- LOGIN
- MemberShip
- 2026-04-11 03:01:27
- Company
- Nubeqa, approval for a combination therapy indication
- by Eo, Yun-Ho Dec 23, 2022 06:06am
- It is expected that the prescription of the combination therapy of the prostate cancer treatment Nubeqa will be possible in Korea. According to related industries, Bayer Korea recently submitted an application to the Ministry of Food and Drug Safety to expand the Taxotere combination therapy indication of an oral androgen receptor inhibitor (ARi) Nubeqa for treating vision nmCRPC. Nubeqa is an androgen receptor inhibitor with a unique chemical structure that binds to androgen receptors to inhibit the growth of prostate cancer cells through strong antagonism. The corresponding indication of this drug has been validated through an ARASENS phase 3 study. The metastatic hormone-sensitive prostate cancer patient group with Nubeqa and ADT and Taxotere on ARASENS 3 demonstrated a statistically significant improvement in total survival compared to the control group with ADT and Taxotere. The patient group using Nubeqa, ADT, and Taxotere showed a statistically significant delay in the period required for the pain to progress. The clinical results were announced at the 2022 Urology Cancer Symposium (ASCO GU) of the American Society for Clinical Oncology held in February, and were published in the medical journal "New England Journal of Medicine." Nubeqa has not yet applied for insurance benefits in Korea, so it is expected to take time to lead to actual prescription activation. The drug has not been registered since the HIRA Cancer Disease Review Committee ruled it unsuitable in February 2021.
- Company
- 1 of 2 patent suits filed this year proceed to the 2nd round
- by Kim, Jin-Gu Dec 23, 2022 06:06am
- It seems that the patent disputes in the pharma and bio industry will continue on in the long term. Among the 20 major rulings made during the first trial, 11, over 50% are being retried in a second trial by the Patent Court of Korea. Both the winning and losing parties of the patent suit that had been filed against Boryung Pharmaceutical’s fixed-dose combination for hypertension, ‘Dukarb (fimasartan and amlodipine),’ opted to continue their dispute in a second trial. In the case of the patent dispute over Novartis’s heart failure treatment ‘Entresto (valsartan+sacubitril),’ the original company appealed after the generic drug companies won the first trial. In the case of the dispute over the DPP-4 inhibitor class antidiabetic ‘Galvus (vildagliptin),’ the agenda is being fiercely tried again from square one after the Supreme Court's ruling of remand after reversal, and Novartis is also further countering by filing a trial for the active confirmation of the scope of rights. ◆Generics companies win first trial on Entresto... Novartis appeals According to industry sources on the 23rd, 21 major trial rulings and judgments have been made this year for patents in the industry. The number excludes cases where the company voluntarily withdrew its suit after requesting judgment. 20 of the cases received a ruling from the Intellectual Property Trial and Appeal Board (first trial), and 1 received a ruling from the Patent Court of Korea (second trial). Of the 20 cases that received a ruling in the first trial, 11 cases chose to continue on to the second trial. This means that more than half of the parties of major disputes did not accept the decision of the first trial. Pic of EntrestoEntresto’s patent dispute is one representative example. The dispute was sparked after its generic companies simultaneously filed suits to invalidate and confirm the passive scope of rights on Entresto’s 6 patents. Starting with Elyson Pharm, 13 companies challenged ▲1 use·composition patent ▲ 1 salt·hydrate patent (un-listed in the patent register) ▲1 use patent ▲1 crystalline form patent ▲2 substance patents for Entresto. The first trial was concluded in favor of the generic companies. Generic companies succeeded in challenging 1 use·substance patent, 1 crystalline form patent, and 2 substance patents. The remaining trials for the use patent and salt·hydrate patent have not been concluded yet. The company of the original drug, Novartis, decided to continue on the fight to the second trial for the crystalline form patent and the use·composition patent in March and July of this year. The company has given up the trials for its other 2 substance patents. Entresto has no drug substance patent, and the role is replaced by the use·composition patent and crystalline form patent. Therefore, Novartis plans to focus its defense strategy around these two patents. ◆Both the winning and loser parties appeal to ruling for Dukarb... long-term dispute inevitable Pic of Dukarb Two conflicting rulings were made in the first trial for Boryung’s Dukarb patent. Among 40 generic companies that challenged Dukarb’s patent, 2 won against Boryung, while the others lost. 29 of the companies filed a new trial on the same patent at the same time. If the first trial they attempted was made to avoid the patent by confirming the passive scope of rights, the companies added a patent invalidation strategy after losing the first trial. From the generic company’s perspective, whether it receives a winning decision from the Patent Court of Korea or a new ruling from the Intellectual Property Trial and Appeal Board, the company will be eligible to release its generic early. This is why companies that challenge the patent are mobilizing all available strategies. Boryung Pharmaceutical also filed a lawsuit against the two pharmaceutical companies that won against the company in the first trial to annul the trial decision. As a result, the Dukarb patent dispute, which began in March last year, is expected to be prolonged to even after the substance patent for Kanarb expires in February next year. ◆Galvus dispute to continue to the end...Novartis files active confirmation of the scope of rights # i3 The patent dispute around Galvus was also inconclusive this year. The analysis is that the conflict has been prolonged for more than 5 years due to the sharp differences in the position of the two parties. The situation had also intensified with Novartis' counteract, filing a trial for the active confirmation of the scope of rights. The conflict began in 2017 when Ahn-Gook Pharmaceuticals and Hanmi Pharmaceuticals claimed part of the term extended for the substance patent of Galvus invalid. The generic companies won the first trial. In the second trial, the court overruled the first court’s decision and ruled in favor of the original company. In the third trial, the Supreme court questioned the qualifications of the original company that filed the appeal and remanded it back to the first trial. With the remandment, the dispute resumed in the first instance court. Unlike in the previous first trial, the Intellectual Property Trial and Appeal Board ruled in favor of the original company, and generic companies appealed once again. Currently, Ahn-Gook Pharmaceuticals and Hanmi Pharmaceuticals are waiting for the second trial ruling. Apart from the dispute, Novartis had filed a trial for the active confirmation of the scope of rights against Kyongbo Pharmaceutical, Ahn-Gook Pharmaceuticals, and Korea United Pharm. The active confirmation of the scope of rights is filed by the original companies to protect their patent rights from generic companies. The suit is rarely filed in the domestic pharmaceutical and bio-industry. Kyongbo and the others have already released their generic versions after they won the first trial. The substance patent for Galvus expired in March of this year. However still, Novartis plans to see the dispute to the end and be recognized for the generic companies’ patent infringement. If Novartis wins, it will be possible to claim damages due to patent infringement. All companies that released Galvus generics will be subject to claims for damages. Also, Roche and Alvogen Korea received mixed results over the 3 use patents for Avastin. Alvogen Korea won 2 trials and Roche succeeded in defending 1 claim. Both companies decided to appeal and take the dispute to the second trial. However, Roche withdrew the lawsuit it had filed for cancellation of the trial decision in September and plans to focus on the case appealed by Alvogen Korea. In the patent dispute over Monterizine Cap, Hanmi Pharmaceuticals filed an appeal for the cacellation of the trial decision against 20 pharmaceutical companies after losing the first trial.
- Policy
- Revlimid's benefit for multiple myeloma maintenance therapy
- by Lee, Tak-Sun Dec 23, 2022 06:05am
- Revlimid, a treatment for multiple myeloma, is eligible for maintenance therapy for hematopoietic stem cell transplantation patients. The effective date is January 1 next year. The HIRA has launched an opinion survey on the revision of the anti-cancer drug standard. According to the revision, benefits will be newly established for maintenance therapy for newly diagnosed multiple myeloma patients who have reacted more than stable stool after transplanting Revlimid into autologous hematopoietic stem cells. The administration starts six months after the end of the transplant, and as a result of the response evaluation, the administration was stopped when the disease progressed. In response, the HIRA said that Lenalidomide, including Revlimid, is a drug licensed to maintain multiple myeloma patients who have been newly diagnosed with autologous hematopoietic stem cell transplants, and that this therapy is mentioned as standard treatment in textbooks, and is also recommended by the NCCN Guidelines (2022) and ESMO Guidelines (2021). In particular, in a random assignment, double-blind, and phase 3 clinical trial (CALGB) for patients with multiple myeloma who received a newly diagnosed autologous hematopoietic stem cell transplant, it was confirmed that it was clinically useful compared to those who did not perform maintenance therapy, and added that benefit standards were set. The clinical trial results showed 57.3 months of progression-free survival median (mPFS) (28.9 months in the control group) and 113.8 months of total survival median (mOS) (84.1 months in the control group). The revised bill also introduced a combination of adjuvant and Fluorouracil + Leucovorin for biliary tract cancer. Pediatric combination therapy among "Dasatinib + chemotherapy" has been modified and supplemented for acute lymphocytic leukemia. This is in response to a request to change the Korean Pediatric Blood Oncology Association. At the same time, Niraparib monotherapy (more than 4th round) was deleted from ovarian cancer, ovarian cancer, and primary peritoneal cancer palliative, while Paclitaxel + Cisplatin (Intra-peritoneal) combination therapy was also deleted for other cancers.
- Policy
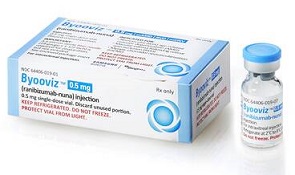
- Lucentis biosimilar price set 63% lower than original
- by Lee, Tak-Sun Dec 23, 2022 06:05am
- Samsung Bioepis Biosimilars of the macular degeneration treatment Lucentis (ranibizumab), which are set to be listed for reimbursement in Korea next month, are expected to be launched at a price much lower than their original. As a result, the burden borne by these patients is expected to be reduced significantly. According to industry sources on the 21st, the Lucentis biosimilars that are expected to be listed with reimbursement next month will be launched at a price 44~63% lower than the original version’s insurance ceiling price. Chong Kun Dang’s LucenBS Inj is set to be listed at KRW 300,000 per vial. The original version with the same ingredient of the same strength, Lucentis Inj 10mg/mL (3mg/0.3mL) is priced at KRW 820,636 per vial. When listed, CKD’s product will be 63.4% cheaper than the original. Samsung Bioepis’s Amelivu Inj is set to be listed at KRW 463,773. The original version with the same ingredient of the same strength, Lucentis Inj 10mg/mL (2.3mg/0.23mL) is priced at KRW 828,166. When listed, Samsung Bioepis’s product will be 44% cheaper than the original. Even if the original drug's ceiling price drops by 30% after the listing of biosimilars, Chong Kun Dang's LucenBS will still be 47.7% cheaper and Samsung Bioepis’s Amelivu 20% cheaper. Lucentis had been considered a drug with a high patient burden due to its high price. In particular, due to the strict reimbursement standards, patients unable to meet the standards had to pay the full amount without reimbursement. The situation had been eased somewhat since 2017 after the limit on the number of uses had been lifted, somewhat reducing the burden on patients. With the listing of cheaper biosimilars, patients are expected to have a broader range of choices and less burden from treatment costs. Lucentis’s annual sales in Korea have recorded KRW 35.1 billion (based on IQVIA 2021). Just one-third of the sales will be KRW 10 billion. This is why the two companies opted to release their biosimilars at a drastically reduced price to penetrate the market. Also, Chong Kun Dang brings its strong sales power, and Samsung Bioepis partners with Samil Pharm, an ophthalmology drug-specializing company, to start full-scale sales and marketing activities.
- InterView
- IL-23 Continued research on inflammatory pathways
- by Dec 23, 2022 06:05am
- Efforts to develop treatments through various studies such as combination therapy and receptor targets. Interleukin (IL) inhibitors used in autoimmune diseases such as psoriasis are evolving as several companies continue to develop them. With the emergence of more effective treatments, psoriasis can expect "PASI 100," which means complete improvement. Interleukin inhibitors are classified according to the mechanism. Stelara, which appeared first, targets IL-12 and IL-23 simultaneously. Cosentyx and Taltz are mechanisms that inhibit IL-17. Recently, drugs that are attracting attention are Tremfya and Skyrizi, which are exclusively targeting IL-23. It is known to have the most powerful effect. As IL-23 pathway research becomes more active, evidence is accumulating that IL-23 inhibitors are inevitably more effective than interleukin inhibitors of other mechanisms. In a recent interview with Dailypharm, Daniel Cua Janssen, vice president of the immune business department, said, "IL-23 inhibitors act on immune cells themselves that activate inflammatory pathways, preventing the underlying cause in the early stages. Recently, it was found that IL-23 reprogrammed a group of T cells that produce IL-17 and found that a fairly strong inhibitor was needed among IL-23 inhibitors, he explained. Daniel Cua is a world-renowned scholar who first discovered the IL-23 pathway 22 years ago. The IL-23 route he discovered was the beginning of the development of IL-23 inhibitors such as Trempier and Skyrich. Studies on the mechanism of how IL-23 causes inflammation are still ongoing. "It is clear that IL-23 is the strongest interleukin associated with autoimmune diseases. Based on this, he added, "In the future, challenges to develop treatments through various studies such as combination therapy and receptor targets will continue." The following is a question-and-answer session with Vice President Cua. -After studying immunology for a long time, we discovered the IL-23 pathway that is the target of autoimmune disease treatment. We are curious about the history. =22 years ago, I was researching a small biotech in Silicon Valley, USA. At that time, research was in full swing to discover substances that control inflammation. The goal was to find an inflammatory control substance, but the IL-23 pathway was found at that time. At that time, not many people believed it because it was an unexpected discovery. It was only a year or two later that the substance that academia wanted to find was IL-23 which we discovered in 2000. It began to be recognized as a very important discovery. Animal models that inhibited IL-23 showed all resistance to several autoimmune diseases. This also affected future research and treatment development. The discovery of the IL-23 pathway is considered to be the starting point of several subsequent studies. - Various interleukin inhibitors such as IL-12/23, IL-17 and IL-23 have been developed as treatments for autoimmune diseases. What differences do differences in mechanisms make? =One of the most frequently asked questions is why blocking only IL-23 works better than blocking IL-12 and 23 together. To put it simply, IL-23 is the standard target we have to hit. Cytokines sometimes cross-regulate each other. Controlling one means that the other rises. IL-12 and IL-23 are. Blocking the two together in a cross-regulation relationship results in a "push and pull" conflict effect of each other's actions at the same time, which reduces the effect. Only IL-23 should be blocked to show the most precise inhibitory effect we want. IL-17 is an interleukin present in a lower stage than IL-23 on the reaction path. It has higher specificity in intestinal epithelial cells and skin epidermal cells. On the other hand, IL-23 acts on immune cells themselves that activate inflammatory pathways. For this reason, IL-23 must be suppressed to prevent the underlying cause of the inflammatory system in a more early stage. In psoriasis, IL-23 shows a clinical improvement effect of 80-90% in more than 90% of patients. - Like immuno-cancer drug combination therapy in cancer, can autoimmune diseases be more effective by combining upper and lower series that suppresses interleukin? = combination therapy is the approach that many researchers and companies in this field are most interested in. The concern is the accumulation of adverse reactions caused by combination therapy. Therefore, research on port therapy is being conducted on the most reasonable and scientific basis. A combination of specific route drugs based on biomarkers is being sought. Although it is still in its early stages, many efforts are being made to find a combination that increases the effectiveness but does not increase the response. So far, it is known that blocking complementary paths may be more effective than targeting only specific paths. For detailed research, the understanding of various paths is improved based on biomarker analysis. -How far has the research on IL-23 progressed? =The level we know is that IL-23 induces the production of receptors called IL-17, IL-22, and GM-CSF. However, it was not clear exactly how IL-23 causes inflammation. It has recently been revealed that IL-23 reprogrammed a group of T cells that produce IL-17. Reprogramming is a fairly powerful action and cannot go back because it is semi-permanent in itself. To prevent this, considerably strong inhibitors among IL-23 inhibitors should be used. In the end, I realized that inhibiting IL-23 has a much stronger effect than inhibiting elsewhere and that it is also effective in preventing lower-level diseases when applying a mechanism that prevents reprogramming at the epigenetic level to new drug development. - What are the targets or pipelines to pay attention to in the subsequent development of interleukin formulations? = First, the receptor. Ligands such as IL-23 bind to receptors, a new drug that blocks the receptor itself to prevent binding. The number of receptors is smaller than that of ligands, so it is easier to block and has precise access, making them a good candidate. The second is the study of RORgamma-t. Many researchers are targeting it, but no one has succeeded yet. Treatments that produce the most accurate and precise effects in this route are expected to be developed in the future.
- InterView
- From domestic sales to the global brand general
- by Dec 22, 2022 05:53am
- Considering healthcare systems around the world such as in Russia, the Middle East, Southeast Asia, and South America, the company will set up a strategy to launch a new anti-cancer drug. It is the main job of Kim Soo-yeon (48), executive director of the International Lung Cancer Brand, at AstraZeneca. Managing director Kim's role has also grown as the number of new anti-cancer drug pipelines from companies such as Tagrisso and Enhertu has increased. Having been working in Singapore for two years, he is neither a study abroad nor a pharmacist or doctor. Executive Director Kim is the only case in a multinational pharmaceutical company where a native Korean, not an expert or a study abroad, has stood as a global marketer. Kim, who met in Singapore, cited four things. "I think confidence, courage, responsibility, and people have grown me this much." It is also something that executive director Kim wants to emphasize the most to his juniors. In fact, she plucked up courage in certain areas. Kim, who set foot in the domestic pharmaceutical industry about 20 years ago with cardiovascular sales at Pfizer Korea, moved to Novartis with the aim of marketing anticancer drugs. At that time, multinational pharmaceutical companies were just starting to set up an anti-cancer drug division. "I had a clear goal. I thought the market for anticancer drugs, which is more grounded and directly affects patients' lives, would grow, and I thought I should be in charge of anticancer drugs. I went to Novartis and launched the first targeted anticancer drug, Glivec, and I did a lot of work to tell them what targeted anticancer drugs were. Another targeted anti-cancer drug, Tasigna, was launched." Based on the successful launch of anticancer drugs such as Glivec and Tasigna, AstraZeneca contributed to the rapid introduction of Tagrisso in Korea. Korea's approval of Tagrisso was the fifth in the world. Kim was not satisfied with her domestic success. It has jumped into unfamiliar markets such as the Middle East and South America. Executive Director Kim was in charge of Tagrisso marketing in all global markets except the United States, Europe, and Japan. The situation was not good because it was a time when COVID-19 was spreading. Even under difficult conditions, Executive Director Kim successfully launched Tagriso and was promoted to the general manager of lung cancer once again after a year and 10 months of promotion. "In fact, the domestic anticancer drug market knows the market to some extent, so I was able to work well. I think it's time to try something new. When I was in charge of global marketing, the Middle East and South America were really unfamiliar. She didn't know anything about culture, economic conditions, government policies, and healthcare systems. It is also greatly affected by the international situation such as COVID-19 and the Russia-Ukraine war. On the contrary, it was new and fun to understand the market. It was also fun to communicate with employees all over the world." She made it his first goal to be responsible and influential in a given role until the end. In countries where the early diagnosis of lung cancer is not well performed, cooperation continued to activate the early diagnosis system. It is not just about introducing products, but about increasing the size of the market. Executive Director Kim's marketing strategy is to lead early diagnosis as an important agenda and create an ecosystem where lung cancer treatments can be used for patients at an appropriate time. Executive Director Kim stressed that such success cannot be achieved alone. As a mother of two children, the support and support of the family became the driving force to focus on work. The good influence of the surrounding seniors and juniors became manure and grew Executive Director Kim. This is why executive director Kim values "people." "People are important in the family, in the company, and everywhere. I was able to come here because my family supported my children when they were young. The company should also help each other so that the seniors and juniors can grow together. I can't succeed alone just because I'm good. With confidence, courage, and responsibility, what matters is people."
- Company
- New HCV drug Epclusa lands in hospitals with reimb
- by Eo, Yun-Ho Dec 22, 2022 05:52am
- The Hepatitis C treatment ‘Epclusa’ is rapidly landing in hospitals for prescriptions after being granted reimbursement in Korea. According to industry sources, Gilead Science Korea’s oral chronic hepatitis C treatment Epclusa (sofosbuvir/velpatasvir) has passed the Drug Committees (DCs) of the Big-5s including the Seoul National University Hospital, Seoul St. Mary’s Hospital, Asan Medical Center, and Shinchon Severance Hospital, as well as other major medical institutions including Ajou University Hospital, Hallym University Sacred Heart Hospital, Pusan National University Hospital, Kyungpook National University Hospital, Chonnam National University Hospital, Chonbuk National University Hospital, Chungnam National University Hospital, and Chungbuk National University Hospital. When considering how the drug has only been listed for reimbursement on the 1st of last month, the drug is quickly landing at hospitals for prescriptions in Korea. Epclusa can be administered with reimbursement in adult and pediatric patients 23 years or older, weighing at least 30 kg, with any genotype of the chronic hepatitis C virus, regardless of the stage of cirrhosis or previous treatment experience. With the approval, Epclusa became the only treatment in Korea that can be prescribed with reimbursement regardless of HCV genotype or stage of fibrosis from November. Epclusa is a fixed-dose combination of sofosbuvir, a nucleotide analog NS5B polymerase inhibitor, and velpatasvir, an NS5A replication complex inhibitor. The drug demonstrated a 99% treatment success rate (SVR12) in the ASTRAL-1 study conducted on patients with chronic HCV infection genotypes 1, 2, 4, 5, and 6 , without cirrhosis or with compensated cirrhosis. Also, in the ASTRAL-4 study, the treatment success rate was 94% in patients with decompensated cirrhosis using Epclusa. Meanwhile, according to the HCV treatment guidelines issued by the Korea Association for the Study of the Liver, Epclusa is recommended for patients with all HCV genotypes, with or without compensated cirrhosis, with or without treatment experience. Also, Vosevi is recommended as a retreatment for patients with chronic hepatitis C that have previously failed DAA treatment.
- Company
- Dong-A ST introduces candidate substance technology
- by Kim, Jin-Gu Dec 22, 2022 05:52am
- Global joint development of immuno-cancer drugs under the mechanism of double fusion antibodies and acquisition of exclusive sales rights Dong-A ST announced on the 21st that it has signed a license agreement with Kanaph Therapeutics to license candidate substances for immuno-cancer drugs under the dual fusion antibody mechanism. Dong-AST will introduce global joint research and development and exclusive sales rights of preclinical immuno-cancer drug candidates held by Kanaph Therapeutics, a domestic pharmaceutical bio-venture. Dong-AST will pay Kanaph 5 billion won in advance and up to 18 billion won in additional payments depending on the development milestone. It also plans to pay up to 180 billion won more depending on sales if it succeeds in commercialization. Royalty is separate and will be paid at a certain rate according to the sales profit after the product is released. According to Dong-AST, the substance is an antibody and cytokine fusion protein. It is a mechanism to activate immunity by transferring cytokines specifically to tumors using antibodies to proteins expressed in the tumor microenvironment. At this time, cytokines are not delivered to normal tissues, so side effects of systemic immune activity can be prevented.
- Product
- Get rid of Imotun & Godex which waste taxpayers' money
- by Dec 22, 2022 05:52am
- Pharmaceutical Society for a Healthy Society (CEO Shin Hyung-geun) has called for the expulsion of Imotun and Godex. In a statement on the 21st, the Pharmaceutical Association for Health Society argued that maintaining health insurance benefits for six-component compounds (Godex) including avocado-soya unexamined (Imotun) and adenine hydrochloride (60 billion won) are not valid in terms of clinical usefulness, cost-effectiveness, and social needs. They said Imotun and Godex are drugs whose clinical usefulness is unclear. "Although Imotun's clinical usefulness is based on the recently revised rheumatology textbook, there is no SCI-listed clinical journal that validates knee osteoarthritis patients over the past four years, and foreign rheumatology textbooks and guidelines related to the International Osteoarthritis Society are all described at the supplement level as preferred." In addition, France, the original developing country that first licensed Imotun as a drug, concluded in 2013 that it does not recommend Imotun as a salary due to its unclear clinical usefulness, and no one in developed countries supports the purchase of Imotun. Chong Kun Dang, a domestic seller, also conducted clinical trials on 300 people at university hospitals in Korea from 2013 to 2016, but it is pointed out that related results have not been disclosed. It is the position of the health drug that Imotun and Godex are not cost-effective and that social demands are low. They said, "Imotun and Godex are basically drugs that are difficult to confirm their effectiveness, and patients have no choice but to take the medicine according to their prescription regardless of their economic situation." Even if it is supported by health insurance benefits, Imotun must spend 3,400 won and Godex 17,000 won per month, he said. "In addition, Imotun is a drug that threatens health insurance finances of 40 billion won per year and Godex is 60 billion won per year, and social needs to be evaluated to delete benefits." The Pharmaceutical Society for the Health Society said, "Over the past few years, we have demanded the removal of drugs that are not clinically useful, including Colin Alpo, but as of December 2022, not a single drug has been removed from health insurance benefits. The reason why this revaluation system was not implemented properly is that the Ministry of Health and Welfare is looking at pharmaceutical companies, he said. "The Ministry of Health and Welfare should review the decisions of Emoton and Godex's Pharmaceutical Evaluation Committee and quickly come up with new measures to control unnecessary drug use."
- Policy
- New reimbursement standards set for Kymriah·Zolgensma
- by Kim, Jung-Ju Dec 22, 2022 05:52am
- New reimbursement standards have been set for high-priced pharmaceuticals like Kymriah and Zolgensma, the so-called ‘one-shot treatments.’ Also, reimbursement standards will be newly established for the second CGRP-targeted therapy for migraine, Ajovy (fremanezumab), and the new tuberculosis drug Dovprela 200mg (pretomanid) that had been the first to be developed in half a century, as the drugs will be listed for reimbursement starting on January 1. The Ministry of Health and Welfare issued a pre-announcement of an administrative notice for the ’Proposed Partial Amendment of Details Regarding the Standards and Methods for Applying Medical Care Benefits (drugs)’ that contain the details stated above, and will be collecting opinions until the 28th. First, a new standard will be established for the reimbursement management of high-priced pharmaceuticals. With ‘one-shot treatments’ Kymriah and Zolgensma being listed this year, a new reimbursement standard was needed for the management of such high-priced drugs. Thus, the government and the Health Insurance Review and Assessment Service established new reimbursement standards that specify those subject to reimbursement management and how they can prepare a medical care benefit expense statement. Under the new standards, the follow-up management period for Kymriah has been set to 1 year when administered for non-Hodgkin lymphoma, and Zolgensma to 5 years. Also, new reimbursement standards were set for the Ajovy autoinjector and Ajovy pre-filled syringe, the CGRP-targeted new therapies for migraine, as well as the new tuberculosis drug Dovprela tab, which are set to be listed on the 1st of next month. In the case of Ajovy, response evaluations with headache diaries and MIDAS should be performed within a month before administration and every 3 months thereafter. If the number of monthly migraine days is not reduced by over 50% from baseline at any response evaluation, administration will be discontinued. The administration period was set to 12 months, and switching between anti-CGRP migraine prevention drugs is not accepted for reimbursement. In the case of Dovprela 200mg, patients must first apply in advance and are granted reimbursement by the KDCA to receive reimbursement. The details for the KDCA’s approval including its procedure, method, and committee member composition will be determined by the KDCA commissioner. The Praluent pen inj 75mg (evolocumab) and other alirocumab injections will be extended reimbursement to patients confirmed with heterozygous familial hypercholesterolemia (heFH) as diagnosed by a score of 6 or higher on the Dutch (2004) diagnostic criteria or ‘definite heFH or possible heFH' under the Simon Broome (2006) standards. The government and HIRA explained that it had received an opinion from a relevant academic society that patients with a high LDL-C level (over 190mg/dL) with a family history should be regarded as heFH patients and be treated accordingly. Clinical literature (RUTHERFORD-2, 2015) showed studies were conducted with patients with definite heFH or possible heFH based on Simon Broome (2006) standards, and NICE had described heFH as “patients with a score of 6 or higher on the Dutch (2004) diagnostic criteria or definite heFH or possible heFH' under the Simon Broome (2006) standards.” Also, with Nuvorozet tab. 40/2.5/5/10mg being newly listed next month, the government and HIRA decided to add the combination to the list of oral fixed-dose combinations that have already been listed for reimbursement. The new combination added is ‘S-amlodipine+telmisartan+rosuvastatin+ezetimibe.’ Also, reimbursement standards for the 25mg strength of K-Cab 50mg (tegoprazan) will be extended with its listing next month. In addition to its already-reimbursed indications as ▲treatment of erosive and non-erosive gastroesophageal reflux disease, ▲ treatment of gastric ulcer, the drug will be additionally reimbursed as ‘maintenance therapy after the treatment of erosive GERD (only the 25mg strength).' 10mg strengths of Lucentis inj and Lucentis prefilled syringe (ranibizumab) are also set to be newly listed for reimbursement. Therefore, reimbursement standards for the ingredient will be modified, and in consideration of the differences in indication by product, a new phrase was added to allow the administration of the drug within the scope of each drug’s indication.