation under risk-sharing agreements and the consequent delays in the domestic introduction of new therapies. As of January of this year, the MOHW improved the evaluation criteria so that even drugs facing administrative sanctions, such as fines, can be considered for ceiling price adjustments to enhance patient treatment access and ensure pharmaceutical access. In cases where drug supply shortages may disrupt patient care, it has become possible to negotiate with the NHIS to raise drug prices. Furthermore, the government implemented the regulatory revision to improve plasma fractionation products' stable supply and cost-effectiveness. After a decision by the Health Insurance Policy Deliberation Committee, a change has been made to the price ceiling for plasma fractionation products that are already listed. If a manufacturer can substantiate, based on additional research into pricing models for raw plasma and similar inputs, that cost-increasing factors exist, then prices may be raised not only to cover past losses but also to account for future cost escalations. The MOHW has also relaxed the criteria for accepting efficacy evidence in high-risk advanced regenerative medicine clinical trials. In order to alleviate the burden associated with such high-risk studies of advanced regenerative medicine, the 'Guidelines for Reviewing and Preparing Advanced Regenerative Medicine Clinical Trial Plans' have been revised to establish exception criteria for safety and efficacy evidence. Through these measures, institutions conducting regenerative medicine trials will be able to choose appropriate testing methods that can adequately demonstrate safety and efficacy, even if there are variations in administration routes or methods, provided that additional supporting data justifying changes in clinical design are submitted in line with the specific characteristics of each study. Monitoring implementation of Bio-Health Training Strategies An assessment of the progress of 81 bio-health training projects, managed by nine ministries in 2024, revealed that a total of 44,800 bio-health professionals have been trained. This figure far exceeds the target of 22,100 reported to the 2nd Bio-Health Innovation Commission last year. The government analyzed that this substantial increase is due to growing interest in the bio-health sector, which has led to the establishment of new courses, increased demand for education, and an expansion of educational institutions. In detail, in one of the four major areas of bio-health human resource development, such as "industry-based school education," enhanced practical training and strengthened industry-academia linkages have produced approximately 16,400 professionals. In addition, around 20,000 individuals have been trained in the area of production and regulatory science, including workforce development (through institutions like K-NIBRT), regulatory science, and continuing education for current employees. Furthermore, to support the NEXT semiconductor leap through core research human resource development, via initiatives in AI-driven drug development, specialized graduate schools, and programs for physician-scientists, about 8,000 professionals have been trained, and projects aimed at stimulating local employment and linking job creation with start-up support have also been successfully advanced. This year, in line with the plans of the various ministries, 10 of the original 81 projects that have either been completed or will not be pursued this year will be excluded, while 7 new projects will be initiated. In total, 26,900 professionals are planned to be trained across 78 projects. Additionally, the government will focus on expanding interdisciplinary education, practical talent development programs, and initiatives for training professionals in new technology sectors within school curriculum. Furthermore, the Bio-Health Innovation Commission has reviewed research findings reflecting input from industry and academic experts on the persistent mismatch between the supply and demand of bio-health professionals, and is now discussing future strategies based on these insights. The research identified the primary factors contributing to the human resource mismatch as a lack of industry input in university curriculum, a skewed focus in human resource development that leaves a gap in understanding specialized fields such as new technologies, and a shortage of expert personnel (professors and instructors). In response, the Commission agreed on the importance of cultivating professionals tailored to corporate needs, training professionals to meet future demands in new technologies, and training globally competitive expert instructors. Going forward, the government plans to publish a '2025 Bio-Health Talent Development Business Guide' in May to boost awareness and participation among job seekers, schools, and educational institutions regarding these talent development initiatives. Going forward, the government plans to publish a '2025 Bio-Health Talent Development Business Guide' in May to boost awareness and participation among job seekers, schools, and educational institutions regarding these talent development initiatives. Director Kim stated, "At today’s meeting, we were able to discuss topics on a government-wide support plan to maximize industry capabilities by reviewing the implementation status of bio-health human resource development projects and improvements in regulatory issues," adding, "We will continue to monitor these issues regularly at the level of the Bio-Health Innovation Commission and strive to ensure that today’s discussions are reflected in government policies, aimed to achieve clear outcomes."
- Policy
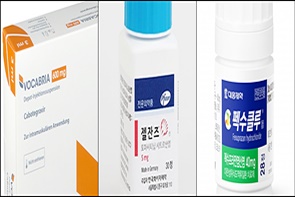
- Vocabria·Rekambys newly listed…expanded reimb for Xeljanz
- by Lee, Jeong-Hwan Mar 26, 2025 06:00am
- Product photos of Vocabria, Xeljanz, and FexuclueThe HIV treatments Vocabria (cabotegravir) and Rekambys (rilpivirine) will be newly added to the National Health Insurance reimbursement list next month. The scope of reimbursement will be expanded for Pfizer's autoimmune diseases treatment Xeljanz (tofacitinib), Novartis' Cosentyx (secukinumab), and Daewoong's erosive gastroesophageal reflux disease treatment Fexuclue (fexuprazan). On March 20, the Ministry of Health and Welfare (MOHW) announced this on the administrative notification board regarding the 'Draft for Partial Revision to the Criteria and Scope of National Health Insurance (Pharmaceuticals).' The revision will be implemented on April 1, and the MOHW will collect opinions until the 24th. For HIV therapies, in addition to the conventional cocktail regimen, PrEP and three products, including Vocabria Tab 30 mg, Vocabria injections, and Rekambys injections, have been added to the National Health Insurance reimbursement list. Reimbursement is applicable only when the drugs are administered within the approved indications. The ceiling prices for Vocabria Tab 30 mg is KRW 16,303 per tablet, for Vocabria injections is KRW 991,802 per vial, and for Rekambys injections is KRW 434,550 per vial. The National Health Insurance reimbursement criteria will be added to oral tofacitinib, such as Xeljanz, to include juvenile idiopathic arthritis. Oral tofacitinib can be used to treat children (age 2 to 17) diagnosed with juvenile idiopathic arthritis according to the ILAR criteria (2001 revision), including ▲Polyarticular arthritis that affects five or more joints ▲Extended oligoarthritis ▲Psoriatic arthritis ▲Those who discontinued treatments due to inadequate response to one or more biological agent or side effects. After 6 months of usage, an additional 6-month usage will be approved if an assessment indicates a decrease of over 30% in the number of inflammatory joints compared to the initial administration timepoint. After that, the evaluation will be carried out every 6 months, and when the assessment result in the first 6 months is maintained, consistent administration will be approved. If a patient diagnosed with juvenile idiopathic arthritis before age 17, beyond the scope, subsequently uses a medication that meets the reimbursement criteria after turning 17, reimbursement will be applied. Cosentyx received an expanded reimbursement scope for chronic severe plaque psoriasis, lowering the age threshold from 18 years or older to 6 years or older. For Fexuclue, the reimbursement criteria for the 10 mg oral formulation has been updated to include "improvement of gastric mucosal lesions in acute and chronic gastritis." Additionally, for orally administered triple combination therapy for antidiabetic medications, 'triple combination therapy containing dapagliflozin+sitagliptin+metformin' has been added to reimbursed ingredients of an SGLT‑2 inhibitor, a DPP‑4 inhibitor or a biguanide product. Furthermore, the reimbursement criteria for the ENT product Ryaltris Nasal Spray Sol (olopatadine+mometasone furoate) will be expanded due to changes in its MFDS-approved labeling. Although the previous approval limited its use to patients aged 12 and older, the age range has now been extended to 6 years and above. Consequently, reimbursement is granted only for seasonal allergic rhinitis in adults and adolescents aged 12 or older, while children aged 6 to 11 will cost 100% copay. As the inclusion of the gastrointestinal drug Irricol Tab to the listing is anticipated, the existing oral product Irribow Tab (ramosetron HCl 2.5 μg and 5 μg) has been added to the reimbursement criteria.
- Policy
- New law proposed for the cancer and rare disease fund
- by Lee, Jeong-Hwan Mar 25, 2025 05:54am
- A bill to establish a new fund for cancer and rare diseases to strengthen patient access to ultra-high-priced drugs has been proposed to the National Assembly. The fund will be raised through transfers and deposits from other funds, such as the lottery fund. On the 24th, National Assembly member Myeong-ok Seo (People Power Party), a member of the National Assembly's Health and Welfare Committee, announced that she had submitted a bill to establish the Cancer Management Fund and the Rare Disease Management Fund. The main points of the bill include ▲the establishment of a cancer management fund for the prevention and treatment of cancer (amendment to the Cancer Management Act), ▲the establishment of a rare disease fund for the prevention and treatment of rare diseases (amendment to the Rare Disease Management Act), ▲the establishment of a basis for the establishment of the fund in the National Finance Act (amendment to the National Finance Act), and ▲the establishment of grounds for the use of the lottery fund (amendment to the Lottery Tickets and Lottery Fund Act). According to the data that Rep. Seo received from the Health Insurance Review and Assessment Service, it took an average of 332 days for an anticancer drug to be listed for reimbursement in Korea from 2014 to 2024. In particular, in the case of anticancer drugs for blood cancer and lung cancer, there were cases where it took 600 to 800 days to listing. To address this, the UK operates an anticancer drug fund and a rare drug fund. Italy also has a rare disease drug fund that is financed with 5% pharmaceutical sales promotional expenses and government funds. Although there have been attempts to establish a new cancer management fund in Korea, the attempts have been frustrated by the opposition of financial authorities, with repeated calls on the need for realistic solutions to raise necessary finances. “Currently, about KRW 85 billion of the National Health Promotion Fund is being spent on cancer prevention and treatment, so it is not impossible to use it as the source of funding as it is while using a portion of the lottery proceeds, which have been increasing rapidly recently,” explained Seo. According to recent reports, lottery sales have risen by an average of KRW 450 billion per year since 2020. Next year, lottery ticket sales are expected to exceed KRW 8 trillion for the first time in history. “In a situation where it is becoming structurally difficult to apply health insurance reimbursement to treatments for patients with cancer and rare diseases, the establishment of the fund is a matter directly related to the lives of the people,” said Rep. Seo. “I hope that the bill will prompt forward-looking discussions.”
- Policy
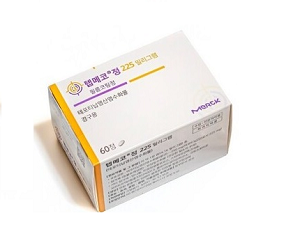
- Tepmetko, Tevimbra granted reimbursement in Korea
- by Lee, Tak-Sun Mar 24, 2025 05:52am
- Tepmetko Tab 225mg (tepotinib, Merck Korea). New anticancer drugs Tepmetko and Tevimbra will be included in the list of reimbursed drugs as of April 1 in Korea. In addition, the economic burden on the patients is expected to be significantly reduced as the co-insurance rate for abiraterone acetate drugs such as Zytiga has been reduced for the first-line treatment of castration-resistant prostate cancer. The Health Insurance Review & Assessment Service has announced a revision to the notice regarding the drugs prescribed and administered to cancer patients and has entered the opinion survey period. The effective date is April 1. According to the revision, Tepmetko 225 mg (tepotinib, Merck Korea) is granted reimbursement for patients with locally advanced or metastatic non-small cell lung cancer with MET exon 14 deletion. HIRA said, “We set the reimbursement criteria by considering factors such as the fact that the applied drug is a drug with a clear target and can provide patients with another treatment option, and is a drug deemed necessary for medical treatment.” Tepmetko is the only MET-mutated anticancer drug that is covered by the National Health Insurance in Korea. According to the diagnosis of 1,020 patients with non-small cell lung cancer in Korea, 1.9% of patients were confirmed to have MET exon 14 deletion. In Phase II clinical trial which added a confirmatory test arm, Tepmetko showed an ORR of 51.4% (95% CI, 45.8-57.1), mPFS of 11.2 months (95% CI, 9.5-13.8), and mOS of 19.6 months (95% CI, 16.2-22.9), confirming its high therapeutic effect. This drug is recommended in major textbooks and guidelines of overseas academic societies. The insurance ceiling price of Tepmetko is reportedly KRW 76,500 per dose. It has signed a refund-type and expenditure cap-type Risk Sharing Agreement (RSA) to share the drug’s financial burden. Tevimbra Inj (tislelizumab, BeiGene Korea) is indicated as a monotherapy for patients with unresectable, relapsed, locally advanced, or metastatic oesophageal squamous cell carcinoma who are unable to continue platinum-based chemotherapy or who have relapsed or progressed within 6 months after receiving prior platinum-based chemotherapy. However, reimbursement is granted for patients who have not received treatment with an immune checkpoint inhibitor such as a PD-1 inhibitor. As an immuno-oncology drug that has a PD-1 inhibitory mechanism of action, its reimbursement listing in April will allow the drug to become the first immuno-oncology drug to be covered for esophageal cancer in Korea. The maximum amount of this drug is KRW 1,206,000 per bottle. Like Tepmetko, it is applied the refund type and expenditure cap type RSA. Meanwhile, the co-insurance rate for abiraterone acetate formulations such as Zytiga will be reduced from 30% to 5% for the first-line treatment of castration-resistant prostate cancer. Abiraterone is available not only as the original Zytiga (Janssen Korea) but also as generic versions supplied by Hanmi Pharmaceutical and Ace Pharmaceutical. This reduction in the co-insurance rate is also related to the entry of generics. The National Health Insurance Service said, “The reimbursement of the ‘next-generation hormone drug (all-trans retinoic acid, ATRA)’ for metastatic hormone-sensitive prostate cancer will gradually decrease the number of patients eligible for this treatment. The price of the drug has been reduced upon the listing of generics, etc, and the coinsurance rate for ‘abiraterone acetate + prednisolone’ as a first-line treatment for castration-resistant prostate cancer will be reduced from 30% to 5%." The ATRA-class next-generation hormone drug is Janssen's Erleada. Erleada was listed for reimbursement in April 2023.