- LOGIN
- MemberShip
- 2026-04-14 06:57:09
- Policy
- Quality issues alarmed the pharmaceutical industry
- by Jun 22, 2021 05:50am
- Recent cases of GMP violations by Korean pharmaceutical companies have served as an opportunity to guard against risks throughout the industry. In addition, there are opinions that it should be improved in the wake of this incident. Until now, actual changes in standards for GMP have been sluggish according to global level. Only a few companies aiming to expand overseas have voluntarily attempted to improve. Looking at the recent cases, most small and medium-sized companies have been commissioned to produce pharmaceutical drugs to the domestic market, but even large companies, which are consignment companies, cannot avoid responsibility. The consignee lacked quality control, and the trustee had to meet the customer's needs. What efforts are being made in the industry? Dailypharm held the 42nd Future Forum under the theme of 'KGMP Ethics and Compliance! How to Lead' on the 16th and listened to industry opinions. Professor Lee Jae-hyun of Sungkyunkwan University is the head of the group, Lee Sam-soo, CEO of Boryung, Chung Myung-hoon, deputy director of the MFDS' drug quality management department, and Kim Eun-young, CEO of Waters Korea, attended the panel to express their opinions. From the left, Lee Jae-hyun, professor at SKKU, Lee Sam-soo, CEO of Boryㅕng, Chung Myung-hoon, director of the MFDS, and Kim Eun-young, CEO of Waters Korea Labor shortages, lack of technology, and lack of awareness are the causes of the situation CEO Lee Sam-soo analyzed that both environmental and technical factors played a role in the frequent occurrence of drug quality problems. According to him, there were 477 pharmaceutical manufacturers in South Korea as of 2019, and each manufacturer produced ₩49.9 billion. Among them, the average of the bottom 80% is only ₩189 billion. More than 100 finished drugs are owned per company. There was a huge shortage of manpower per plant. There are about 500 factory managers that should be mandatory, and 500 QM and manufacturing managers are also needed. The generic licensing process is also easier than that of advanced countries. CEO Lee said, "In the U.S. and Europe, generics should also have all CTD (Common Technical Document) formats, but in Korea, drugs for data-based re-evaluation are only required." There was a lack of research on quality factors or processes to be considered from the product design stage, consideration of subtle changes that occur while scaling up, and reflection of changes in pharmaceuticals, standards, and equipment. "Let's restore confidence in pharmaceuticals." No matter how important quality management is, it is useless without the will of management. Therefore, the new Drug Quality Management Innovation TF, which was established at the KPBMA, is noteworthy. The TF will be joined by Lee Sam-soo, CEO of Boryung, Kwon Ki-beom, vice-chairman of Dongkoo, Ildong's CEO Yoon Woong-seop, and Huons Global vice-chairman Yoon Sung-tae, and Hee-mok Won, chairman of the KPBMA. The team is based on owners and representative directors who can make decisions. The main role of TF is to identify each company's permission and cases of non-compliance with GMP, analyze the causes, and present solutions. It will also find matters that need to be improved. It also has a working group of TF that focuses on GMP experts from 15 businesses such as factory manager, QM manager, and research institute head. They listen to the field's opinions and share their opinions with the MFDS. Industries are actively supporting amendments to 1+3 Pharmaceutical Act by actually operating TF. The industry agreed with the MFDS that the introduction of Quality by Design (QbD) and data integrity should be emphasized to improve quality control. The purpose of the TF is to achieve various education, meetings, information sharing, and system improvement to realize these goals. CEO Lee said, "We intend to expand human resources education by opening online seminars and curricula of the association, visit each other among manufacturing plants, and share data." "We will also achieve quantitative and qualitative improvements by supplementing QA/QC personnel and supporting regular training." In order to spread the introduction of QbD, it should be accompanied by support measures. QbD, the industry believes that carrot-and-stick policies are needed to achieve a common goal of data completeness. CEO Lee said, "QbD is key to both risk assessment and statistics, and in order to become a smart factory, auxiliary tools such as PAT must be applied together." Real-time analysis does not follow simply by doing QbD. "It will cost tens of billions of won to install analysis equipment and sensors for auxiliary tools, so it will not be easy to introduce unless it is a blockbuster item." As a result, industries are strengthening regulations on quality management while also requesting support measures. It applies tax benefits for facilities/equipment necessary for QbD or data completeness, rapid review of related items, and preferential treatment of drug prices. "We will discuss with innovative TFs and actively consider operating a developmental GMP system," said Chung Myung-hoon, an official who attended as a representative of the MFDS. "We will listen to various opinions such as difficulties of small and medium-sized companies so that we can actively improve the constitution of the industry."
- Policy
- Closing the opinion gap important for the generic '1+3 bill'
- by Lee, Jeong-Hwan Jun 21, 2021 05:51am
- With the bill limiting the participating generic makers to three consignees per consignor when conducting joint biological equivalence tests or clinical tests awaiting review by the National Assembly’s Legislation and Judiciary Committee., whether the opinion gap between the large pharmaceutical companies and small- and medium-sized pharmaceutical companies can be closed is gaining attention. Despite the unanimous decision made by the Health and Welfare Committee, concerns have been rising on the influence the divided positions held by different sized pharmaceutical companies, which, if not resolved, may have on the process and result of the review conducted by the Legislation and Judiciary Committee. On the 20th, the pharmaceutical industry has been fighting a psychological battle behind the scenes on the legislation of the ‘1+3 restriction of generics’ bill The Health and Welfare Committee that passed the bill agreed that the 1+3 bill will reduce the excessive amount of generics and drugs requiring data submission and significantly improve the structure of the pharmaceutical drugs in Korea, however, the pros and cons are still fiercely being judged in the industry. While the large pharmaceutical companies that own the capability and economic scale to develop new drugs are strongly in favor of the 1+3 bill, the small- and mid-sized pharmaceutical companies that profit from generics and data submission drugs have been complaining that the bill will serve as leverage to promote the collapse and job cuts in their companies. In particular, the small- and mid-sized pharmaceutical companies have also criticized that, regardless of its purpose, legislating a bill to reduce the excessive number of items is out of order as they have been selling the data submission drugs according to the regulations set by the government. As a result, the logic that the excessive number of generic items have made it difficult for the government to respond or manage GMP violations or detection of impurities such as NDMA and that a regulation tailored for large pharmaceutical companies that does not reflect the reality of small and medium-sized companies will be clashing at the Legislation and Judiciary Committee’s review. According to the National Assembly Act, the Legislation and Judiciary Committee only owns the right to review a bill(right to review the system and order self-revision) that has been already reviewed and passed by the relevant standing committee, however, in reality, there exists cases where the bill passed by the relevant standing committee remains halted at this stage without being processed. For example, the bill to revoke the license of a doctor who was sentenced to imprisonment or higher has been on hold for more than two months after the Health and Welfare Committee’s vote due to disputes between the ruling and opposition parties during the judiciary committee's review. In other words, whether the large and small- and mid-size companies will be able to reach a consensus on the bill may act as one variable for passing the bill. The Legislation and Judiciary Committee plans to review the 1+3 bill passed by the Health and Welfare Committee in July. If the bill passes this review, the final legislative process will likely be completed at the plenary session that will be held in the same month. An official who is in charge of development at a top domestic pharmaceutical company said, “The small- and mid-sized companies are arguing that the law should allow 4 or more pharmaceutical companies to jointly develop and produce the data submission drugs as its clinical trial costs 5 to 15 billion won. However, the fact is a clinical trial that costs 10 billion won is uncommon. If the companies are really opposing due to the burden of cost, they may receive recognition as a 1+3 bill exception according to the exception clause specified in the bill. The companies need to understand how serious the excessive generic and data submission drugs issue is and work to make remedies to their system.” Another official from a small- and mid-sized pharmaceutical company said, “Regulation on generics is necessary, but I oppose to the regulations being set on data submission drugs. The small- and mid-sized companies with insufficient power to develop new drugs yet need to have some possibility of joint development to be able to maintain management and make a cash cow with incrementally modified drugs. The bill is just another order for company restructuring. The bill will directly have an impact on reducing the number of pharmaceutical companies rather than the number of generic companies. This would inevitably lead to job cuts in the companies, so I cannot agree with a bill that disregards this reality.”
- Company
- Competition between PCSK9 Inhibitors has just begun
- by Eo, Yun-Ho Jun 21, 2021 05:51am
- The competition for prescription of PCSK 9 inhibitors began more than four years after the domestic approval. Sanofi-Aventis' Allepatadine (Olopatadine) was listed on the 7th. It was approved in January 2017. This was the first time in South Korea that PCSK's 9th suppression system was introduced. Later in April of the same year, Amgen's Repatha (Evolocumab) was approved, with a stronger willingness to register benefits. Repatha was first registered as Homozygous Family Hypercholesterolmia (HOFH) in August 2018. Although there were two drugs in PCSK 9 inhibitors, Repatha was the only option. PCSK 9 inhibitors are drugs that have excellent efficacy but had price issues. There were problems not only at home but also at a global level, and Amgen lowered the price of drugs held by 60% in October 2018 and Sanofi in February 2019. Sanofi voluntarily withdrew Praluent's application for registration in October 2018, before the drug price was lowered, and it was registered about two years later. PCSK.9 inhibitors had a wide range of requirements for HOFH and in January 2020, Repatha succeeded in extending the reimbursement standard to patients with atherosclerotic cardiovascular disease (ASCVD) high risk, Heterozygous Family (HeFH), and Statinability. The two drugs are believed to compete in the ASCVD and HeFH. Praluent added HoFH in the United States in April. Praluent has tended to reduce the risk of all-cause death, and is a licensed drug with Praluent 75 mg and Praluent 150 mg, allowing patient-specific dose selection with reference to patient status and LDL-C levels. Repatha, which is first listed, is already prescribed by medical institutions. It can be prescribed at major medical institutions across the country, including the Big 5 general hospitals, including Samsung Medical Center l, Seoul St. Mary's Hospital, AMC, and Sinchon Severance Hospital.
- Policy
- Gov will proactively improve system for severe psoriasis
- by Kim, Jung-Ju Jun 21, 2021 05:50am
- With a year left before the re-registration of special exemption of insurance calculation for severe psoriasis, the government, payer, and patient group gathered to discuss improvement. The issue discussed was that despite reimbursement approved for severe psoriasis drugs, patients are not being properly covered as the eligibility standards for the special calculation system are too strict, which increases the actual cost borne by patients. The patient group appealed to the government and payers that the standards for severe psoriasis should be set at the same level as other severe incurable diseases rather than solely relying on expert opinions, and the authorities said that they will take a proactive stance in resolving the issue. The Korea Alliance of Patients' Organization and the Korea Psoriasis Association had raised the issue at its 'Patient Shouting Cafe' event held on the 18th. Immediately after the event, the Ministry of Health and Welfare and the National Health Insurance Service held the roundtable and expressed their positions on improving the special exemption of insurance calculation for severe psoriasis. A patient group has requested improvement of the registration criteria for the exempted calculation of health insurance for severe psoriasis that will be up for re-registration in one year to the government and payers. Pic. from the hosted by the Korea Alliance of Patients Organization The point raised by KAPO was clear and simple. Patients with severe psoriasis have to bear a very high cost of treatment that can only be relieved through the special exemption of insurance calculation. However the entry criteria for the special calculation, the new and re-registration standards, are too strict, and only 17,500 out of the 22,000 severe psoriasis patients are unable to receive any benefit from the system. In other words, for most patients, the benefit is just a 'pie in the sky.' As the re-registration of special exemption of insurance calculation for severe psoriasis will start next year, unless the system is improved now through discussions, the issue is doomed to reoccur. However, the biggest barrier to the improvement is that the experts' opposition to making changes in the registration criteria. In fact, at the National Assembly's Health and Welfare Committee plenary session on the 16th, Yong-ik Kim, president of NHIS had answered Rep. Chun-sook Jeong's inquiry that as the payer, the organization had to follow the experts' judgment as its experts opposed to the change of new registration standards for special exemption of insurance calculation for severe psoriasis. On this, the patient group emphasized that when setting registration standards for special exemption of insurance calculation, policy judgments should be made based on equity with other disease and social consensus rather than simply by reimbursement standards. They argued that the special exemption system is different from medical insurance benefit standards that require consideration of cost-effectiveness and efficacy and that the government is hiding behind experts' judgment. High-priced biologics for severe psoriasis are already being reimbursed. However, contrary to other severe diseases like Crohn's disease, ankylosing spondylitis, and severe atopic dermatitis that apply special calculations immediately for high-priced biologics, the special calculation does not apply to severe psoriasis despite the reimbursement approved for its treatment. The high out-of-pocket payment borne by the patients greatly affects the livelihood and life of the patients. On this, the Korea Psoriasis Association, a direct stakeholder to the system, suggested that the NHIS transparently discuss this issue at the Special Exemption of Calculation Committee meeting, a discussion body for various stakeholders rather than blindly follow the judgment (medical judgment) made by the expert advisory committee. MOHW's Insurance Benefits Division and the NHIS responded that they will address the issue of new registration standards of severe psoriasis in a proactive manner and identify and find solutions for the issue that was discussed at the meeting.
- Policy
- Yuhan's Raboni-D has been licensed
- by Lee, Tak-Sun Jun 21, 2021 05:50am
- Yuhan, which had a high dependence on sales for imported drugs, has recently been speeding up with the commercialization of new drugs such as Lazertinib and IMD. In particular, Yuhan refrains from entrusting or entrusting developing products and is building market competitiveness with its own products. The MFDS approved "Raboni-D," Yuhan's combined osteoporosis IMD, on the 17th. It is a combination drug of Raloxifene HCl, an existing osteoporosis treatment drug, and Cholecalciferol Concentrated Powder, known as a vitamin D. There are already six products, including Hanmi Pharmaceutical's "Rabone D" and Alvogen Korea's "Ebistrar Plus," that combine the two ingredients. However it is evaluated that it has secured safety by lowering the amount of Raloxifene that was developed by Yuhan. Raboni-D is Raloxifene 45mg, while conventional products are Raloxifene 60mg. Raboni-D is used for the treatment and prevention of osteoporosis in postmenopausal women, and is given oral administration one tablet a day. Pregabalin SR, a pain reliever, is also a self-developed item by Yuhan Corporation. 15 items, including Pfizer's Lyrica CR, are licensed. Yuhan was also granted Yuhan Pregabalin SR in 2019. However, safety and effectiveness of Yuhan Pregabalin SR have not been confirmed in patients with new functional disabilities and those aged 65 or older compared to Lyrica CR. Yuhan is focusing on securing competitive edge in products that are as good as original by expanding clinical trials. In this clinical trial, pharmacokinetic characteristics and safety will be evaluated for patients with new disabilities and healthy people who have not been established safety and validity in the past when administering YHD119 single oral administration. Yuhan has been steadily decreasing its sales share of new drugs due to the expansion of its own development items. As of the end of the first quarter, sales of products stood at 56.6%, down 3.9% points from 60.4% in the same period last year. Analysts say that at this rate, the proportion of new drugs introduced in a few years will fall below 50%. The new drugs vary greatly with copyright agreements with developers, so there is a risk of maintaining sales. Leclaza was approved early this year, starting with the commercialization of the first IMD Duowell in 2014. Attention is focusing on whether Yuhan, which generated ₩1 trillion in sales for the first time in the pharmaceutical industry, will also gain fame as a new drug development company.
- Product
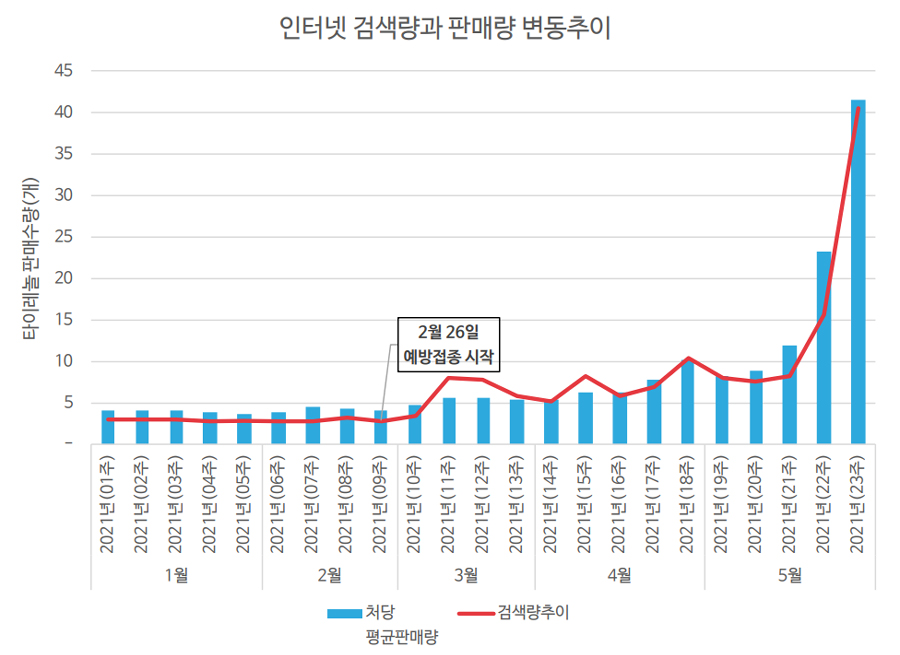
- Tylenol sales increased 10 times to peak in May
- by Jun 21, 2021 05:50am
- Pharmacies' difficulties have reached their peak due to the surging demand for Tylenol, which is said to be "more precious than COVID-19 vaccine." This is because Tylenol has become so precious that it is rarely found in pharmacies. Then how popular was Tylenol? Analysis of POS data from 185 pharmacies showed that sales of Tylenol 500 mg and Tylenol ER 8HR increased 10 times compared to the beginning of this year. According to a survey of sales at 185 pharmacies, Care Insights recorded a peak in May after steadily increasing in March and April since the COVID-19 vaccination began on February 26. In particular, demand more than quadrupled compared to the average year in May. According to this year's sales, 12,467 in January, 12,222 in February, 20,756 in March, 29,447 in April and 54,340 in May. From January to May, 129,232 units were sold. Compared to 126,590 in 2018, 138,327 in 2019, and 173,687 in 2020, the company sold a year's worth in five months. The market share of Tylenol in Pharmacies was also overwhelming with 92.4%. Other APAP formulations accounted for only 7.6%. Considering that the average Tylenol sales share was 82.6% last year, the market share itself has risen further. An official from Care Insights said, "The more Tylenol is mentioned through the media, the more Tylenol sales tend to increase." The official said, "Although 100 to 200 Tylenols are supplied per pharmacy, this is believed to be the amount that can only be sold for about 15 days," and added, "As the vaccination rate is increasing, it seems necessary to come up with measures."
- Company
- JAK inhibitor ‘Olumiant’ seeks reimb for atopic dermatitis
- by Eo, Yun-Ho Jun 18, 2021 05:54am
- The JAK inhibitor ‘Olumiant’ is seeking extended reimbursement benefit in atopic dermatitis. According to industry sources, Lilly Korea has submitted an application for the reimbursement of Olumiant (baricitinib) in ‘the treatment of adult patients with moderate to severe atopic dermatitis who are candidates for systemic therapy’. The company had rapidly carried out the listing application process after receiving approval for this additional indication last month. As a new drug for moderate to severe atopic dermatitis, Olumiant’s reimbursed price is expected to be set at a more economic price than recently listed new treatment for atopic dermatitis, ‘Dupixent (dupilumab).’ Olumiant selectively and reversibly inhibits JAK1 and JAK2 to reduce the expression of inflammatory cytokines and has an anti-inflammatory effect. Olumiant demonstrated significant treatment effect as well as safety as monotherapy and as combination therapy with a topical corticosteroid (TCS) in adult patients with moderate to severe atopic dermatitis compared to placebo in 3 clinical trials - BREEZE-AD1, BREEZE-AD2, and BREEZE-AD7. In the three studies, patients treated with Olumiant showed an improvement in their symptoms such as itching, which severely deteriorates the patients' overall health state and quality of life, as early as in the second day of treatment. Dr. Chang-Wook Park, Professor of Dermatology at the Severance Hospital said, “The rapid improvement of symptoms that were observed from the second day of Olumiant treatment in the patient-reported outcome gives hope to atopic dermatitis patients in Korea who previously had limited treatment options.” Park added, “With its strengths in rapidly improving itch symptoms, convenient oral administration that improves patient compliance, and long-term safety profile that has been confirmed as a rheumatoid arthritis treatment, I expect Olumiant will be able to address patients' unmet needs in the field of atopic dermatitis.”
- Policy
- An exception to the 1+3 Bill for IMD
- by Lee, Jeong-Hwan Jun 18, 2021 05:54am
- A letter from a pharmaceutical representative to the National Assembly affected the process of the National Assembly's Health and Welfare Committee's handling of generics, drug for data-based re-evaluation "1+3 bill." It was reflected in the revised schedule when a representative of company A sent a petition to 24 members of the National Assembly's Health and Welfare Committee that IMD, which has decided to jointly develop more than four pharmaceutical companies, could have a negative impact on development and launch. The Pharmaceutical Affairs Law, approved by the National Assembly's Health and Welfare Committee on the 16th, contains the effective date, application case, and progress measures for medicines undergoing clinical trials. The most controversial part was the transitional provision of drugs already approved, or drugs already undergoing clinical trials, regardless of the Bill. If the bill goes into effect, pharmaceutical companies that exceed the four pharmaceutical companies regulated by the bill have pointed out that it is difficult to release drugs under joint development. In line with the implementation of the 1+3 bill, the welfare committee added an additional rule to apply the previous regulations to drugs under joint development after four pharmaceutical companies were approved by the MFDS. In this case, however, data proving the decision to jointly develop must be reported to the MFDS within one month from the enforcement date of the law. The amendment also reflected claims contained in a petition letter from the representative. In a letter, he explained that IMD, an arthritis drug that the company is jointly developing with several other pharmaceutical companies, is on the verge of being suspended with the 1+3 bill. He added that IMD, which requires billions of won, is conducting Phase 3 clinical trials by investing development costs jointly with multiple domestic companies because it is difficult for small and medium-sized pharmaceutical companies to invest R&D costs alone. During the development process, the 1+3 regulation bill passed the welfare committee on April 29, and he stressed that his company was in danger of having to stop developing IMD research and development because of the bill. "We can accept the general regulations to prevent the disturbance of generic drugs," he said in a letter. "The IMD's regulations can be difficult to develop without a certain period of delay," he said. "The IMD has already received development costs from multiple domestic companies and is in the process of Phase III clinical trials." The welfare committee partially accepted his intention of IMD and established an additional clause that submits and reports evidence that he is jointly developing it to the MFDS within a month of the enforcement of the law. An official of the welfare committee said, "We collected opinions to minimize confusion among pharmaceutical companies that have been preparing for IMD development through joint development contracts regardless of the bill." "We have resolved unreasonable parts and improved the completion of the bill with the provision of transitional measures for medicines undergoing clinical trials," he explained.
- Policy
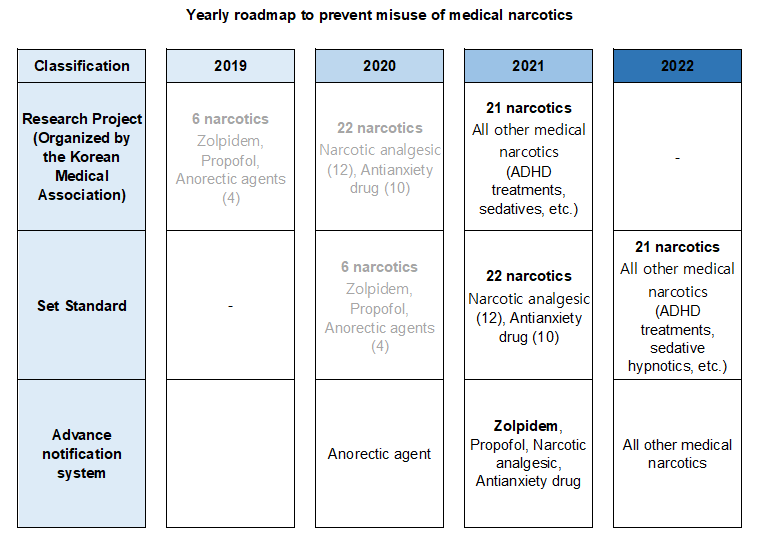
- 559 doctors warned for wrongful prescription of Zolpidem
- by Lee, Tak-Sun Jun 18, 2021 05:53am
- After analyzing the prescription information on the Narcotics Information Management System (NIMS), the Ministry of Food and Drug Safety announced its decision to issue written ‘warnings’ to 559 doctors that have continuously prescribed or used Zolpidem beyond the safe use standards to prevent abuse and promote proper use of the medical narcotic, Zolpidem. This measure is the second step taken by regulators based on analysis results of the 2-month prescription and use history that was collected after notifying the 1,720 doctors who prescribed and used Zolpidem beyond the safe use standards on March 2nd, through the advance notification system as the first step. MFDS said that the doctors' compliance rate of medical narcotics safety guidelines had improved after implementing the advance notification system. The number of doctors who prescribed and used Zolpidem beyond the set safe use standards after receiving the first step warning in March decreased 68% from 1,720 to 559, and the total number of prescriptions also decreased 51% from 5,593 to 2,724. Also, the ministry added that it plans to conduct on-site inspections and take administrative actions (suspension of handling narcotics) to restrict prescriptions of doctors who do not correct their actions in prescribing medical narcotics beyond the scope set under the safe use standards even after the second written warning. An MFDS official said, “The advance notification system that has been implemented to anorectic agents in December last year will also be applied to analgesics and anti-anxiety drugs this year. By next year, we plan to expand the system to all narcotics, through which we aim to continue our efforts in preventing misuse and abuse whiling encourage safe use of medical narcotics in Korea.”
- Policy
- “The 1+3 bill” was passed by the Welfare Committee
- by Lee, Jeong-Hwan Jun 18, 2021 05:53am
- The 1+3 bill passed a plenary session of the National Assembly's Health and Welfare Committee on the morning of the 16th and will be reviewed by the legislation and judiciary committee. The resolution reflected the revision of the supplementary provision, which excludes the report from the MFDS within a month from the enforcement date of the law, a drug for data-based re-evaluation, which was approved before the enforcement of the law. The welfare committee has passed on a number of the pharmaceutical affairs law to the judiciary committee, including the bill, mandatory preparation and submission of expenditure reports on pharmaceutical CSO, designation of drug days as a legal anniversary, and mandatory safety vulnerable braille and voice codes. A total of 94 bills were handled by the welfare committee on the same day. The most interesting legislation is the 1+3 bill. The main point of the bill is to allow up to three consignees per trustee to conduct joint biological equivalence tests and clinical trials necessary for the development of drug for data-based re-evaluation with generic. Regardless of the revision of the bill, the welfare committee partially reflected the transitional measures for medicines that are already undergoing clinical trials. They say that drugs that have signed a joint development contract and received approval for a clinical trial plan from the head of the MFDS do not apply the 1+3 regulation. In this case, however, pharmaceutical companies that have been approved for clinical trials must submit data proving the joint development of drugs to the MFDS within one month from the enforcement date of the law. Like pharmaceutical companies, CSO, which is in charge of promoting drug sales, has also been designated as a drug supplier, imposing obligations to prepare and submit expenditure reports for doctors and pharmacists, and regulating those who illegally purchased Rx drugs. The welfare committee passed on amendments to the Infectious Diseases Act, the Medical Devices Act, the Infectious Disease Prevention and Control Act and the Health Insurance Act to the legislation and judiciary committee.The welfare committee introduced 114 new bills to the plenary session to continue its review at the legislation and judicial committee. They included bills that surveys and public announcements on the status of illegally licensed pharmacies, hospitals run illegally by the office manager and illegally licensed pharmacies( they will be excluded from health insurance care institutions), vaccine development status sharing, paid leave after vaccination, expanded national coverage for side effects, and prohibition of the establishment of a medical institution for child sex offenders.