- LOGIN
- MemberShip
- 2026-04-14 00:37:01
- Company
- Keytruda's sales in Q1 of the year are ₩9 trillion
- by Aug 05, 2021 08:46pm
- (Top left clockwise) Yervoy, Opdivo, Keytruda, Imfinzi, and Tecentriq MSD's Keytruda (Pembrolizumab)'s global quarterly sales surpassed $4 billion for the first time, ranking first in immuno-cancer drugs. According to related industries on the 2nd, the total global performance of five Checkpoint inhibitors (Keytruda, Opdivo, Tecentriq, Imfinzi, and Yervoy) in the first half of this year was $15.558 billion, up 17.7% year-on-year. All immuno-cancer drugs, except Opdivo, recorded 20% year-on-year growth. Keytruda posted the highest sales of $8.075 billion. It is a 21% increase compared to the same period last year-on-year. Keytruda surpassed $4 billion for the first time in the second quarter of this year, recording $4.176 billion due to increased sales every quarter. Keytruda, which added six new indications, including bladder cancer, skin cancer, colorectal cancer, lymphoma, triple negative breast cancer, and solid cancer with TMB, expanded its scope to esophageal cancer and early triple negative breast cancer this year. Keytruda was the second-highest selling drug after Humira with annual sales of ₩14.4 billion last year. It is expected to beat Humira next year. BMS and Ono's Opdivo made $3.63 billion in sales. Opdivo made up for the second quarter of this year. Sales in the second quarter rose 15.5% year-on-year to $1.91 billion. However, due to stagnant sales in the first quarter, the growth rate in the first half of the year was only 6.2%. Yervoy (Ipilimumab), a CTLA-4 family used as a combination therapy with Opdivo, posted $966 million in sales, up 26.4% from the previous year. Sales of Roche's Atezolizumab and Astrazeneca's Imfinzi are also increasing. Tecentriq rose 22.7% year-on-year to $1.727 billion in the first half of the year, while Imfinzi expanded 21.6% to $1.16 billion. Keytruda, the No. 1 market share, is also growing by a similar margin. Keytruda accounts for more than half (51.9%) of the five Checkpoint inhibitors. Meanwhile, Merck-Pfizer's most recently licensed "Bavencio" still stands at $60 million to $70 million in quarterly sales, less than ₩100 billion.
- Company
- Sales of PPI antiulcer agents rise 30% in 2 years
- by An, Kyung-Jin Aug 05, 2021 06:03am
- The ranitidine impurity issue gave wings to the rising prescription of proton pump inhibitors (PPIs). The share of PPI prescriptions in the antiulcer agent market grew 30% in only 2 years after the drugs containing ranitidine – which used to occupy the largest share of the H2 receptor antagonists - were pulled from the market. Hanmi’s incrementally modified drug ‘Esomezol’ gained marked influence over the antiulcer drug market with the impurity issue and COVID-19 as momentum. ◆PPI prescriptions increase 5%...reaps benefits despite prolonged COVID-19 crisis According to the pharmaceutical market research institution UBIST on the 4th, outpatient prescription of PPIs recorded ₩616.7 billion in Q2 this year, which is a 5.0% year-on-year increase from the same period of the previous year. Compared to Q2 2019, sales had risen 29%. Despite the overall contraction of the prescription drug market due to the prolonged COVID-19 pandemic, PPI prescriptions have maintained quarterly sales of over ₩160 billion during the past year. The cumulative PPI prescriptions in the first half of this year amounted to ₩323.6 billion, which was a 7.3% year-on-year increase. Proton pump inhibitors (PPIs) block the final step of acid secretion and maintain a high pH level in the stomach to treat peptic ulcer disease and gastroesophageal reflux disease (GERD). Since recording ₩335.8 billion in outpatient prescription sales in 2015, sales of PPIs have increased over 10% each year to account for the largest share of anti-ulcer drugs prescribed in Korea. The rate of increase became greater since 2019. The ranitidine impurity issue was pointed to as the reason for the surge in PPI prescriptions. Analysts believe the suspended sales of 'ranitidine' products among H2 receptor antagonists that used to hold the largest proportion of prescriptions and had a similar prescription target to PPIs, led to PPIs receiving the benefit. ◆Sales of Esomeprazole jump 33%·rabeprazole 22%...polarization intensifies in PPI prescriptions The ranitidine impurity first led to an improvement in the prescription performance of all the 7 PPI ingredients. However, after two years, the different PPI ingredients saw mixed results. Prescription of esomeprazole and rabeprazole, which accounted for a large proportion of prescriptions, have continued to rise after their prescription performance surged in Q4 2019, while the upsurge in the rest of the ingredients was only temporary, showing the intensifying gap between the ingredients. The esomeprazole ingredient that is prescribed the most has continued to make double-digit growth despite the COVID-19 crisis. Outpatient prescriptions of esomeprazole amounted to ₩70.4 billion in Q2. This is a 4.0% increase from the previous year and a 33.3% increase from the year before the last. In Q3 2019, esomeprazole sold around ₩53.7 billion, however, after ranitidine was removed from the market in Q4 2019, its sales rose sharply to reach ₩67.9 billion. The upward growth continued afterward to reach ₩70.8 billion in Q4 last year. The rabeprazole ingredient sold ₩38.1 billion in Q2, marking a 4.5% year-on-year increase. Rabeprazole’s sales in Q4 2019 rose to ₩36.5 billion and continued its rise to reach ₩ 41 billion in Q4 last year. Compared to 2Q 2019, prescriptions rose 22.0% in just 2 years. Although the increase was not as prominent as in esomeprazole or rabeprazole, Ilaprazole also benefited from the market withdrawal of ranitidine. Ilaprazole’s outpatient prescriptions reached ₩8 billion in Q2. This was an 8.8% year-on-year decrease, but a 2.3% increase from 2 years before. The other PPI ingredients received less impact from the impurity issue. Omeprazole sold ₩6.4 billion in Q2. This was a 10.2% and 6.6% decrease from the same period a year and 2 years ago. In the same period, pantoprazole sold ₩9.6 billion, an 11.2% and 6.5% decrease from the previous year and 2 years ago. Outpatient prescriptions of lansoprazole in Q2 was ₩3.9 billion. This was an 11.6% year-on-year increase, but a 12.2% decrease from the 2 years ago, showing less performance than usual. ◆Prescriptions of Hanmi’s 'Esomezol' rise 45%...Products see mixed results from COVID-19 The key PPI products have experienced a sharp change in their prescription performance during the past 2 years, with the lessening impact of the impurity crisis and the increasing impact of the COVID-19 crisis. Hanmi Pharmaceutical’s ‘Esomezol’ showed the most marked growth. In 1H this year, Esomezol sold ₩23.3 billion in outpatient prescriptions. Despite the COVID-19 crisis, prescription of the drug increased 14.2% year-on-year to exceed ‘Nexium’ and become the leader in the PPI prescription market. Esomezol is a salt-modified drug of esomeprazole. The impurity issue led to an upsurge in prescription demand for Esomezol, with outpatient prescriptions rising 44.8% in just 2 years. AstraZeneca’s Nexium sold ₩18.7 billion in outpatient prescriptions in Q2. This is a 17.6% decrease from the year before and a 5.8% decrease from 2 years ago. Due to the COVID-19 crisis, prescriptions of Nexium decreased significantly, enough to hand over its long-held lead in the PPI prescription market. Nexium is the original esomeprazole brand that is distributed and sold by Daewoong Pharmaceutical in Korea. The other items were also not free from the impact of COVID-19. Il-Yang Pharmaceutical’s ‘Noltec’ sold ₩16.2 billion in 1H this year. This was a 4.3% year-on-year decrease, but also a 4.7% increase from 2 years ago. Noltec was able to maintain its 3rd place among prescriptions of single-agent PPIs. Noltec, an ilaprazole PPI agent developed by Il-Yang Pharmaceutical, was released as the nation’s 14th new indigenous drug at the end of 2009. Takeda Pharmaceuticals Korea’s ‘Lanston LFDT’ also rose to the ranks in H1 this year, selling ₩13.8 billion in outpatient prescriptions. In the same period, Daewon Pharm's ‘Eswonamp’ sold ₩8.3 billion. After the ranitidine issue, Eswonamp had enjoyed a profit with the annual prescription amount exceeding ₩20 billion for the first time, but prescription performance fell 17.3% this year. In addition to the impurity issue, analysts believe that the COVID-19 pandemic has acted as a variable that increased the gap between prescriptions of the products. HK Inno.N’s release of the antiulcer agent ‘K-Cab (tegoprazan) that has a new mechanism of action, has also intensified the change in the dominion of antiulcer drug prescriptions.
- Product
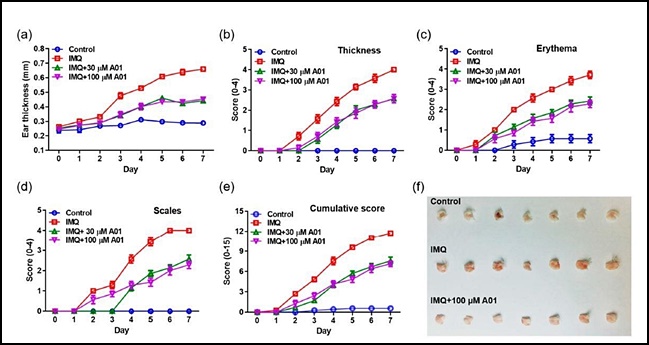
- "Inhibiting ANO1 activity has effect on treating psoriasis"
- by Lee, Jeong-Hwan Aug 05, 2021 06:02am
- A study result has shown that inhibiting the activity of the Anoctamin-1 (ANO1) protein may be effective in treating psoriasis, an intractable skin disorder. The results are expected to aid further research in developing ANO1 inhibitors for the treatment of intractable diseases such as psoriasis and cancer. On the 4th, the joint research team of Gyeonggi-do Regional Research Center and Professor Young Duk Yang’s team at the CHA University School of Medicine announced the results of the study that contained these findings. Psoriasis is an intractable skin condition that causes a build-up of extra dead cells and inflammation that occurs due to abnormal function of the immune system. The joint research team found that effectively blocking one of the ion channels that exist in the body – the ANO1 ion channel – reduced key symptoms of psoriasis such as rash, erythema, white scales, etc. while reducing inflammation-inducing substances that cause psoriasis Accordingly, the teams plan to expand their scope of research to develop new ANO1 inhibitors for the treatment of intractable diseases such as psoriasis and cancer. ANO1 is a membrane protein that acts as a channel for chloride ion transport. Professor Young Duk Yang said, “Psoriasis is an intractable skin disorder with no identified cause that is difficult to treat and recurs well. The study results may be used as a new breakthrough in the development of treating psoriasis.” The study results were published in the International Journal of Molecules Sciences in July 2021.
- Company
- Restylane Kysse has been approved in Korea
- by jung, sae-im Aug 05, 2021 12:41am
- Galderma Korea announced on the 2nd that Hyaluronic acid filler Restylane Kysse was approved by the MFDS on July 26 for the purpose of temporarily expanding the volume of lips for adults aged 21 or older. Restylane Kysse, which received Korea's first permission for lip fillers, was developed by Galderma as Optical Balanced Technology( OBT). Customized procedures can be performed to suit the patient's skin condition, and lip volume can be restored to create natural and beautiful lip volume. More than 78% of all patients who participated in Restylane Kysse's clinical trial said they were still satisfied with the results of the procedure a year later. The effect of Restylane Kysse was confirmed by a clinical trial. A total of 270 people were evaluated for the validity and satisfaction of the product by measuring changes after the lip enlargement procedure every eight weeks for 12 months. Clinical results showed that it lasted up to a year and showed high treatment satisfaction not only in middle-aged people who lost lip volume due to aging but also in young patients who wanted natural and thick lips. Based on this, it was approved by the FDA on March 26 last year as a lip filler. Lee Jae-hyuk, executive director of Galderma Korea's Aesthetic Division, said, "We reaffirmed the technical skills of Galderma, which developed the Hyaluronic acid filler Restylane Kysse. We will continue to make efforts as a helper to complete each person's skin through products like Restylane Kysse. We have been constantly innovating for healthy and beautiful skin."
- Company
- Roche’s Polivy can be prescribed at general hospitals
- by Eo, Yun-Ho Aug 05, 2021 12:40am
- The new lymphoma drug Polivy can be prescribed at general hospitals. According to related industries, ADC that combine with conventional BR therapy (Bendamustine/ Rituximab) treatments for diffuse large B-cell lymphoma (DLBCL) that have poor non-responsive prognosis of Roche. However, Polivy is still a non-reimbursed drug. Roche applied insurance benefit earlier this year, but failed to pass HIRA's Cancer Drugs Benefit Appraisal Committee in April. Roche is attempting to register Polivy in the future. Diffuse large B-cell lymphoma, which accounts for the highest percentage of non-Hodgkin lymphoma, is a type of aggressive lymphoma that requires immediate treatment due to its fast progression. More than half of patients have good treatment response rates to reach related levels, but 30 to 40% of patients do not respond to the standard therapy, R-CHOP, or experience recurrence after the first treatment. Most patients experience recurrence within two years, and the disease is fatal enough to last only six months. Recurrent and nonresponsive diffuse large B-cell lymphoma has been an area that lacks effective treatment options. Polivy is the first ADC drug to target CD79b, which binds to CD79b expressed in B cells and has the mechanism of inducing apoptosis. Polivy was validated by phase 1b/2 clinical trials in 80 patients with recurrent or diffuse large B-cell lymphoma patients who could not receive hematopoietic stem cell transplants and had more than one treatment experience. It was designated as a rare drug in Korea in October last year. Studies have shown that the Polivy co-therapy group extends CR, Complete Response and OS and Overall Surviva compared to the BR therapy group. Ko Young-il, a professor of hematology at Seoul National University Hospital, said, "Polivy has confirmed its usefulness through clinical use and is a treatment option recommended by international guidelines, which is significant in that it has proposed a new alternative to re-use or diffuse large B-cell lymphoma treatment.
- Policy
- No prior approval req. filed for Soliris, 47 for Ultomiris
- by Lee, Hye-Kyung Aug 04, 2021 06:01am
- ‘Ultomiris (ravulizumab),’ a follow-on drug of ‘Soliris (eculizumab)’ that was approved for reimbursement from June, has taken away all new prescriptions for the treatment of paroxysmal nocturnal hemoglobinuria (PNH) from Soliris. Reimbursement for Ultomiris, like Soliris, needs to be authorized in advance through a prior authorization application process to the NHIS before administration at a medical care facility equipped with the personnel and facilities as specified in the ‘Criteria on the medical care benefit for hematopoietic stem cell transplantation.' According to the eligibility for reimbursement of Soliris and Ultomiris deliberated in July by HIRA’s Healthcare Review and Assessment Committee, new approval applications filed by PNH patients were 0 for Soliris and 47 for Ultomiris. The doctors and new PNH patients were seen to have selected Ultomiris, which can be administered every 8 weeks as maintenance therapy 2 weeks after the initial dose, over Solaris, which needs to be administered biweekly. Ultomiris received approval from the U.S. FDA in December 2018, and from the EMA in July 2018. The drug received marketing authorization in Korea on May 21st, 2020. After receiving approval, Handok applied for the insurance benefit listing on August 31st last year. After receiving approval from HIRA’s Pharmaceutical Benefit Assessment Committee, going through NHIS’s drug pricing negotiations, and deliberation by MOHW’s Health Insurance Policy Deliberative Committee, Ultomiris was listed at ₩5,598,942 per vial from June 7th. Based on the clinical trial results, Ultomiris was found to be clinically non-inferior to its substitute Soliris. Also, Ultomiris was considered cost-effective if its price is set at Solaris's weighted average price of ₩5,598,942 or less. Meanwhile, 43 PNH patients receiving Soliris that were monitored received approval to continue administration of Soliris. 6 cases that were filed to newly administer Soliris for atypical hemolytic uremic syndrome (aHUS) were all disapproved, and 2 monitoring cases for Soliris were approved and the other 2 were disapproved.
- Policy
- Will changes be made to Hemlibra’s reimbursement standards?
- by Lee, Jeong-Hwan Aug 04, 2021 06:01am
- As the Anti-Corruption & Civil Rights Commission (ACRC) in addition to the National Assembly raised the need to ease the reimbursement standards for the hemophilia treatment Hemlibra (emicizumab), the growing sense of urgency on the Ministry of Health and Welfare (MOHW) and the Health Insurance Review and Assessment Service (HIRA)’s behalf to take follow-up actions is expected to increase. The NA and ACRC requested the standards be changed so that pediatric patients under the age of 12 can use the subcutaneous injection Hemlibra immediately without prior use of the intravenous drug treatment ‘immune tolerance induction (ITI) therapies. Sun-Woo Kang of the Democratic Party of Korea had first criticized the inappropriateness of Hemlibra’s reimbursement standards in June, then the ACRC had also made recommendations to the MOHW to change the reimbursement standards. The NA member pointed out that the existing reimbursement standards that acknowledge reimbursement of Hemlibra in pediatric patients aged under 12 only after they fail intravenous injections that accompany great pain should be changed. The ACRC also decided that it was harsh to recognize insurance benefits for Hemlibra only after the pediatric patients that have weak blood vessels undergo ITI therapy that require intravenous injections for 2-3 years. The NA and ACRC’s move was influenced by the appeals made by guardians of pediatric patients with hemophilia that were posted on the National Petition board of Cheong Wa Dae. A total of 4 requests to change the reimbursement standards of Hemlibra had been posted on the national petition board from November 2019 to date. 2019 was when JW Pharmaceutical had received marketing authorization for Hemlibra. JW Pharmaceutical received approval for Hemlibra from the Ministry of Food and Drug Safety on January 20th, 2019. In the earlier petitions, the request was focused on approving reimbursement for Hemlibra, but from last year, the content of the petitions changed to request modification or ease of reimbursement standards for patients aged less than 12 years. Recently, the caregivers of patients (parents) had requested MOHW and HIRA to delete the mandatory ITI therapy prerequisite for Hemlibra’s reimbursement in pediatric patients aged 1 to 12 years old. They criticized that the current reimbursement standards violate human rights by forcing pediatric patients to receive intravenous injections even though Hemlibra, a subcutaneous injection option, is available. Upon receiving such criticism, the MOHW and HIRA are making a move to improve the reimbursement standards. HIRA had held an expert advisory meeting on the efficient operation of ITI therapies for hemophilia A, and the MOHW is planning to amend the reimbursement standards for pediatric patients based on HIRA’s discussion results. In particular, the need to improve the standards grew with the NA strongly criticizing Hemlibra’s reimbursement standards as “standards that approve reimbursements based on the pain and suffering of our children.” Also, the fact that other countries do not limit reimbursement of Hemlibra by age raises the validity on the need for the health authorities to improve the benefit standards. The U.K .approves reimbursement of Hemlibra when the patient satisfies any one of the following conditions: those who fail ITI therapy, or cannot control bleeding, or administers the drug as maintenance therapy or bleeding therapy with bypassing agents, or as maintenance therapy to prevent bleeding symptoms during ITI therapy. In response to the continued criticism from the NA, ACRC, and caregivers of pediatric patients, the MOHW and HIRA are expected to present a bill to revise the benefit standards in the near future. On Sun-Woo Kang’s criticism, the Minister of Health and Welfare Kwon Deok-Cheol said that he would “comprehensively review the issue with experts and discuss how to improve the reimbursement standard of Hemlibra in consideration of the characteristics of pediatric patients.”
- Policy
- Daewoong has added a line-up of osteoporosis treatments PO
- by Lee, Tak-Sun Aug 03, 2021 08:27pm
- Daewoong's Evimax 45mg was approved on July 29. It is generic for Raloxifene HCl 45mg. The original for Raloxifene HCl is Evista from Alvogen Korea. Evista contains Raloxifene HCl 60 mg. Raloxifene HCl 45mg was first developed by Yuhan. In September 2019, Yuhan was granted 'Raboni 45mg', a product containing Raloxifene HCl 45mg. The company explains that safety has been increased by lowering the content of Raloxifene HCl by improving its bioavailability. Daewoong's approved product is also manufactured and supplied by Yuhan. In June, Daewoong was previously granted "Evimax D," a combination of Raloxifene HCl 45mg and a concentrated vitamin D powder. This is also an item produced by Yuhan. As a result, Daewoong will have three Raloxifene preparations, including Daewoong Raloxifene HCl 60mg, which was approved in March last year. Daewoong has been selling Daewoong Zoledronic Acid as a treatment for osteoporosis and Ricen Plus as an oral medicine. The performance is not bad either. Based on IQVIA last year, Daewoong Zoledronic Acid recorded ₩11.2 billion and Ricen Plus, which is a combination of Risedronic acid and Cholesterol, recorded ₩1.5 billion. In terms of sales, oral products are less competitive than injection products. For this reason, Daewoong needed the latest treatment for oral osteoporosis. It is believed that the company introduced Raloxifene HCl 45mg developed by its competitor Yuhan first. In the past, Daewoong sold original osteoporosis drugs such as Fosamax Plus D and had the largest share in the oral market. However, due to the termination of the contract for MSD's Fosamax, it suffered from losing its main items. Zoldronic Acid, which had little sales at the time, was successful by co-promoting with Sandoz. Since then, the Zoldronic Acid injection has been manufactured by Daewoong by purchasing copyrights. Considering Daewoong's business competitiveness in the osteoporosis treatment market, the introduction of Raloxifene HCl 45mg is also likely to be successful. Attention is focusing on how much sales Daewoong will make in the osteoporosis treatment oral market through the launch of a new product.
- Policy
- The number one treatment for mild COVID is Kaletra
- by Kim, Jung-Ju Aug 03, 2021 08:24pm
- The number one drug used to treat mild COVID in the first half of this year was AbbVie's Kaletra, which amounted to ₩596 million. The total amount of drugs spent on the entire treatment, including severe cases, amounted to ₩12.1 billion. Among the data on "COVID-19 Treatment Expenditure in the first half of the year," the "Status of Drug Administration by severe cases" showed this. According to the current status, the amount of drugs used to treat COVID-19 in the first half of this year amounted to ₩12.1 billion, or 2.5% of the total medical expenses. AbbVie's Kaletra (₩596 million) is the number 1 based on drug costs . Then, Chong Kun Dang's Tazoperan 4.5g , ₩107 million, Samsung's Combicin 3g, ₩98 million, Ukchemipharm's Trison Kit 2g is ₩88 million, Boryung's Maxipim 1g is ₩83 million, SK Chemical's Huthan 50mg is ₩72 million, Bayer Korea Avelox 400mg, ₩68 million, and Yuhan's Meropen 0.5g, ₩65 million. Following oxygen, cost of AbbVie's Kaletra is the most at ₩128 million, Yuhan's Meropen 0.5g, ₩112 million, Chong Kun Dang's Tazoperan 4.5g, ₩99 million, Maxipim 1g, ₩72 million, Huthan, ₩51 million, Avelox 400mg, ₩47 million, and Clexane, ₩45 million, Vfend 200mg, ₩4400 million. Excluding oxygen, Fresofol 2% of Fresenius Kabi Korea was the most used, followed by Meropen 0.5g of Yuhan with ₩238 million and Huthan 50 with ₩1.9 million. Greencross Human Serum Album 20%, ₩173 million, Human Serum Album 20%. ₩127 million, Vfend 200mg, ₩120 million, Boxter's Phoxilium Phosphate Solution 1.2 mmol/L, ₩85 million, and JW Life Science's Winuf, ₩83 million, respectively.
- Company
- Will regulations on ERP be established?
- by Aug 03, 2021 08:24pm
- Presidential candidate Lee Nak-yeonLee Nak-yeon, a presidential candidate for the Democratic Party of Korea, mentioned the need to draw up regulations on the Early Retention Program (ERP) of foreign-invested companies, including foreign pharmaceutical companies. Attention is focusing on whether frequent ERP regulations in the pharmaceutical industry will be possible. He made the remarks at a recent meeting in Gwangju Metropolitan City, "How to improve the working environment and secure the right to survive for workers in office research jobs and foreign-invested companies." "Recently, foreign-invested companies, including multinational pharmaceutical companies, have unilaterally implemented corporate changes such as division, acquisition, merger, and transfer of operations, and the government's investment in the name of internal restructuring. He agreed, "Changing legislative and public policies for new types of workers with different working conditions from existing production service jobs is an important task." "We need additional protective measures to regulate reckless abuse of voluntary retirement," he said. "We will make efforts to respect the value of labor in the future and not ignore the legal system for workers who are not legally protected in blind spots other than existing production, service, and even those who are not properly protected," he said. ERP of foreign pharmaceutical companies is being implemented more frequently following the outbreak of COVID-19. Sanofi, Lilly, and Roche conducted ERP in the second half of last year, and five to six companies, including Viatris, Astellas, GSK and Roche, conducted ERP in the first half of this year. He also said, "The comprehensive wage management is a structure in which it is difficult to reflect the opinions of workers because there is a possibility that users will unilaterally decide," adding, "We need to think about ways to increase the bargaining power of office research." The meeting, hosted by LG Electronics' human-centered office workers' union and the Korea MSD labor union, and organized by Daesang LLF, drew attention from unions of three pharmaceutical companies as representatives of foreign investment companies. They emphasized the reality that Korean branch employees are harmed by unilateral M&As at the global level and the need to pass legislative proposals for safety measures.