- LOGIN
- MemberShip
- 2026-04-03 16:43:01
- Opinion
- [Reporter’s View]Biotech’s responsible approach to failure
- by Cha, Jihyun Jul 15, 2025 06:06am
- Cases of terminated licensing deals and halted clinical trials are on the rise in Korea's biotech sector. Recently, IntoCell announced that the licensing agreement for its ADC (antibody-drug conjugate) platform with ABL Bio had been canceled due to patent overlap issues. Prior to that, Orum Therapeutics voluntarily decided to halt clinical development of its lead pipeline after the occurrence of serious adverse events (SAEs). This series of announcements quickly translated into investor losses. IntoCell’s stock plummeted immediately after the termination of its licensing deal was made public. The following day, the company’s share price closed at a price 25.9% lower compared to the previous trading session. On the day of the disclosure, IntoCell hit the lower limit in after-hours trading (3:40–8:00 p.m.), falling to KRW 28,900. When Orum Therapeutics announced the suspension of its clinical trial, its stock price also fell to the lower price limit, showing a similar trend. Ironically, both companies had recently gone public via Korea’s special technology listing track (Tech Exception IPO). Less than two months after listing on the KOSDAQ, they released negative disclosures. This has fueled suspicions that the licensing deals may have been used to inflate valuations and justify higher offering prices during the IPO process. Critics have also raised concerns that the Korea Exchange (KRX) failed to properly vet these companies during the tech exception listing review process. However, it is necessary to take into account the fact that failure is inevitable in the biotech industry. New drug development is inherently a high-risk, high-reward industry with a high failure rate. Even global big pharma companies often succeed in developing only one out of dozens of candidate substances. Midway terminations of licensing agreements or discontinuation of clinical trials are not uncommon, both in Korea and abroad. In other words, failure itself cannot be regarded as the core issue. Due to the structural nature of the biotech industry, it is also unrealistic to expect the stock exchange to filter out all risks in advance. However, this does not mean that all failures should be excused. Companies must maintain a minimum level of responsibility for their technology and decision-making processes. This responsibility is even greater for publicly listed companies, as their actions can undermine trust in the entire domestic biotech industry. In fact, investors have already witnessed multiple instances where poor handling by individual companies has led to a collapse in overall market confidence. In the end, what truly matters is how a company responds after failure. The real test lies in how transparently it discloses the reasons behind a terminated clinical trial or a canceled deal, the technical limitations involved, and what alternative plans it puts forward. The greater risk is not the failure itself, but the inability to provide a convincing explanation for it. To earn a second chance in the market, companies must be willing to thoroughly explain the causes of failure and demonstrate a proactive attitude — even amid uncertainty. In this regard, IntoCell's response was noteworthy. For two days after the contract termination, IntuCell issued two official statements, sharing details on the structure and analysis of the patent issues that led to the termination, the results of legal reviews, the connection to its current pipeline, and plans to secure additional technology. The company also pledged to restore market communication and trust by having its CEO purchase KRW 10 billion worth of company shares and expanding investor relations (IR) activities. IntoCell's actions stand in contrast to some biotech companies that have remained silent or offered vague explanations in the face of failure. It remains to be seen how faithfully IntoCell will follow through on its commitments and whether this communication approach will help restore market trust in the future. Hopefully, this case will serve as a turning point for the Korean biotech industry—prompting a broader reflection on how companies confront and manage failure.
- Company
- Various immunotherapies attempt to treat Alzheimer’s
- by Son, Hyung Min Jul 15, 2025 06:06am
- A series of immunotherapy candidates are showing promise in the field of Alzheimer's disease treatment. With the limitations of existing antibody therapies becoming apparent, multinational pharmaceutical companies are attempting approaches targeting new immune pathways. In particular, pipelines targeting innate immune regulation, such as NK cell therapies and TREM2 antibodies, are gaining attention. According to industry sources on the 12th, US biotech company NKGen recently administered its autologous NK cell therapy candidate ‘troculeucel (SNK01)’ for the first time to patients with mild Alzheimer's disease who did not respond to existing antibody therapies. This is a case approved under the U.S. Food and Drug Administration's (FDA) Compassionate Use program, applied to patients whose cognitive decline persisted despite treatment with the amyloid-targeted therapy lecanemab. Troculeucel, NKGen’s cell therapy developed by using non-genetically modified ex vivo expanded autologous NK cells, is currently in a Phase IIa clinical trial in the U.S. targeting moderate patients. NKGen stated, “The therapy demonstrated efficacy in improving amyloid, tau, and alpha-synuclein levels in cerebrospinal fluid (CSF) by crossing the blood-brain barrier (BBB), as well as reducing neuroinflammatory markers such as GFAP.” In the previously conducted Phase I clinical trial, troculeucel demonstrated cognitive function stabilization and improvement in approximately 70% of patients, along with changes in biomarkers such as reduced pTau181 and GFAP levels in CSF. Treatment responses were dose-dependent, and no significant adverse reactions were observed. While existing antibody therapies have only slowed the progression of cognitive decline, NK cell therapy targets both inflammation control and protein aggregation resolution, opening up the possibility of a new mechanism of action for use. In particular, its use in patients unresponsive to existing drugs suggests potential as part of a combination strategy or as a new treatment option. NKGen Major global pharmaceutical companies have also begun full-scale development of antibody and small-molecule therapies targeting TREM2. Novartis recently began a multinational Phase II clinical trial of VHB937, a TREM2 antibody-based drug candidate, in multiple countries, including South Korea. This study is designed as a randomized, placebo-controlled trial to evaluate the efficacy and safety of VHB937 in patients with early-stage Alzheimer's disease over 72 weeks. VHB937 increases the expression of TREM2, a surface receptor on dendritic cells, and inhibits its shedding. According to the company, this strategy aims to enhance intracellular signaling, thereby simultaneously targeting the phagocytic function of microglia and anti-inflammatory effects. Sanofi recently acquired Vigil Neuroscience, a company developing a TREM2 small molecule agonist, and entered the field in earnest. VG-3927, which Vigil is developing, binds only to cell membrane receptors and does not bind to soluble TREM2, enabling more precise regulation of microglia function, which is a key differentiator. Sanofi plans to begin Phase II clinical trials of VG-3927 in the third quarter of this year. Targeting TREM2 is a mechanism of action that leading companies such as AbbVie and Takeda have experienced a series of failures. However, latecomers are shifting their focus to stabilization strategies and small molecule mechanisms after carefully analyzing the reasons for previous failures. New mechanisms continue to be developed in Korea as well In the field of Alzheimer's disease drug development, innovative approaches that go beyond the existing methods of removing amyloid beta and tau proteins and aim to regenerate and protect brain neurons are emerging. SNR1611 (generic name: trametinib), developed by the Korean drug development company Genuv, is one representative example. SNR1611 is a repurposed small-molecule compound derived from the already-approved anticancer drug trametinib. It works by inhibiting MEK1/2 enzymes within the MAPK/ERK signaling pathway. Through this mechanism, it suppresses the aberrant activation of MAPK signaling observed in neurodegenerative diseases such as Alzheimer’s disease, thereby promoting neural stem cell differentiation and the generation of new neurons. Recent preclinical studies have shown that SNR1611 promotes the expression of specific genes involved in brain neuron differentiation, resulting in a more than 2.7-fold increase in brain neural regeneration and approximately a twofold improvement in memory. These findings were published in “Experimental & Molecular Medicine,” a sister journal of the prestigious scientific journal Nature, thereby establishing scientific evidence. When SNR1611 was administered to an animal model with Alzheimer's disease, neural regeneration increased by approximately 176% and 295% in the dentate gyrus and subventricular zone, respectively, which are critical regions for memory formation. Additionally, neural regeneration effects were confirmed in the cerebral cortex, and memory improved by nearly twofold in the treatment group. Notably, SNR1611 has drawn attention for its ability to induce neurogenesis—a process previously considered nearly impossible in the adult brain—through pharmacological intervention. Genuv is also developing SNR1611 as a treatment for amyotrophic lateral sclerosis (ALS). In ALS animal models, administering SNR1611 reduced abnormal protein aggregation through autophagy activation and confirmed motor neuron protection effects. Currently, Phase 1/2a clinical trials for ALS are underway at five major hospitals in South Korea. AriBio is developing AR1001, a new drug candidate for Alzheimer's disease. In March, the company successfully transferred the technology to China's Q-BioTech. AR1001 targets multiple causes of Alzheimer's disease, including PDE5 and toxic proteins. This new drug candidate is an improved oral treatment based on mirodenafil (Mvix), a medication similar to Viagra for erectile dysfunction. Recent findings suggesting that PDE5 inhibitors like Viagra and Cialis may prevent Alzheimer's disease provide further evidence supporting the mechanism of AR1001. AR1001 is currently in Phase III clinical trials. AriBio has initiated global Phase III clinical trials in the United States, where the first patient dosing began in December 2022, as well as in Korea, the United Kingdom, Germany, France, Spain, Italy, Denmark, the Netherlands, the Czech Republic, and China.
- Company
- Omjjara’s reimbursement progress gains attention
- by Eo, Yun-Ho Jul 14, 2025 06:04am
- Attention is turning to whether progress will be made in the reimbursement listing process for the new myelofibrosis drug, Omjjara. Omjjara (momelotinib), a myelofibrosis treatment developed by GSK Korea, passed the Health Insurance Review and Assessment Service's Cancer Disease Review Committee in March and is currently awaiting submission to the Drug Reimbursement Evaluation Committee. It remains to be seen whether the HIRA process will be completed within the second half of this year. The indication currently under review is for the “treatment of myelofibrosis in intermediate- or high-risk adults with anemia.” Omjjara has a triple mechanism of action – it blocks 3 key signaling pathways, not only JAK1 and JAK2 that were inhibited by existing therapies, but also the ACVR1 (activin A receptor type). In the treatment of myelofibrosis, inhibition of JAK1 and JAK2 may contribute to improving systemic symptoms and reducing splenomegaly in patients, while inhibition of ACVR1 may help alleviate anemia by inducing a reduction in hepcidin expression. Anemia management is one of the unmet needs that remain in the treatment of existing myelofibrosis patients. Anemia, which increases blood transfusion dependency, is not merely an issue of dizziness as commonly perceived, but can lead to a life-threatening condition depending on its severity. Omjjara demonstrated significant improvements in key symptoms such as splenomegaly and a reduction in transfusion dependency in patients with anemia-associated myelofibrosis, regardless of prior JAK inhibitor treatment history, in the Phase III SIMPLIFY-1 study and the MOMENTUM study. In the SIMPLIFY-1 study, which evaluated the clinical efficacy and safety of Omjjara in comparison with Jakavi (ruxolitinib) in patients with myelofibrosis and had no prior JAK inhibitor treatment experience, Omjjara demonstrated non-inferiority to ruxolitinib in the primary endpoint of spleen volume response at 24 weeks of treatment. The proportion of patients in each arm who were transfusion-free was 66.5% in the Omjjara arm and 49.3% in the ruxolitinib arm, with significantly lower transfusion dependence (better transfusion independence) in the Omjjara arm. Seo-Yeon Ahn, Professor of Hematology at Chonnam National University Hwasun Hospital, said, “While JAK inhibitors previously used in the treatment of myelofibrosis demonstrated efficacy in reducing splenomegaly and alleviating systemic symptoms, they also posed unmet needs such as worsening anemia or increasing blood transfusion dependency. Omjjara has demonstrated significant clinical value in managing anemia, which is closely linked to the prognosis of patients with myelofibrosis. With its domestic launch, it is expected to contribute to improving treatment outcomes and quality of life for more patients."
- Company
- Aftermath of Januvia's price cut…MSD-CKD compensation fight
- by Son, Hyung Min Jul 14, 2025 06:04am
- Chon Kun Dang Regarding transaction for price difference compensation following the drug price reduction for the diabetes treatment Januvia, Chong Kun Dang and MSD Korea have not reached agreement during negotiation for nearly two years. While the distribution industry continues to demand compensation for losses incurred due to the drug price cut, the lack of clarity on who is responsible for the settlement is exacerbating confusion and burden in the field. According to industry sources on July 14, Chong Kun Dang recently sent an official letter to the Korea Pharmaceutical Distributors Association (KPDA), officially announcing its stance on the distribution industry's request for price difference compensation related to the drug price reduction of 'Januvia Family' products, including Januvia·Janumet. The distribution industry has been requesting price difference compensation from pharmaceutical companies for price differences on inventory since the sequential drug price reductions of Januvia (sitagliptin) on September 2, 2023, Janumet XR (sitagliptin·metformin) on the same date, and Janumet (sitagliptin·metformin) on October 1, 2023. The maximum price drops by dosage for each product indicate that Januvia 100mg was reduced from KRW 846 to KRW 592, Janumet XR 100/1000 mg from KRW 831 to KRW 572, and Janumet 50/1000 mg from KRW 520 to KRW 420. The maximum price difference per tablet amounted to KRW 259. However, as no compensation has been made to date, complains are growing within the industry. In response, Chong Kun Dang recently sent an official letter to the KPDA, stating that discussions regarding the responsible party have not yet concluded. According to the official letter, Chong Kun Dang acknowledges that the drug price reductions for Januvia and Janumet, due to patent expirations, were implemented sequentially between September and October 2023, and that distributors have been consistently requesting compensation. Chong Kun Dang explained that the reason no proper compensation has been made yet is a complex situation where Chong Kun Dang demanded price difference compensation from MSD Korea, the marketing authorization holder (MAH) at the time of the drug price reduction, but MSD Korea refused. Januvia is a DPP-4 inhibitor, containing sitagliptin, used to treat diabetes and is developed by MSD. Following Januvia's launch, MSD and Chong Kun Dang have been jointly promoting these products since 2016. However, in 2023, MSD Korea reorganized its chronic disease business unit to focus on its oncology and vaccine businesses, transferring the rights for the Januvia Family to Chong Kun Dang. Chong Kun Dang has been exclusively handling the domestic sales of Januvia as of July 15, 2023. MSD Korea's position is that since the marketing and revenue rights were already transferred at that time, they bear no responsibility for compensation related to the drug price reduction after September 2023, when the price cut occurred. An MSD Korea official stated, "We transferred all domestic rights, including marketing and manufacturing rights, for Januvia products to Chong Kun Dang in July 2023, transferring exclusive sales and marketing authority," and added, "Accordingly, Chong Kun Dang is responsible for price difference compensation due to drug price reductions that occurred after that point." The official added, "We are willing to compensate for inventory that we sold before July 2023, before Chong Kun Dang acquired the marketing rights." The official further explained, "It is true that the transfer of MAH was delayed until July 2024, but this was because time was needed for actual transfer preparations, such as changing product labels," and added, "At the time of the drug price reduction in September 2023, we were not engaged in actual revenue-generating activities, and all related rights had been transferred to Chong Kun Dang. The company was only the MAH on paper." Negotiations ongoing due to discrepancy between marketing rights and MAH...Distribution industry "we're troubled" The key issue is related to the division of roles and accountability between the MAH and the actual operating entity at the time of the drug price reduction. MSD Korea maintains that since it transferred domestic marketing and revenue rights for Januvia to Chong Kun Dang as of July 2023, Chong Kun Dang is responsible for price difference compensation arising from subsequent drug price reductions. Conversely, Chong Kun Dang stresses that MSD Korea still held the marketing authorization at the time of the drug price reduction. Januvia's marketing authorization was indeed transferred to Chong Kun Dang on July 23, 2024, after which the business transitioned to a global direct import structure. Chong Kun Dang believes that "Shifting the responsibility for compensation without the official change of the marketing authorization holder is unfair." However, given the sensitivity of the issue, they are taking a reserved stance by stating that it's difficult to issue an official statement and that discussions with MSD Korea are still ongoing. In 2016, MSD Korea and Chong Kun Dang have signed an agreement to jointly promote treatments for diabetes and dyslipidemia, including Januvia. However, as negotiations delay, the burden of price difference compensation due to the drug price reduction is ultimately being passed on to pharmaceutical distributors. The pharmaceutical distribution industry has consistently demanded compensation for the drug price difference of Januvia family products since the second half of 2023. However, settlement has been delayed due to the ambiguity of accountability between the two companies. The recent official letter sent by Chong Kun Dang to the KPDA directly mentioned this dissatisfaction from the distribution industry. The letter stated, "Discussions between the two companies are ongoing, but as the compensation discussion has not been finalized, distributors are facing difficulties," adding, "We will proactively work towards a swift resolution with responsibility." An official from a pharmaceutical distribution company pointed out, "The pharmaceutical companies that should be responsible for settling the price difference due to the government's drug price reduction are pushing responsibility onto each other," and added, "Distributors compensate pharmacies for the price difference but receive no compensation from the pharmaceutical companies, thus bearing the entire burden in the middle." The industry widely expects the transfer of Janumet XR's marketing authorization in November, raising concerns that the same problem could recur. If the discussion over responsibility drags on while the drug prices of major sitagliptin-based diabetes treatments like Januvia, Janumet, and Janumet XR are sequentially reduced. In that case, distributors will inevitably continue to bear the settlement burden. Ultimately, this discussion goes beyond individual products and extends to the industry-wide challenge of how to resolve systemic blind spots that can arise from the separate structures of marketing rights and marketing authorization.
- Policy
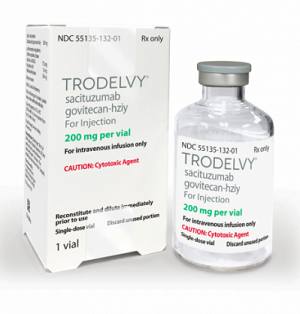
- How Trodelvy was applied flexible ICER for innovativeness
- by Lee, Tak-Sun Jul 14, 2025 06:04am
- The ADC breast cancer drug Trodelvy (sacituzumab govitecan, Gilead), which was reimbursed in June for triple-negative breast cancer, was the first case in which the ICER (incremental cost-effectiveness ratio) threshold was flexibly applied in recognition of its innovation. So how did the Drug Reimbursement Evaluation Committee (DREC) of the Health Insurance Review and Assessment Service at the time evaluate the innovativeness of this drug? According to the recently released evaluation results, Trodelvy was found to satisfy all criteria for innovative treatment, including the absence of alternative treatments, clinical improvement, and the criteria for expedited review. According to industry sources on the 11th, in the recently released DREC evaluation results for Trodelvy, the committee members recognized its reimbursement as appropriate despite the high cost-effectiveness ratio, taking into account the drug’s innovativeness." In August last year, the Health Insurance Review and Assessment Service established new requirements for the innovation of drugs subject to flexible ICER threshold assessment. According to the newly established criteria, the innovation of a new drug is recognized when ▲there are no substitute products or treatments with equivalent therapeutic positions, ▲significant clinical improvement can be recognized in the final outcome indicators, such as prolonged survival, ▲the new drug is a new drug approved by the Ministry of Food and Drug Safety through an expedited review process in accordance with Article 35-4, Paragraph 2 of the Pharmaceutical Affairs Act, or a drug recognized by the committee as equivalent to such a drug. All three requirements must be met for a new drug to be recognized as innovative. Trodelvy became the first drug to be recognized as innovative under these criteria. At the time, the DREC members evaluated that Trodelvy satisfied all three requirements. First, they determined that it satisfied the first requirement as the first antibody-drug conjugate (ADC) approved for triple-negative breast cancer, with no products or treatments with equivalent therapeutic positions. In addition, they evaluated that the second requirement was also satisfied, as clinical improvement (mOS HR=0.51) was recognized in the final outcome indicators, such as prolonged survival. Finally, considering that it was approved by the FDA as a breakthrough therapy and designated as a Global Innovative product for Fast Track item by the MFDS, the committee deemed that the third requirement was also satisfied. In terms of cost-effectiveness, the committee concluded that Trodelvy is significantly more cost-effective than existing anticancer drugs and breast cancer drugs that have been reviewed in the past. However, considering that it is a drug used for diseases that threaten survival in the third or further line of administration, that it has been recognized for improving the social burden of disease and quality of life, and that it is the first antibody-drug conjugate (ADC) approved for triple-negative breast cancer, the committee accepted its reimbursement as adequate. In other words, the high price was accepted in recognition of its innovation. Another factor was that the number of patients eligible for treatment was small, so the drug’s financial impact was not significant. According to the DREC evaluation results, Trodelvy is a drug used by approximately 200 patients, and the decision was made after comprehensive consideration of factors such as the number of patients eligible for treatment and the financial impact. In the end, Trodelvy was listed for reimbursement under two types of risk-sharing agreements (RSA) with the National Health Insurance Service. Among the types of RSA contracts that were applied, one was a Refund-Type agreement, where the pharmaceutical company reimburses a certain percentage of the claimed amount to the NHIS, and the other was the Utilization Cap/Fixed Cost per Patient Refund Type agreement, where the usage limit per patient is set in advance, and a certain percentage of the excess amount is reimbursed to the NHIS if the limit is exceeded. The listed price (maximum amount) is KRW 1,052,300 per vial, but due to the dual pricing system applied under the reimbursement-type contract, the actual price is higher. The estimated number of patients receiving Trodelvy annually is approximately 282, with an estimated fiscal expenditure of approximately KRW 12.5 billion. However, the actual fiscal expenditure is expected to be lower if the risk-sharing agreement is applied. At a 5% coinsurance rate, patients will only need to pay approximately KRW 2.21 million per year for its administration.
- Company
- Daiichi Sankyo and Ono Pharmaceutical see surge in sales
- by Son, Hyung Min Jul 14, 2025 06:03am
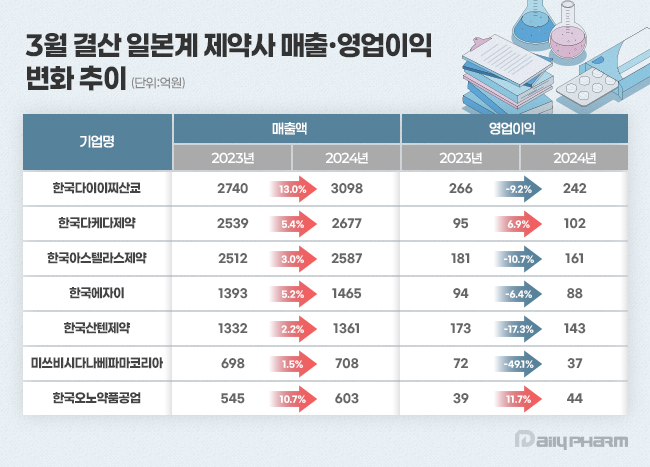
- Japanese pharmaceutical companies operating in South Korea saw overall sales growth last year. All seven companies surveyed achieved year-on-year sales growth, with Daiichi Sankyo Korea and Ono Pharma Korea leading the way with double-digit growth rates. However, in terms of operating profits, the companies showed mixed emotions. According to the Financial Supervisory Service's electronic disclosure system on the 11th, the performance of 7 Japanese pharmaceutical companies with March fiscal year-ends totaled at KRW 1.2499 trillion, up 6.3% from KRW 1.1759 trillion the previous year. During the same period, operating profit decreased by 11.2% from KRW 92 billion to KRW 81.7 billion. Major Japanese pharmaceutical companies' Korean subsidiaries follow the Japanese fiscal year, recognizing the period from April last year to March this year as their 2024 annual sales. #SB Daiichi’s sales surpass KRW 300 billion for the first time... Ono’s sales double in four years Last year, Daiichi Sankyo Korea's sales increased by 13.0% year-on-year to KRW 309.8 billion. During the same period, operating profit decreased by 9.2% from KRW 26.6 billion to KRW 24.2 billion. Daiichi Sankyo Korea's sales have been steadily increasing since 2020. The company surpassed KRW 200 billion in sales for the first time in 2020 with KRW 217.9 billion, followed by KRW 245.4 billion in 2021, KRW 253.2 billion in 2022, and KRW 274 billion in 2023, continuing its upward trend. In particular, it is analyzed that the company achieved synergistic effects by collaborating with the domestic pharmaceutical company Daewoong Pharmaceutical on certain cardiovascular products such as Lixiana and Sevikar. Daiichi Sankyo Korea signed a co-marketing agreement with Daewoong Pharmaceutical for Sevikar in 2013 and Lixiana in 2015, and has maintained its partnership with the company to date. Among these, the highest-selling product is the direct-acting oral anticoagulant (DOAC) Lixiana. According to market research firm UBIST, Lixiana's prescription sales last year reached KRW 117.5 billion, an 11.6% increase from KRW 105.3 billion in 2023. The three-component hypertension combination drug Sevikar HCT also maintained its growth momentum. Last year, Sevikar HCT's prescription sales reached KRW 42.1 billion, up 4.0% from the previous year. Daiichi Sankyo Korea generated approximately KRW 140 billion in prescription sales from olmesartan-based hypertension treatments, including Sevikar HCT (KRW 42.1 billion), Olmetec (KRW 30.6 billion), and Sevikar (KRW 68.8 billion). Daiichi Sankyo Korea is working to expand its business from cardiovascular drugs to anticancer drugs. In particular, the company is focusing on new growth areas such as antibody-drug conjugates (ADCs) by concentrating its R&D capabilities. Following the approval of Enhertu, the company is preparing to launch various treatments, including five ADC strategies such as Datroway, Patritumab deruxtecan, DS-7300, and DS-6000. Last year, Takeda Pharmaceutical Korea's sales increased by 5.4% year-on-year to KRW 267.7 billion. Operating profit rose by 6.9% to KRW 10.2 billion. Anticancer drugs such as Zejula, Alunbrig, and Ninlaro contributed to the sales growth through steady growth. According to the market research firm UBIST, Alunbrig recorded prescription sales of KRW 12.1 billion last year. Since its launch in 2019, its prescription sales have continued to increase. This treatment is gaining market share for its indication in ALK-positive non-small cell lung cancer. Alunbrig is in fierce competition with Pfizer's Lorviqua and Roche's Alecensa. However, However, Alunbrig offers the advantage of once-daily, single-tablet dosing, and can be taken regardless of food intake. This simplicity reduces the number of factors patients need to consider when taking the medication. The ovarian cancer treatment Zejula also recorded KRW 14.3 billion in sales last year, a 51.6% increase in prescriptions from the previous year. In October last year, Zejula was added to the health insurance reimbursement list for the treatment of HRd-positive ovarian cancer. Until then, Zejula had been reimbursed only as maintenance therapy for BRCA-mutated ovarian cancer patients who responded to platinum-based therapy as a first-line treatment for ovarian cancer. The growth of Ono Pharma Korea was also noteworthy. Ono’s sales last year were KRW 60.3 billion, up 10.7% year-on-year. Operating profit in the same period increased 11.7% from KRW 3.9 billion to KRW 4.4 billion. The sales and operating profit recorded by Ono last year are the highest figures since the company began submitting audit reports in 2017. Ono recorded sales of over KRW 40 billion in 2021 and surpassed KRW 50 billion for the first time the following year. Comparing the KRW 60.3 billion recorded by Ono last year with the KRW 31 billion in sales in 2020, sales increased by 94.5% over four years. The immune-oncology drug Opdivo drove Ono’s sales growth. Opdivo is an anti-PD-1 class immuno-oncology drug jointly developed by Ono and BMS, and was approved in Korea in 2015. Ono holds development and sales rights in Asia, including South Korea, Japan, and Taiwan. In particular, the combination of Opdivo and BMS's CTLA-4 targeted immunotherapy Yervoy has demonstrated efficacy in various solid tumors such as melanoma, renal cell carcinoma, and hepatocellular carcinoma, contributing to the increase in market share. According to market research firm IQVIA, Opdivo surpassed KRW 100 billion in domestic sales in 2022. All showed external growth, but with mixed operating profits Astellas Pharma Korea, Eisai Korea, and Santen Pharmaceutical Korea all saw slight increases in sales, but their operating profits all declined. Astellas Pharma Korea reported sales of KRW 258.7 billion last year, a 3.0% increase from the previous year. Harnal, Betmiga, Prograf, and Xtandi performed well, driving sales growth. Harunal, a treatment for benign prostatic hyperplasia, recorded prescription sales of KRW 66.2 billion last year, a 0.8% increase from the previous year. Betmiga, a treatment for overactive bladder, and Prograf, an immunosuppressant, have consistently recorded prescription sales of over KRW 30 billion even after their patents expired. Last year, prescription sales for Betmiga and Prograf were KRW 33.4 billion and KRW 33.6 billion, respectively, up 2.6% and 3.9% year-over-year. Operating profit decreased by 10.7% year-on-year to KRW 16.1 billion. A slight increase in the cost of sales contributed to the decrease in gross profit. Astellas Pharma Korea is pinning its hopes on its next-generation anticancer drugs. The company's Padcev, its ADC anticancer drug, and Vyloy, its new gastric cancer targeted therapy targeting Claudin 18.2, have entered the domestic market. Both drugs have shown remarkable results in clinical trials. Currently, Astellas Pharma Korea has completed its application for insurance reimbursement for both drugs. Eisai Korea's sales rose 5.2% from KRW 139.3 billion in 2023 to KRW 146.5 billion last year. Operating profit for the same period decreased 6.4% to KRW 8.8 billion. Sales of existing products such as the anticancer drug Lenvima, the gastroesophageal reflux disease treatment Pariet, and the Alzheimer's disease treatment Aricept remain steady. The three products—Aricept (KRW 95.9 billion), Pariet (KRW 17.9 billion), and Lenvima (KRW 12.9 billion)—combined for over KRW 100 billion in sales last year. Operating profit decreased slightly due to factors such as increased cost of sales. EisaiEisai is expecting strong performance from its new Alzheimer's disease drug, lecanemab. Lecanemab is an Alzheimer's disease treatment developed by Eisai and Biogen that selectively binds to amyloid beta, a substance believed to cause the disease, and has been proven to slow the progression of the disease and delay cognitive decline. It is reported that lecanemab is currently being prescribed at various general hospitals and semi-general hospitals in Korea. Santen Korea reported sales of KRW 136.1 billion last year, a 2.2% increase from the previous year. However, operating profit decreased by 17.3% to KRW 14.3 billion. Santen specializes in ophthalmic products and holds various formulations, including diquafosol. However, in South Korea, hyaluronic acid eye drops are primarily used, and the company has not significantly increased its prescription volume in Korea. As of last year, the product with the highest sales among Santen Pharmaceutical's portfolio was Cosopt S eye drops, used for glaucoma. Cosopt S’s prescription volume last year was KRW 33.9 billion, an increase of 2.6% compared to the previous year. Mitsubishi Tanabe Pharma Korea's sales last year were KRW 70.8 billion, up 1.5% from 2023, but operating profit fell 49.1% in the same period. The decrease in operating profit was due to an increase in selling, general, and administrative expenses, such as reimbursement and retirement benefits, as well as an increase in cost of sales.
- Policy
- Orphan drug 'Bylvay Cap' wins reimb approval after reeval
- by Lee, Tak-Sun Jul 14, 2025 06:03am
- 'Bylvay Cap,' which is being considered for the expedited reimbursement listing process as a designated drug for 'Pilot Project for Integration of Product Approvals, Reimbursement Coverage Reviews, and Drug Price Negotiations,' received reimbursement appropriateness decision after reevaluation. Consequently, Bylvay Cap will be subjected to negotiation with the National Health Insurance Service (NHIS) before its inclusion in the reimbursement list. Health Insurance Review and Assessment Service (HIRA) stated that it has held the 7th Drug Reimbursement Evaluation Committee (DREC) meeting on July 10 and decided as such. During the meeting, the DREC decided on the reimbursement appropriateness for Bylvay Cap 200, 400, 600, and 1200 μg (odevixibat sesquihydrate). Drug Reimbursement Evaluation Committee (DREC) Bylvay Cap is used to treat progressive familial intrahepatic cholestasis (PFIC). This drug was designated as the first concurrent approval-evaluation-negotiation pilot project in 2023. The criteria for the concurrent approval-evaluation-negotiation pilot project were drugs with sufficient efficacy intended for treating cancer or rare diseases with a life expectancy of less than one year, as well as those proving superior survival·treatment effectiveness for over two years when alternative treatments are not available. Bylvay Cap obtained marketing authorization from the Ministry of Food and Drug Safety (MFDS) in August 2024 and entered into the reimbursement evaluation process. Yet, the reimbursement evaluation process has been uneasy. A complaint was heard from an expert who attended the reimbursement review meeting from the evaluation stage. In the DREC meeting held last April, a conclusion was not reached, and it was decided to re-evaluate. It was only today, three months later, that it passed the DREC review. As drugs under the concurrent approval-evaluation-negotiation pilot project undergo a one-stop process without following separate stages, Bylvay is likely to have already entered negotiations with the NHIS. If the NHIS negotiations are completed, patients will be able to receive National Health Insurance coverage for this drug immediately after review by the Health Insurance Policy Review Committee. Meanwhile, the DREC decided on HLB Pharma's Citrelin ODT to receive a conditional approval, stating that reimbursement appropriateness if the company accepts a price below the evaluated amount. This drug is used for 'improving ataxia caused by spinocerebellar degeneration.'
- Company
- Bimzelx may be prescribed at general hospitals in KOR
- by Eo, Yun-Ho Jul 11, 2025 06:12am
- The new psoriasis drug Bimzelx may be prescribed at general hospitals in Korea. According to industry sources, UCB Pharma Korea's Bimzelx (bimekizumab) has been approved by the Drug Committees (DCs) of major hospitals nationwide, including tertiary hospitals like Seoul Asan Medical Center and Severance Hospital as well as major hospitals including Kyungpook National University Hospital, Pusan National University Hospital, Seoul National University Bundang Hospital, Chonnam National University Hospital, Jeonbuk National University Hospital, and Hanyang University Hospital. The company appears to be rapidly working to expand Bimzelx prescriptions following its reimbursement listing in June. i1Bimzelx is the first plaque psoriasis treatment that dually inhibits interleukin-17A and 17F (IL-17A and 17F). IL-17A and IL-17F are key cytokines that trigger the inflammatory process in psoriasis, and Bimzelx selectively and directly targets and inhibits both simultaneously. This dual inhibition mechanism against IL-17A and 17F is what makes the drug’s introduction significant. Despite the emergence of various psoriasis treatments, there remained an unmet need in practice due to resistance and other factors, which is why health professionals saw the introduction and reimbursement of Bimzelx encouraging. Yong-Beom Choi, President of the Korean Society for Psoriasis (Department of Dermatology, Konkuk University Medical Center), said, “Psoriasis is an intractable disease that recurs and improves repeatedly, and the quality of life of patients with severe psoriasis in particular tends to deteriorate significantly, affecting their mental health. Therefore, the reimbursement and launch of Bimzelx, which has been proven to be highly effective, is very meaningful for both medical professionals and patients.” He added, “Based on its next-generation mechanism of action, Bimzelx is expected to be an excellent treatment option for both new patients and those who have not seen sufficient results with existing treatments.” Meanwhile, in the Phase III BE READY trial, 90.8% of patients in the Bimzelx group achieved PASI 90 at Week 16, and 68.2% of patients achieved PASI 100. In another clinical trial that compared Bimzelx with another biological agent, there was a clear difference in the percentage of patients who achieved complete clearance of skin lesions at Week 16, or 'PASI 100'. Specifically, PASI 100 for the drug and control drug were as follows: ▲BE VIVID: Bimzelx 59%, ustekinumab (Stelara) 21% ▲BE SURE: Bimzelx 60%. 8%, adalimumab (Humira) 23.9% ▲BE RADIANT: Bimzelx 61.7%, secukinumab (Cosentyx) 48.9%, etc.
- Opinion
- [Reporter's View] US tariffs…variable that must be faced
- by Whang, byung-woo Jul 11, 2025 06:12am
- Tension is growing in the domestic pharmaceutical and biotech industry as President Donald Trump recently announced the potential imposition of tariffs on imported pharmaceuticals. If the tariff hike is imposed, it is expected to directly impact not only the procurement costs of raw materials and sub-materials for Korean companies but also their price competitiveness when exporting finished products. According to the announcement, the tariffs are projected to be imposed approximately 1 to 1.5 years from now. While opinions within the industry are divided on the realistic feasibility of applying tariffs as high as 200%, the burden companies would face, even if tariffs are partially imposed, would be substantial. In particular, the U.S. market is crucial for Korean pharmaceutical and biotech companies as an export destination. For domestic companies that have rapidly grown in areas such as biosimilars and active pharmaceutical ingredients (APIs), securing global competitiveness, tariff barriers pose a significant threat. If a deterioration in profitability and a decline in price competitiveness materialize, significant disruptions to companies' global strategies are inevitable. Furthermore, Korean companies that have relied on the Contract Manufacturing Organization (CMO) and Contract Development and Manufacturing Organization (CDMO) industries as key growth pillars could face direct pressure, such as demands to relocate production sites or re-evaluate contract structures. Although specific implementation plans have not yet been released, long-term and strategic responses are required. Thus, there is a growing need to establish various tariff imposition scenarios and prepare stepwise countermeasures starting now. This tariff issue is likely to affect not only the pharmaceutical and biotech industries but also industries across the board, including automobiles, electronics, and steel. Therefore, individual corporate responses alone have apparent limitations. Ultimately, the government and businesses must collaborate to develop various scenarios based on potential tariff increases and formulate corresponding phased countermeasures. At the government level, while diplomatic efforts are necessary to reduce the likelihood of tariff increases, it is also essential to implement policy support measures, such as financial assistance and tax benefits, to minimize the impact on companies. Especially given that the government has identified the pharmaceutical and biotech sector as one of the future growth engines, neglecting this anticipated threat or delaying a response would be a strategic misstep. The pharmaceutical and biotech industries, by their nature, require long-term preparation and investment. The pharmaceutical and biotech industries, which are highly susceptible to the negative effects of trade conflicts, demand even more concrete and proactive preparation. It is crucial to recognize that this President Trump-initiated tariff issue is not merely a political discussion but a significant variable that will determine the overall competitiveness and survival of the industry. Therefore, discussions on a pan-governmental response must be initiated fully. The pharmaceutical and biotech industries should use this tariff issue as an opportunity to comprehensively re-evaluate the structural risks associated with global market entry and formulate new strategies. The government must actively consider and implement consultative bodies in collaboration with the private sector to prepare for tariff increases and long-term competitiveness strategies.
- Policy
- Will drugs subject to reimbursement reevals be expanded?
- by Lee, Tak-Sun Jul 11, 2025 06:11am
- The government is reportedly planning to tighten the selection criteria ahead of the second phase of the drug reimbursement adequacy reevaluations, which is set to begin next year. As a result, it is expected that the number of ingredients subject to reassessment may be expanded. According to industry sources on the 10th, the Health Insurance Review and Assessment Service (HIRA) is currently collecting feedback from the pharmaceutical industry before finalizing its second-phase drug reimbursement adequacy reevaluation plan. The first phase will conclude this year. Since the pilot reevaluation was conducted for choline alfoscerate in 2020, HIRA has completed four full reevaluation rounds from 2021 to 2024, and the fifth round is currently underway. Thus far, the reassessment has focused on ingredients listed for reimbursement from 1998 to 2006, before the positive listing system was implemented, and ingredients currently undergoing clinical reevaluation by the Ministry of Food and Drug Safety. Also, ingredients with an annual total reimbursement claim exceeding 0.1% (approximately KRW 20 billion), and those reimbursed in only one foreign country among the A8 reference countries was the criteria. Industry observers anticipate that the selection criteria stated above will change significantly from the second phase of reevaluations. In particular, the strengthened criteria may lead to the inclusion of ingredients that were previously excluded from reevaluations. It is reported that authorities are considering expanding the annual claims threshold to identify ingredients that have remained in a regulatory blind spot. The criteria under discussion include lowering the reimbursement claim threshold from KRW 20 billion to KRW 10 billion, and expanding the eligibility from being listed in only one A8 country to fewer than three. As in the first phase of reevaluations, the second phase will also continue to target drugs listed before the implementation of the positive listing system, making it likely that previously excluded products will be included. If the criteria are expanded as proposed, pharmaceutical companies will likely begin reviewing their product portfolios to identify those that may be subject to reevaluation. Unlike in the first phase, drugs with lower claims may also be included, posing new challenges for the companies preparing for reevaluations. However, as the selection criteria are still under review, companies are expected to limit their response until the final criteria are confirmed. Given the delay in finalizing the criteria and scope for the second phase, the industry is calling for the 2025 revisions to be postponed to allow sufficient preparation time.