- LOGIN
- MemberShip
- 2026-04-13 23:21:32
- Policy
- No. of refunded RSA drugs become 23 with addition of Onivyde
- by Lee, Hye-Kyung Aug 09, 2021 06:00am
- With the new addition of Servier Korea's pancreatic cancer treatment 'Onivyde (irinotecan liposome)' that was approved for reimbursement, a total of 23 drugs are now being covered under the Risk-sharing Agreement (RSA) scheme. Patients who take RSA drugs need to bear the full cost of the drug, then request refunds to the applicable pharmaceutical company. Recently, the National Health Insurance Service (NHIS) provided information on drugs eligible for the refund and the contact information of relevant reimbursement officials in each company for patients who paid a 100/100 copayment for the use of RSA drugs. 23 drugs are currently maintaining the RSA contract. 5 drugs whose RSAs were terminated - 'Pirespa tab,' 'Revlimid cap.,' 'Xalkori cap.,' 'Soliris inj.,' and 'Naglazyme inj.,' - may also apply for refunds if the patients received the drugs within the RSA term. NHIS monitors the RSA claims data regularly and notifies the pharmaceutical companies of the RSA refund amount and the financial expense each company has incurred according to their RSA scheme. Refunds of the additional cost borne by patients who paid 100/100 copayment for their drugs are directly conducted by the pharmaceutical companies or their agencies. In 10 of the 23 companies, the patients may directly contact the company to receive the refund, however, for the other 13 drugs including 'Erbitux,' patients need to ask an agency or academic society for reimbursement. The NHIS receives a monthly notice on the details of refunds from pharmaceutical companies and sends a notice to patients who did not receive refunds after comparing the refund details with the 100/100 copayment claims records. Meanwhile, when a medical care institution such as a hospital or pharmacy prescribes or dispenses an RSA drug with 100/100 copayment by the patient, the doctor or pharmacist responsible should file a claim under Section code U (Health insurance 100/100) according to the 'Method on claiming medical care benefit expenses, and form and tips for filling out review claims and statements.' ▶RSA drugs eligible for a refund for the f00/100 patient copayment cost Erbitux Inj., Xtandi Soft Cap., Stivarga Tab., Pomalyst Cap., Opdivo Inj., Keytruda Inj., Ibrance Cap., Kyprolis Inj., Cyramza Inj., Darzalex Inj., Spinraza Inj., Perjeta Inj., Dupixent Prefilled Inj., Imfinzi Inj., Strensiq Inj., Verzenio Tab., Bavencio Inj., Kisqali Tab., Benlysta Injection, Ninlaro Cap., Hemlibra S.C Inj., Defitelio Inj., Onivyde Inj.
- Policy
- It will spend ₩2.2 trillion to become a vaccine power
- by Lee, Jeong-Hwan Aug 09, 2021 06:00am
- President Moon Jae-in has announced that he would leap forward into a 'global vaccine production, the Big Five' by 2025. It also announced plans to select vaccines as one of the 'three major national strategic technologies' along with semiconductors and batteries and invest ₩2.2 trillion over the next five years. On the 5th, President Moon presided over the "K-Global Vaccine Hub Vision and Strategy Reporting Conference" at Cheongwadae and made the remarks. He plans to foster the vaccine industry as a new growth engine for the nation's economy. It said it will strengthen its global cooperation system and support the rapid development of domestic vaccines while creating an ecosystem for the vaccine industry. President Moon said, "By 2025, Korea will become one of the top five global vaccine production powerhouses and select vaccines as the top three national strategic technologies along with semiconductors and batteries." "We will invest ₩2.2 trillion over five years," he said. "Without sufficient supply of vaccines to all countries, continuous mutations and the spread of COVID cannot be prevented. After all, the fundamental solution is to dramatically increase the supply of vaccines. President Moon said, "We will greatly expand tax support related to vaccine R&D and facility investment and self-sufficiency of essential materials, parts, and equipment to maximize production capabilities of Korean companies. We will establish K-Bio Lab Hub and designate high-tech investment zones to provide various incentives." In order to strengthen the global cooperation system, President Moon said he would strengthen the vaccine partnership with foreign countries. "We will expand our vaccine partnership with other countries such as Germany and the U.K. and strengthen communication and cooperation with international organizations such as the WHO, global vaccine institutes, and companies, and prepare a stage for vaccine hubs such as revitalizing foreign investment and attracting global companies," President Moon said. He also mentioned the rapid development of domestic vaccines. He said, "Quick development of domestic vaccines to secure vaccine sovereignty is also very important. "COVID vaccine developed by domestic companies will enter phase 3 clinical trial this month," he said. "We are expected to commercialize the first vaccine in Korea in the first half of next year and are speeding up the development of the next-generation mRNA vaccine." "Domesticization of core production technologies is taking place. The government will provide medical expenses for the rapid development of domestic vaccines, and operate various support systems such as localization of raw and subsidiary materials and patent analysis. The Global Vaccine Hub Promotion Committee should take this opportunity to develop and finish the mRNA vaccine," he ordered. Prime Minister Kim Bu-gyeom and other related ministries attended the reporting meeting, which was also the first meeting of the "Global Vaccine Hub Promotion Committee," which was launched on the same day. In the private sector, 12 civilian members of the Global Vaccine Hub Promotion Committee and representatives of Korean companies that produce vaccines such as SK Bioscience, Samsung Biologics, and ST Pharm were present. The Global Vaccine Hub Promotion Committee is headed by the Prime Minister, but President Moon's direct presidency reflects his commitment to developing domestic vaccines and global vaccine production hubs to secure vaccine sovereignty.
- Policy
- Attention is focused on licensing domestic vaccines
- by Lee, Jeong-Hwan Aug 09, 2021 06:00am
- As the supply price of COVID vaccines, which relies entirely on imported products, is confirmed to increase next year's supply price in Korea, it is necessary to develop the vaccine within this year. Attention is focusing on the development status of Korean pharmaceutical bio companies, which are about to undergo phase 3 clinical trials, will be able to complete clinical trials as planned and obtain marketing permits within this year or in the first half of next year. According to the pharmaceutical bio industry on the 4th, five companies approved by the MFDS, including Genexine, SK bioscience, GeneOne, Cellid, and Eubiologics. The goal of the five companies is to obtain approval within this year for phase 3, which is evaluated as a phase just before commercialization, with phase 2 in progress or completed. It takes time for Curatis and HK Inno.N to get a commercial license after receiving a relatively recent vaccine approval last month. The development of a domestic COVID vaccine is urgently needed as the number of new confirmed cases of COVID patients in Korea is spreading and countries around the world are forced to distribute their own vaccine supplies. U.S. pharmaceutical companies Pfizer and Moderna have also decided to raise the supply price of their mRNA vaccines. Therefore, domestic supply prices are expected to rise next year. However, there is a long way to go to develop a domestic vaccine. Major foreign media reported on the 1st that Pfizer and Modena will increase the price of a single dose of vaccine supplied to the European Union by 25% and 10%, respectively. Pfizer raised its contract price from €15.5 to €19.5. The price of a single Modena rose from €19 to €21.47. The development of domestic vaccines is to ensure that Korea is not affected by such price hikes and fluctuations in volume of imported vaccines. SK Bioscience is the fastest development among domestic pharmaceutical bio companies that are close to clinical phase 3, the final step for permission. SK Bioscience submitted a clinical phase 3 plan for "GBP510," a synthetic antigen coronavirus development substance, to the MFDS in June. SK Bioscience, which received approval for phase 2 and phase 3 in Indonesia last month, aims to receive commercial approval and release it in the first half of next year after receiving phase 3. Genexine also received phase 2 and 3 of its DNA vaccine substance "GX-19N" in Indonesia last month. Along with Genexine history SK Bioscience, it is evaluated to be the closest to clinical phase 3. Cellid, which is developing the virus vector vaccine, Eubiologics and DNA vaccine developer GeneOne are also planning to enter phase 3 in the second half of this year and release the vaccine as early as the first quarter and second quarter of next year at the latest. It is not yet clear whether Korean vaccines will be available as scheduled because phase 3 clinical trials take a long time. Health authorities also have high expectations for domestic vaccine developers. The MOHW has successfully deliberated on the National Assembly by deploying ₩98 billion in phase 3 support from five companies that have started developing domestic vaccines in the second revised budget this year. Earlier in the year, Kwon Deok-chul, Minister of Health and Welfare, was convinced that domestic vaccines, which have been developed by the five companies since the beginning of this year, will enter commercialization with permission to market by the end of this year or early next year. The government has allowed domestic vaccine developers to enter clinical trials relatively quickly. The advantage of comparative clinical trials is that the number of subjects can be reduced to thousands of people compared to existing clinical trials, which require tens of thousands of people, significantly reducing clinical costs and time. If domestic vaccines enter the commercialization stage of inoculation after obtaining market permits, they will have a positive impact on the stabilization of supply and demand in Korea and the reduction of confirmed cases. It will not matter if the price of overseas development vaccines increases or supply and demand of supplies is unstable. "In the mid- to long-term, securing vaccines in Korea is very important," said Sohn Young-rae, head of Central Disaster Management. "In addition to the price issue, there are only a few vaccine supply pharmaceutical companies worldwide, and all countries are willing to purchase them, so buyers are inferior to suppliers in the negotiation process," he said.
- Company
- Sales of 'Stillen' have declined in two years
- by Chon, Seung-Hyun Aug 08, 2021 06:50pm
- Sales of gastritis treatments containing Artemisia Herb as an active ingredient have decreased. Sales had increased due to reflective profits from withdrawal of Ranitidine, but fell in two years. It is analyzed that Artemisia Herb's sales have decreased as the prescription market has shrunk due to the prolonged COVID-19 outbreak and PPI drugs have replaced Ranitidine. According to UBIST, a pharmaceutical research agency on the 5th, the amount of outpatient prescriptions for gastritis treatments including Artemisia Herb in the first half of last year fell 9.3% year-on-year to ₩59 billion. It fell 9.7% from ₩65.3 billion in the second half of last year. Artemisia Herb is a natural drug containig mugwort. Dong-A ST's Stillen is the original. It is the first time in two years that the Artemisia Herb gastritis treatment market has shown a decline since the first half of 2019. Sales of Artemisia Herb rose at the end of September 2019 with the impurity crisis of Ranitidine. The prescription amount of Artemisia Herb in the second half of 2019 was ₩54.9 billion, up 30.6% from a year earlier. Artemisia Herb was not the same as Ranitidine, which is used in gastric acid, heartburn, gastric ulcers, and reflux esophagitis, but it is analyzed that it was actively prescribed. The size of Artemisia Herb's outpatient prescription market in 2019 when Ranitidine was expelled increased 17.4% from the previous year to ₩87.5 billion. The Artemisia Herb market fell every year from ₩105.5 billion in 2014 to ₩74.5 billion in 2018, but it rebounded for the first time in a while. The prescription amount of Artemisia Herb in the first half of last year was ₩65.1 billion, up 53.7% from the first half of 2019 before Ranitidine was expelled. Prescriptions for the second half of last year rose 19.1% year-on-year to ₩65.4 billion. However, sales have declined since this year. Prescription amount of PPI drugs in the first half of this year was ₩323.6 billion, up 7.3% from the previous year. Compared to a 23.4% year-on-year increase in prescriptions in the first half of last year, the rise was slightly slower. The prescription amount of H2 receptor antagonist in the first half of the year was ₩68.6 billion, up 8.7% from a year earlier. Two years after Ranitidine crisis broke out, sales of Artemisia Herb decreased, unlike PPI and H2 receptor antagonists. Major Artemisia Herb drugs also saw their prescription performance drop. The amount of prescription for Dong-A ST's Stillen 2X in the first half of the year fell 15.3% year-on-year to ₩6.2 billion. Artemisia Herb gastritis treatments has 60 mg and 90 mg. Stillen TwoX is Artemisia Herb 90mg. Daewon's Otillen F prescription amount fell 8.6% year-on-year to ₩5 billion in the first half of last year, while Jeil's Nexillen S recorded ₩3.7 billion, down 22.8% from the first half of last year.
- Company
- Celltrion's developing next-generation mRNA vaccine platform
- by Lee, Seok-Jun Aug 08, 2021 06:49pm
- Celltrion has started developing next-generation mRNA vaccine platform with TriLink Biotechnology in the U.S. According to the company on the 4th, TriLink is a contract development and manufacturing organization (CDMO) based on the mRNA platform located in San Diego, U.S. It has its own Vactor and Gen3 CleanCap, which are essential for the development of mRNA vaccines, and has the ability to provide technology transfer along with clinical materials. TriLink plans to produce and supply substances that can proceed with Phase 1 and Phase 2 clinical trials to Celltrion by utilizing its own capping technology along with verification of antigen sequences. It also provides Celltrion with a GMP production scale-capable mould vector and mRNA process technology. Celltrion is expected to develop next-generation vaccines independently that have preventive effects against various mutated viruses, including COVID, and at the same time accelerate the development of mRNA platforms using its own patented technology. Celltrion is focusing on developing the next generation COVID vaccine using various mutated virus antigens, rather than developing and commercializing vaccines using the existing COVID antigens. It is planning to produce large-scale clinical phase 3 substances by establishing mRNA process facilities along with development of vectors that avoided patents. Through the development of the mRNA platform, it is planning to expand its technical scope beyond COVID to other diseases such as anticancer.
- Company
- Pfizer seeks generation change in the ALK market
- by Aug 08, 2021 06:48pm
- ALK target treatment Xalkori and Pfizer’s LorviquaPfizer, which developed the first ALK-targeted treatment, has released a third-generation new product. A new option has emerged in the ALK targeted treatment market, where subsequent drugs were not appropriate. Third-generation drugs are also expected to compete with second-generation products as a primary treatment. Pfizer was approved by the MFDS on the 29th of last month to treat ALK-positive non-small cell lung cancer. Lorviqua can be used at least once to treat patients who have been treated with conventional first-generation or second-generation treatments. The third-generation drug, Lorviqua, can be an alternative to the treatment of patients with resistance after the first-generation drugs Xalkori and the second-generation drugs Zykadia, Alecensa and Alunbrig. If Xalkori is used in the first generation, it can be used in the second generation and then in the second generation, it can be used in the second generation, and chemotherapy had to be used if secondary drugs developed resistance. G1202R is the most common type of resistant mutation that occurs after the second generation medication, and depending on the drug, F1174L (Zykadia), I1171T/N/S (Alecensa), and E1210K (Alunbrig). Lorlatinib is known to have an effect on all known resistant mutations. In the second phase of 275 previously treated patients, including the second generation ALK TKI, Lorviqua showed 47% ORR and 63% ORR in patients with brain metastasis. Lorviqua quickly became effective after taking. The time median to the first response and the time median to the first two in-reactions were 1.4 months, respectively. In the sub-analysis classified by second-generation drugs, Lorviqua maintained a consistently significant effect.The objective response rates in Alecensa, Alunbrig and Zykadia groups were 40.3%, 37.5%, and 40.4%, respectively. Attention is focusing on whether Pfizer, which has weakened its position in the ALK TKI market due to the advent of second-generation drugs, will succeed as third-generation Lorviqua. Pfizer has developed the first ALK targeted treatment called Xalkori, but sales continue to decline as second-generation drugs enter the primary treatment option. Xalkori, which had more than ₩10 billion in sales in the quarter of 2018 based on IQVIA, started to see a sharp drop in sales in 2019 due to the expansion of benefit for the second generation Alecensa and the termination of the risk-sharing contract. Annual sales fell 59% from ₩49.6 billion in 2018 to ₩20.3 billion in 2019. Last year, it also dropped 28% year-on-year to ₩14.6 billion. A three-phase CROWN study identified the primary treatment effect of Lorviqua. In this clinical trial, Lorviqua showed an improved OR (73% vs. 58%). It also reduced the risk of disease progression and death compared to Xalkori by 72%. The U.S. Food and Drug Administration (FDA) added an indication in March to use Lorviqua as a primary drug. It is expected that it will soon obtain additional indications in Europe. It is also expected that Lorviqua will expand its adaptation in Korea. However, competition between second-generation drugs is expected to continue for the time being because many steps have to be passed before applying domestic benefits.
- Policy
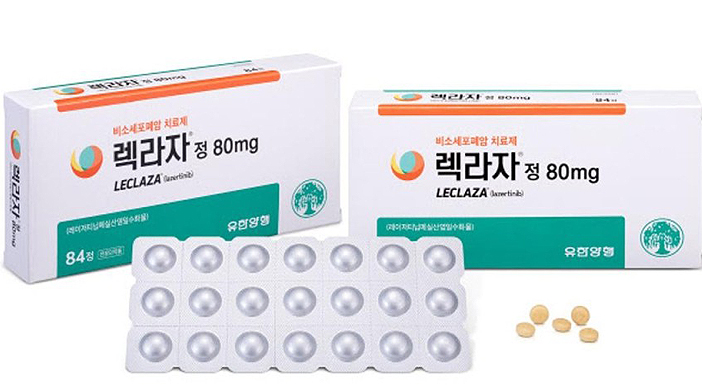
- PBAC results on Leclaza that was listed in 165 days show...
- by Lee, Hye-Kyung Aug 06, 2021 06:05am
- ‘Leclaza tab. (lazertinib mesylate monohydrate),' the 31st novel drug to be developed in Korea that received approval for insurance benefit in only 165 days after its approval, was reviewed as a 'new treatment alternative for non-small cell lung cancer patients' in the evaluation process for assessing the reasonableness of the medical care benefits for drugs. The Health Insurance Review and Assessment Service (HIRA) had recently disclosed the results and minutes of the benefit evaluation that was conducted on Leclaza by the Pharmaceutical Benefit Assessment Committee on April 8th. Leclaza was approved by the Ministry of Food and Drug Safety on January 18th as a ‘treatment for patients with EGFR T790M mutation-positive, locally advanced or metastatic NSCLC who were previously treated with an EGFR-TKI, then received reimbursement from July 1st after the notice was issued on the ‘amendment to the drug benefit list and the maximum ceiling price table’ on July 1st. Yuhan Corp had applied for the insurance benefit listing of Leclaza on December 30th, 2020 with the ‘approval-benefit appraisal linkage system.’ Through the system, the drug received insurance benefits at an unprecedented pace - in only184 days from its listing application and 165 days since receiving the marketing authorization. According to data disclosed by HIRA in 2017, it generally takes 1030 days for anticancer drugs, 475 days for rare disease drugs, 550 days for general drugs to receive notification for benefit listing since MFDS approval. Compared to the average, Leclaza shortened the listing registration period by 30 months. As a domestically developed novel drug that received its first approval this year, Leclaza’s approval status in the A7 countries, listing status in drug price reference books, and data on the clinical effectiveness in textbooks and clinical practice guidelines were not available for evaluation. In the PBAC evaluation results disclosed, the committee members concluded that “it is difficult to see a difference between Leclaza, which was approved for the treatment of patients with EGFR T790M mutation-positive, locally advanced or metastatic NSCLC who were previously treated with an EGFR-TKI, and the alternative drug.” Despite the review, the reason why the domestic new drug was able to be listed for reimbursement in only 165 days since its approval was because its reasonableness of receiving medical care benefits was recognized from the cost-effectiveness aspect, as the drug ‘is therapeutically equivalent to Tagrisso (osimertinib) while being cheaper than Tagrisso when using the RSA scheme proposed by the pharmaceutical company.’ The comparator in the evaluation was Tagrisso (osimertinib), and PBAC emphasized that the “evaluation results and conditions of Tagrisso’s RSA scheme should be considered in Leclaza’s pricing negotiations.” Also, relevant academic societies including the Korean Cancer Study Group, Korean Society of Medical Oncology, Korean Association for Lung Cancer added their support, saying that, “Leclaza showed a similar effect to the already-approved Tagrisso, and demonstrated an acceptable safety profile and low cardiac toxicity. We can fully expect Leclaza to become a viable substitute to Tagrisso” The societies requested reimbursement for Leclaza stating that “The drug can bring new opportunity to patients with EGFR T790M mutation-positive, locally advanced or metastatic NSCLC who had limited treatment options.” At the Cancer Disease Deliberation Committee meeting that was conducted on February 24th before the PBAC review, the committee had set the standard - ‘medical care benefit will be recognized for patients with Stage IIIA or higher NSCLC as second-line or later therapy that belong to one of the subject numbers (other than No.22,23,24)' for Leclaza. After deliberation and decision by the PBAC and the negotiations with NHIS that followed from April 23rd to June 8th, Leclaza was applied the RSA refund-type and expenditure cap type and listed for medical care benefit at a ceiling price of ₩68,964 per tablet.
- InterView
- “Olumiant provides rapid AD symptom improvement"
- by jung, sae-im Aug 06, 2021 06:04am
- The introduction of JAK inhibitors in the field of moderate-to-severe atopic dermatitis (AD) treatment was like a welcome rain in the drought of treatment options in AD. In 2018, the approval of the biologic agent ‘Dupixent’ transformed the AD treatment paradigm, however, many patients still feel an unmet need existed due to the limited reimbursement conditions and high drug price. JAK inhibitors are expected to widen the scope of treatment options for AD patients with features different than those offered by Dupixent. The first JAK inhibitor to receive approval for the AD indication was Eli Lilly’s ‘Olumiant (baricitinib)’ The treatment effect and safety of Olumiant were identified in the three clinical trials - BREEZE-AD1, BREEZE-AD2, BREEZE-AD7 –as a monotherapy and combination therapy with a topical corticosteroid (TCS) in adult patients with moderate to severe AD. In particular, Olumiant rapidly improved itching that severely deteriorates the quality of life from Day 2. Professor Sang Wook Son (Department of Dermatology, Korea University Ansan Hospital), who has been prescribing Olumiant from the clinical stages said, “Olumiant will play an important role in improving the symptoms and quality of life of patients. The various new drugs that have been introduced to the AD treatment environment have brought a ‘revolution’ to the AD treatment environment.” Dailypharm met with Professor Son to hear about the adult atopic dermatitis treatment environment and the role of Olumiant in Korea. Professor Sang Wook Son -Clinical trials on Olumiant have shown that it rapidly improves symptoms from the second day since its first administration. What does this mean for moderate to severe AD patients?” = For AD patients, managing pruritus, or itching, is very important. However, this symptom was difficult to control, as the inflammatory response or immune response, which are mechanical aspects that cause itching, are difficult to control. We use immunosuppressants and antihistamines for pruritus treatment, but it was difficult to expect dramatic effects with these treatments. This lack of a rapid and effective treatment option for severe itching had made treating AD patients quite difficult, and Olumiant’s data on its rapid effect was the most awaited aspect of the new drug. I have been experiencing this rapid symptom improvement effect of Olumiant in clinical practice, and believe this feature will continue to be one of the greatest strengths of Olumiant. - Olumiant’s oral formulation is also considered beneficial. For which patients would the oral Olumiant tablet be appropriate? =According to Olumiant’s indication, patients with moderate to severe AD, in other words, those whose Eczema Area and Severity Index (EASI) score is between 16-23 or over may use Olumiant. However, as there is only little experience accumulated in the actual clinical field with Olumiant, there is still no consensus or guideline on which treatment should be used for which patient. However, Olumiant has the benefit of being easy to carry as an oral formulation. Patients may conveniently carry around and take Olumiant from their bag according to their dosing schedule. Also, they can flexibly apply doses, reducing the dose or taking a resting period when symptoms improve. Also, patients who fear needles would also prefer oral forms of treatment. -AD is a chronic disease that requires long-term use of treatments. Therefore, the safety of long-term administration is also an important aspect in selecting treatments. I heard there were safety concerns in using JAK inhibitors. How about Olumiant? =Olumiant has around 7 years’ worth of long-term safety data accumulated in the field of rheumatoid arthritis. No special adverse events were identified as an issue during that period. Based on these results, there are expectations that Olumiant will have fewer side effects than existing treatment options not only in clinical studies but also in actual clinical settings -Olumiant can be administered alone or in combination with a topical corticosteroid (TCS) =Combination therapies are considered a sort of principle in AD treatment as it requires the use of TCS and other topical immunomodulators to achieve full effect. In particular, data has shown that the efficacy of treatment improves when using Olumiant with TCS in AD. -What role do you expect Olumiant will play in the treatment of adult AD? = I felt more confident treating patients after Olumiant was added as an AD treatment option. Compared to existing treatments, Olumiant has little burden of safety has a positive therapeutic effect. As patients can experience dramatic changes in various aspects including treatment satisfaction, improvement of quality of life, and improvement of itching and lesions, I have high expectations for Olumiant's role in the field of AD treatment.
- Policy
- Last year, the drug trade surplus was achieved
- by Lee, Tak-Sun Aug 05, 2021 08:47pm
- Domestic medicine achieved its trade surplus for the first time last year. This is because biosimilar led the export. The MFDS announced on the 1st that it analyzed the production, export, and import performance of drugs, quasi-drugs in 2020. Drugs achieved the trade surplus (₩1.394 trillion won) for the first time since 1998, when the MFDS compiled statistics. The main reason for this is that exports of finished drugs, which accounted for 79.6% of total exports (₩9.96 trillion), increased 92.3% (₩7.9308 trillion) compared to 2019. Quasi-drugs showed a 124% increase in production performance of anti-COVID-19 products (₩3.7149 trillion) compared to 2019. This is largely due to the increase in demand for products to prevent infectious diseases, with production performance of masks and disinfectants rising by 818% and 926% respectively compared to 2019. The average annual growth rate of drug production is 6.9%, which is six times higher than the manufacturing average. In 2020, drug production increased 10.1% year-on-year to ₩24.565.5 trillion, while export performance increased 5.2% year-on-year to ₩9.96 trillion ($8.44 billion), 62.5% year-on-year and import performance to ₩8.57 trillion ($7.26331 billion). Its production performance was 1.2% compared to GDP and 5.1% compared to domestic manufacturers. The MFDS explained that the average annual growth rate over the past five years is 6.9%, which is more than six times higher than the total domestic manufacturing production (1.1%), indicating the possibility of the pharmaceutical industry as Korea's future industry. In 2020, there were two companies with production performance of more than ₩1 trillion, while Celltrion rose 149.2% year-on-year to ₩1.476.9 trillion, followed by Hanmi with ₩1.14.3 trillion, similar to that of 2019. Although the size of the pharmaceutical market fell 4.7% year-on-year to ₩23.17 trillion in 2020, the size of the pharmaceutical market has grown 1.6% annually over the past five years. It was attributed to a significant increase in export performance to 62.5%, respectively, compared to the increase in production performance and import performance (10.1%, 5.2%, respectively). Remsima, Herzuma and Truxima exported the most to Germany In 2020, the drug trade balance recorded ₩1.394 trillion due to rising drug export performance, the first time since 1998, when the MFDS compiled performance statistics such as drug production, export and import. The main characteristic of the domestic pharmaceutical market in 2020 is that the export performance of finished drugs, which led to the ▲ trade surplus, increased by 92.3% and ▲ biosimilar exports are active amid strong performance in the production and export of bio pharmaceuticals,▲ and this is because the proportion of Rx drugs production has been maintained and the production of new drugs in Korea has steadily increased. In the case of finished drugs, which accounted for 79.6% (₩7.9308 trillion) of total drug exports in 2020, exports increased by 92.3% compared to 2019, and were larger than 62.5% of total drug exports, leading to a trade surplus. Among the finished drugs, the top three products in the size of exports are all bio pharmaceuticals, and bio pharmaceuticals account for a large portion of all domestic pharmaceutical exports. Among the finished drugs, Remsima was the No. 1 export, followed by Herzuma 150mg with ₩98.6 billion ($80million) and Truxima with ₩75.3 billion ($60million). Germany ($1.855.96 million), the U.S. ($780.61 million), and Turkey ($589.55 million) are the countries that Korea exports the most finished drugs. And, the largest importers of finished medicines were the United States ($977.61 million), Germany ($806.65 million), and China ($887.74 million). As a result, the U.S. and Germany have been identified as the largest trading partners. Twelve of the top 20 items in 2020 were bio-medicine products, and eight out of the 12 items accounted for 79.7% ($1.69.65 billion) as bio-similar products. Exports of bio pharmaceuticals have increased significantly in Europe and North America, which is analyzed to have been approved for new item licenses or listed on the prescription list in Europe and the United States in the second half of 2019. The production performance of Rx medicines (₩17.84 trillion) accounted for 84.9% of the finished medicines, 83-84%, which has been maintained for the past five years. Six new domestic drugs produced more than ₩10 billion, and 19 new domestic drugs produced ₩322.1 billion, up 38.2% from 2019. Meanwhile, the production performance of quasi-drugs in 2020 increased by 124% to ₩3.7149 trillion from 2019 (₩1.657 trillion), the highest growth in the past three years, and the trade balance of quasi-drugs recorded a surplus of ₩214 billion in 2020.
- Company
- Generic share in the Tamsulosin market has surpassed 60%
- by Kim, Jin-Gu Aug 05, 2021 08:46pm
- Harnal-DIn the Tamsulosin prostate hypertrophy treatment market worth ₩170 billion per year, Rx amount of generics increased by 10% compared to last year. During the same period, the original decreased by 10%, with generic accounting for more than 60% of the total market. According to UBIST, a pharmaceutical market research institute on the 2nd, the market size of Tamsulosin for prostate hypertrophy in the first half of last year is ₩83.7 billion. Compared to ₩82.6 billion in the first half of last year, it increased by 1%. Considering that prescriptions for prostate hypertrophy treatments are usually concentrated in the third and fourth quarters, it is expected to expand to more than ₩170 billion by the end of this year. Original Rx performance is decreasing, and generic performance is gradually increasing. The original Hannal-D by Astellas recorded ₩33 billion in prescription in the first half of this year. Compared to ₩36.6 billion in the first half of last year, it decreased by 10%. Generics increased 10% from ₩46 billion to ₩50.7 billion during the same period. Its market share is 61%. It surpassed 50% for the first time in the second half of 2019, and exceeded 60% in a year and a half. Since the expiration of the original patent in 2015, generic has joined the market in earnest. Currently, 107 companies are licensed for generics. Among them, 86 companies are competing in the market. Among generic companies, Hanmi posted ₩13.6 billion in prescription records in the first half alone with Hanmi Tams and Hanmi Tams OD. Analysts say that the release of Hanmi Tams 0.4mg and Hanmi Toms OD, which are high-capacity products in line with market demand, was significant. Genuine Science, DongKoo, Kyung Dong, and Celltrion posted more than ₩2 billion in prescriptions in the first half of the year. A variable in this market is combination formulations. Recently, pharmaceutical industries are developing a variety of combination formulations centered on Tamsulosin. GSK's Duodart was approved in May, a compound that combines Tamsulosin with another ingredient in the treatment of prostate hypertrophy. This compound is the first licensed drug in Korea. Given that there is a combination of two components in the prostate hypertrophy treatment market, attention is being paid to whether a compound including two components will reorganize the market composition. Development of a compound targeted at both prostate hypertrophy and irritable bladder is also in full swing. Ildong and Jeil are the hypersensitivity bladder therapy ingredients for Tamsulosin. Clinical treatment for a combination drugs that combines Solifenacin has been completed. DongKoo and Kyung Dong are developing a combination of Tamsulosin and Mirabegron, an irritant bladder treatment ingredient. Because there are many patients suffering from both diseases at the same time, it is explained that the possibility is sufficient. It is also developing combined drugs that targets prostate hypertrophy and erectile dysfunction at the same time. Hanmi's Gugutams (Tamsulosin+Tadalafil) was approved. Chong Kun Dang is also undergoing phase 3 of the compound clinical trial, which combines the same ingredients. Dongkuk and Yuyu are developing a compound that combines Dutasteride with Tadalafil.