- LOGIN
- MemberShip
- 2026-04-16 08:12:13
- Company
- Korean companies take drug patent strategy to next level
- by Kim, Jin-Gu Sep 22, 2020 06:26am
- The South Korean pharmaceutical companies are evolving their patent strategies. In the first half of the year, the companies set the historic record of registering the most number of patents. Apparently, their number of patents is on par with multinational pharmaceutical companies’. According to Ministry of Food and Drug Safety (MFDS) and Korean Intellectual Property Office (KIPO) on Sept. 17, total 134 pharmaceutical and bio related patents were newly registered in the first six months of the year. And 67 patents were applied by Korea companies. In other words, the multinational companies had 67 patents applied. For the first time in the history, the Korean companies had the number of registered patents even with the multinational companies. Number of patent registration in half a year by Korean companies (Unit: cases) Source: MFDS and KIPO So far, patenting was mostly owned by multinational pharmaceutical companies. From 2014 through the first half of 2020, total 1,331 patents were registered, but only the 26 percent, or 341 patents, are registered by the Korean companies. As the multinational companies had more originals, naturally they had more patents. But as more Korean companies have succeeded in developing new drugs recently, their number of registered patents has increased as well. But because there are more Korean companies disputing over patents, the Korean companies with original drugs feel the need to prepare for patent defense strategy. As for this year, Chong Kun Dang, SK Chemicals, Boryung Pharmaceutical and LG Chem led the wave of patent registration. Especially, Chong Kun Dang registered total 15 patents associated with Telminuvo (4), Certirobel (4), Raparobell (3), Eso Duo (3), and Tenofobell (1). SK Chemicals had total 13 patents for Renexin (3), Qudexy (4) and Ongentis (6). Boryung Pharmaceutical had 10 patents with Dukaro, Jeil Pharmaceutical had eight patents with Lonsurf, and LG Chem had five patents each for Zemiro and Zelief Extended Release. Sources also confirmed Kolon Pharma registered three patents, Daewon Pharmaceutical, GL Pharma, Korea United Pharm, Halim Pharm have registered two patents, respectively, and Daewoong Pharmaceutical, GL Pharm Tech, Taejoon Pharm and Hanmi Pharmaceutical have registered one patent each. The industry experts say they have to see the statistics for the latter half of the year is to name such surge in patent registration by Korean companies as either a temporary phenomenon or a new trend. Nevertheless, the number of registered patents for last three months from July through September shows the Korean companies have registered 17 patents (40 percent) out of 42 and multinational companies registered 25 patents (60 percent). Compared to previous years, the ratio of Korean companies’ patents has gone up. An insider from the Korean pharmaceutical industry noted, “Lately, Korean companies are more aggressively registering patents,” and “they are expanding the patent scope by registering several patents for one pharmaceutical substance.” Ratio of patent registration by Korean companies vs. multinational companies (Unit: %) Source: MFDS and KIPO The total number of new patents in half a year hit the historic high in the first half of 2020 since 2014, due to the boost from the Korean companies. The movement to register Korean patents registration took off from 2012, when the KORUS FTA was signed. For two years from 2012 through 2013, the exploding number of patents applications was submitted. In year 2012 and 2013, 563 patents and 868 patents were registered, respectively. Experts say the patents left unregistered until the KORUS FTA were all registered in the two years of time. Since 2014, the number of patent registration has been around 200 per year. From 2014 through 2019, 181 patents, 234 patents, 223 patents, 223 patents, 126 patents and 210 patents were registered, receptively. In the first six months of this year, total 134 patents were registered. It was the highest number of patents registered in six months time. The pharmaceutical patent experts explain the Korean companies have become assertive with patent registration as a defense mechanism against generic companies’ patent challenges. Number of patent registration by half a year (Unit: cases) Source: MFDS and KIPO
- Company
- Sales in the outpatient prescription drug market are active
- by Chon, Seung-Hyun Sep 21, 2020 06:13am
- It has been shown that the outpatient prescription drug market has not been hit even with the recent spread of COVID-19) patients. Even though sales activities contracted due to level 2 social distancing, the total prescription drug volume recorded a growth trend last month. According to UBIST, a drug research institute on the 20th, the amount of outpatient prescriptions last month was ₩1.2491 trillion, an increase of 3.2% from the same period last year. There is no significant difference, compared to the amount of prescriptions that rose 4.4% from the previous year in August last year. This means that the conventional prescription drug market has continued to grow. Monthly outpatient prescription amount trend (Unit: ₩100 million, Source: UBIST) Last month, social distancing was strengthened due to the spread of COVID-19. Since 103 new cases were recorded on the 14th of last month, the spread of new infections showed a trend. At the end of last month, the number of new confirmed cases expanded to 400. From the 23rd of last month, the government raised social distancing to the second stage. Although external activities declined due to reinforced social distancing, the prescription drug market was not affected. After the second stage of social distancing, pharmaceutical companies have been working from home. While the number of salespeople's visits to medical institutions has been minimized, normal business activities have contracted. This year, the prescription drug market has been showing ups and downs. In January, prescriptions decreased by 4.4% from the previous year, but in February they increased by 13.0%. As the Lunar New Year holidays this year was pulled to January, which is earlier than the previous year, it is analyzed that the increase and decrease rate of prescriptions in January and February showed a deviation according to the increase or decrease of the number of business days.. Prescription performance in April and May decreased by 8.7% and 9.4% from the previous year, respectively. The industry believes that the slowdown in the prescription drug market in April and May was the impact of COVID-19. It is analyzed that a considerable prescription gap occurred in the second quarter as patients with chronic illnesses, who are reluctant to visit medical institutions, were prescribed in large quantities of necessary medicines. In fact, when the number of COVID-19 patients surged, it is known that the number of cases receiving prescriptions for 3 to 6 months in advance increased significantly. Monthly outpatient prescription amount increase/decrease from the same period last year (Unit: %, Source: UBIST) In fact, from June, the prescription drug market has shown a recovery trend. In June, the prescription size was ₩1.2667 trillion, up 11.7% from the same period last year, and in July, it recorded a growth rate of 1.8%. As the use of medicines continues to increase due to the increase of the elderly population and the number of chronic diseases, the entire industry will not shrink due to short-term issues such as infectious diseases. Unlike the tourism and cultural industries, which fell into extreme crises after COVID-19 outbreak, the pharmaceutical industry is affected by the demand of patients rather than the external environment, so it is difficult to lead to a sudden downturn. Until last month, the total accumulated prescription amount was ₩9,8876 trillion, an increase of 0.6% from the same period last year. It was estimated that the prescription drug market was superior to other industries, although it was slower than the cumulative prescription growth rate of 6.7% until August last year.
- Opinion
- [Reporter’s Note] Will KRPIA appoint a Korean chairman?
- by Eo, Yun-Ho Sep 21, 2020 06:12am
- While a few multinational pharmaceutical companies have confirmed their foreign executives would be leaving the position, the Korean Research-based Pharmaceutical Industry Association’s (KRPIA) board of director (BOD) is also expected to undergo some changes as well. The two foreign executives bidding farewell are MSD Korea’s CEO Avi BenShoshan and GSK Korea’s Vice President and General Manager Julien Samson. Currently, CEO Avi BenShoshan is the Chairman of KRPIA and VP and General Manager Julien Samson is a Vice-chair. The focus of the industry lies on the possibility of expanding the Korean executives among the KRPIA BODs. Considering Chairman Avi BenShoshan’s term is to last until the end of the year, KRPIA would likely to appoint an acting-chairman. Except for Vice-chair Julien Samson, Kay Bae (Sanofi-Aventis Korea) and Oh Dong-wook (Pfizer Korea) are the other two vice-chairs, who could be appointed to take the acting-chairman position. With the two foreign executives leaving, the existing BOD roster with seven Koreans and six foreigners may increase the ratio of Korean executives. The members of the organization have been raising their voices that appointing a Korean as the chairman would make more sense, because the organization’s integral role is to communicate with drug pricing-related government bodies as the multinational companies mainly supply new drugs to the Korean market. Moreover, the government affairs-related skills have been highly sought after lately to receive reimbursement on high-cost drugs the multinational companies’ recent pipelines are more focused on. In fact, Ministry of Health and Welfare’s (MOHW) drug pricing and listing divisions tend to prefer Korean executives. Chairman Avi BenShoshan was actually a first foreign executive appointed as the chairman, after seven years since 2011 the position was served by Korean executives including Lee Dong-soo (former President of Pfizer Korea), Kim Jin-ho (former GM of GSK Korea), and Kim Oak-yeon (former CEO of Janssen Korea). Having Korean executives in BODs does not always mean better. But, surely for KRPIA, this is the critical time to have more Koreans to lead the organization. Going beyond the one-dimensional goal of receiving satisfying level of drug pricing, hopefully KRPIA would appoint BODs, who can rationally and cleverly find a middle ground with the Korean health authority to ultimately ‘improve patient’s treatment access.’
- Policy
- Acromegaly and Huntington’s disease drugs win approval
- by Lee, Tak-Sun Sep 21, 2020 06:12am
- The South Korean health authority granted licenses for Pfizer Pharmaceutical Korea and Teva Handok, respectively. Pfizer’s acromegaly treatment and Teva Handok’s Huntington’s disease treatment would be now accessible to the patients in need of treatment. The Ministry of Food and Drug Safety (MFDS) granted approval on Somavert (pegvisomant) by Pfizer Pharmaceutical Korea and Austedo (deutetrabenazine) by Teva Handok as of Sept. 16. Somavert injection is an incrementally modified drug with evidence submitted, whereas Austedo tablet is a novel drug. Somavert is used to treat adults with acromegaly and intolerance or resistance in normalizing insulin-like growth factor 1 (IGF-1) with somatostatin analogues, and who respond inadequately to surgery or radiation. Acromegaly is a hormonal disorder that enlarges hands, feet, forehead, and jaw as excessive level of growth hormone is excreted even after the growth plates have been closed. About 60 out of one million people are diagnosed with the rare diseases, and usually the diagnosis is made from forties. The main cause of the disorder is excess growth hormone produced by benign tumor in the pituitary gland. However, relevant studies have not found the cause of the tumor, yet. The recommended treatment is to remove the tumor in the pituitary gland with a surgery, but when the tumor is too big and cannot be removed completely, the patient has to receive radiation therapy and growth hormone-inhibiting medication simultaneously. But many of the patients do not respond well to growth hormone-inhibiting drug, so the demand for more fundamental treatment is high. The newly approved Somavert is a recombinant analogue of the human growth hormone, which blocks the growth hormone receptor and impedes signal transduction. During a clinical trial conducted with 112 acromegaly patients, Somavert demonstrated statistically significant improvement in overall symptoms and relevant indicators than the placebo. And approximately 93 percent of trial participants reached normalized IGH-1 level for 42.6 weeks. Also approved on the same day, Austedo is a new drug used to treat symptoms of Huntington’s chorea, a kind of inherited diseases. The Huntington’s disease is a rare progressive brain disorder caused by a mutated gene on chromosome 4. According to Health Insurance Review and Assessment (HIRA), there were 286 patients with Huntington’s disease reported in South Korea as of 2018. The most common symptom, also known as chorea, is abnormal motor behavior like sudden involuntary muscle spasms. Approximately 90 percent of the patients with Huntington’s disease show the symptom. Austedo conducted a clinical trial with 90 Huntington’s disease patients having chorea, but who can walk by themselves. The trial compared the drug against placebo and confirmed the drug’s statistically significant effect with the evaluation indicator score. The patient group taking Austedo reached 4.4 points, while the placebo group marked 1.9 points.
- Policy
- Cancer Fund Act is promoted
- by Lee, Jeong-Hwan Sep 21, 2020 06:12am
- A bill is being promoted to strengthen the guarantee of expensive anticancer drugs for cancer patients by establishing a cancer management fund. The goal is to improve the reality that patients are deprived of treatment opportunities due to delays in health insurance payments for innovative treatments such as immune anticancer drugs due to limitations in health insurance finances. On the 17th, Lee Jong Seong, a member of People Power Party, announced that he had proposed a partial amendment to the Cancer Control Act. The bill was submitted to the National Assembly on the 16th. He pointed out that if the national security of innovative anticancer drugs is not properly secured, treatment opportunities and options vary depending on the economic level. His opinion is that the priority of health insurance for anticancer drugs with high social demand should be raised, and separate financial support should be made if financial resources are insufficient. Accordingly, he proposed a bill to strengthen support for cancer patients and establish a cancer management fund as a source of funding for cancer screening, medical expenses, and cancer research and treatment projects. He said, "Protecting the lives and safety of the people is the basic duty of the government, and having a therapeutic safety net that meets the level of the Korean economy should be the core oriented value of health policy."
- Company
- Lilly's Olumiant, is clinically approved for COVID-19
- by Kim, Jin-Gu Sep 18, 2020 06:30am
- Olumiant Eli Lilly's autoimmune disease treatment 'Olumiant (Baricitinib)' has begun phase III clinical trial targeting COVID-19 patients in Korea. It is the second clinical trial in Korea related to COVID-19 after Remdesivir by Gilead Science. On the 7th, the MFDS approved a phase III clinical trial plan of Baricitinib (LY3009104) in Lilly Korea for COVID-19 patients. This clinical trial is part of Lilly's global phase III clinical trial. It will be held from this month to next October, and the size of the clinical trial is 600 sujects. Among them, 15 domestic patients are expected to receive medication at Ajou University Hospital. Lilly has already started clinical trial in the US and Europe since last June. According to the company, Olumiant is expected to be able to reduce cytokine storms, one of the symptoms of infection complications caused by COVID-19. In clinical trials previously conducted in Italy, etc., positive results were found in improving the symptoms of COVID-19 patients. The most representative study was conducted in 7 hospitals in Italy from February 20 to March 15 this year. It was published in 'The Journal of Infection' on the 23rd of last month. The researchers administered Olumiant and Kaletra (Lopinavir + Ritonavir) together to 113 patients for 2 weeks in 192 COVID-19 patients, and only Kaletra to the remaining 78 patients. Olumiant Kaletra Lopinavir / Ritonavir As a result, the mortality rate, the intensive care unit admission rate, the discharge rate, and COVID-19 virus detection rate were all positive in the group administered with Olumiant. As for the mortality rate, there were no dead patients in the Olumiant co-administration group, while 5 (6.4%) died in the control group. There was also no intensive care unit admission rate in the co-administration group, whereas 14 patients (17.9%) in the control group occurred. As for the discharge rate, 88 out of 113 patients in the co-administration group were discharged, recording 77.9%, whereas in the control group, 10 out of 78 patients were discharged, which was 12.8%. Two weeks after treatment, the detection rate of COVID-19 was 12.5% in the co-administered group and 40% in the control group. Results of a study on the therapeutic effect of COVID-19 in the combination of Olumiant + Kaletra (Data The Journal of infection) Remdesivir is the only treatment for COVID-19 officially recognized in Korea so far. It is expected to be the second treatment if Olumiant finishes phase III clinical trial and obtains approval as a treatment for COVID-19. Remdesivir In the case of Remdesivir, the effect of shortening the hospital stay has been proven in clinical trials. However, it failed to prove the improvement in mortality. If Olumiant proves an improvement in mortality in this clinical trial, it is expected to receive more attention than remdesivir.
- Opinion
- [Reporter’s Note] “Loonshots” in new drug development
- by An, Kyung-Jin Sep 18, 2020 06:30am
- I finally caught up to read the bestselling book, “Loonshots” that Bill Gates praised as ‘a book to carry around in your bag to read.’ The book is written by Safi Bahcall, who received a doctorate degree in physics from Stanford University and co-founded ‘Synta Pharmaceuticals.’ The author defines “Loonshots” as “a neglected project, widely dismissed and its champion written off as unhinged.” To use such ‘crazy idea’ in a formula of corporate innovation and growth, the physicist applies theories of physics like ‘phase transition,’ ‘phase separation,’ and ‘dynamic equilibrium.’ After creating a biotech company and working there for a decade as a CEO, he figured the scientific principles are not so different from corporate management. The book explains of the distinction between a ‘start-up’ and a ‘conglomerate’ with the basic concept of physics—more is different. As a scale of an organization can completely change its nature, a good design of an organization can only maintain the dynamic equilibrium, which enables the organization to keep a balance and internally communicate. But unfortunately, an organization tends to dismiss the loonshots, when they grow in scale and get stabilized. Basically, he claimed, for an organization to nurture the innovative idea that seem ridiculous in the moment, it would be more efficient to separately form groups either freely My abrupt book review is to argue that the loonshot nurturing rules and the new drug development procedure seem to share a lot in common. A life-saving new drug is also dismissed as a so-called crazy idea or loonshots in the initial development stage. The one of most well-known dyslipidemia medicine, statin had a commercialization process that shows the realistic barrier against an innovative idea. Statin has originated from teal-colored fungus found by a researcher Endo Akira working at a Japanese-based pharmaceutical conglomerate Sankyo. But because of risk of adverse reaction, the medicine was discarded by the company, and rather a U.S.-based conglomerate MSD took it over. Taking the tips from Endo, MSD finally released the first statin drug Mevacor in 1987 after conducing tens of clinical trials. With other follow-on statin drugs, the U.S. company has become one of the most successful global pharmaceutical company. In present day, many of South Korean pharmaceutical and bio companies have declared they would develop unprecedented innovative new drugs. Interestingly, the loonshot rules have been applied to an increasing number of new drug developments. One of most prevalent cases is a notion of developing a ‘spin-off’ that lets a R&D department to focus on the new drug pipeline. In 2016, Yuhan Corp co-founded a joint venture ‘ImmuneOncia’ with Sorrento Therapeutics to strategically develop new anticancer treatment. SK Chemicals spun off their new drug development division and founded ‘Tiumbio.’ Late last year, Ildong Holdings found new drug development-specializing company ‘Idience’ and transferred its new anticancer drug pipeline. Daewoong Pharmaceutical split off from its own subsidiary ‘iN Therapeutics’ in last May, and transferred ion channel new drug development platform and non-opioid analgesics, hearing loss treatment and brain disease treatment. Summing it up, the conventional pharmaceutical giants are applying the ‘phase separation’ concept to prevent halting a promising pipeline. The rules from the book may not be the correct answer. Nevertheless, we welcome the Korean companies doing all they can to achieve innovation and growth. Hopefully, their effort would result in a satisfying outcome.
- Company
- Julien Samson will be relocating to the UK headquarters
- by Eo, Yun-Ho Sep 18, 2020 06:29am
- Julien Samson According to related industries, GSK Korea held a town hall meeting on the 16th, and Julien Samson, announced that his current term of office as the head of the Korean subsidiary will be ended in this November. Julien Samson will be relocating to the UK headquarters in December. The successor to lead the Korean subsidiary was Robert Kempton, vice president of the current North Carolina branch. He joined GSK as a field vice president, with responsibility for sales of GSK's ELLIPTA portfolio in 2017 and is currently serving as vice president, global commercial lead, specialty medicine related to infectious diseases from 2018 to the present. Julien Samson, who has been leading GKS Korea since February 2018, was the first foreign president of a domestic corporation. The company has appointed President Hong Yoo-seok in 2014 after former chairman Jin-ho Kim, who has been leading the company since GlaxoWellcome, and CEO Julien Samson was appointed as CEO of GSK Canada's pharmaceutical business (treatment and vaccine) corporation in 2018. After working in public hospitals and government agencies in France, he joined GSK in 2012, and recently, he was in charge of establishing a global sales marketing strategy at the GSK respiratory disease business headquarters in the UK headquarters. Meanwhile, the head of MSD's Korean subsidiary is also replaced. Avi BenShoshan (49), the current CEO, will move to the US headquarters as of November, and MSD Korea will appoint Kevin Peters, the current president of MSD Thailand, as its successor president. With the overseas appointments of Avi BenShoshan and Julien Samson, the composition of the KRPIA’s council is also predicted to change. Abi Benshosan is currently chairman of KRPIA, and Julien Samson is vice chairman.
- Policy
- Sinopharm COVID-19 vaccine, not considered to be introduced
- by Kim, Jung-Ju Sep 18, 2020 06:29am
- Although China is in the midst of developing COVID-19 treatments and vaccines, the government said that it has no plans to import domestically unless the safety and efficacy of these products are secured. Far from being included in the plan to secure 30 million domestic patients, it means that domestic introduction itself is impossible. Kang-lip Kim, Vice Health Minister for the Central Disaster and Safety Countermeasure Headquarters for COVID-19, clarified this position through a Q&A briefing on the morning of the 16th. In order to accelerate the end of COVID-19, the government is currently approaching the 'two-track' method of supporting domestic development and securing quantities of foreign products in relation to treatments and vaccines. When it was introduced in Korea, it also planned to secure 30 million patients. China is also strengthening R&D for the development and export of domestic drugs and vaccines, among which, interest is emerging as to whether it is possible to introduce a vaccine developed by Sinopharm in Korea. Regarding this, Kim said, "Vaccines are much more difficult to verify for safety than other drugs. As we have not yet secured safety and efficacy data for vaccines being developed by Chinese pharmaceutical company Sinopharm, we will not review them in detail." Since there is no data on the safety and effectiveness of Sinopharm, it is not even reviewed whether it is introduced or not, and if the review is complete, it is possible to review domestic introduction or securing of quantity itself. He said, "although the securing of 30 million people has not yet been specifically structured, and it is not in the stage of confirming the so-called 'portfolio', various consultations are ongoing with pharmaceutical companies developing vaccines."
- Product
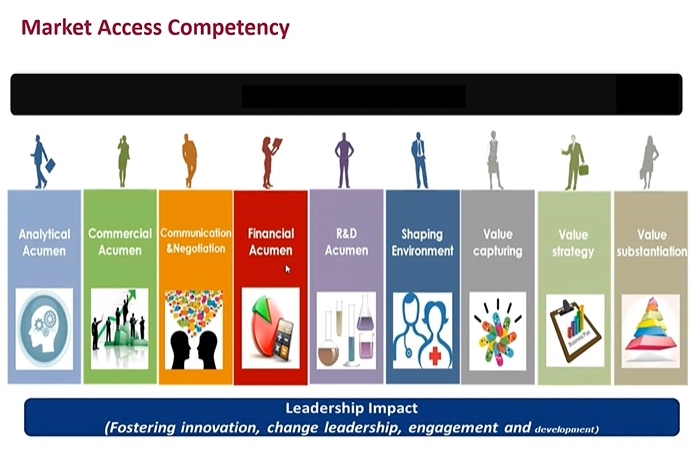
- “MA and marketer need experience, analytic mind, acumen”
- by Kim, Min-Gun Sep 18, 2020 06:29am
- “For a Market Access (MA) specialist, the most important skill set is to analyze the clinical trial data and to recreate them as a valuable message. They need to find any missing data required for pharmaceutical reimbursement review, and prepare the evidences prior to the review, if need be. If you are a pharmacy school student and want to become a MA specialist, you need to acquire rich experience and knowledge.” That was an advice from MA Director Lim Kyungwha at Jansen Korea to the students in college of pharmacy, who are looking into getting a job in the MA sector. The Pharmaceutical Marketing Professional Leaders (PPL) convened a sixth pharmaceutical industry seminar on Sept. 5 with the theme of ‘the Wave of Pharmaceuticals.’ At the seminar, Director Lim gave a lecture on the current industry status, and the importance of MA specialists. MA Director Lim Kyungwha at Jansen Korea Until few years back, MA specialists played a role of applying for reimbursement listing after a new drug approval. But things have changed—now the MA specialists participate from the clinical designing stage. In the initial clinical stage, new drug development and market access departments come together and talk about strategy as an ‘integrated MA’ to ponder on what the government would stress on when listing for reimbursement, areas to better reflect new drug’s value than other global alternative options, and which clinical outcomes would win the healthcare reimbursement. Director Lim said, “Nowadays, pharmaceutical companies do not consider MA as a single department, but rather treats it like a strategy. It’s because MA can intervene from the initial clinical stage to accelerate the reimbursement listing procedure and affect the entire drug approval and market access strategy.” In other words, MA is a key player in building a strategy for a new drug seeking for a proper acknowledgement of its value and to enter the market fast. Specialized and diversified MA specialist also has to serve two roles of communicating and negotiating with the government. So what kind of skill sets should a MA specialist have? Director Lim said, “MA specialist has to understand clinical data and capture the value in them. The reason why most of the companies hire master’s degree or doctorate degree graduates majoring in public health or social pharmacy is because we need people, who have wide experience and knowledge, skill to analyze finance and market, and understanding of R&D.” “Understanding the general pharmaceutical industry is crucial, as necessary strategy can be designed by understanding of the business. I also finished my MBA, but I went back to school to study social pharmacy,“ Director Lim added. Market access competency She encouraged the students and said, “Students who want to become a MA specialist need to acquire abundant knowledge, experience and strategic mind. As the industry is short of well-experienced MA specialists, the companies time to time hire entry level applicants, fresh out of university or graduate school, with potential seen during an interview.” She also reiterated, “Only because you choose your career path once, does not mean you would stay on it forever. Having richer experience is more important as all companies prefer people with more experience.” ’Acumen’ is critical asset for a marketer, while knowledge in pharmacology and toxicity is essential Executive Director Park Kwang-kyu at Gilead Science Liver Disease Business Department, emphasized ‘acumen’ is the most important factor in a student who dreams of being a marketer, who has to communicate with consumers in the sales department. They have to be highly empathetic to grasp when to be soft or firm during a conversation. Executive Director Park Kwang-kyu at Gilead Science Executive Director Park also mentioned insight, communication and leadership are important as well. He pointed out, “Even the marketers on the job struggle to distinguish ‘insight’ and ‘summary.’ Often the marketers have to deal with big data. And they need to digest the vast amount of data into a concise message,” so “they should be able to say easily in a single sentence to describe ‘what the company should do’ based on those data.” “We need people who are not afraid of challenges with confidence to work well in a multinational company. When it comes down to marketing, your own skill is more valuable than your degree,” he noted. However, Executive Director Park said the number of marketing jobs in multinational companies is declining. He said, “Two decades ago, a pharmacist-turned-marketer was treated as a specialist with expert knowledge. But now we have marketers from liberal arts, biology, pharmacy and nursing background, while [work] has gotten diversified and enhanced. But the company still thinks highly of pharmacy school graduates, so it would be worth it to knock on their doors.” Senior Director Kang Byung-chul at COSMAX BIO Senior Director Kang Byung-chul at COSMAX BIO leading both marketing and R&D departments recommended the participating students to make sure they study about pharmacology test and toxicity test before graduating to apply for a job in a pharmaceutical company. Senior Director Kang highlighted, “In any department, pharmacology and toxicity are the basics. And we all assume a pharmacy school graduates have full understanding of those.” The senior director did not recommend applying for the Business Development (BD) department for an entry level applicant. He said, “Only after working over a decade in R&D or marketing department, you can become a fully functioning member of the BD department. I would recommend transferring to BD department, after becoming a specialist in respective area for 10 years.”