- LOGIN
- MemberShip
- 2026-04-14 06:57:08
- Policy
- Will Rosuvastatin OD be released?
- by Lee, Tak-Sun Jun 30, 2021 05:55am
- Crestor (Rosuvastatin)Attention is focusing on whether a treatment for hyperlipidemia, Rosuvastatin OD (Orally Disintegrated Tablet) will be released. Rosuvastatin OD was approved in Japan in 2016, but has not yet been released in Korea. According to industries on the 25th, BCWORLD Pharm recently applied for approval of Rosuvastatin OD to the MFDS. AstraZeneca's Crestor is the original for Rosuvastatin. Generics have been released in Korea since 2009, and 941 cases are currently approved. However, there is no formulation of ODT that is melted with the tongue without water. In Japan, AstraZeneca and Shionogi were approved for Crestor OD in 2016. It is said to have been developed by Shionogi. ODT is useful for patients who have difficulty swallowing tablets or who need to refrain from drinking water. However, since most patients are familiar with regular tablets, there will not be much demand for ODT formulation. Last year, Crestor recorded ₩85.5 billion in outpatient prescriptions in the country. Therefore, if ODT is released for the first time, it is enough to attract attention regardless of market size. However, it is too early to discuss the success of commercialization. When a new drug is applied for permission, the MFDS decides whether to approve the sale after safety and the review.
- Company
- Amitiza can be prescribed at general hospitals
- by Eo, Yun-Ho Jun 29, 2021 05:46am
- Amitiza, a chronic constipation drug, can be prescribed at general hospitals. According to related industries, Amitiza(Lubiprostone), which is conducting exclusive promotion activities in Korea through contracts with Takeda Pharmaceuticals, passed the drug committee (DC) of 42 medical institutions nationwide, including AMC and Severance Hospital. Amitiza is used to treat certain types of constipation (chronic idiopathic constipation, irritable bowel syndrome with constipation). Chronic idiopathic constipation has an unknown cause and is not due to diet, other diseases, or drugs. Lubiprostone is also used to treat constipation caused by opioid medications in people with ongoing pain due to medical conditions other than cancer. and was released in February last year. Amitiza, co-developed and marketed by Takeda with Sucampo Pharmacutics, obtained market approval from the U.S. FDA in 2006. Amitiza facilitates CIC-2 chloride channels to increase fluid secretion in the small intestine to relieve constipation symptoms. Clinical trials showed that 60% of constipation patients succeeded in natural bowel movements within 24 hours of taking Amitiza. Amitiza was sold about ₩688.2 billion last year alone in major countries including Japan, the United States and Europe. It is an oral bicyclic fatty acid that selectively activates type 2 chloride channels in the apical membrane of the gastrointestinal epithelium, resulting in increased fluid secretion Lee Kwang-jae, Professor of Gastroenterology at Ajou Medical School (chairman of the Korean Society of Neurogastroenterology and Motility), said, "As it is the first new mechanism in Korea, we expect it to be a primary treatment option for chronic constipation patients who experience daily discomfort."
- Company
- Select the right initial treatment for kidney cancer
- by Jun 29, 2021 05:46am
- Cancer immunotherapies are receiving the spotlight in the treatment of kidney cancer. Demonstrating superiority over existing therapies, these drugs are quickly gaining ground in the field of kidney cancer treatment. Recently, MSD’s cancer immunotherapy ‘Keytruda (pembrolizumab) received approval as first-line treatment in combination with Inlyta(axitinib). Afterward, the immuno+immunotherapy combo ‘Opdivo (nivolumab)+ Yervoy (ipilimumab)’ also received approval as a first-line treatment option. Dr. Chan Kim, Professor of Medical Oncology/Hematology at CHA Bundang Medical Center believes that the use of cancer immunotherapies will broaden in the field of kidney cancer. And there’s ample grounds to support Kim’s belief. Keytruda in combination with a different targeted therapy, ‘Lenvima (Lenvatinib)’ has recently produced even more superior data in a clinical trial. However, limitations do exist. Currently, cancer immunotherapies are approved only for the first-line indication. Therefore, if the patient had previously used other treatments, they may not be prescribed cancer immunotherapy drugs. Therefore, patients with Stage IV kidney cancer must carefully consider and select their first treatment. Dailypharm met with Professor Kim to seek his insight on the significance and prospect of cancer immunotherapy drugs in the field of kidney cancer. Professor Chan Kim-How has the kidney cancer treatment environment changed with the introduction of cancer immunotherapies? = Kidney cancer is a field known for its poor response to general chemotherapy or radiotherapy treatment. Due to this, cytokine-based immunotherapy has been used as an option for over 20-30 years in the field, but even this option has not been broadly used because its objective response rate is only around 10-20%, with serious side effects. This treatment paradigm for kidney cancer has completely changed with the introduction of cancer immunotherapy drugs (immune checkpoint inhibitors). Cancer immunotherapy drugs have fewer side effects than previous drugs and can be used safely even in elderly patients. Most of all, it showed a good therapeutic effect. However, immunotherapy drugs by themselves were not enough to produce satisfactory results as the objective treatment response rate of immunotherapy drugs alone was around 25-30%. As a result, attempts to use immunotherapies in combination with targeted therapies have been made, and clinical studies on this combination were released for kidney cancer in 2019. Results showed that the immuno+targeted therapy combination achieved a twice higher ORR than monotherapy, with an ORR of 55-60%. The uncrossed barrier of 30% was finally overcome. -Were many combination therapies implemented in practice after the data was presented on the immuno+targeted therapy combination? How effective is it in practice? =Use of the combination increased explosively after the presentation. In Korea, Keytruda in combination with the targeted therapy Inlyta is most commonly used. Much treatment data has been accumulated on the combination since its introduction in September 2019. Our hospital presented data on 42 kidney cancer patients who were treated with the Keytruda+Inlyta combination from September 2019 to March 2021 at the 47th Annual Meeting of the Korean Cancer Association that was held from the 17th to 18th this month. One key thing to note is that at the data cut-off point (June 3rd, 2021), the ORR was 54.3%, an excellent rate that is comparable to the clinical trial results. Also, among the patients, 2.9% achieved a complete response and 51.4% achieved a partial response. The median progression-free survival was 12.4 months, and the median OS was not reached in our study. In particular, tumor size in patients with aggressive tumors whose symptoms are difficult to control reduced significantly. 85% of the patients survived, and 63% are still continuing their treatment with the combination therapy. The 1-year survival rate is expected to be around the 90% range. In general, Real World Data (RWD) often does not produce as good a result as clinical trials that were conducted in controlled environments. In this context, the fact our results in the field showed a 90% survival rate that is similar to the clinical trial is encouraging news. -How would you interpret the 42 month long-term follow-up data on Keytruda+Inlyta that was recently presented in the 2021 ASCO Annual meeting? =The academic society had been unsure whether the combination would work in the long-term The data that was presented at the ASCO meeting holds significance as it demonstrated the long-term effect of Keytruda+Inlyta. More specifically, in the KEYNOTE-426 study, the overall survival of the combination therapy was 45.7 months, significantly longer than the 40.1 months when using the standard treatment, ‘Sutent (sunitinib).’ PFS also showed a significant difference of 14.7 months vs 11.1 months. OS at month 42 was 57.5% vs. 48.5%, respectively, and PFS at the same period was 25.1% vs. 10.6%, for the combination therapy group and Sutent group. ORR was 60.4% for the combination therapy and 39.6% for sunitinib. CR was also higher in the combination therapy group, being near 10%, compared to the 3.5% of sunitinib. -The Keytruda+Inlyta combination is currently approved only as a first-line treatment. Does this mean patients who already used other drugs do not have the chance to use the combination? =Yes. Patients often visit our hospital wanting to use Keytruda after using targeted therapies for a month or so. It is unfortunate, but these patients are not allowed to use Keytruda because it is only approved as first-line in Korea. This is why selecting the right initial treatment is ever so important for stage IV kidney cancer patients. If you hastily decide on using a treatment, it is highly likely that you may miss a good opportunity, so I ask the patients to make an informed decision after carefully collecting ample information in advance. -Recently, the Keytruda+Lenvima combination had also shown excellent, differentiated data results from previous kidney cancer treatments. =We have high expectations for the Keytruda+Lenvima combination. In the Phase III CLEAR study, the combination showed a median PFS of 23.9 months, which is over twice longer than the PFS shown in the Sutent group’s 9.2 months. The risk of mortality also decreased by 34% compared to the Sutent group. ORR of the combination therapy was 71%, and 16.1% of the patients reached complete response. The combination is expected to address all the limitations in effect and side effects that arose from previous treatments. If this combination is also approved in the future, 4 treatment options (targeted therapy, immuno+immuno combination therapy, Keytruda+Inlyta, Keytruda+Lenvima) will become the established options used to treat kidney cancer. Among these, the Keytruda + Lenvima combination is expected to be the most anticipated option in the field. -What role would cancer immunotherapies play in the field of kidney cancer in the future? =Cancer immunotherapies are already a well-established treatment option in kidney cancer, and its use will continue to expand in the future. I believe the new treatment trend would be for low-risk groups to first use targeted anticancer therapies and for mid-to high-risk groups to consider the use of immuno+targeted combination therapies. As the combination is yet non-reimbursed, not enough patients are benefiting from this effective treatment option. I hope the government will provide support as soon as possible in consideration of its excellent treatment effect.
- Policy
- Janssen Vaccine for 10,800 people has been approved
- by Lee, Tak-Sun Jun 29, 2021 05:46am
- The MFDS said it approved the nation lot release on the 25th (Friday) for 108,800 people of "Covid-19 Vaccine Janssen" applied by Janssen Korea. Nation lot release is a system in which the state checks the quality of the vaccine once again before it is distributed on the market by comprehensively evaluating the MFDS' test results of the vaccine. The MFDS explained that it has been thoroughly preparing for rapid national lot release of "Covid-19 Vaccine Janssen" by verifying its own test method since early this year and introducing equipment needed for testing such as enzyme analyzers. Test methods established by the MFDS include implantable gene expression, viral gene verification, viral protein verification, vector content, and purity. The effectiveness, safety, and quality were confirmed through the test and manufacturing and test data review of Covid-19 Vacine Janssen Inj and the lot release was decided because it met the national shipping approval criteria. This product was tested for the effectiveness of vaccines such as ▲ antigen protein expression, viral gene confirmation indicating effectiveness, and viral gene and transporter (vector) content by conducting a ▲ antigen test, verification test, etc. ▲It was confirmed that the product was not contaminated by conducting aseptic test, endotoxin test, and purity test. ▲ The MFDS explained that quality consistency was confirmed by conducting quality, pH, and practicality tests and reviewing quality test data issued by the manufacturer's quality assurance manager. Covid-19 Vaccine Janssen Inj is a virus vector vaccine manufactured by incorporating COVID-19 surface antigen gene into the adenovirus mold, the same platform as the country's first licensed AstraZeneca Korea Covid-19 Vaccine Inj. The MFDS stressed that it will do its best to thoroughly verify COVID, which will be introduced in Korea, by making the most of the related infrastructure necessary for the national lot release. Janssen vaccine, which was approved for lot release this time, is not a vaccine donated by the U.S. government, but an individual contract with Janssen.
- Policy
- Leclaza was listed at ₩68,964 per tablet
- by Kim, Jung-Ju Jun 29, 2021 05:46am
- Leclaza 80mg (Lazertinib) made with domestic technology passed the final step to insurance benefits. Leclaza finally chose this track, considering its similar clinical utility to the alternative drug Tagrisso, but cheaper with the Risk Sharing Agreement (RSA) Refund and Expenditure Cap. The date will be started on July 1, which means that the company succeeded six months after the approval in January. The MOHW announced on the 25th that the revised "Pharmaceuticla benefit list and upper price limit table" was proposed as a sub-issue of the 15th Health Insurance Policy Committee. Leclaza is a third-generation epithelial cell growth factor receptor (EGFR) tyrosynkina inhibitor (TKI) and received "conditional approval" from the MFDS on January 18 this year as a treatment for local progressive or metastatic non-small cell lung cancer. At that time, the company applied for insurance registration at the HIRA on December 30 last year, just before being licensed using the drug approval-patent linkage system. On 24 February, the HIRA held the Cancer Drugs Benefit Appraisal Committee to discuss the drug, and was judged on 8 April after deliberation by the Pharmaceutical Benefits Advisory Committee. The Pharmaceutical Benefits Advisory Committee determined that the clinical usefulness was similar to that of Tagrisso, an alternative drug, and that it was therapeutically equivalent to Tagrisso, which is currently being applied to RSA. Leclaza also concluded that following the RSA track would be cost effective at a lower cost than Tagrisso. Korean Cancer Study Group, Korean Society of Medical Oncology, and Korean Association for Lung Cancer suggested these opinions that similar effects have been shown compared to Tagrisso, and it is an additional treatment alternative that demonstrates safety results such as decreased cardiac toxicity and is more effective than treatment. It chose refund and expendature cap among RSA. The RSA Refund type is that a pharmaceutical company reimburses the NHIS for the full amount of pre-determined drug claims, and the Expenditure Cap reimburses the NHIS for a certain percentage of the excess if the actual claims exceed the pre-set annual expected cap. Since then, the MOHW has ordered negotiations on drug prices between the NHIS and the company and agreed on the estimated amount of claims for Refund and Expenditure Cap from April 23 to the 8th of this month. It has agreed that it will make $18.5 million (₩14.1 billion). The NHIS predicts no additional financial needs because there is an alternative drug, Tagrisso. Leclaza was insured from authorization in just half a year. The price is ₩68,964 per tablet, and the benefit will begin in earnest on the 1st of next month.
- Company
- Attention focused on children’s NIP flu vaccine supply
- by Whang, byung-woo Jun 29, 2021 05:46am
- With the bid for the 8 million doses of influenza vaccine for seniors under the National Immunization Program nearly compete, the pharmaceutical industry is intently focused on the supply amount and bid that will be set for the children’s NIP flu vaccines that will follow. According to the Korea Disease Control and Prevention Agency and the industry, the expected NIP quantity of influenza vaccines for those aged 6 months to 12 years will be around 4.6 million doses and will be supplied under a third-party Unit Price Contract. The reason why the pharmaceutical companies are paying attention to the NIP influenza vaccine (hereinafter referred to as flu vaccines) supply for children and adolescents is because the bid amount could affect the number of vaccines that will be released to the private sector in the future. Last year, due to concerns over a ‘Twindemic,’ where COVID-19 and influenza spread simultaneously, the subjects for NIP flu vaccines were temporarily expanded to include those in the 13 to 18-year range, increasing the supply by 1.54 million people’s worth. From this year's perspective, this means that if the flu vaccines for adolescents are removed from the NIP supply this year, that amount will now return to the private sector. Generally, the third-party unit price contract for children’s flu vaccines under the NIP are made without a procurement contract. When the pharmaceutical company supplies flu vaccines to vaccinating medical institutions, the institutions vaccinate eligible children and receive expenses for the vaccination and execution from their local public health centers. The pharmaceutical companies also receive the price for the vaccine from public health centers. When the pharmaceutical company supplies flu vaccines to vaccinating medical institutions, the institutions vaccinate eligible children and then receive expenses for the vaccine and its administration from their local public health centers. The pharmaceutical companies also receive the price for the vaccine from the public health centers. Currently, around 5.2 million children and adolescents are eligible to receive the NIP flu vaccine based on the resident registration statistics in May. The NIP inoculation rate is expected to be around 80% of the eligible population, which means that NIP will roughly require 4.2 million doses. Adding the 500,000 that need to be inoculated twice to the supply, the total amount required will be around 4.7 million doses. Most domestic companies will take part in the bid as NIP suppliers for the pediatric flu vaccines, and Sanofi, which is known to have ramped up its flu vaccine volume by nearly 30% in the absence of SK Bioscience, is also expected to participate in the bid. The pharmaceutical industry expects the total vaccine supply, including the NIP amount, will be around 26 million doses this year. Taking into account the various factors, some believe that the competition among pharmaceutical companies will intensify as the number of flu vaccines released into the private market will be much greater than expected compared to the previous year. However, there also lies the possibility that these expectations will be reversed, as the eligibility age for NIP may continue to be expanded this year in line with the probable approval of the supplementary budget. Analysts predict that supplementary budget requests this year will not be rejected as the Ministry of Economy and Finance had underestimated last year's tax revenue by around 32 trillion won and will be reflecting this amount in the budget. At least, the NIP vaccine age expansion plan will unlikely be rejected due to the lack of a budget. In addition, NA member Jong Sung Lee of the People Power Party had pointed out that NIP for adolescents was virtually nonexistent except for the supplementary budget bill approved in 2020 to inoculate adolescents aged 14 – 18 years old. This pressure from the NA may also be positively reflected in the supplementary budget request. An official from a domestic pharmaceutical company said, “The total NIP amount will be determined according to whether the NIP age range will be expanded or not. The rest will be distributed in the private market. It’s not about whether we agree or disagree to the age expansion plan – the nay or yay needs to be sorted out as soon as possible so we can plan measures accordingly.” Also, the pharmaceutical industry believes that COVID-19 vaccination will be an important variable in establishing a competitive strategy for the flu vaccine market. “This year’s flu vaccine market size may be determined by the COVID-19 vaccination rate this year," a Sanofi official said. "The government will need to provide guidelines for inoculations as the COVID-19 vaccination completion period coincides with the flu vaccination period that was announced by the government."
- Policy
- Following Gilead's hepatitis C tx Epclusa, Vosevi was filed
- by Lee, Tak-Sun Jun 28, 2021 05:50am
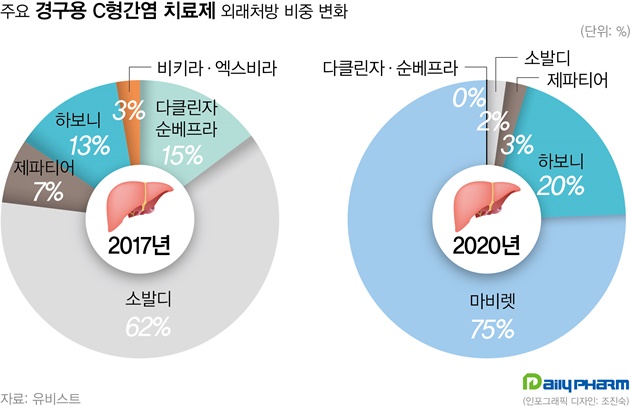
- This year, hepatitis C drugs Epclusa and Vosevi have been filed by the MFDS in Korea. These items are expected to replace Sovaldi. According to the industry on the 27th, Gilead's Vosevi (Sofosbuvir/Velpatasvir/Voxilaprevir) has recently been filed and will begin screening in earnest. Vosevi is expected to be a Pangenotypic DAA preparation that can be used in patients who fail to treat existing Direct-Acting Antiviral (DAA) drugs. AbbVie's Mavyret, a phangenotypic inhibitor, is now the market leading item. And, Mavyret is applicable to both chronic hepatitis C virus genotypes 1, 2, 3, 4, 5 and 6. For Gilead's Sovaldi, it is only valid for type 1, 2, 3 and 4 and must be administered simultaneously with Ribavirin. For this reason, Mavyret currently occupies 75% of the domestic hepatitis C treatment market based on UBIST last year. Sovaldi, by contrast, stood at 2%. Sovaldi had a 62% market share in 2017. Gilead's other hepatitis C drug, Harvoni, came in second with a 20% share after Mavyret last year. Epclusa (Velpatasvir + Sofosbuvir) was also received. Epclusa is also a pangenotypic treatment that can be administered regardless of genotype, a competitive drug for Mavyret. If the two drugs are registered in Korea, it is predicted that they will replace performance of Sovaldi and Harvoni. Although the market for hepatitis C drugs is decreasing in size due to the decrease in the number of patients due to effective drugs, it is attractive for pharmaceutical companies as Mavyret achieved ₩32.6 billion in outpatient prescription last year. As Gilead prepares a new drug, the domestic hepatitis C treatment market is expected to enter fierce competition again.
- Policy
- 16 drugs including Venclexta receive reimbursement in 1H
- by Kim, Jung-Ju Jun 28, 2021 05:50am
- A total of 14 products were newly listed on the insurance benefit list to improve patient access in the first half of this year. Also, two drugs benefited from the reimbursement criteria expansion that was applied to already-listed new drugs. This expanded coverage is interpreted as a result of the government’s decision to flexibly apply coverage for drugs according to the patients’ needs and social importance. The government estimates that around 760,000 patients in Korea will be benefiting from the change. The coverage will financially cost around 115.6 billion won per year, which indicates that the coverage expansion for drugs is being carried out faster than the previous year. During the first half of this year, from January to June, a total of 16 drugs (based on each drug’s representative strengths) were listed on the drug benefit list. These include new drugs that were newly listed and those whose criteria (indication, administration criteria, etc) were expanded to enhance coverage. New drugs that were newly listed this month include the hemophilia A treatment Afstlya inj.; bacteremia treatment Daptocin inj., Boryung Daptomycin inj., and Dapto Inj.; nocturnal hemoglobinuria treatment Ultomiris inj.; and hyperlipidemia treatment Praluent Pen inj.. Also, the reimbursement criteria for the already-listed choric lymphocytic leukemia treatment Venclexta tab. was expanded to enhance coverage. The number of domestic patients expected to use the drugs that were newly listed or received expanded reimbursement varies greatly by product. For the Venclexta tab., the number of patients that will benefit from the expanded reimbursement is expected to be around 75. Also, the number of patients to benefit from the newly listed Ultomiris is 92. Contrary to such drugs that are expected to benefit less than 100 patients, the glaucoma treatment Eybelis Eye Drops is expected to benefit around 45,000 patients. Also, the blood sugar regulator Xultophy Flex Touch inj. that was newly listed in May is expected to benefit 12,756 patients, and Parkinson treatment Equfina Film Coated tab. that started its reimbursement process in January this year is expected to benefit 7,000 patients. The number of beneficiaries greatly varies due to policies that now allow flexible expansion of NHI coverage to high-price drugs that are used for a small number of rare diseases, that were established based on the increased social maturity that can now accept such policies. Due to the new listings and expanded benefit criteria, the government and payer are expected to spend around 115.6 billion won a year to grant access to around 76,769 patients.
- Company
- MET inhibitor ‘Tabrecta’ to overcome Tagrisso resistance?
- by Jun 28, 2021 05:50am
- A study that attempts to address the resistance issue by using the first-ever MET inhibitor ‘Tabrecta’ in combination with the EGFR-TKI ‘Tagrisso,’ will be conducted in Korea. According to the Ministry of Drug and Food Safety on the 25th, Novartis Korea’s application to conduct a Phase III trial on its non-small cell lung cancer treatment ‘Tabrecta (capmatinib)’ was approved that day. Tabrecta is the first-ever MET inhibitor to be approved by the U.S. FDA and was approved in June last year. In Korea, the drug has been designated as an orphan drug by the MFDS in the pre-approval stage. The new clinical trial that will be conducted by Novartis will be studying Tabrecta in combination with Tagrisso (Osimertinib). More specifically, the study will evaluate the treatment effect of Tabrecta+Tagrisso in comparison to platinum-based chemotherapy in patients with non-small-cell lung cancer (NSCLC) with epidermal growth factor receptor (EGFR) mutation, T790M negative, mesenchymal-to-epithelial transition factor (MET)-amplified who progressed following treatment with 1st/2nd generation EGFR tyrosine kinase inhibitors (TKIs) or Tagrisso. MET amplification is one of the known causes of EGFR TKI resistance, in which an EGFR-inhibited cancer cell activates alternative signaling pathways to escape inhibition. It is found in around 25% of all patients treated with Tagrisso and is considered the most commonly observed alteration with C797S mutations associated with resistance to Tagrisso. No treatment that targets the abovementioned mutations currently exists, therefore, patients who develop resistance have no option but to use cytotoxic agents. Thus, work to address these Tagrisso-resistant mutations has been continuing around the world. AstraZeneca is currently conducting a Phase II study on combination therapy using Tagrisso with its new MET inhibitor drug substance ‘savolitinib (product name: Orpathys).’ Savolatinib is a new lung cancer drug in development by AstraZeneca and the Chinese pharmaceutical company, Hutchison China MediTech (Chi-Med). The drug received its first approval in China on the 24th. In the first cohort study that was conducted initially on 46 patients, the combination was associated with an objective response rate (ORR) of 52% with 24 partial responses The median duration of response (DOR) was 7.1 months. In the second, the combination yielded an ORR of 28%, with 12 partial responses. The median DOR was 9.7 months However, the savolitinib+Tagrisso combination showed more side effects in the cohort study. Jassen and Yuhan Corp. is also developing a combination of Rybrevant (amivantamab) and Leclaza (Lazertinib) that targets MET amplification. According to a paper recently published at the 2021 American Society of Clinical Oncology (ASCO) Annual Meeting, the combination resulted in a 36% ORR, with one complete response and 15 partial responses. Rybrevant and Leclaza are each currently approved in the U.S and Korea.
- Policy
- Billing for α-GPC is managed
- by Lee, Jeong-Hwan Jun 28, 2021 05:50am
- The HIRA plans to consider the need to manage claims by selecting Choline alfoscerate, which is under controversy over reducing adaptation certificates, as the "selection focused item." The MFDS plans to periodically monitor pharmaceutical companies that have been clinically reassessed, block unnecessary clinical extensions, and take administrative action against pharmaceutical companies that have failed to reassess. On the 24th, the HIRA and the MFDS responded to Nam In-soon's criticism of the Democratic Party of Korea about Choline alfoscerate. She ordered the HIRA to review and implement follow-up measures to prevent Choline alfoscerate from prescribing drugs based on reduced benefit due to the lack of proven efficacy and effectiveness of other than dementia. The HIRA said it will collect opinions from the public and medical circles and decide whether to select and manage Choline alfoscerate as a selective target after deliberation and resolution process of the central screening coordinator committee. It is a proactive screening system that selects items that need to improve medical trends, such as increased medical expenses, screening problems, and social issues, and makes intensive screening after prior notice to induce improvement in autonomous medical trends of medical institutions. The HIRA will make efforts to make sure that Choline alfoscerate is not prescribed for other than dementia in front-line medical institutions. In addition, the HIRA said it is currently monitoring claims, amounts, and actual number of employees after the suspension of execution of Choline alfoscerate's benefit standard notice. "We will continue to monitor Choline alfoscerate's claims and come up with follow-up measures with related agencies," The HIRA said. "We will continue to re-evaluate the benefit adequacy of the system to streamline spending structure for health insurance fiscal sustainability of health insurance." She ordered the MFDS to block pharmaceutical companies from indiscriminately extending the clinical re-evaluation period of Choline alfoscerate and start managing pharmaceutical companies that have not submitted clinical plans. The MFDS said it will monitor the progress of clinical re-evaluation periodically and speed up the process of changing or canceling permits if it is deemed ineffective or insufficient. The MFDS said 11 items from eight companies that did not participate in the clinical re-evaluation are being subject to secondary administrative measures under the Pharmaceous Affairs law. The first disposal will be suspended for two months, the second disposal will be suspended for six months, and the third disposal will be revoked. The MFDS announced on the 10th that it had previously notified the procedure of changing permission to delete "emotional and behavioral changes, senile caustic depression," which is not included in the scope of clinical re-evaluation. The intention is that there is a need for management only for "secondary symptoms and metamorphic or degenerative cerebral substrate syndrome caused by cerebrovascular deficits" where clinical re-evaluation has been decided. The MFDS said, "To reevaluate the validity quickly, we adjusted and approved the clinical trial period after consulting experts based on the progress of clinical trials of similar adaptive items and data on insurance claims by the HIRA." The MFDS replied, "We will closely review the re-evaluation process and take administrative action against non-submitted pharmaceutical companies."