- LOGIN
- MemberShip
- 2026-04-13 16:19:35
- Company
- Lynparza was approved for pancreatic cancer/ prostate cancer
- by Oct 08, 2021 05:56am
- PARP inhibitor "Lynparza (Olaparib)" has expanded its indications to pancreatic and prostate cancer. AstraZeneca Korea announced on the 7th that it has been approved by the MFDS for Lynparza's indications for pancreatic and prostate cancer. As a result, Lynparza can be used for ▲ maintenance therapy in adult patients with gBRCA mutated pancreatic cancer who have not undergone chemotherapy for at least 16 weeks after receiving 1st platinum-based chemotherapy and ▲ for treating adult patients with BRCA mutated metastatic castration-resistant prostate cancer who have previously progressed after treatment of new hormone treatments. The approval for pancreatic cancer and prostate cancer indications was based on clinical studies of POLO3 and PROfound4, respectively. In a POLO phase 3 study evaluating the efficacy of Lynparza maintenance therapy in pancreatic cancer patients with reproductive cell BRCA (gBRCA), the median progression-free survival period (PFS) of the Lynparza administration group was 7.4 months, nearly doubling compared to placebo (3.8 months). Approval for prostate cancer indications was based on the results of sub-analysis of patients with BRCA1/2 mutation in a PROFUND phase 3 study [3] in patients with HRR mutation metastatic castration resistant prostate cancer. In this study, Lynparza reduced the risk of disease progression and death by 78% in patients with BRCA1/2 mutation among the target patients, and the median radiological progression-free survival period (rPFS) was 9.8 months, showing improved results than enzalutamideAAbiraterone of 3.0 months. In addition, the median overall survival period (OS) of the enzalutamideAAbiraterone group was 14.4 months, while the Lynparza administered group recorded 20.1 months, lowering the risk of death by 37%. "Lynparza is the first ARP inhibitor to expand indications in two carcinomas, pancreatic cancer and prostate cancer, showing clinical usefulness in two carcinomas that had high incomplete demand," said Myung Jin, executive director of the anticancer drug division. In particular, metastatic pancreatic cancer and metastatic castration-resistant prostate cancer are all the more meaningful in that treatment options have been limited, he said.
- Policy
- Pfizer vaccines from Romania not transported in official box
- by Lee, Jeong-Hwan Oct 08, 2021 05:56am
- Criticism has been raised that the 1,053,000 doses of Pfizer’s COVID-19 vaccine that the government received from Romania last month were not stored in transport boxes that were officially authorized by its manufacturer, Pfizer. Pfizer’s vaccines require ultra-low-temperature storage for quality maintenance and should be stored in Pfizer’s custom shipping box that ensures cold chain storage. However, the criticism is that the government used private boxes, which could have compromised the quality of the vaccines. On the 7th, National Assembly member Jong Hean Baek of the People Power Party raised the issue based on the data submitted by the Korea Disease Control and Prevention Agency. According to the data, while transporting Pfizer's vaccines from Romania to Korea via Incheon airport in September, the Korean government used vaccine shipment boxes provided by a separate private transport company. The background is that the Romania government had returned Pfizer’s custom shipment back to Pfizer after directly purchasing the vaccines. Baek said that using private storage boxes for sensitive vaccines that should be transported in specially designed ultra-low-temperature storage containers is the issue. Although they look like regular delivery boxes, Pfizer’s custom boxes use new plastic material or polymeric compound-coated corrugated cardboard and are strong against moisture, and have excellent thermal insulation. Baek added that Pfizer meticulously packs its vaccines in custom boxes in its Belgium facility before shipping them to Europe. This is to maintain the ultra-low temperature required to ensure the quality of the vaccine, and the box is packed in two layers and filled with refrigerants in between. Baek added that in Korea, Pfizer Korea receives and the vaccines from the custom shipment boxes, takes out the vaccines, and returns the custom boxes to Belgium for reuse. In other words, Baek’s view is that the vaccines should have been shipped after obtaining Pfizer's custom shipment boxes to maintain the vaccines’ quality and warranty. As of the 5th, 4,031 cases of adverse events were reported after being vaccinated with the Pfizer vaccine provided by Romania. The government is investing in the causality of the events and said that the vaccines’ temperature has been well maintained in the course of bringing the vaccines to Korea. Baek said, “While conducting the casualty investigation for the Pfizer vaccines from Romania, the government should analyze the cause by looking specifically at whether the initial cold chain was compromised in any way. Also, the government should transparently disclose whether the manufacturer will warrant after-sale service for Romania’s vaccine quality issue.”
- Company
- Latecomer JAKi ‘Rinvoq’ now has the most indications
- by Oct 08, 2021 05:56am
- The latecomer JAK inhibitor, Abbvie’s ‘Rinvoq,’ is seeking to overturn the market landscape by greatly expanding its indications. Whether the drug can break the two-way lead battle between Xeljanz and Olumiant, is gaining attention. According to the Ministry of Food and Drug Safety, Abbvie’s ‘Rinvoq (upadacitinib)’ received additional approval for psoriatic arthritis, ankylosing spondylitis, and atopic dermatitis indications. With the original rheumatoid arthritis indication, Abbvie’s drug is now approved for 4 indications in total. More specifically, Rinvoq is indicated for the treatment of adults and patients over the age of 12 with moderate to severe atopic dermatitis. In ankylosing spondylitis, Rinvoq can be used to treat adult patients who show inadequate responses to existing treatments. Also, in psoriatic arthritis, the drug can be used as monotherapy or combination therapy in patients who are intolerant or do not respond appropriately to disease-modifying anti-rheumatic drugs (DMARDs). In particular, the drug became the second JAK inhibitor after Olumiant to be approved for atopic dermatitis, a condition that has limited treatment options. Unlike Olumiant, which can only be used in adults, Rinvoq can also be used in adolescents aged 12 or older. With the addition of Rinvoq, biologic treatment options for atopic dermatitis have now increased to three in the market that used to be dominated by Sanofi Aventis’s ‘Dupixent.’ Abbvie has been actively targeting the atopic dermatitis treatment market, conducting a head-to-head trial between Rinvoq and Dupixent. In the Phase 3b Heads Up study, 71.0% of patients treated with Rinvoq achieved EASI 75 at week 16, which was higher than the 61.0% in the Dupixent-treated group. More recently, the patients’ eczema area was divided into 4 (head and neck, body, arm, leg) to assess the rate of EASI 75 response at week 16 in the areas. At EASI 76 at week 1 in all 4 areas was higher for Rinvoq, and lasted so until week 16. In other words, the study indicated that Rinvoq can relieve symptoms faster than Dupixent, regardless of the affected area. Rinvoq is a latecomer JAK inhibitor that was introduced in Korea in June last year. This is 2.6 years later than Olumiant and 6 years later than Xeljanz. During this period, Xeljanz had overtaken the market by expanding the area to rheumatoid arthritis, ulcerative colitis, psoriatic arthritis, and releasing an extended-release formulation. Then, Olumiant started to show strength in rheumatoid arthritis and divided the JAK inhibitor market into a two-way race. Also, Olumiant became the first to receive approval for atopic dermatitis, pioneering a new market. According to the market research institution IQVIA, Xeljanz enjoyed the sole lead old 14.7 billion won in sales in 2019. Its sales in 2020 were 16.2 billion won. Olumiant only sold 2.2 billion in 2019, but its sales rose greatly to 9 billion last year. In the first half of this year, Xeljanz (including the XR formulation) sold 7.8 billion, and Olumiant sold 5.6 billion, greatly narrowing the gap. Rinvoq, which started to record sales at the end of last year, sold 600 million won in the first half of this year. Its sales have been rising with the rapid reimbursement, however, the degree of growth is yet too small to catch up with Xeljanz nor Olumiant. However, Rinvoq suddenly increased its indications to 4 and change the future landscape. With the added indications, Rinvoq now owns the most amount of indications among JAK inhibitors. Pfizer plans to approach the atopic dermatitis market with a different JAK inhibitor other than Xeljanz. Unlike Xeljanz, which mainly inhibits JAK3, ‘Cibinqo (abrocitinib)’ mainly targets JAK1. Pfizer is known to have applied for Cibinqo’s approval and is being reviewed. However, the safety concern that remains in all JAK inhibitors is the issue. Based on the post-marketing safety study of Xeljanz, the US FDA concluded that it may increase the risk of severe heart disease such as heart attacks, etc. The FDA did not limit the boxed warning requirement to just Xeljanz, but expanded the alert to all JAK inhibitors and requested all three products to contain black box warnings. If this safety concern is not resolved, and the scope of treatment becomes restricted, this may affect and shrink the entire JAK inhibitor market.
- Policy
- Expanding the submission of opinions reflecting permission
- by Lee, Tak-Sun Oct 07, 2021 05:54am
- Opportunities for submitting opinions from industries will be expanded when reflecting permits based on the results of the reexamination. A pre-announcement procedure is added to the previous opinion inquiry. The MFDS announced that since the 27th of last month, the procedure for reflecting permits based on the results of the reexamination has been improved and applied. There was a 14-day period of submitting opinions through "Inquiry on Opinions on Permit Change" before reflecting the results of the retrial in the permission. An opportunity to submit opinions will be given one more time. An official from the MFDS said, "It is expected that opportunities for the industry to submit opinions will expand through a pre-announcement period." A total of 28 days of opinion submission period will be given. At the end of the pre-announcement period, a change order will be issued as before. After the change order, the changes in permission matters within three months must be reflected in the product insert. In this improvement plan, in order to distinguish between PMS survey results and general post-marketing adverse effect analysis evaluation, it will be divided into domestic post-marketing adverse effect analysis results and domestic post-marketing adverse effect analysis evaluation results. The results of the analysis and evaluation of the adverse effect after domestic marketing are the same as the existing re-examination method. An official from the MFDS explained, "We distinguish between the results of the adverse effect analysis collected in the post-market survey and the results of the adverse effect analysis reported after the market and reflect them in the permit, but we revised the phrase to clarify the analysis results." The improvement of the method of writing the improvement plan will be applied from the change of permission after the 27th of last month, and the pre-announcement period will be given from the items for re-examination received after the same day.
- Policy
- ’With Covid’ scheme unclear with over 5,000 cases expected
- by Lee, Jeong-Hwan Oct 07, 2021 05:53am
- Concerns have been raised that Korea will be unable to adopt the ‘With Corona’ scheme due to the public’s distrust in vaccinations, the government’s non-acceptance of casualties of adverse reactions from vaccines, and the surge in daily COVID-19 cases, etc. Based on the mathematical model that took into account current incidence, the transmission of COVID-19, and the vaccination rate, the authorities expect that the number of new COVID-19 cases may increase on average up to 5,000 cases per day if the fourth wave of the COVID-19 pandemic persists. The National Assembly member Jong Hean Baek of the People Power Party announced the above results on the 6th based on the data submitted by the Korea Disease Control and Prevention Agency. According to KDCA data, if the current incidence, transmission, and vaccination rate are applied to a mathematical model, around 5,000 new COVID-19 cases are expected to arise per day. As for the reason, Baek pointed to the public distrust in vaccines despite inoculation being required for the living ‘With Corona’ scheme, the government’s non-acceptance of casualties of adverse reactions from vaccinations, and the lack of responsibility of the nation in taking care of those who suffer from misinoculations or side effects pre- and post-vaccination. The actual recognition rate of vaccine’s side effects was 53.4% in general patients and a mere 0.3% in deaths. More specifically, KDCA’s immunization surveillance investigation team’s assessment report showed that causality of 1,764 in 3,305 cases, or 53.4% of the cases were recognized. However, among the 678 cases of deaths after vaccination, the causality of only 2 was recognized. Also, 2,014 cases of vaccine misinoculations arose due to the government's neglect, but none were compensated for the damages. Baek said, “To live ‘With Corona’, we must first address the public’s distrust in vaccinations and prepare post-management measures. The citizens are anxious and disappointed by the government’s irresponsible response to the deaths from vaccinations.” He added, “I do not understand why the government is irresponsible in dealing with this life-threatening issue for administrative convenience. The government needs to show responsibility so that the people can trust the authorities and receive their vaccinations.”
- Policy
- Nexviazyme has been applied for domestic permission
- by Lee, Tak-Sun Oct 07, 2021 05:53am
- Sanofi plans to release a new Pompe's disease treatment in Korea. It is known that Nexviazyme, which was approved by the U.S. FDA in August, recently applied for permission from the MFDS. According to the MFDS on the 1st, Sanofi Aventis Korea submitted a report on the results of clinical trials by Nexviazyme (Avalglucosidase alfa-ngpt) and applied for permission. This drug is a Pompe's disase treatment. Pompe's disease is a genetic disorder in which respiratory failure and myocardial disease appear due to muscle strength loss and muscle atrophy. It is a rare disease that is reported to be about one person per 40,000 people worldwide, and it is known that there are about 1,300 patients in Korea. Pompe's disease is divided into IOPD, a fast-onset infant disease in infancy, and LOPD, a late-onset disease that develops at all ages and gradually weakens muscles. As muscles are damaged, heart failure, respiratory failure, exercise disorders, and sleep disorders also occur. The disease administers drugs targeting M6P receptors that allow GAA enzymes to migrate to intracellular lysosomes to treat GAA gene abnormalities. Existing treatments include Sanofi's Myozyme. Nexviazyme, which applied for permission this time, reportedly increased the M6P content by about 15 times compared to Myozyme. Nexviazyme was approved by the U.S. FDA in August. The MFDS designated the drug as a rare drug for patients to use before it was officially approved in June. If designated as a rare drug, the drug can be purchased through the KODC.
- Policy
- Introduction of pre-registration is difficult
- by Kim, Jung-Ju Oct 07, 2021 05:53am
- The government said it is difficult due to concerns over weakening NHIS' drug price negotiation power, while various fields are proposing the introduction of a system that is first registered and evaluated later for access to treatments for severe rare and intractable diseases. Regarding referring to Korean drug prices such as China, the government also said it is actively responding by expanding RSA to prepare for "China risks" such as "Korea Passing." The MOHW recently submitted the "2020 National Assembly Processing Results Report on the Requirements for Correcting and Processing the National Assembly" with such contents. First of all, the National Assembly previously demanded the MOHW to consider introducing a "pre-registration and post-evaluation system" to access treatments for severe rare and intractable diseases. The MOHW said that it is difficult to operate a realistic system for post-registration evaluation. The MOHW replied, "It is expected that it will be difficult to operate a realistic system and manage reasonable drug spending, so careful review is needed." This is because it is difficult to adjust drug prices if pharmaceutical companies do not accept the evaluation results after being registered, and the NHIS' drug price negotiation power may weaken, causing problems. They would expand RSA on countermeasures against so-called "Korea Passing" concerns due to China's drug price system. Overseas countries such as China are deciding their own drug prices by referring to Korean drug prices. Due to the current strict drug price system in Korea, it can lead to " In response, the MOHW replied, "We have introduced RSA since 2014 to respond to delays in insurance registration due to reference to foreign drug prices," adding, "We have been expanding the applicable drugs since October 2020." Regarding the demand to raise funds through the National Health Promotion Fund and the primary benefit of the immuno-cancer drug Keytruda, he replied in July that the HIRA is in the process of expanding benefits of Keytruda and Tecentriq. However, the MOHW said that the National Health Promotion Fund needs to be carefully reviewed by comprehensively considering the target, scope, and allocation of required resources within the scope of support.
- Company
- Roche and Lilly to launch RET targeted cancer drugs in Korea
- by Eo, Yun-Ho Oct 06, 2021 06:06am
- Two types of RET targeted anticancer therapies are concurrently seeking domestic entry. According to industry sources, Roche Korea's Gavreto (pralsetinib)’ and Lilly Korea ‘Retevmo (selpercatinib)’ are under review for domestic approval. Both are anticancer drugs that target RET (Rearranged during transfection) gene fusions. The drugs not only inhibit primary RET fusions and mutations but also secondary RET mutations that cause resistance to treatment, and are receiving much attention on whether they will be able to address unmet needs in various types of cancer. Retevmo became the first to receive global approval by a few months. Retevmo received marketing authorization from the US FDA in May last year, and Gavreto in September. Retevmo was approved for the Non-small Cell Lung Cancer (NSCLC) and thyroid cancer indications, whereas Gavreto was first approved as a lung cancer treatment, then approved for the thyroid cancer indication in December of the same year. Although the drugs were initially approved for lung cancer and thyroid cancer indications, the companies are expected to actively pursue more indications for RET inhibitors in the future. RET gene fusions are present at a low frequency in colorectal cancer, breast cancer, pancreatic cancer, and EGFR-positive NSCLC. Like NTRK (Neurotrophic tyrosine receptor kinase) targeted therapies like ‘Rozlytrek (entrectinib)’ etc., RET inhibitors have the potential to be used as tumor-agnostic, personalized treatment. Meanwhile, Retevmo’s efficacy was verified through the Phase I/II LIBRETTO-001 study. Retevmo’s overall response rate (ORR) was 64% in 105 RET-gene positive patients with small cell lung cancer who have previously received platinum-based chemotherapy. Also, 81% of the patients who showed treatment response showed a response for at least 6 months. Also, Retevmo recorded a high ORR of 84% in 39 RET-gene positive, treatment-naive patients. In 143 medullary thyroid cancer patients that have previous treatment experience with ‘cabozantinib’ or ‘vandetanib,’ etc., the ORR was 69%. And 76% of the patients maintained treatment response for at least 6 months. In other RET-gene positive thyroid cancer, the ORR of patients with radioactive iodine-refractory (RAI) was 79%, 87% of which showed continued treatment response for over 6 months. Gaverto was initially approved as a lung cancer treatment based on the ARROW study. In the study, Gavreto showed an ORR of 57 % and a complete response (CR) rate of 5.7 % in NSCLC patients that were previously treated with platinum-based chemotherapy (87 patients). Also, ORR was 70% and CR 11% in newly treated patients (27 patients).
- Company
- Evrysdi (PO) is differentiated due to its low price
- by Oct 06, 2021 06:06am
- Roche's "Evrysdi (Risdiplam)," the second treatment for spinal muscular atrophy (SMA) in Korea, signaled its launch after about a year of approval. With the differentiation of the only PO drug and low prices, fierce competition in the SMA market was predicted. "Evrysdi is the first oral drug among SMA treatments and is applicable to patients with difficulty in treating SMA, and has confirmed its effectiveness and safety in patients of a wide range of ages and types," Roche Korea stressed at an Evrysdi meeting on the 5th. Evrysdi (Risdiplam) is a prescription medicine used to treat spinal muscular atrophy (SMA) in adults and children 2 months of age and older. Spinal muscular atrophy (SMA) is a rare genetic disease in which the SMN1 gene is inherently deficient or mutated, resulting in gradually shrinking muscles. It is known that about one per 10,000 newborns worldwide, and about 30 patients (based on 300,000 newborns) occur every year in Korea. Three new drugs were released in Korea in three years, starting with Biogen's Spinraza in SMA diseases, which had no cure so far. Evrysdi is the second new drug approved in November last year and is the only PO drug. It has been almost a year since the license was granted, but it has not yet been released on the market. As a result, it was delayed than Novartis' Zolgensma, which was belatedly approved. Novartis applied for Zolgensma's benefit in June. Evrysdi began the registration process only in July, eight months after the permit. Roche is expected to officially release Evriesdi after completing the registration process. It is expected to be reimbursed well as Spinraza, which is already more expensive, has been reimbursed. Evriesdi is also known to be likely to follow RSA ( the Expenditure Cap) applied to Spinraza. Lee Seung-hoon, director of Roche Korea, said, "We are currently trying to get reimbursed within the same range as Spinraza." He explained, "I think access to insurance is more important than anything else because receiving rapid treatment for patients has a great impact on the prognosis." Roche explained that it is the only PO drug and that the relatively low cost is Evrydi's strength. Spinraza prices exceed 90 million won, and Zolgensma, which is negotiating benefits, is a "one-shot" treatment that costs more than 2 billion won per time. Evrysdi's final drug price has not been determined, but it is much lower than the prices of Zolgensma and Spinraza. In particular, it is expected to be cheaper for infants and toddlers. Jung-hyun Chang of Roche Korea said, "Unlike other products, Evrysdi's dosage is determined by age and weight, so we expect infants and toddlers under the age of 2 to be treated at a much lower price than adults, greatly reducing the burden of drug costs." Evriesdi can be self-administered at home, which can reduce the socioeconomic burden, he added. Attention is also being paid to which drugs medical staff and patients will choose. Lee Yoon-jung, a professor of pediatrics at Kyungpook National University, emphasized "patient accessibility." Fast diagnosis and treatment are of paramount importance because SMA's symptoms deteriorate rapidly within a short period of time and the slower the treatment, the less effective it is. Professor Lee said, "No matter how active treatment is, the disability often remains no matter how active the treatment is," adding, "As diagnosis and initial administration are very important, we need to consider what drugs can speed up the initial administration as much as possible."
- Policy
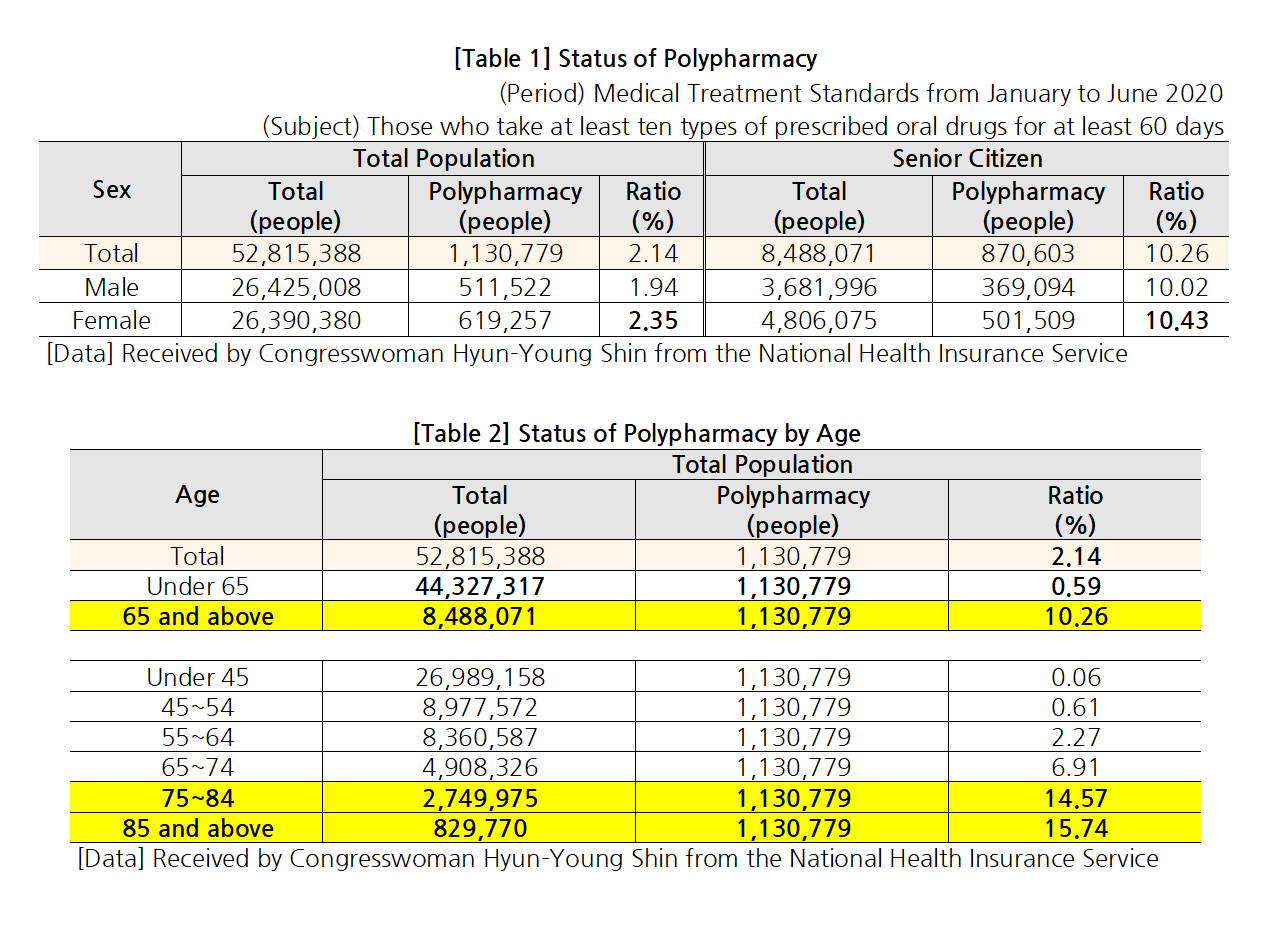
- “Elderly polypharmacy rate Korea 70% vs OECD countries 48%"
- by Lee, Jeong-Hwan Oct 06, 2021 06:06am
- Statistics showed that a high rate - 70.2% - of older adults (aged 75 or over) in Korea chronically take ‘more than 5 drugs for over 3 months.’ The average of 7 OECD countries other than Korea that submitted the same data was only 48%. In other words, concerns over the current polypharmacy status of elderly patients in Korea have been reaffirmed. On the 4th, NA member Hyung-young Shin of the Democratic Party of Korea announced the results above after analyzing OECD data. The medication rate of elderly patients in Korea has been globally high. According to the OECD’s data on the ‘rate of patients aged 75 or over who chronically take 5 or more drugs for over 3 months,’ the average of the 7 countries that submitted the data was 48.3%, compared to the much higher rate of 70.2% in Korea. However, Using an unnecessarily high amount of drugs in older adults can have a negative impact on their health. According to the National Health Insurance Service Ilsan Hospital's report, patients aged 65 or older who take 5 or more drugs have an 18% increased risk of hospitalization and 25% increased risk of death than those who take 4 or fewer drugs. According to the data that Shin received from the NHIS, 2.14% of the total population (1.13 million) in Korea takes 10 or more drugs. Among those, the elderly accounted for the largest proportion with 10.26%, and the polypharmacy rate was higher in women (2.35%) than men. The rate and number of patients using multiple drugs also increased with age. The polypharmacy rate was 10.26% for people over 65 but 15.74% for people over 85. By insurance premium quintile, polypharmacy rate was higher (12.52%) in medical aid beneficiaries than NHI policyholders. The polypharmacy rate in elderly medical aid beneficiaries was 22.57%, which roughly translates to one in 4 to 5 patients. Also, the proportion of the elderly was highest in the 1st and 10th insurance premium quintile (9.88%). Furthermore, the polypharmacy rate was higher in patients with underlying diseases such as diabetes, heart disease, cerebrovascular disease, asthma/COPD, chronic renal failure, pulmonary tuberculosis, etc. By population, the polypharmacy rate was highest in patients with chronic renal failure (18.38%), heart disease, (15.36%), cerebrovascular disease (13.86%). The order was the same in elderly patients but at a higher rate - chronic renal failure (23.80%), heart disease (20.97%), cerebrovascular disease (18.31%). Shin said, “As the rate of patients who take 10 or more drugs increases with age, we need to review whether the patients are taking unnecessary drugs due to over-prescription or redundant prescriptions. In other words, institutional support on the appropriate use of drugs is needed, and a system that assigns primary care physicians for the elderly should be introduced to manage the elderly’s use of multiple drugs, as well as provide customized support on the appropriate use of healthcare.”