- LOGIN
- MemberShip
- 2026-04-10 05:23:34
- Policy
- Patent protection for 42 yrs for Humira/32 yrs for Keytruda
- by Lee, Hye-Kyung May 16, 2023 09:07pm
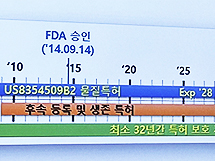
- It was analyzed that Humira maintained its patent protection period for at least 42 years and Keytruda for at least 32 years after filing for material patents with the Evergreening patent strategy. Evergreening refers to extending the term of a patent or extending the term of a patent for more than 20 years in the case of a patent to obtain more exclusive rights during the patent protection period. The types of evergreening strategies are representative of salt compounds, solvates, crystalline forms, optical isomers, dosage forms and pharmacokinetic data, manufacturing methods, and uses. Kim Tae-Kwon, responsible for the Korea Institute of Patent Technology Advancement, held on the last day of the 'Bio Korea 2023' event held on the 12th, 'Strategies for responding to original drugs and late-comer drugs according to the expiration of blockbuster drug patents'. Introducing the patent strategy. Kim Tae-kwon, head of the Korea Institute of Patent Technology Promotion The reason Humira and Keytruda are cited as examples is that the two medicines, excluding vaccines, respectively, ranked first and second in global sales in 2021, with sales of Humira at $20.694 billion and Keytruda at $17.186 billion that year. Humira applied for a substance patent in the United States in 1996, was registered as a patent in 2000, and received FDA approval in 2002. Since then, it has received FDA approval for additional indications such as psoriatic arthritis, ankylosing spondylitis, and Crohn's disease, and the substance patent period expired in 2016. So far, Humira has confirmed 746 patents based on 79 original patent applications. By type, 23 family groups (29.11%) for medicinal use and manufacturing method were distributed the most, followed by other 9 (11.39%), 8 diagnoses (10.13%), 7 formulations (8.66%), and 4 compositions. (5.56%), and 2 material improvements (2.53%). Responsibility Kim said, “Before the substance patent of Humira expired, we continuously extended the patent period with patents for medicinal use, formulation and composition patents, manufacturing method patents, diagnostic patents, automatic administration device patents, and pharmaceutical improvement patents.” It maintained its monopoly." In other words, Humira used an ever-greening patent strategy, such as forming a relatively superior patent barrier in the field of pharmaceutical use and manufacturing methods and forming a patent barrier in the pharmaceutical field as well. As a result, Humira was able to have a protection period of at least 42 years after applying for a substance patent and 35 years after FDA approval. After applying for a material patent in 2008, Keytruda registered a patent in 2013 and received FDA approval in 2014. Based on 67 original applications for patents, a total of 345 patents were confirmed. Looking at the distribution by type, 48 (71.64%) of the medicinal uses showed an overwhelming distribution. Next, 7 drugs (10.45%), 4 double antibodies (5.97%), 3 substances (4.48%), 3 diagnoses (4.48%), and 2 others (2.99%) are shown. Kim said, "There was also a case where the dosage and usage were changed as a barrier patent for the evergreening strategy of Keytruda products." "There was also data that it was to block the development of biosimilars as the patent term expires in 2028." and interpreted. In the case of Keytruda, it used the evergreening patent strategy, and it was analyzed that it had a protection period of at least 32 years after the substance patent and about 26 years after FDA approval. Kim explained, "As a result of the analysis of patent applications by type of biopharmaceutical, material patents must be filed, and based on material patents, applications are filed in the order of pharmaceutical use, composition, formulation, manufacturing method, diagnosis, and material improvement patents." As a result of comparing the evergreening strategy of biopharmaceuticals and synthetic drugs, there are patents for use, manufacturing methods, and formulations after substance patents, but synthetic drugs have many improved patents such as crystal forms, optical isomers, polymorphs, and intermediate patents for chemical formulas. However, as for biopharmaceuticals, there were patents for improving antibody or protein drugs. In addition, substance patents are important, but in the case of manufacturing method patents, it was noted that the distribution of biopharmaceuticals was high and the distribution of synthetic drugs was low. Kim said, "One thing to look carefully at is the expiry of the duration of a biopharmaceutical, but we need to look at changes in the patent for formulation, usage, and dosage in the use patent."
- Company
- Crysvita, the first fast-track drug by Yoon gov, is expected
- by Eo, Yun-Ho May 16, 2023 09:06pm
- It is now possible to prescribe 'Crysvita', the No. 1 rapidly registered drug of the Yoon government. This drug recently passed the Drug Committee (DC) of medical institutions such as SNUH. Kyowa Kirin Korea's (XLH rickets treatment Crysvita is a pediatric treatment that has been clinically proven effective in the field of diseases for which there is no equivalent drug or treatment. ) was listed on the list of insurance benefits. Since it is a drug that requires prior approval for reimbursement, it will take more time until the first prescription comes out. Crysvita's reimbursement criteria are for patients with X chromosome-linked hypophosphatemic (XLH) rickets aged 1 to 12 years who have not been controlled despite continuous administration of existing treatments (active vitamin D preparations, etc.) for more than 6 months. Symptoms such as growth retardation, dental abnormalities, lower extremity bone deformity, premature cranial fusion, and increased intracranial pressure are confirmed, and 'RSS level is 2 points or more, hypophosphatemia (less than 3.0 mg/dL) and loss of renal phosphorus (TmP/GFR reference value) less) must be proven. Until the registration of Crysvita, pediatric XLH rickets was treated with large doses of phosphate and vitamin D, but it was administered 4 to 6 times a day, and about 30% required surgery. Kang Hee-kyung, a professor of pediatrics at Seoul National University Hospital, said, "The existing treatment method of directly administering insufficient phosphate has a limit in that a vicious cycle occurs in which 'FGG23' is further activated and the degree of phosphate reabsorption further decreases. Crysvita is not a method of adding phosphate, It is a fundamental treatment that inhibits FGG23.”
- Company
- JAKi Cibinqo owns strengths in dosage adjustments
- by Jung, Sae-Im May 16, 2023 05:40am
- Pfizer's JAK inhibitor ‘Cibinqo (abrocitinib)' has embarked on a full-fledged journey to expand its prescriptions. After landing in major general hospitals at the end of last year, the drug is likely to be registered for reimbursement within the first half of this year. Cibinqo is a Janus kinase 1 (JAK1) inhibitor approved by the Ministry of Food and Drug Safety in November 2021. It is the 4th JAK inhibitor introduced to Korea and the second JAK inhibitor drug introduced by Pfizer after Xeljanz. Unlike Xeljanz, which is only used for ulcerative colitis, Cibinqo is used to treat severe atopic dermatitis. Treatment options have increased significantly for severe atopic dermatitis starting with the introduction of the injection-type biologic medication and oral JAI inhibitors. In particular, a total of 3 oral JAK inhibitors are available for patient use in Korea. Olumiant (baricitinib) and Rinvoq (upadacitinib) have first entered the market and are being used with reimbursement, followed by Cibinqo. Although Cibinqo is a latecomer, it owns a differentia table property from existing drugs. Dailypharm met with Jung-Im Na, Professor of Dermatology at Seoul National University Bundang Hospital to hear about its differentiable properties in the field. Jung-Im Na, Professor of Dermatology at Seoul National University Bundang Hospital Its first advantage is in its free dose control. Cibinqo comes in three doses: 50, 100, and 200mg. Although the recommended starting dose is 200mg, the dosage can be adjusted to 100 mg or 50 mg depending on the progress of treatment. If symptoms worsen with dose reduction, the doctor can increase treatment response again by increasing the dose and using local treatment together (JADE REGIMEN study). Professor Na said, “JAK inhibitors are fast and effective, but its exit strategy is considered a problem after the patient’s condition improves. In other words, ending treatment after seeing an effect is difficult with the use of JAK inhibitors. However still, due to its free dosage adjustments, it is attractive that the dosage can be reduced step by step from 200mg to 50mg.” He added, “No drug can be used for the rest of one’s life. Therefore, how to complete the treatment well is an important factor, and dose plays an important role. In particular, JAK inhibitors generally have a short half-life, and therefore disappear quickly from the body, leading to recurrence. Therefore, you cannot terminate treatment at once,” emphasizing the importance of dosage control. Major efficacy endpoint results in the JADE DARE trial Another advantage is in how it holds grounds based on a head-to-head trial for switching to the biological drug Dupixent. Among Cibinqo’s 7 Phase III clinical trials, the JADE DARE study is a head-to-head clinical trial comparing Cibinqo with Dupixent. As a result of separately administering Cibinqo 200mg and dupilumab 300mg in combination with a topical treatment for 26 weeks, the proportion of patients who achieved an improvement of 4 points or larger in the Peak Pruritus Numerical Rating Scale (PP-NRS4) at 2 weeks was 48.2% in the Cibinqo group, higher than the 25.5% of the Dupixent group. The proportion of patients who achieved a 90% improvement in the Eczema Area and Severity Index (EASI-90) at Week 4 was also significantly higher in the Cibinqo group (28.5%) than in the Dupixent group (14.6%). Moreover, the company also conducted the JADE EXTEND study, which studied the effect of switching patients who used Dupixent to Cibinqo. Patients were divided into those who were responsive to Dupixent and those that were non-responsive to Dupixent. 93.5% and 90.2% of patients who showed a response to Dupixent achieved EASI-75 after switching to Cibinqo 200mg and 100mg, respectively. PP-NRS4 was 89.7% and 81.6%, respectively. Among patients who did not respond to Dupxient, the proportion of those that reached EASI-75 was 80% and 67.7%, respectively, and PP-NRS4 was 77.3% and 37.8%, respectively. Efficacy of switchin gto Cibinqo(Abrocitinib) among patients thatresponsive to Dupxient(left) and those that were unresponsive (Data: Phase 3 efficacy and safety of abrocitinib in adults with moderate-to-severe atopic dermatitis after switching from dupilumab). The study provided evidence that patients who did not see an effect with Dupixent can expect an effect when they switch to Cibinqo. Previously, a clinical trial was also conducted with the JAK inhibitor Rinvoq on switching therapy from Dupixent. However, Cibinqo is the only drug that analyzed the effect of switching administration according to the patient’s presence or absence of an effect with Dupixent. This is why the trials have raised expectations of JAK inhibitors receiving expanded reimbursement as a replacement therapy when switching from Dupixent. Professor Na said, “The reason why the JADE EXTEND clinical trial is significant is that there are many patients who do not see an effect while using Dupixent, even though it is a good drug. However, these patients have to discontinue treatment and use immunotherapies for 3 months and see a deterioration in their condition before switching to a different treatment to receive reimbursement due to limited reimbursement standards. As Cibinqo has evidence prepared with clinical trials, the trials may be grounds to extend the drug’s reimbursement to switching medications. Also, For those who have not seen any effect after using Dupixent, the evidence is there to switch to Civinco.”
- Opinion
- [Reporter's view] AI drug development, should face reality
- by Hwang, Jin-joon May 16, 2023 05:39am
- "Artificial intelligence (AI) drug development will be useful, but there is a problem with only talking too much hope. Domestic AI drug development is still at an early stage, so more R&D is needed." These are the words the representative of an AI drug development company attending a recently held event related to digital healthcare threw to the debaters in the audience. It seems to point out that although new drug development using AI is being carried out in various parts of the world, companies that have achieved tangible results in Korea have yet to be seen. According to the Ministry of Science and ICT, new drug development is one of the projects that take an average of 10 years or more for a new drug to come to the market. It takes about 5 years to discover 5,000 to 10,000 promising candidates. Among the discovered candidate substances, 10 to 25 substances are entering preclinical trials. Of these, nine substances are approved for phase 1 clinical trial plans. The success rate is so low that there are only 5 in Phase 2, 2 in Phase 3, and only 1 in the market. In order to overcome the low success rate of new drug development and entry barriers that require long-term and high costs, a plan to utilize AI in the early R&D stage has been proposed. For traditional new drug development, a method of selecting a target disease and searching for a candidate substance by a research team reviewing about 400 to 500 related papers is used. AI is expected to have the advantage of being able to search for more than 1 million papers and hundreds of thousands of chemicals so that dozens of researchers can do the work that they have to do for 1 to 5 years more quickly. The AI drug development market is estimated to have high growth potential. The AI drug development market is expected to grow from 636.2 billion won in 2019 to 4.7693 trillion won by 2027, growing at an average annual rate of 28.63%. Global pharmaceutical companies such as AbbVie, Amgen, AstraZeneca, Boehringer Ingelheim, GSK, Janssen, Eli Lilly, MSD, Novartis, Pfizer, Sanofi, and Roche have also actively collaborated with AI drug developers to discover candidate substances. In 2021, a candidate substance for the treatment of pulmonary fibrosis discovered and designed through AI by Insilico Medicine in Hong Kong entered clinical trials. In October of last year, Verge Genomics, a US AI drug developer, started clinical trials of candidate materials for amyotrophic lateral sclerosis (ALS, Lou Gehrig), which were secured using AI's new drug discovery platform Converge. Verge Genomics is a biotech company that has received investments from Eli Lilly, MSD, WuXi AppTec, and BlackRock. In Korea, the Ministry of Science and ICT invested 25.8 billion won for three years from 2019 to 2021 to develop new AI drugs. The main goal was to build a customized AI platform for each stage of new drug development. It aimed to lead the development of three areas: candidate substance discovery, drug re-creation, and smart pharmacovigilance. Through a three-year support project in the field of AI new drug development, it developed an AI model that can be applied to neurodegenerative diseases and new anticancer drugs. The developed AI model is loaded into the public platform KAIDD and is openly operated so that researchers from industry, academia, and research institutes can utilize it. As a follow-up project that started in September of last year, the company plans to improve the data sharing and utilization environment by advancing the AI new drug development model by 2026. We plan to develop new drug candidates that can be applied for a clinical trial plan. There are several areas that need improvement in clinical prediction and targeting of clinical patient groups. It is also an important issue to establish social consensus and specific action plans that can utilize personal medical data classified as sensitive information. It is problematic that the AI drug development industry is not securing tangible results such as sales while talking only about the ideal use of technology, overseas cases, and the growth potential of the global AI market. It is necessary to face reality and solidify internal stability to be competitive.
- Company
- Samil, a series of love calls from multinational companies
- by Lee, Seok-Jun May 16, 2023 12:16am
- Samil Pharmaceutical is receiving a series of love calls from multinational companies. This time, it has decided to exclusively distribute and sell all Sandoz products, including the central nervous system (CNS). The CNS division, which was newly established in 2021, will gain momentum for business expansion. It is evaluated that Samil Pharmaceutical's expansion of partners is because it has met global standards in sales and marketing. Even before Sandoz, the company has partnered with global pharmaceutical companies such as GSK, Abbott, AbbVie, Mundipharma, and Beatrice. It is also affiliated with domestic companies such as Samsung Bioepis. Samil Pharmaceutical exclusively distributes and sells all products, including Sandoz CNS (Central Nervous System). The two companies recently signed such an agreement. Sandoz changed its Korean business to a 100% third-party distribution model. Sandoz is a subsidiary of Novartis in the generic and biosimilar business. Samil Pharmaceutical plans to strengthen the recently expanding CNS field while holding Sandoz products. In order to broaden the CNS lineup in 2021, the company established a new CNS division for sales of the existing neurology team and psychiatric products. In the same year, it signed an exclusive distribution and sales contract for Beatrice's Xanax, Zeldox, and Zoloft in the Korean market. A company official said confidently, "Samil Pharmaceutical will provide the best treatment options for domestic patients by further strengthening its powerful central nervous system disease lineup through the distribution and sales of Sandoz medicines." Sandoz currently has about 40 items in stock. In addition to CNS, it deals with chronic disease products such as Sandoz Amlodipine and Sandos Atorvastatin. This is also exclusively distributed and sold by Samil Pharmaceutical. This is where Samil Pharm's external expansion is expected. The company's sales last year were 179.7 billion won. It is expected to break through 200 billion won this year. Samil Pharmaceutical’s factory in Vietnam Samil Pharmaceutical is receiving a series of love calls from multinational companies in addition to Sandoz. Prior to Sandoz, it partnered with global pharmaceutical companies such as GSK, Abbott, AbbVie, Mundipharma, and Beatrice. There are also domestic companies such as Samsung Bioepis. It is evaluated that it is because it has met global standards in sales, marketing, and distribution. Sandoz Korea also explained that the decision to partner with Samil Pharmaceutical was made considering △company size, △professionalism in supplying medicines in ophthalmology, liver and stomach, musculoskeletal system, and CNS areas, and long-term collaboration experience and capabilities with many global companies. Samil Pharmaceutical has been making efforts to grow into a global company for several years. △ Established a subsidiary in Vietnam in 2018, △completed the construction of a CDMO plant for eye drops in Vietnam in 2022, △Established a North American subsidiary in Canada in 2022, and △Secured future growth engines by signing contracts for new drug candidates in the global phase 3 clinical trials such as Aramchol and Lorecivivint △ Domestic introduction of excellent foreign medicines, etc. In particular, the Vietnam global eye drop CDMO plant, which had a completion ceremony in November last year, is expected to serve as a stepping stone for global expansion. The factory can produce about 330 million eyedrops per year. It is aiming to obtain GMP approval for DAV in Vietnam and MFDS in Korea by the end of the year at the earliest. Within the next two to three years, the company plans to supply high-quality medicines to the global market through US FDA cGMP, European EMA EU GMP, and Health Canada GMP approvals.
- Policy
- Biopharmaceutical CDMO annual average of ↑31%
- by Lee, Hye-Kyung May 15, 2023 05:41am
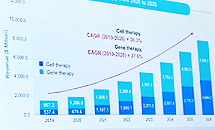
- Kwon Soon-jae, managing director of ENCell, is giving a presentation on the current status of the CDMO market at Bio Korea 2023.Globally, the CDMO market for biopharmaceuticals is growing at an average annual rate of 31%. If this trend continues, the size of the Cell&Gene Therapy CDMO market is expected to reach 10 billion dollars in 2026. Kwon Soon-jae, managing director of ENCell, announced this at the 'CDMO Partnership for Acceleration of Biopharmaceutical Development session held at the 'Bio Korea 2023' event held on the 10th. Director Kwon explained, "Cell & Gene Therapy is expected to grow 5.5 times in 2026 compared to 2019, and gene therapy is expected to grow 8.7 times." He explained, "If you look at the high CAGR from 2019 to 2026, it will account for 36.3% and 27.6%, respectively." The demand for CMOs and CDMOs has increased due to the COVID-19 pandemic, and Director Kwon said, "Small companies use CMOs and CDMOs to reduce costs and time, while large companies use CMOs and CDMOs to reduce marketing and R&D costs." It plays a part," he said. However, in the case of domestic CGT treatment, the manufacturing technology is complicated and the number of platforms is not large, so it was inevitable to create a GMP facility with an 'in-house' concept rather than CDMO service, and have many in-house processes and services. Director Kwon said, "However, as the requirements of the Ministry of Food and Drug Safety become stricter, infrastructure, raw materials, facility costs, labor costs rise, and technology becomes more complex, outsourcing instead of in-house is becoming a trend." Looking at the domestic CGT CDMO market, Lonza, Samsung Bio, SK, CJ, Lotte, and Medipost have announced their entry into the CMO/CDMO business following Thermoficer in 2017. Director Kwon said, "Most of the 30 CGT companies in Korea are major companies, and 80% of them are trying to develop AAC, adenovirus, CAR-T, etc., and only 30% of them are investing more than 2 million dollars." "If you look at the CGT market alone, it's still the first step, the introductory stage," he said. Director Kwon said that the present, when the first step of CDMO in the CGT market was taken, is an important point in determining the future. Director Kwon said, "More than 100 companies worldwide have entered the CDMO cell gene therapy market, and price, location, and regulations are challenges to be resolved." It looks like I'll have to give it a try," he said.
- Company
- New diabetes treatment guidelines released
- by Kim, Jin-Gu May 15, 2023 05:41am
- The status of GLP-1 analogues and SGLT-2 inhibitors has risen further in Korea’s new diabetes treatment guidelines. In the revised guidelines, when considering options to use in combination with injection therapy, GLP-1 analogues were recommended over basal insulin, and SGLT-2 inhibitors were recommended first for diabetic patients with heart failure, kidney disease, or cardiovascular disease. Kyu-Chang Won, Chairman of KDA (Endoctriology, Yeungnam University Hospital) is introducing KDA On the 12th, Korean Diabetes Association introduced the key contents of its ‘KDA clinical practice guidelines for diabetes: 8th edition’ at the 2023 Spring Conference of the Korean Diabetes Association that was held at the Kimdaejung Convention Center in Gwangju. The KDA publishes treatment guidelines for diabetes every 2 years. The association organized a clinical practice guideline committee around Director Min Kyong Moon (Internal Medicine, Seoul National University Medical School) and developed the new practice guidelines from January last year to April this year. The most notable change observed in the new guideline is the use of GLP-1 analogues and SGLT-2 inhibitors. In general, the two classes of drugs were more strongly recommended. GLP-1 analogues currently available in Korea include Trulicity (dulaglutide) and Ozempic (semaglutide), and SGLT-2 inhibitors include Forxiga (dapagliflozin) and Jardiance (dapagliflozin). ◆GLP-1 analogues = The new clinical practice guidelines ‘recommend GLP-1 analogues over basal insulin when considering injection-based combination therapy.’ Previously, treatments in combination with injection therapy were recommended as priority for potent blood sugar-lowering effect. In addition, GLP-1 analogues in combination with basal insulin were recommended to enhance blood sugar control. The newly revised guidelines went a step further from the previous guidelines to recommend GLP-1 analogues first. Jung-Hyun Noh, professor of internal medicine at Inje University, said, “In a study that directly compared the results of using GLP-1 analogues+oral medications to separate use of GLP-1 analogues or insulin, there was no significant difference in their HbA1c level reduction effect. However, the use of GLP-1 analogues was found to maintain the lowering effect longer.” In addition, the content that if the target blood sugar level cannot be achieved with either the GLP-1 analogue or basal insulin alone, the two drugs shall be used together, was newly added to the guideline. It was also added that if the target blood sugar level is not reached even with GLP-1 analogue or basal insulin treatment, insulin potentiation therapy should be attempted. ◆SGLT-2 inhibitors = SGLT-2 inhibitors were also more strongly recommended. The revised guideline recommends, "'in case of accompanying heart failure, the use of SGLT-2 inhibitors with proven heart failure benefits is recommended in priority regardless of HbA1c level, and the treatment should be continued unless contraindications or side effects arise.' In the case of renal disease as well, the revised guideline recommend 'in case of albuminuria or decreased estimated glomerular filtration rate, SGLT-2 inhibitors with proven renal benefits should be used first regardless of HbA1c level, and be maintained unless contraindications or side effects arise." Previous guidelines have recommended ‘treatment including SGLT-2 inhibitors to be considered first in patients with heart failure.’ Here, the phrase 'regardless of the HbA1c level’ was newly added with the revision. Professor Noh said, “New studies have demonstrated the benefit of SGLT-2 inhibitors in heart failure and renal disease. The effect was consistent in patients without diabetes. This is why we recommended the use of SGLT-2 inhibitors if there is a risk of disease, regardless of HbA1c level." In addition, it was recommended that "treatment including GLP-1 analogues or SGLT-2 inhibitors with proven cardiovascular benefits should be prioritized in patients with atherosclerosis cardiovascular disease. Previously, SGLT-2 inhibitors were recommended as monotherapy for such patients, but this time, GLP-1 analogues were added to the recommendations."
- InterView
- ‘Even a 0.2 vision is a miracle to some’
- by Eo, Yun-Ho May 15, 2023 05:41am
- Professor Suk Ho Byeon The reason for the slow development of new drugs in a specific disease can usually be attributed to one of the following two reasons. Low disease awareness or difficulty in developing the drug itself. The one-shot gene therapy Novatis’s ‘Luxturna (voretigene neparvovec)’ is a drug that overcame both barriers. Luxturna, which is a treatment for IRD (Inherited Retinal Dystrophy) caused by the mutation in both copies of the RPE65 gene, is the first treatment option developed for the difficult rare genetic condition. IRD is a rare intractable disease caused by a mutation in the gene responsible for the structure and function of retinal visual cells. It includes over 20 types of ophthalmologic conditions, and around 270 causal genes are known to be implicated in IRD. RPE65-IRD, caused by the mutation in both copies of the RPE65 gene, causes abnormalities in the visual cycle of the retina that converts visual information into a neural signal and delivers it to the brain. The mutation in the RPE65 gene reduces the RPE65 protein essential to the visual cycle and destroys the retinal cell, gradually narrowing the field of vision to eventually result in loss of vision. With only 6 patients found with IRD in Korea, patients with the condition reach legal blindness in their adolescence, at about 16 to 18 years of age, then progress to complete blindness. Due to the lack of a fundamental cure, only conservative treatment that could temporarily delay symptoms was available until now. WIth its release, Luxturna became the first drug introduced into the field that could prevent blindness. Dailypharm met with Professor Suk Ho Byeon, Department of Ophthalmology at Sinchon Severance Hospital to hear about the significance of RPE65-IRD and Luxturna. Professor Byeon had recently coauthored a consensus paper on RPE65-IRD published in the Korean Journal of Ophthalmology, an English journal published by the Korean Ophthalmological Society. -The consensus paper you released recently seems to have covered the whole content on RPE65 mutation-associated IRD starting from its concept. It felt more like a clinical practice guideline. Does the fact that this paper was published signify the lack of content on the diagnosis and management of RPE65-IRD in Korea? It can be said so. Due to the characteristics of the disease, treatment is rare in any form of IRD. Not many doctors have experience using drugs to treat the disease. So we tried to relay the existence of such a disease and the need to look for the disease. With the first drug released fRPE65-IRD, hope is rising among all IRD patients including those with genetic mutations other than the RPE65 mutation. However, since there was little information arranged on the disease itself as well as its diagnosis and treatment, we decided to bring together experts who had experienced or were familiar with the disease in Korea to arrange the information on the disease. The consensus paper includes information on what RPE65-IRD is, what kind of patients are considered to have IRD, its global epidemiology and epidemiology in Korea, to which extent genetic mutations are reported, and how and whom should receive gene tests for the disease. -As you’ve mentioned, the consensus paper includes content on finding the subjects that should receive genetic testing. Which parts should the doctor check during gene testing? One of the most prominent characteristics in these patients is that they had poor eyesight from an early age. This may be difficult to notice during infancy, but over time, the patient’s eyesight decreases significantly compared to normal people and is accompanied by night blindness. However, with so many areas bright at night, night blindness is sometimes discovered late these days. Patients with severe symptoms also experience eye tremors. However the features of the disease can vary even among patients affected with the same genetic mutation. Some patients show fewer symptoms and some do not have night blindness. Therefore, the disease is difficult to identify based solely on symptoms. Therefore, in pediatric patients, I think it is better to proceed and conduct a genetic test even if at the smallest suspicion. -So Luxturna was released in the field that had no available drugs. The drug received attention as the first gene therapy for ophthalmic diseases. What is your opinion on the value of the drug? That would be difficult for general ophthalmologists to judge. I can better explain its significance because I have experience treating patients with Luxturna. Patients with poor eyesight ever since childhood rarely visit the hospital, so doctors often do not know how these patients are faring. In that sense, it is difficult to estimate how much a patient's life would have improved with improved eyesight. Ophthalmologists measure both visual acuity and visual fields. This index of visual acuity and field of view measures how far off the patients’ eyesight is from those of normal people. Patients who were treated with Luxturna showed much improvement in the visual field and visual acuity. The improved visual acuity was about 0.2. For non-patients, visual acuity of 0.2 may feel like a poor condition, but for those that had almost no vision, even a 0.2 vision can be of great benefit. I was very surprised to see a patient I treated working part-time at a coffee shop. A patient who could go blind escaped the danger with Luxturna. In this sense, the effect of such treatment should not be judged based on standards set for normal eyes. - Luxturna received the non-reimbursement decision last month from the National Health Insurance Review and Assessment Service’s Drug Reimbursement Evaluation Committee. The committee pointed to how the condition is not life-threatening and is a high-priced drug as a barrier to reimbursement. There also seems to be a difference between the government and pharmaceutical companies regarding reimbursement standards. Reimbursement is a complex issue that requires broad and serious considerations. Since Luxturna is a gene replacement therapy, I know there have been disagreements on the remaining target cell and the criteria for recognizing its effect. As a doctor, I can definitely say that there can be no crystal-clear standard for evaluating living cells. Some cases can only be determined after treatment. Since it is such a rare condition, it is difficult to even estimate how many cases there will be.
- Company
- MSD Korea begins voluntary retirement
- by Jung, Sae-Im May 15, 2023 05:40am
- MSD Korea, which announced a reduction in manpower due to the abolition of the Januvia division, has begun a full-fledged reduction in personnel by disclosing the conditions for ERP. The labor union of MSD Korea warned of a tough response, saying, "We cannot accept the company's attempt to reduce manpower." On the 12th, MSD Korea announced the conditions for voluntary retirement for GM division employees. Employees who apply for voluntary retirement are paid a monthly basic wage equal to the number of months equivalent to the value of adding 10 to twice the number of years of service as a basic severance pay (2n+10). The payment limit is limited to a maximum of 48 months. In addition, an additional 20 million won is paid as severance compensation. For example, if an employee with 10 years of service applies for voluntary retirement, he or she will receive a monthly base salary equivalent to 30 months and an additional 20 million won. However, if the total severance pay exceeds the 'amount calculated by multiplying the number of remaining months until retirement by the monthly basic salary', the excess amount is deducted. This voluntary retirement program is aimed at about 100 employees in the GM division. The company began reorganization by handing over all rights of the diabetes treatment 'Januvia series' sold by the GM division to the domestic pharmaceutical company Chong Kun Dang. The plan is to get rid of the GM division, which mainly sold chronic disease treatments, and leave only three divisions: anticancer drugs, vaccines, and hospital specialties. The downsizing of the division was also accompanied by a reduction in personnel. The labor union strongly opposed the company's attempt to cut personnel. Since there was no urgent management problem to abolish the business unit, it is a position that it cannot agree to all attempts to reduce personnel, such as ERP, individual meetings, external career support programs, and layoffs. The union said, "According to the Labor Standards Act and Article 18 of 'Employment Security' of the MSD Korea Collective Agreement, the union and all members will not participate in any sessions, including one-on-one meetings scheduled for the future." In addition, the union said, "The union and all union members will fight strongly by mobilizing all available capabilities to defend the right to live."
- Company
- Advate leading hemophilia treatment
- by Nho, Byung Chul May 12, 2023 05:45am
- In the field of hemophilia non-antibody and antibody treatment, Takeda Korea Pharmaceutical and JW Pharmaceutical are gaining attention as they are strengthening their positions. Korea Takeda Pharmaceutical's 'Advate/Adynovate', and JW Pharmaceutical's Hemlibra's sales last year were 26.2 billion won and 7.6 billion won, respectively, ranking first in related markets. Of particular note is that the hemophilia antibody treatment market is fluctuating in line with the launch of Hemlibra in 2020. Due to the emergence of strong competing products, the external expansion of traditional powerhouses Novoseven RT and Faiva is drawing a steep downward curve. Novoseven RT·Faiva's sales from 2018 to 2022 show a decreasing trend from '19.5 billion - 5.4 billion won' to '6.1 billion - 3.6 billion won'. In the non-antibody treatment market, six domestic and foreign pharmaceutical companies, including GC Green Cross, Pfizer Korea, Sanofi, and Bayer Korea, are competing fiercely with Takeda at the forefront. Based on the sum of pharmaceutical product lines, Takeda's 'Advate/Adynovate -19.4 billion won/6.7 billion won' and GC Pharma's GreenMono/Greengene F -6.5 billion won/2.7 billion won' ranked first and second, respectively. Pfizer's Xyntha Solofuse, Sanofi's Eloctate, and Bayer's Kogenate FS achieved 5.1 billion won, 3 billion won, and 1.8 billion won last year. The newly released CSL Bering Korea Afstyla in 2021 showed a performance of 40 million won in the first year and about 100 million won last year. The peculiarity is that except for Adynovate and Eloctate, the performance of most non-antibody treatment products is showing a downward trend, and the market also decreased by nearly 5 billion won last year compared to 50 billion won in 2018, five years ago. The antibody treatment market also recorded a sales peak of 25.7 billion won in 2018, followed by a 35% decline with sales worth 16.7 billion won in 2022. Hemophilia is largely divided into hemophilia A (factor VIII deficiency), hemophilia B (factor IX deficiency, also known as 'Christmas disease'), hemophilia C (factor XI deficiency), para hemophilia (factor V deficiency), and pseudo hemophilia. (pseudo hemophilia) or von Willebrand disease (von Willebrand factor deficiency). The incidence of hemophilia A patient is about 1 in 5,000 to 10,000 normal-born males, and hemophilia B is about one-fifth of this. Von Willebrand disease occurs in both men and women and occurs in about 1 in 1000 cases. It is estimated that there are 600,000 hemophilia patients worldwide, and about 12% of all hemophilia patients are hemophilia B patients. In Korea, the number of patients with hemophilia A is the highest at 2,000, followed by 400 patients with hemophilia B, 100 patients with von Willebrand disease, and 20 patients with hemophilia C.