- LOGIN
- MemberShip
- 2026-04-11 03:01:27
- Policy
- Largest-ever drug pricing reeval to be conducted this year
- by Kim, Jung-Ju Jan 02, 2023 06:04am
- The health and pharmaceutical industry is busy preparing various systems and policies for the new year. The reimbursement adequacy reevaluations for listed drugs that had been initiated as a pilot project on choline alfoscerate products settled as an annual policy project and are expected to start full-scale reevaluations on previously notified products. High-priced drugs for pediatric patients will now be eligible for pharmacogenomic evaluation data exemptions (PE exemptions) and subjects for the special calculation system will also be expanded. In addition, the 3rd Comprehensive Plan to Foster the Pharma and Bio Industry will be initiated, and the task force for the development of the service industry that includes the healthcare industry will also be launched within the month and impact the medical and pharmaceutical industry. The National Assembly will discuss the abolition of the sunset clause on state funding for national health insurance and legislation for public late-night pharmacies. Dailypharm prepared a summary of notable changes in the system as well as policy projects that will be made in 2023 in the pharmaceutical and biopharmaceutical industry. First, the conversion factor for medical care facilities will be raised with the start of the new year. The increase rate is 1.6% for hospitals, 2.1% for clinics, 3.0% for oriental medicine hospitals, and 3.6% for pharmacies. Along with this, the health insurance rate will also rise to 7.09% based on those employed. For drug pricing coverage and control, high-priced drugs for pediatric patients may be allowed PE exemptions starting this month. PE exemption may be allowed for drugs used to treat pediatric patients that are therapeutically equivalent or has no available treatment option; and demonstrates improvement in quality of life or are otherwise approved by the committee. The adjustment in the reference countries used to calculate the foreign adjusted drug price that raised concerns in the pharma and bio industry has been made. The government added Canada to the existing A7 (Japan, France, Germany, Italy, Switzerland, the UK, and the US) to A8. Australia, the country that the industry worried would be included as a reference country, was excluded. The reimbursement reevaluations planned for the year are expected to be the largest ever. The government announced that HIRA will be reevaluating reimbursement on a total of 8 ingredients - Rebamipide, Limaprost alpha-cyclodextrin, oxiracetam, acetyl L carnitine, loxoprofen sodium, levosulpiride, epinastine hydrochloride, sodium hyaluronate eye drops. When adding the efficacy reevaluation set to be conducted on sterepto drugs that received conditional approval for 1 year, the number of drugs subject to reevaluation is increased to 9. The discount rate for the drugs will be finalized in the coming December. In February this year, ingredients subject to reimbursement reevaluations in 2024 will be decided upon and reviewed by the Health Insurance Policy Deliberation Committee. Also, a performance evaluation procedure and management plan for high-priced drugs such as Kymriah and Zolgensma, the so-called 'one-shot treatments', will be prepared within the month. Also, the negotiation period for drugs eligible for the Risk Sharing Agreement (RSA) among PE exemption drugs will be shortened by 30 days through the introduction of a prior discussion system, improving accessibility. Also, the scope of drugs eligible for special calculations will be expanded. A total of 42 new rare diseases and artificial kidney dialysis patients with chronic renal failure will also be applied to special calculations. Also, the administrative dispositions used for the drug serial number reporting system will be raised. From January, HIRA will raise the serial number reporting rate standard for administrative dispositions from 85% to 90% at the time of shipment by wholesalers (including manufacturers and importers that supply third-party licensed items). Therefore HIRA will request administrative dispositions to be made to local governments if the reporting rate becomes less than 90%. Also, the ‘ Regulation on Manufacture and Sales Management of Biological Products, Etc.’ that applied to all biological products will not be categorized into 3 product types. The MFDS plans to make the amendments to the regulation before the 17 of this month. The 3rd Comprehensive Plan to Foster the Pharma and Bio-Industry which had been set as a 5-year project will be implemented this year to support all areas ranging from R&D to business development, investment exports, job creation, and institutional infrastructure. The 'Service Industry Development TF', which sets policy directions to revitalize the service industry, including the field of healthcare, will also be launched in earnest this month. The Ministry of Economy and Finance that oversees the project plans to disclose its 5-year plan for innovating the service industry in March through the TF. During the H1 of the year, a pilot project for the ‘Approval Evaluation-Negotiation Linkage System' will be carried out, and subject drugs will simultaneously undergo three tracks of the drug approval process – MFDS’ safety and efficacy evaluations for marketing authorizations, HIRA’s reimbursement adequacy evaluations, and NHIS’s drug pricing negotiations. In the same period, the National Assembly will discuss whether to abolish the sunset clause for state support of health insurance finances and whether to legislate public late-night pharmacies. Although the contract sales organization (CSO) reporting system is expected to be discussed at the plenary session in the H1 of the year, as the effective date for the law is 1 year and 6 months after the promulgation, its actual application is expected to be possible only in the 2024 2H at the earliest, even if the revisions are made rapidly.
- Policy
- SK Chemicals Riluzole is listed
- by Lee, Tak-Sun Jan 02, 2023 06:04am
- Only tablet type is available for Riluzole, and suspension type is expected to be an alternative for patients with difficulty in swallowing. According to industries on the 1st, SK Chemical's Teglutik Suspension was listed at 134,970 won per bottle. Riluzole, the main ingredient of Teglutik, is used in the treatment of Amyotrophic Lateral Sclerosis (ALS), which is known to delay symptoms by several months. Riluzole is the first drug approved for Lou Gehrig's disease and is most commonly used. There are two products in Korea: Rilutek and Yooritek by Riluzole. Based on IQVIA in 2021, Rilutek showed sales of 3.7 billion won and Yuritech 2.5 billion won. Despite such identical products, Teglutik is introducing them to the domestic market because there is a demand for suspension. Patients with Lou Gehrig's disease have early symptoms of weak tongue and neck muscles, making it difficult to chew and swallow. It was difficult for patients who felt these symptoms to take two tablets of Riluzole daily. Teglutik is a suspension that can be easily taken through oral injections. Patients who are difficult to take directly can be injected through a PEG tube. The drug was developed by the Italian pharmaceutical company Italco Pharma and approved by the U.S. Food and Drug Administration (FDA) in 2018. SK Chemicals has completed patent registration since it was introduced in Korea. The upper limit of the benefit 134,970 won per bottle, is 15 days' worth, which is the same as the existing refining price (4,499 won per day) when converted into daily doses. Since there is no price difference, Teglutik is expected to be useful for patients with symptoms of dysphagia. On the other hand, Sanofi and Yoo Young, which divided the market, are expected to focus on protecting the market this year because they have difficult competitors.
- Policy
- High conc Ultomiris Inj 100mg/mL also approved in Korea
- by Kim, Jung-Ju Jan 02, 2023 06:04am
- Handok received marketing authorization for the orphan drug ‘Ultromiris 100mg/mL(ravulizumab)’ that is used to treat Paroxysmal nocturnal hemoglobinuria (PNH) and atypical hemolytic uraemic syndrome (aHUS) and made a step towards supplying the drug in Korea. On the 28th, the Ministry of Food and Drug Safety announced that it had granted marketing authorization for the drug. The drug is a humanized monoclonal antibody (mAb) that specifically binds to the C5 protein and inhibits complement-mediated inflammation and hemolysis, etc. More specifically, the drug is indicated for the treatment of adults with paroxysmal nocturnal hemoglobinuria (PNH), and as a treatment for adult and pediatric patients with atypical hemolytic uraemic syndrome (aHUS) and inhibits complement-mediated thrombotic microangiopathy (TMA). The MFDS explained that the product approved this time is a high-concentration version of the already-approved Ultomiris Inj and has the benefit of being able to reduce the IV infusion time in patients. The MFDS said, “We will continue to make efforts to allow for the prompt provision of treatments with confirmed safety and efficacy based on regulatory science."
- Product
- How is the non-face-to-face tx/delivery going overseas?
- by Jan 02, 2023 06:04am
- With the platform industry and the medical community preparing first ahead of the institutionalization of non-face-to-face treatment next year, calls for measures to be prepared for drug delivery are gaining momentum. A recent study report titled "Non-face-to-face treatment prerequisites" by the Korean Medical Association's Medical Policy Institute (Director Woo Bong-sik), which argued that ▲ face-to-face treatment principles, ▲ non-face-to-face treatment, and non-face-to-face treatment should be an auxiliary means ▲ national health security medical association. "Most countries allow drug delivery," the Korea Institute for Medical Policy said. "The government is also in a position that it is difficult to proceed with non-face-to-face treatment and drug delivery in a separate policy, but recently, it is necessary to address drug concerns and devise measures to increase the effectiveness of the policy." It also introduced overseas cases such as Japan, Australia, Britain, France, the United States, Canada, China, and India. In fact, it was found that drug delivery was allowed in Japan, Australia, the United States, and China due to COVID-19. In Japan, as the amount of non-face-to-face treatment increased after COVID-19, drug delivery was officially allowed through joint notification by the Health, Labor and Welfare Ministry's Medical Policy Bureau and the Pharmaceutical and Household Sanitation Bureau on April 1, 2020. Japan is a country in the form of voluntary division of labor, and when prescribed in the hospital, it can be delivered directly from the medical institution to the patient's home For out-of-house prescriptions, the prescription must be faxed to the pharmacy, and the original prescription must also be sent separately to the pharmacy. After consulting with the patient on medication guidance, pharmacists should prepare appropriate ways to deliver drugs that need special attention and drugs that need rapid delivery, or ask the patient's family to visit the pharmacy. From April 17, 2020, Australia will also be able to receive prescriptions by text message, email, or fax after non-face-to-face treatment. The prescription allows the patient himself or his guardian to go to the pharmacy or receive the medicine home after sending it to the pharmacy he or she wants. If you do not know which pharmacy to send the prescription to, the doctor who treated the patient can suggest a pharmacy in the area or the patient can directly search for a pharmacy in the area using the Health Direct website to select a pharmacy. NHS Digital in the UK abolished the paper prescription issuance system in November 2019 and began issuing electronic prescription services (EPS). After GP treatment, the patient can receive the medicine at the pharmacy by sending the prescription to the pharmacy designated by the patient. When taking drugs regularly, repeated prescriptions are issued, and even if you take all of them, you can order them when necessary without follow-up treatment with your GP until the next drug prescription review. However, only if the GP sets these prescriptions, the patient can request the drug delivery to the pharmacy, and if the drug delivery service is provided by the pharmacy designated by the patient, the drug can be received by courier. France provides drug delivery services to non-face-to-face patients. After non-face-to-face treatment, use the Ordo Express app to scan the prescription and send an electronic signature. The app is sent in the form of an e-mail and an encrypted prescription is sent to the pharmacist selected by the patient to ensure safety. Drugs can be delivered to the patient's home or workplace, and drugs can be delivered to all types of drugs, except drugs, regardless of the presence or absence of prescription. At this time, patients can track their drug delivery process in real time and contact the delivery agent in charge through a real-time chat system to shorten the delivery process. In the United States, doctors are required to send prescriptions to pharmacies selected by patients using e-mail or smartphones after non-face-to-face treatment. After Amazon Pharmacy, an online pharmacy, was officially launched in 2020, a prescription drug delivery service was launched, and the medicine is delivered to the patient's home on the same day. In addition, pick-up services through drive-through were actively introduced to pharmacies in the wake of COVID-19, and the unmanned autonomous vehicle "Nuro R2" was used to deliver medicines to reduce unnecessary face-to-face contact and ensure accurate and safe delivery of necessary medicines. In February 2020, the U.S. federal government implemented special measures to allow unmanned autonomous vehicles to enter public roads in order to deploy autonomous vehicles in the COVID-19 situation, allowing them to receive drug delivery, starting with Nuro headquarters in California. Canada is one of the countries where non-face-to-face treatment is active. Canada requires doctors to send prescriptions to the pharmacy they want by fax or e-mail after non-face-to-face treatment. When a patient is ready to pick up the drug, he or she can be notified through a non-face-to-face treatment app or delivered directly to the patient's home. Among Canadians, it was found that one in three people who used non-face-to-face treatment were using non-face-to-face treatment for prescribing and delivering drugs. In China, the ban on online sales of drugs was lifted in 2018, and the policy of using Internet hospitals was announced. It is a government-level intervention, and the State Council, the National Health Commission, and the National Insurance Service have changed their policy of delivering drugs after non-face-to-face treatment, stressing the importance of actively expanding and succeeding in Internet hospital work covered by health insurance. In China, if a doctor issues a prescription after receiving medical treatment at an Internet hospital designated by a state-run health agency in each province, a pharmacist in the in-house screening department can check electronic prescription and deliver the medicine to the patient or receive it directly from a nearby pharmacy. In China, online re-examination prescriptions can be issued only for general and chronic diseases diagnosed with the disease in face-to-face first-time visits in advance. In China, Internet hospitals have become a service platform that integrates online treatment and counseling, prescription and drug delivery, and online and offline diagnosis and treatment. India's Ministry of Health has allowed the delivery of medicines listed in e-mail prescriptions sent from hospitals to pharmacies to the patient's front door after the announcement of a notice on drug delivery permits for non-face-to-face care in March 2020. However, certain antibiotics and psychotropic drugs cannot be delivered. The Institute for Medical Policy said, "In order to achieve the purpose of the non-face-to-face treatment policy, drug delivery should be allowed. "Since there are many concerns and opposition from the pharmaceutical society, it is necessary to devise measures to address the concerns of the pharmaceutical society and increase the effectiveness of the policy," he explained.
- Company
- U.S. first possible NASH therapy
- by Jan 02, 2023 06:04am
- ↑Expectations for the first NASH treatmentThe U.S. Madrigal Pharmaceuticals has succeeded in phase 3 clinical trials of Resmetirom, a new drug candidate for non-alcoholic fatty hepatitis (NASH). When new drugs are approved, Resmetirom appears as the first NASH treatment aimed at the 33 trillion won market. Yuhan Corporation, Hanmi Pharmaceutical, and LG Chem are also speeding up the development of NASH new drug candidates through technology transfer or its own clinical progress. On the 20th, Madrigal Pharmaceuticals announced that it has secured a positive top line in phase 3 clinical trials (MAESTRO-NASH), which studies Resmetirom as a NASH treatment. Resmetirom is a thyroid hormone receptor (THR)-beta agonist administered orally. NASH is a disease in which fat accumulated in the liver develops into inflammation, causing liver fibrosis and cirrhosis. It is highly likely to develop in obese and diabetic patients. According to Evaluate Pharma, a pharmaceutical market research company, the NASH treatment market is expected to grow more than 20 times from 277.1 billion won this year to 6.723 trillion won by 2026. It is expected to expand to 33 trillion won by 2030. Phase 3 clinical trials are a study that analyzes efficacy and safety for up to 54 months after administering Resmetirom 80 mg/100 mg and placebo for 52 weeks in 955 patients with confirmed NASH. The first evaluation index is "the standard for resolving more than two points of fibrosis without worsening liver fibrosis" and "the standard for improving the first stage without worsening fibrosis." It was effective in both the 80 mg and 100 mg groups. The clinical trial was successfully completed by obtaining statistical significance compared to the placebo group. The secondary evaluation indicators are 'whether LDL cholesterol decreases at week 24' and 'based on liver enzymes'. The 80 mg group and the 100 mg group reduced LDL cholesterol by 12% and 16%, respectively, to obtain statistical significance. The placebo group increased by 1%. A significant decrease was also confirmed in the liver enzyme standard. Compared to placebo, it was confirmed that fibrosis decreased through biomarkers and imaging tests in the Resmetirom treatment group. Resmetirom was safe and drug resistant in both 80 mg and 100 mg doses. The frequency of severe side effects (SAEs) was similar across the target group, with each group showing around 12%. Paul Freedman, CEO of Madrigal Pharmaceuticals, said, "This clinical trial is reasonably likely to predict Resmetirom benefits and has achieved both major indicators proposed by the U.S. Food and Drug Administration," adding, "We will submit an application for a new drug to approve resmetirom acceleration in the first half of 23." Domestic pharmaceutical companies are also speeding up the development of NASH treatments. Yuhan Corporation signed a technology transfer contract with global pharmaceutical companies Beringer Ingelheim and Gilead, respectively, in 2019. NASH's new drug candidate "YH25724," which was transferred to Beringer Ingelheim, is undergoing phase 1 clinical trials in Europe. YH25724 is a candidate material developed by Yuhan Corporation. In this process, it is a fusion protein that combines "Hybrid FC" (HyFc), an antibody fusion protein platform technology of bio company Genexine. There are two types of synthetic materials transferred to Gilead, Yuhan Corporation and Gilead will jointly conduct non-clinical research, and Gilead will conduct global clinical trials. Hanmi Pharmaceutical transferred NASH's new drug candidate "Dual Agonist" to MSD in 2020. MSD has been undergoing phase 2 global clinical trials since last year. Another candidate substance, LAPSTriple Agonist, was also designated by the FDA as a fast track development drug for NASH treatment. If it is designated as a fast-track drug, it can have close consultations with the FDA at each stage of development and receive full support. LG Chem is developing its own NASH new drug candidate "LG203003" through the LG Chem Life Science Innovation Center in Boston, USA. In March, the FDA approved a phase 1 clinical plan (IND). In July, phase 1 was launched for 88 healthy adults and NASH patients to observe safety and drug resistance, pharmacokinetics, pharmacology, and changes in fat volume in the liver. LG203003 is a mechanism that suppresses fat accumulation in liver cells by selectively interfering with the activation of DGAT-2, a neutral fat synthase. LG Chem also has another NASH candidate, 'LG303174. It inhibits the expression of VAP-1 protein, which is known to affect inflammation in the liver. In August 2020, LG Chem signed a technology transfer contract from TransThera Biosciences in China to introduce LG303174's right to global development and commercialization.
- Company
- The dismissal of 18 employees of Zuellig Pharma is unfair
- by Jan 01, 2023 10:43pm
- According to the pharmaceutical industry on the 29th, the 11th Civil Affairs Department (chief judge Park Tae-il) of the Seoul Western District Court ruled in favor of some of the plaintiffs in a lawsuit filed by 18 fired Zuellig Pharma SSK against the company on the 22nd. According to the Democratic Pharmaceutical Union, the first trial court judged that "the company's layoffs themselves are invalid due to illegal and unfair dismissal in violation of the current Labor Standards Act." In order for layoffs to meet the prescribed requirements of the Labor Standards Act, they must meet ▲ urgent management needs ▲ company's efforts to avoid dismissal ▲ fair dismissal standards, and target selection ▲ consultation procedures 50 days in advance. The court said, "The layoffs did not meet the urgent management needs and did not make efforts to avoid dismissal." Considering the fact that workers endure unpaid leave at a time when operating losses in 2020 are insignificant, only a small number of customers can operate their business units, and labor costs are mostly operating costs, it is difficult to say that they are in an urgent management crisis. In addition, it was difficult to say that the government took measures to minimize the scope of dismissal, given that it decided to abolish its business unit immediately just a month after the strike ended, which led to the departure of the customer base, which led to the layoffs. Zuellig Pharma SSK, Zuellig Pharma's digital marketing division specializing in healthcare, is divided into a marketing division and a PC (Patient Card) division. The layoffs were made for the PC division. Zuellig PharmaSSK announced a voluntary retirement and dismissal plan in December 2020 along with the abolition of the PC division due to management difficulties. The layoffs took place on March 31, 2021, about three months later. The Democratic Pharmaceutical Union claimed that the company's layoffs were intended to suppress the union. When members of the Zuellig Pharma SSK branch went on strike due to the breakdown of wage negotiations, they fired all members by abolishing the business unit. Four months after the layoffs, SSK CEO Irwan Wolf left Korea after his term in office, making it difficult to hold him accountable. In response to the court's ruling on unfair dismissal, the Democratic Pharmaceutical Union said, "The company seems to be willing to proceed with the appeal, but it will continue its legal struggle until the end for the legitimate reinstatement of laid-off employees."
- Company
- Pharmaceutical shares capitalization of ₩56 trillion
- by Chon, Seung-Hyun Jan 01, 2023 10:42pm
- This year, pharmaceutical bio companies' stock prices were sluggish. It has never recovered from its peak on the first day of the stock market opening. The market capitalization of major companies fell by more than 56 trillion won. More than two out of ten have seen their market capitalization shrink by less than half in a year. According to the Korea Exchange on the 29th, the KRX Healthcare Index closed this year at 2,634.49. It fell 29.2% in a year from 3721.17 at the end of last year. The KRX sector index is an index that divides listed stocks in the securities market and KOSDAQ market into 17 industrial groups and selects and calculates representative stocks for each industry group. KRX Healthcare consists of 83 major pharmaceutical bio shares selected by the exchange. This means that stock prices of major pharmaceutical bio companies listed in Korea fell 29.2% on average. This year, pharmaceutical bio companies' stock prices have remained sluggish throughout the year. The KRX Healthcare Index recorded 3751.82 on January 3, the first day of its opening this year, the highest this year. The KRX Healthcare Index fell 27.4% in six months to 2,723.45 on June 23 but recovered 20.6% to 3,284.61 on August 9. However, it fell again, falling to 2,463.10 on October 13. It fell 34.3% in nine months compared to January 3. The KRX Healthcare Index has been on a slight rise since November but failed to make up for the previous decline. As a result, the KRX Healthcare Index fell sharply for the second consecutive year. The KRX Healthcare Index soared 89.3% from 2915.31 to 5517.31 in 2020 but fell 32.2% to 3742.91 last year. At the end of this year, the KRX Healthcare Index shrank 52.3% compared to two years ago. This year, most of the market capitalization of major pharmaceutical bio companies fell sharply from the end of last year. As of the end of last year, the market capitalization of 92 pharmaceutical bio companies that made up the KRX Healthcare index totaled 222.3191 trillion won, down 55.834 trillion won to 166.4851 trillion won in a year. The market capitalization of major pharmaceutical bio companies has decreased by 25.1% in a year. 86 out of 92 major pharmaceutical bio companies saw their market capitalization decrease compared to the end of last year. SK Bioscience evaporated 11.5689 trillion won from 17.2125 trillion won in market capitalization at the end of last year to 5.6436 trillion won in a year. In June, SK Bioscience was the first Korean company to receive a license for SKY Covione multi, a vaccine for preventing COVID-19, as the 35th new drug developed in Korea. However, as SKY Covione was not used much and the size of the COVID-19 vaccine consignment business of multinational pharmaceutical companies decreased, the stock price fell significantly. The stock price of SK Bioscience was 73,500 won, down 67.3 percent from 225,000 won at the end of last year. Celltrion's market capitalization was 22.5992 trillion won, down 4.7143 trillion won from 27.3135 trillion won at the end of last year. Celltrion Healthcare decreased by 3.2552 trillion won from 12.43 trillion won to 9.1781 trillion won during the same period. Celltrion Pharmaceutical's market capitalization has decreased by KRW 2.246 trillion over the past year. SD Biosensor, SK Biopharm, Seehene, Mezzion, Hanmi Science, Samsung Biologics, Alteogen, BNC Korea, and GC Pharma saw their market capitalization fall by more than 1 trillion won this year. The market capitalization of 20 out of 92 major pharmaceutical bio companies has decreased by more than 50% in a year. Mezzion's market capitalization fell 82.0% from KRW 1.932 trillion to 347.6 billion won. BNC Korea, Anterogen, Cellid, Ubiotics, Eyegene, and Aptabio saw their market capitalization fall by more than 70% in a year. Enzychem, Genexine, SK Bioscience, Medpacto, Prestigebiologics, Korea Pharma, Geneone Life Science, Seegene, GC Cell, PCL, United Pharmaceutical, and Olivia Pharma have seen their market capitalization fall by less than half compared to 1 year ago.
- Policy
- Australia, excluding drug price reference countries
- by Lee, Tak-Sun Jan 01, 2023 10:40pm
- Australia's addition to the drug price reference country, which faced opposition from the pharmaceutical industry, failed. The HIRA initially decided to take a step back from adding Australia and Canada to the drug price reference country and add only Canada to the reference country. The HIRA released the "Detailed Evaluation Standards for Drugs Subject to Negotiation, including New Drugs" on the 28th on the work portal of nursing institutions. The evaluation criteria include the contents of PE drugs announced by the HIRA in August and the expansion of drug price reference countries announced in November. According to the revised detailed evaluation criteria, Canada was included in the existing countries of Japan, France, Germany, Italy, Switzerland, the United Kingdom, and the U.S. A7 countries, making it A8. Australia, which was announced in November, was not included in the reference country. In the case of Australia, the domestic pharmaceutical industry strongly protested, saying that the price of generic drugs could be lowered if they are used for re-evaluation of post-registration materials due to low generic drugs. The KRPIA also issued a statement of opposition, fearing that the price of new drugs would be lowered. The HIRA includes Canada, which is acceptable to all stakeholders, to secure the validity of the amendment, and Australia is eventually excluded. The HIRA official said, "Australia was added to the drug price reference country due to similar geographical access and economic conditions, but as the pharmaceutical industry's opinion, we decided to exclude it in terms of industrial similarity." If it is possible to omit the submission of PE data, it will be evaluated as the lowest among the national adjustment prices of eight foreign countries of similar drugs when registered in more than three A8 countries. In addition, even if it is difficult or possible to select foreign similar drugs, 10% of the highest price of alternative drugs is added to the list of less than A83 countries, and the adjustment price of countries excluding similar drugs is used for evaluation. The drug A8 is also referred to in RSA drugs. The detailed evaluation criteria also reflected that "a small number of target patients" were among the requirements for the omission of submission of PE data. The number of patients is judged based on the expected number of patients subject to benefits (in Korea) of the indication, but the current status of the expected number of patients at the time of evaluation of the drug, which has been considered essential for treatment, is considered. For drugs that can be used for both adults, the submission of PE data can be omitted only if the main indication is children. This reflects the HIRA's prediction in August. In this regard, there were many objections, saying that the PE exemption system has retreated to limit the number of patients and exempt PE only from pediatric indication treatments. However, the HIRA applied the original plan as it is, saying that these regulations can be flexibly applied in the evaluation process.
- Policy
- New formulations of narcotics will be as strictly reviewed
- by Lee, Jeong-Hwan Dec 30, 2022 06:33am
- Regulations on narcotics for medical use, such as narcotic appetite suppressants and propofol, which is used for general anesthesia, that the government is restricting new approvals for, are expected to become stricter than before. Until now, even narcotic medications for which new approvals are restricted by the government were allowed to receive new approvals when developed into new formulations, but these new formulations will not be approved in the future if they have a high risk of misuse or abuse or is rejected by the deliberation committee. On the 19th, the Ministry of Food and Drug Safety announced that it had publicized the narcotic medications subject to restrictions that contain the restrictions above. More specifically, the MFDS had made the notification on the 28th of last month, upon which the notice immediately took effect. The drugs subject to restrictions are amfepramone and mazindol-containing drugs, GHB and its isomer or drugs that contain its sodium salt, phentermine, phendimetrazine, and propofol-containing drugs. The MFDS had already restricted approvals for the substances on August 14, 2020. One point of attention in the new announcement is that even narcotic medications subject to restrictions that were developed into a ‘new formulation’ may be subject to deliberations by the ‘Narcotics Safety Control Deliberation Committee’ if the MFDS determines the drug to have a high risk of misuse or abuse. Therefore, if the new formulation does not pass the ‘Narcotics Safety Control Deliberation review, that new drug may not receive marketing authorization in Korea. The MFDS announced such restrictions on narcotic medications due to intermittent applications filed for marketing authorizations by companies after changing only the formulation of their drugs to receive approval in categories where new marketing authorizations are restricted. As the authorities restricted new approvals in the above areas to eradicate misuse and abuse of narcotic medications, the MFDS needs to ponder whether to approve the new formulations when a pharmaceutical company requests approvals for developing new formulations. In the case of propofol, a restricted narcotic, the company applied for marketing authorization after changing its injection formulation into a prefilled syringe type. The drug was Fresenius Kabi’s ‘Fresofol MCT Prefilled Syringe.’ As a result, the MFDS issued a public notice and implemented an administrative measure to clearly prohibit approval of new narcotic medications with new formulations, etc., if the drug has a high possibility of misuse or abuse through. Also, by making the drugs subject to the Narcotics Safety Control Deliberation Committee review, the approvals of narcotic medications with new formulations will now be reviewed more professionally and objectively. In other words, narcotic medications that wish to obtain new approvals for their new formulations would need to have little risk of misuse or abuse and pass advisory review from the Narcotics Safety Control Deliberation Committee. An MFDS official said, “Among the narcotic medications that were restricted permission to minimize misuse and abuse, there were cases where companies sought new approvals based on new formulations. We made the announcement to clarify the need to restrict such approvals if their risk of misuse or abuse is high." The official added, “The notice further clarifies our regulations on how narcotic medications, even with new formulations, will not be approved if at high risk of misuse or abuse, or fails to pass deliberations by the Narcotics Safety Control Deliberation Committee.”
- Policy
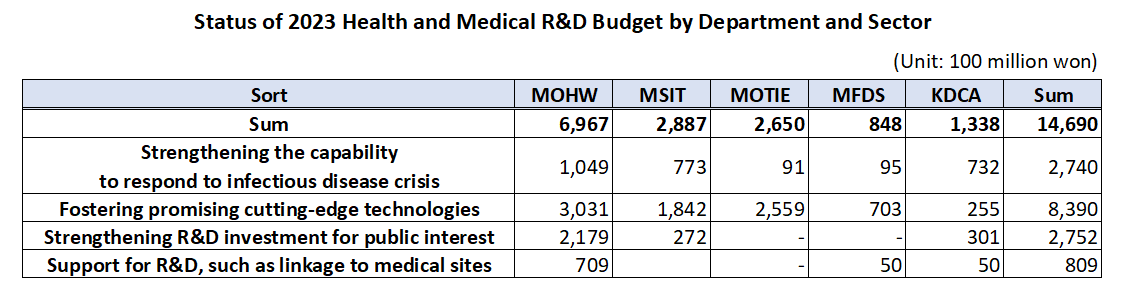
- Gov allocates KRW1.47 trillion budget for healthcare R&D
- by Kim, Jung-Ju Dec 30, 2022 06:33am
- The government’s budget for next year’s healthcare R&D including new drugs, medical devices, and digital transformation to AI-based biohealth is estimated to be around KRW 1.47 trillion. This is the total amount of budget that will be supported by the Ministry of Health and Welfare, Ministry of Science and ICT, Ministry of Trade, Industry, and Energy, Ministry of Food and Drug Safety, and Korea Disease Control and Prevention Agency, an amount similar to this year and 5% of the government’s total R&D budget. According to the MOHW on the 29th, the government plans to support KRW 149.5 billion in new tasks and KRW 1.32 trillion on ongoing tasks, for a total of 128 projects around 4 major areas including ▲reinforcing the ability to respond to infectious diseases, ▲fostering promising advanced technologies in biohealth, ▲increasing R&D investment for public interest including overcoming diseases, etc., and ▲support for R&D linked to the medical field. ◆Reinforcing ability to respond to infectious diseases = The government will be supporting KRW 274 billion for 26 select projects next year to strengthen Korea’s capacity in responding to infectious disease crises. The authorities plan to establish health security by supporting R&D to secure main technology capabilities for vaccines and treatments that can protect public lives and health from future infectious diseases. By ministry, the MOHW will be investing KRW 3.75 billion in developing antiviral treatments in preparation for RNA virus infections (Disease X), and MSIT has allocated KRW 13.3 billion for the Bio & Medical Technology Development Program and the establishment of the National Preclinical Trial Support System. The government also allocated a budget to advance the disease control and prevention system. Based on the lessons learned from the COVID-19 pandemic, the government plans to establish a safe society from new infectious diseases by promoting research on the whole disease control and prevention, etc to advance Korea’s disease control and prevention system. The MOHW allocated KRW 1.6 billion, KDCA KRW 1.3 billion, and MOTIE KRW 0.7 billion for the project to advance the pan-ministerial infectious disease control and prevention system. ◆Fostering promising advanced technologies= The government plans to support a total of KRW 839 billion in 66 projects to foster promising advanced technologies. First, the government will be improving the quality of healthcare by developing data and AI-based technology and prompting the digital transformation of biohealth, and providing personalized healthcare. In terms of new projects that will be initiated by each ministry, MOHW will be investing KRW 6.25 billion in R&D for technology to utilize healthcare MyData and its demonstration project. Also, the MOHW allocated KRW 7.5 billion to the project for developing healthcare technology based on virtual patients and hospitals, and KRW 7.5 billion for the demonstration and introduction of medical institution-based digital healthcare. The authorities will also be investing in the discovery of promising next-generation areas. It plans to continue the search for future drivers of biohealth by supporting R&D in unexplored areas such as microbiome, and in areas that can enhance industry competitiveness by improving the self-sufficiency of core technologies such as advanced medical devices. In terms of new projects that will be initiated by each ministry, the MOHW and KDCA allocated KRW 3.8 billion and KRW 1.3 billion each for the hospital-based human microbiome R&D project. The MFDS has also newly allocated KRW 7.4 billion to support regulatory science for pan-ministerial medical device regulations, and the MOTIE allocated KRW 2.1 billion for the development of interventional medical device technology based on advanced manufacturing technology. The projects also include R&D in regenerative medicine. The government plans to support research to establish a basis for the commercialization of regenerative medicine, such as xenotransplantation R&D projects, to ultimately become a global leader in advanced regenerative medicine technology by securing core and basic source technologies. In terms of new projects that will be initiated by each ministry, the MOHW will invest KRW 6 billion in xenotransplantation R&D projects, the MOTIE will invest KRW 4.5 billion in bio, medical technology development, and stem cell ATLA-based treatment technology for incurable diseases. ◆Strengthening R&D investment for public interest = The government will also allocate KRW 275.2 billion in 28 select projects to invest in R&D for public interest such as overcoming diseases. First, the government plans to ease the socioeconomic burden by focusing on the development of healthcare technologies to overcome diseases that bring high burden to the public such as brain diseases, mental health, and cancer. In terms of new projects that will be initiated by each ministry, the MOHW allocated KRW 4.95 billion in the development of technologies to address issues in the clinical field for diseases related to brain and nervous diseases, KRW 9.63 billion in an R&D project and its demonstration for the development of evidence-based personalized healthcare for cancer survivors, and KRW 0.5 billion for the development of technology for metaverse-based mental health management in the National Center for Mental Health. The government plans to preemptively conduct R&D in healthcare technologies where public demand is expected to surge in line with social changes including aging and the low birth rate, that will contribute to resolving social issues and enhancing the real sense of R&D in public health. For this, the MOHW will support KRW 3.9 billion in R&D of consumer-focused care robots and their service demonstration. ◆ support for R&D linked to the medical field= The government plans to support KRW 80.9 billion in R&D after selecting 8 projects linked with the medical field. First, it plans to reinforce Korea’s global competitiveness by fostering professional manpower that can drive innovation in biohealth, such as expanding research support for new physician-scientists and continuing training for regulatory science experts. For this, MOHW has allocated KRW 4.05 billion for the global research cooperation support project. Seong-ho Eun of the Bureau of Advanced Health Technology Policy MOHW said, “The government will continue to promote relevant policies and continue increasing R&D investments so that the healthcare R&D provided by the government serves as a basis for better quality healthcare services. We will also further activate the Health and Medical Technology Policy Deliberation Committee so that related ministries and the private sector for more organic cooperation and communication.”