- LOGIN
- MemberShip
- 2026-04-07 01:06:40
- Company
- Breast cancer drug Trodelvy receives DREC deliberations
- by Eo, Yun-Ho Aug 29, 2024 04:31am
- The ADC breast cancer drug Trodelvy has made a step toward insurance reimbursement after 9 months of wait. Gilead Sciences' triple-negative breast cancer (TNBC) drug Troldelvy, whose reimbursement request received 100,000 consents in a public petition, will be presented for deliberation to the Health Insurance Review and Assessment Service's Drug Reimbursement Evaluation Committee on the 29th. The drug’s reimbursement application had remained pending for some time since its reimbursement standard was set by the Cancer Disease Review Committee in November last year. Therefore, the industry’s eye will be focused on the outcome of the committee review. The key issue will be the drug price, especially the ICER threshold value. Trodelvy is already listed in about 30 countries around the world. Trodelvy is already listed in about 30 countries around the world. Taiwan, which has a single-payer healthcare system similar to South Korea's, began reimbursing Trodelvy in February this year. The global rush to improve patient access to Trodelvy has been driven by the poor treatment environment for metastatic triple-negative breast cancer and the clinical value of Trodelvy. Triple-negative breast cancer is an aggressive form of breast cancer that recurs and metastasizes rapidly. Patients with metastatic triple-negative breast cancer who have metastasized despite treatment have a life expectancy of only a few months even with chemotherapy. However, chemotherapy has long been the standard of care due to the lack of targets that can effectively kill cancer cells. Trodelvy, the first Trop-2-targeted antibody-drug conjugate (ADC), is the only treatment for metastatic triple-negative breast cancer in the second-line or higher setting that has been shown to prolong survival compared to chemotherapy and has settled as the global standard of care since its introduction. Currently, major guidelines in the U.S. and Europe specify Troldelvy as the preferred agent for patients with previously treated metastatic triple-negative breast cancer. In a Phase III study, the overall survival of the chemotherapy arm was 6.9 months, compared to a nearly one-year survival. (11.8 months) in the Troldelvy arm, In addition, Troldelvy demonstrated an effect in controlling symptoms and pain caused by cancer and improving patients' quality of life by improving their overall health status. Trodelvy was awarded the highest possible score of 5 points on ESMO-MCBS, the European Society for Medical Oncology's (ESMO) scale used to rate the value of anticancer drugs. A score of 5 indicates that a drug is effective not only in prolonging patient survival but also in improving quality of life, and Troldelviy is the only treatment for metastatic triple-negative breast cancer to receive a score of 5 on ESMO-MCBS. In fact, the U.K. has detailed the rationale behind its assessment, stating that the state’s reimbursement decision was based on the severity of metastatic triple-negative breast cancer and the survival benefit of Troldelvy. Similar to Korea, the U.K. uses the incremental cost-effectiveness ratio (ICER) to evaluate new drugs for health insurance coverage. While the UK has one of the highest reimbursement barriers for new drugs, it applies flexible pharmacoeconomic evaluation criteria for innovative drugs used for serious conditions to improve patient access. In the UK, Troldelvy was granted preferential economic evaluation because it prolonged survival in terminally ill patients with less than 2 years life expectancy, whose population is even smaller than those of rare diseases. As a result, Trodelvy gained access with an ICER threshold that was approximately twice higher than that of general drugs. Meanwhile, Troldelvy has been the subject of a series of petitions this year, gathering more than 100,000 consents online. The Korean Alliance of Patients' Organizations also responded to the desperate pleas of patients and their caregivers when the petition was abandoned due to the expiration of the 21st National Assembly's term. In May, the organization submitted a letter directly to the Ministry of Health and Welfare requesting a prompt review of the reimbursement of drugs with high patient demand, including Trodelvy.
- Policy
- Will RWD-based reimb agreements be more beneficial?
- by Lee, Tak-Sun Aug 29, 2024 04:31am
- On the 28th, HIRA held an international symposium on Drug reimbursement contracts satisfy all parties involved when they achieve three goals: The first is patient access, the second is sustained revenue for the pharmaceutical company, and the third is budget management for the payer. “A fair price is one that can satisfy both the seller and the buyer,” said So-young Lee, Director of Health Insurance and Assessment Service’s Pharmaceutical Performance Assessment Division. ”A fair price should guarantee transparency, R&D, production costs, and innovation.” So, what kind of reimbursement agreement can satisfy all three conditions? Experts say that a reimbursement agreement based on RWD (real-world data) can be a good alternative. On the 28th, HIRA held an international symposium on 'RWD-based performance assessment of high-priced drugs' at the Ambassador Seoul Pullman Hotel in Jangchung-dong, Seoul. At the symposium, Lee delivered a keynote address on the topic of 'Sustainable access to high-cost drugs through RWD-based cooperation'. The use of the performance-based assessment system using RWD is in its infancy in Korea and other countries. In Korea, the system was first introduced in 2022 for the rare disease drug Kymriah. In order to reduce the uncertainty of drugs that received an exemption from pharmacoeconomic evaluations, the system applies post-reimbursement patient evaluation to determine whether or not to reimburse the drug. Until now, the cost-effectiveness evaluations of new drugs were determined based on clinical trial data rather than real-world patient data, but there is a global movement to use RWD data of high-priced drugs as clinical trial results do not resolve uncertainties. The UK established a real-world evidence (RWE using RWD) framework in February 2022 and has been continuing to update its guidance on valuation using RWE. “Ultra-high-priced drugs present uncertainties due to disease characteristics and ethical issues,” said Lee, adding, “ It is difficult to standardize clinical trials, and there may not be a comparator drug or enough follow-up data.” In such cases, it is difficult to analyze the cost-effectiveness, and even then, it is difficult to ensure reliability. Among the high-priced drugs listed for reimbursement in Korea, there are already 11 drugs that cost more than KRW 100 million per year. For these drugs, performance-based managed reimbursement contracts are being implemented as their pharmacoeconomic evaluation are difficult and clinical uncertainty high. To date, 5 drugs are receiving patient-level performance-based evaluations and 1 drug-level evaluation. “The industry is negative about the outcome-based drug evaluation,” Director Lee said, ”The industry believes that the system is not objectively valid or reliable.” However, Lee emphasized that RWD, if utilized well, can be used to ensure reliability and fair pricing that satisfies patients, pharmaceutical companies, and the payer. “The advantage of RWD is that evidence can be gathered from the R&D stage to benefit patients, and the incompleteness of the system can be reduced if pharmaceutical companies and the insurer can work together to link a lot of data. If we can ensure full-cycle collaboration based on transparency, the system will not shrink the pharmaceutical industry while ensuring access to good drugs for patients.” Of course, there are limitations. Data analysis needs to be done while protecting patient privacy, and transparency and uniformity of data still remain a challenge. However, standardization efforts are underway in each country. “There is still a lack of confidence in the system using RWD to evaluate cost-effectiveness, as well as limited transparency, data quality issues, and bias,” said Dr. Vandana Ayyar Gupta, Scientific Advisor of NICE in the UK, another keynote speaker at the event, ”This is why NICE is working to update its guidance and create an RWE framework for its support.” “Real-world data and real-world evidence are being used by NICE in a variety of ways, and the number of use cases is expanding,” said Dr. Gupta. ”The RWE framework will continue to be updated through communication between developers and NICE based on rapidly evolving methodologies and technological advances.”
- Opinion
- [Reporter’s View]Support domestic COVID-19 drugs
- by Lee, Hye-Kyung Aug 28, 2024 05:52am
- The recurrence of COVID-19 has sparked interest in COVID-19 drugs. After the pandemic turned into an endemic, the government set a budget of KRW 179.8 billion for COVID-19 drugs this year, which was a 53.2% cut from last year, and the number of COVID-19 drugs introduced in Q1 and Q2 of this year was 179,000, half of the 341,000 that the government procured during the same period last year. The government claimed that the budget cut was due to preparations for the reimbursement of COVID-19 treatments, but the government ultimately set aside KRW 326.8 billion in emergency reserves to purchase an additional 262,000 doses of treatment after failing to predict the resurgence of COVID-19. The amount of the reserve is nearly equivalent to the KRW 384.3 billion budget the government had set for the purchase of COVID-19 drugs last year. Due to the lack of a Korean treatment option, Korea has to rely on global pharmaceutical companies for all supplies of treatments in the event of a COVID-19 outbreak. This is because there are currently only 3 COVID-19 drugs available in Korea - Pfizer's Paxlovid Tab, MSD's Lagevrio Cap, and Gilead's Beklury Inj. If there were locally developed COVID-19 treatments, not just global pharmaceutical companies' products, the government could have addressed the outbreak faster. To stabilize the supply and demand of cold medicines, the government asks domestic pharmaceutical companies to increase production through a public-private consultative body to balance the supply and demand when medical organizations report unstable supply. The recent shortage of COVID-19 drugs is likely to have been affected by Korea’s reliance on imported products. Curing the COVID-19 pandemic, there has been a movement toward self-sufficiency of drugs. Celltrion developed Regkirona Inj and received marketing authorization in September 2021. However, in 2022, the company suspended new supplies due to its low effectiveness against omicron mutations. Following this, Ildong Pharmaceutical's Xocova, which was developed in collaboration with Japan's Shionogi Pharmaceutical, and Hyundai Bioscience's Xafty tried to cross Korea’s threshold through the emergency use authorization pathway but failed. The MFDS pointed to the small number of clinical trial subjects and requested Phase III clinical data for the domestic approval of each drug. COVID-19 treatments need to accumulate clinical data on COVID-19 patients, and this is why domestic pharmaceutical companies have been struggling to recruit patients in the endemic. The need for homegrown drugs and vaccines has been emphasized during the outbreak. The government has also provided various support such as emergency use authorization and expedited review. However, since COVID-19 was declared endemic, many people lost interest in domestic COVID-19 treatments. But the resurgence of COVID-19 this time has made everyone realize that a second or third wave could come at any time. The endemic is not the end of COVID-19. We need to expand our support for domestic drugs to ensure self-sufficiency of COVID-19 treatments in Korea.
- Company
- "Switching of atopic dermatitis treatments not yet allowed"
- by Hwang, Byung-woo Aug 28, 2024 05:52am
- As new drug entries shift the market for atopic dermatitis, the public onion demands changes to reimbursement assessment, such as considering the comprehensive factors to use increased treatment options efficiently. Opinions have been suggested to consider factors related to patient quality of life, such as improving itchiness, in addition to the Eczema Area and Severity Index (EASI)-75 achievement rate, which is a typical criterion for evaluating the effectiveness of atopic dermatitis treatment. The view is that since switching between treatments is the major focus of the guidelines for atopic dermatitis treatment revised for the first time in 9 years and focus on switching between treatments, the Korean government must align with the global trend. During Pfizer Professor Ahn Ji Young, affiliated with the Department of Dermatology at the National Medical Center, and Professor Jang Yong Hyun, affiliated with the Department of Dermatology at Kyungpook National University, discussed the matter while sharing Korea's latest treatment trend of severe atopic dermatitis during the '2024 Pfizer Press University.' Regarding the necessity of switching between atopic dermatitis treatment, Ahn stressed the nature of the disease, which accompanies various factors. Ahn explained, "The treatment of atopic dermatitis is difficult because of genetic and allergic factors and accompanying disease," and "Patients experience improved and worsened symptoms. Therefore, the disease can progress to chronic disease, possibly leading to decreased patient quality of life and healthcare shopping." Ahn stresses the need to consider various factors for reimbursement assessment, such as itchiness, in addition to the previously defined EASI-75 score achievement. Ahn emphasized, "EASI-75 is a familiar indicator because reimbursement coverage is provided when patients with moderate-to-severe atopic dermatitis reach the score during the treatment. However, there are other important factors as well. Subjective symptoms such as itchiness must also be considered when evaluating reimbursement." To reflect on the current situation, experts emphasize switching between medications. Patient-optimized treatment options, depending on patient conditions, must become available. Jang stressed, "Biological agents and JAK inhibitors are currently used to treat atopic dermatitis. However, not all medications work for each patient," and "It is necessary to provide medication that suits patient's clinical outcomes, itchiness, and dermatological pathology. If it doesn't work, another medication must be allowed." (From left) Professor Ahn Ji Young, affiliated with the Department of Dermatology at the National Medical Center, and Professor Jang Yong Hyun, affiliated with the Department of Dermatology at Kyungpook National University. Academics suggest that the basis for switching between treatments is currently sufficient. While the approval criteria do not require the distinction between first-line treatment and second-line treatment, it has been suggested that a comprehensive factor such as the patient's condition must be considered when prescribing medications in clinical practices. Jang stated, "Academics stress the need for prescribing optimized medication to patients with severe symptoms after considering the patient's condition," and "Another medication must be allowed to be used when a patient does not benefit from one treatment, showing insufficient response." According to the pharmaceutical industry, unlike the initial request for authorization of switching therapy, the government may be more willing to bring changes now. For instance, the Health Insurance Review and Assessment Service (HIRA) has recently requested supplementary documents. Therefore, switching therapy may be considered based on academic's suggestion for patients with insufficient response. Jang said, "We were told there will be a meeting with the HIRA to discuss switching therapy."
- Policy
- Price of 17 items cut 30% during Type C PVA negotiations
- by Lee, Tak-Sun Aug 28, 2024 05:52am
- The National Health Insurance Service (NHIS) has announced that it has achieved health insurance financial savings of KRW 52.1 billion (USD 52.1 million) through ‘Type C’ Price-Volume Agreement (PVA) negotiations this year. This is an 85.5% increase from the KRW 28.1 billion in the previous year. In particular, the revision of the detailed operating guidelines contributed to a 36% increase in the price reduction rate of high-cost drugs that billed more than KRW 30 billion per year compared to before the guidelines were revised. The National Health Insurance Service (President: Ki-suck Jung, NHIS) announced that it has completed negotiations for 63 product groups (207 items) subject to the 2024 PVA 'type Type c negotiations, of which the price of 162 items will be reduced unilaterally as of September 1, and the remaining 45 items have signed a one-time refund agreement. ‘Type C’ negotiation is held once a year, and this year's negotiation was conducted for drugs whose claims amount in 2023 increased by '60% or more' compared to 2022, or '10% or more and the increased amount exceeds KRW 5 billion', and the pharmaceutical companies and the NHIS negotiated to reduce the drug price or make a one-time reimbursement of the claimed amount based on the reduction rate. This year's negotiations were the first to apply the 'Detailed Operating Guidelines for PVA Linkage Negotiations,' which was revised in May after collecting opinions from the 2023 System Improvement Council, which included the Ministry of Health and Welfare, NHIs, and the pharmaceutical industry. The main improvements included ▲differentiation of the reference formula used to link the claims amount ▲raising the exclusion criteria to improve the acceptability of pharmaceutical companies and encouraging R&D ▲one-time reimbursement contract ▲introduction of a reduction rate reduction system, to practically improve the financial savings. As a result, the price reduction rate of high-costing drugs that billed more than KRW 30 billion per year increased by 36%, compared to how guideline revision due to the improvement of the reference formula linked to the amount of charges. The NHIS also reported that the efficiency of system operation was improved by raising the criteria for claims that are eligible for exclusion of pricing negotiation, which excluded 64 items, helping to resolve difficulties faced by small and medium-sized pharmaceutical companies. In addition, the NHIS claimed that it applied a one-time reimbursement contract for 45 items whose usage temporarily increased due to unavoidable reasons, such as the COVID-19 pandemic crisis, instead of reducing drug prices to ensure a stable supply of medicines. In addition, the NHIS explained that it enhanced the sustainability of the pharmaceutical industry ecosystem by setting a price reduction rate of 30% for 17 items that had undergone negotiations 3 times within 5 years among innovative pharmaceutical companies. As a result, this year's 'type c’ negotiations resulted in health insurance financial savings of KRW 52.1 billion, up 85.5% from the 28.1 billion won in the previous year. id. “Considering the increasing trend of health insurance drug costs due to the aging population and chronic diseases, the importance of PVA negotiations in the post-management of drug prices is becoming increasingly important,” said Yoo-kyung Yoon, Director of the pharmaceutical management division at NHIS. ”We will continue to strive to improve the sustainability of health insurance and reduce the actual burden of drug costs on the public through effective PVA negotiations.”
- Company
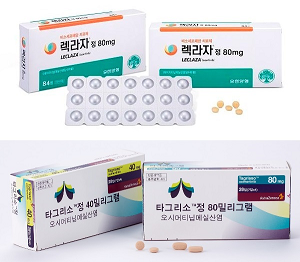
- K-made Leclaza monotherapy to be showcased at global meeting
- by Son, Hyung-Min Aug 28, 2024 05:52am
- Korean biopharmaceutical companies continue to gain R&D achievements for targeted anti-cancer drugs. Companies have prepared to present their Korean-made anti-cancer drugs, including Leclaza and Rivoceranib at the 2024 World Conference on Lung Cancer, which will be held September 7-10 in San Diego, USA. Yuhan Pharmaceutical will present the clinical results comparing the efficacy of its proprietary Leclaza monotherapy, a non-small cell lung cancer (NSCLC) treatment, to Tagrisso. This month, Leclaza plus Rybrevant combination therapy passed the U.S. Food and Drug Administration (FDA) approval. A shift in the market for the first-line treatment of EGFR-positive NSCLC is expected if Leclaza monotherapy obtains better clinical results than Tagrisso monotherapy. HLB group and Jiangsu Hengrui Pharmaceuticals will present the clinical results of their targeted anti-cancer drug, Rivoceranib. The companies attempted to win FDA approval for Rivoceranib in combination with camrelizumab, a PD-1 immune checkpoint inhibitor. However, they received a complete response letter (CRL) request in May. HLB group will retry for approval by demonstrating effectiveness in various solid cancers, including liver cancer. Will present a follow-up study for the MARIPOSA trial, comparing Leclaza vs. Tagrisso monotherapies Treatments for NSCLC.According to industry sources on August 27th, the results of the study comparing the efficacy and safety profile of Leclaza monotherapy and Tagrisso monotherapy will be presented at WCLC 2024, to be held on September 4th. Leclaza and Tagrisso are treatments for targeting NSCLC harboring mutations of EGFR in exon 19 and exon 21 (L858R). Based on the Phase 3 MARIPOSA study results, disclosed last year, Leclaza plus Rybrevant combination therapy showed improvement in progression-free survival (PFS) compared to Tagrisso monotherapy. The study included patient treated with Leclaza monotherapy as a reference group. The results showed that the PFS for the Leclaza monotherapy group was 18.5 months, longer than 16.6 months for the Tagrisso monotherapy group. The company will present secondary results for the MARIPOSA study during the upcoming conference. The study is the first investigative analysis evaluating two third-generation EGFR-TKIs using randomization and a double-blind method. The study focused on 216 patients treated with Leclaza and 429 patients treated with Tagrisso. The primary endpoints included a blinded independent central view (BICR) PFS, duration of response (DOR), overall survival (OS), and safety. According to the disclosed abstract, BICR ORR for the Leclaza group was 88%, and for the Tagrisso group, it was 85%. However, the secondary analysis showed that Leclaza provided more clinical benefits than Tagrisso monotherapy in patients with biomarkers of high-risk disease, including TP53, circulating tumor DNA (ctDNA), and brain metastasis. In most solid cancers, patients with brain metastasis, TP53 mutations, and ctDNA detection tend to show poor prognosis. The median PFS for patients with a history of brain metastasis was 16.4 months for Leclaza, whereas Tagrisso's was 13.0 months. Among patients with detectable ctDNA, the median PFS for the Leclaza group was 18.4 months, and for the Tagrisso group, it was 14.8 months. Among patients with TP53 mutations, the Leclaza group had a median PFS of 14.6 months, which was longer than 12.9 months for the Tagrisso group. Two treatments had similar safety profiles, with moderate side effects between Grade 1 and Grade 2. Tagrisso group was more likely to have adverse reactions like diarrhea, thrombocytopenia, and reduced white blood cells. The Leclaza group was more likely to have rashes and paresthesia. The research team evaluated that "The efficacy and safety were comparable between Leclaza and Tagrisso. The results indicate that Leclaza can be a new treatment option for patients with EGFR-mutated advanced NSCLC and high-risk patients." Rivoceranib plus camrelizumab seen as a potential treatment for early lung cancer On September 8th, clinical trial results of the combination of the HLB group's Rivoceranib and Jiangsu Hengrui Pharmaceuticals' camrelizumab as perioperative neoadjuvant therapy. HLB group and Jiangsu Hengrui Pharmaceuticals, the developers of these two treatments, are evaluating the clinical efficacy of the combination of Rivoceranib, a vascular endothelial growth factor receptor-2 (VEGFR2) inhibitor, and camrelizumab, an immune checkpoint inhibitor. The current Phase 2 clinical trial evaluated the effectiveness and safety of Rivoceranib plus camrelizumab plus chemotherapy in patients with Stage 3 NSCLC. The patients received surgery after the combination therapy. The primary endpoint was the major pathological response (MPR) rate. The secondary endpoint included pathological complete response (pCR) rate, ORR, and safety. Based on the disclosed abstract, the combination of Rivoceranib plus camrelizumab plus chemotherapy as perioperative adjuvant therapy increased the rate of success for lung cancer surgery. The clinical results involving 29 patients showed that 19 patients had complete cancer removal. The combination therapy demonstrated 86.2% ORR, the percentage of patients with a confirmed objective response. The most common adverse reactions were white blood cell reduction (31.0%), thrombocytopenia (17.2%), rash (17.2%), and fatigue (13.8%). OS has not been reported due to incompleteness. The research group said, "The combination of Rivoceranib plus camrelizumab plus chemotherapy showed clinically meaningful anti-tumor activation and manageable safety without blood toxicity," and added, "It seems to be a potential treatment option for patients with resectable Stage 3 NSCLC." HLB group and Jiangsu Hengrui Pharmaceuticals plan to investigate potential in various fields, including liver cancer, gastric cancer, adrenocortical carcinoma, colorectal cancer, ovarian cancer, bile duct cancer, and esophagus cancer, in addition to early NSCLC.
- Policy
- MFDS to review side effects relief imposed on companies
- by Lee, Jeong-Hwan Aug 28, 2024 05:52am
- The Ministry of Food and Drug Safety (MFDS) comments on the The Ministry of Food and Drug Safety (MFDS) announced that it would try to set the reasonably sized 'Payment for Benefits for relief of Injury from Side Effects of Drugs, (hereafter referred to as payment for benefits for relief) ' which is currently charged to pharmaceutical companies. MFDS' audit of the Korea Institute of Drug Safety and Risk Management (KIDS) indicated excessive charging of pharmaceutical companies. As the committee members of the Health and Welfare Committee of the National Assembly pointed out, MFDS seems to be promising to evaluate the appropriateness. On August 27th, the MFDS announced such in a statement submitted after last week's plenary session. The committee members of the Health and Welfare Committee commented that the appropriateness of the rate of payment for benefits for relief must be continuously examined. Additionally, the committee members questioned the MFDS about devising a solution to improve the payment awareness, such as strengthening promotions, expanding the percentage of relief, and recognizing the side effects. Such comments seem to stem from the recent discovery by MFDS during the KDIS audit. The MFDS discovered that KIDS overcharged pharmaceutical companies the payment for benefits for relief. The MFDS promised to devise an appropriate size of the payment for benefits for relief charged to pharmaceutical companies. It also explained that the MFDS has been reducing the percentage of charges covering the payment for benefits for relief since 2018. In fact, MFDS' rate of charge is decreasing, with 0.027% for 2018-2020, 0.022% for 2021-2023, and 0.018% for 2024-2026. The MFDS also mentioned that in the early stages of the system implementation, relief for adverse effects initially covered only death benefits but has since been expanded to include funeral expenses, disability relief, and medical costs. The MFDS stressed, "While death relief was only provided in the early stages of the system implementation, we have expanded the relief size to funeral expenses, disability relief, and medical costs," and added, "Since 2018, we have been reducing the rate of payment by setting the rate of payment yearly in the early stages of the system implementation and every three years since from 2018." The MFDS announced that it will enhance the promotion of the relief providing system. The MFDS added, "MFDS will continue to improve awareness of the system so that citizens can benefit from the relief," and, "We will post promotions on public transportation, buildings, and online platforms. We will also create and distribute informational posters and medicine bags to provide information on the relief."
- Company
- Challenges in the evolving lung cancer treatment landscape
- by Son, Hyung-Min Aug 28, 2024 05:51am
- The head-to-head battle between the epidermal growth factor receptor (EGFR)-mutated non-small-cell lung cancer drugs (NSCLC) Leclaza and Tagrisso has carried on to competition of each drug's combination therapy regimens. This month, the U.S. Food and Drug Administration (FDA) approved Leclaza+Rybrevant as a first-line treatment for EGFR-positive NSCLC. This is the first time a targeted therapy plus targeted therapy combination has been approved. Tagrisso was approved in Korea and the U.S. this year after confirming its efficacy in combination with platinum-based chemotherapy. While attention is focused on whether combination therapies will find a place in the first-line treatment market for EGFR-mutated NSCLC, there is a consensus that the choice of a first-line treatment should be based on the patient's comprehensive needs, including side effects, frequency of hospital visits, and quality of life. Leclaza+Rybrevant combo improved overall survival compared with Tagrisso monotherapy NSCLC drug Leclaza Yuhan Corp and its partner Janssen have confirmed the efficacy of the combination of the Leclaza+Rybrevant combination therapy. Leclaza is a third-generation tyrosine kinase inhibitor (TKI) that targets exon 19, and exon 21 (L858R) in EGFR-positive NSCLC. Rybrevant is a targeted treatment option that targets the exon 20, MET mutation. The two companies successfully secured FDA approval for Leclaza+Rybrevant with the MARIPOSA Phase III trial. Specifically, the FDA approval was based on clinical results presented by Janssen at the European Society for Medical Oncology Annual Congress (ESMO 2023) last year. The results showed a median PFS of 23.7 months in the Leclaza+Rybrevantcombination arm, which was longer than the 18.5 months in the Leclaza monotherapy arm, and 16.6 months in the Tagrisso monotherapy arm. Leclaza+Rybrevant combination was associated with a 30% lower risk of disease progression and death than Tagrisso monotherapy. The interim OS analysis showed a trend favorable to Leclaza+Rybrevantover Tagrisso monotherapy. PFS2 results showed a 25% lower risk of disease progression or death in the Leclaza+Rybrevant combination arm compared with Tagrisso monotherapy arm. Tagrisso+platinum-based chemotherapy is approved in Korea and the U.S. NSCLC drug TagrissoTagrisso was approved in Korea based on the results of the FLAURA2 clinical trial. In May, the Ministry of Food and Drug Safety approved Tagrisso+platinum-based chemotherapy as a first-line treatment for EGFR-positive non-small-cell lung cancer. Tagrisso is a third-generation TKI developed by AstraZeneca. The Phase III FLAURA2 trial enrolled 557 patients with locally advanced or metastatic NSCLC who had received no prior systemic therapy and were positive for EGFR exon 19 deletion or exon 21 mutation. The study evaluated the efficacy and safety of Tagrisso combination therapy versus Tagrisso monotherapy. Results showed that Tagrisso+platinum-based chemotherapy reduced the risk of disease progression or death by 38% compared to Tagrisso monotherapy. Median investigator-assessed PFS was 25.5 months, an 8.8-month extension compared to 16.7 months with Tagrisso monotherapy. Median PFS by blinded independent central review (BICR) was 29.4 months, compared with 19.9 months in the Tagrisso monotherapy arm. Also, inpatients with the L858R mutation, Tagrisso+platinum-based chemotherapy showed a median PFS of 24.7 months, 10.8 months longer than the 13.9 months achieved in the Tagrisso monotherapy arm. Use of combination therapies boosts efficacy, but managing side effects remains key#EB Both Leclaza and Tagrisso have been granted additional regulatory approvals for their combination therapies, heating competition in the first-line treatment market for EGFR-mutated NSCLC. However, side effects management will be a key issue due to its combined use with new drugs. Rybrevant is an intravenous (IV) formulation that requires dosing once every three weeks. Adding an IV formulation to the existing oral formulation, Leclaza, could double the effectiveness but reduce dosing convenience. Janssen is also developing a subcutaneous (SC) formulation to significantly reduce dosing time and address concerns about infusion-related side effects. Recently published clinical results showed that the combination of the subcutaneous formulation of Rybrevant SC+Leclaza achieved similar outcomes to Rybrevant IV+Leclaza. At a median follow-up period of 7 months, the Leclaza+Rybrevant SC was non-inferior to Leclaza+Rybrevant IV. “The Leclaza+Rybrevant combination therapy has continued to generate positive data in brain metastases, L858R mutation, etc., and therefore can be a viable first-line treatment option for EGFR-mutated NSCLC,” said a professor of medical oncology at a university hospital. ”It is difficult to say that use of the combination therapy is good for all patients, as the use of combination therapy is associated with more side effects than using either drug as monotherapy. This is why prescribing Rybrevantto to older patients can be challenging.” “The use of the intravenous formulation of Rybrevantrequires much attention. While a subcutaneous formulation is currently in development, the existing intravenous formulation is associated with frequent rashes, paronychia, and infusion-related adverse events. When selecting a treatment, we should consider not just the efficacy but the patient's overall experience, including side effects, frequency of visits, and quality of life. So there will be an active debate about which patients will benefit most from the early use of the combination therapy.” Concerns also rise on the lack of later-line therapies following the use of Tagrisso+platinum-based chemotherapy The use of Tagrisso and platinum-based chemotherapy as first-line treatment may lead to a shortage of treatment options for patients who develop resistance. In the past, EGFR-mutated NSCLC targeted therapies have been used as follows: 3rd generation TKIs were used on patients who were confirmed to be T790M-positive during biopsy after using 1st and 2nd generation TKIs, and platinum-based chemotherapy (Alimta plus carboplatin/cisplatin) was used on T790M-negative patients. So if a 3rd-generation TKI monotherapy is used in the first line, platinum-based chemotherapy is often used as a second-line treatment as they cannot use 1st or 2nd-generation TKIs due to resistance. This suggests that the use of both Tagrisso and platinum-based chemotherapy in the first line may lead to a lack of later-line treatment options. After developing a resistance to the combination, only taxane drugs such as docetaxel and paclitaxel and immuno-oncology drugs targeting PD-L1 will remain as treatment options. “After failing treatment with EGFR-TKIs, we usually use platinum-based chemotherapy, Alimta+cisplatin/carboplatin,” says a professor of medical oncology at a university hospital. “Combining these drugs with 3rd-generation TKIs is certainly therapeutic. However, we should also consider the lack of later-line treatment options after the patient develops TKI resistance if we pull the next line of treatment in advance amid a shortage of treatment options.” “As 3rd-generation TKIs have settled as the first-line standard of care, the role of 1st- and 2nd-generation TKIs will continue to diminish, and it is likely that it will ultimately become a competition between 3rd-generation TKI monotherapy and combination therapy.”
- Company
- Value of new drugs should be evaluated based on efficacy
- by Hwang, Byung-woo Aug 27, 2024 05:50am
- 'Receiving recognization of the value of new drugs' is one of the key challenges for pharma and biotech companies. With the proportion of new drugs being introduced into Korea rising and the number of homegrown new drugs increasing, the need for a drug pricing policy that recognizes the value of these new drugs has been growing. Recognizing the value of new drugs can lead to a virtuous cycle that drives profits and motivation for subsequent research and development (R&D) in the industry. The 'Measure to Improve the Drug Pricing System to Reflect the Innovative Value of New Drugs' being discussed in this context has raised industry expectations, promising improved access to innovative new drugs and preferential treatment during pharmacoeconomic evaluations. However, as the system is being implemented for the first time, a gray zone where the interpretation of the regulations may differ does exist, raising concerns about the system’s practicality. In the long run, it will be interesting to see if the government's intention to reflect the innovative value of drugs translates into the real world. 'Innovation' condition added to the flexible ICER threshold... Will the threshold exceed KRW 55 million? On the 16th, the National Health Insurance Review and Assessment Service (HIRA) released the 'Detailed Evaluation Criteria for Drugs Subject to Negotiation, including New Drugs', which was revised based on the deliberations of the Drug Reimbursement Evaluation Committee (DREC). The revisions include ▲ establishment of ICER threshold elasticity evaluation criteria for drugs ▲ addition of severe diseases to the risk-sharing program ▲ omission of the Drug Evaluation Committee when expanding the coverage of risk-sharing drugs under 1.5 billion won ▲ and new conditions for submitting clinical evidence such as RWD and RWE when renewing risk-sharing programs. The most notable part is that the 'innovativeness of the new drug' standard had been specified in the criteria to flexibly evaluate the ICER (incremental cost-effectiveness ratio) threshold. Previously, the regulations referred to the use of an ICER threshold as “instead of using an explicit threshold, flexibly refer to the results of previous deliberations considering the severity of disease, social burden of disease, impact on quality of life, and innovativeness.” The government’s In the specified regulations, a new drug is now regarded as ‘innovative’ when it satisfies all of the following criteria: ▲ there is no substitutable or therapeutically equivalent product or treatment ▲ a significant clinical improvement, such as prolonged survival, is recognized in the final outcome, ▲ the new drug is approved by the MFDS under Article 35(4)(2) of the Pharmaceutical Affairs Act through expedited review or is recognized as equivalent by the committee. The question now is how much of the innovativeness of a new drug can be recognized. The industry believes that Enhertu's case will serve as one reference point. Although there is no clearly documented figure, it is generally accepted that the maximum ICER granted for reimbursement in Korea is KRW 50 million. And even the KRW 50 million threshold is known to have been granted very few times. The ICER threshold that the government had set for Enhertu is likely in the low KRW 50 million range. In other words, the drug was recognized as cost-effective despite exceeding the ICER threshold. In this regard, the pharmaceutical industry believes that the ICER threshold should be more flexibly applied given the effectiveness of each drug. A representative from a multinational pharmaceutical company said, “If a drug has such a good effect that it prolongs survival, the ICER is bound to increase because it is more costly to administer. This should be recognized to some extent, but if the flexible ICER range is in the KRW 55 million range, these good drugs will still have difficulty passing the threshold.” For example, the ICER threshold should be more reflective of the innovativeness of a new drug that comes out after decades of lack of treatment, or a treatment that has a much better hazard ratio during survival than existing treatments and improves survival by over twofold. KRPIA’s research on the R&D Investment Status of Global Pharmaceutical Companies Actual cases of flexible ICER application is key...KRPIA, “Flexible approach adopted by major countries should be considered” In particular, the industry predicts that actual cases of application will be important, especially given that many drugs that should be eligible for flexible application of the ICER threshold are awaiting evaluation. “I think it's positive that the government explicitly stated their intention to recognize innovation compared to how such a wording was non-existent regarding the application of the ICER thresholds,” said an industry representative. “There are about 8 drugs that would want to be recognized for their innovativeness, so we have high hopes for the new direction.” “However, if the government’s range of flexibility is adding a mere KRW 5 million to the current upper ICER limit of KRW 50 million, a big difference in perspective will remain. The company's efforts are important, but for real-world application, it remains to be seen whether the government will be willing to recognize an ICER threshold of an unconventional level, whether it is KRW 70 million or KRW 100 million.” The industry pointed out that there are many difficulties for drugs in passing pharmacoeconomic evaluations, such as the reduction in the price of alternative drugs due to various follow-up measures and DREC’s repeated requests for supplementary data until the desired price level is reached. It is unlikely that a small increase in ICER, which fluctuates greatly depending on the assumptions made, will be enough to get an innovative new drug approved quickly. The ICER threshold desired by multinational pharmaceutical companies is reflected in the 'KRPIA Policy Proposal for Improving Public Healthcare' that was released by the Korean Research-based Pharmaceutical Industry Association (KRPIA). In the 'Prospects for New Drug Access Policy' part of the policy proposal, KRPIA suggested that it is not easy to objectively evaluate the effectiveness and innovativeness of new drugs and that it is necessary to consider the flexible new drug evaluation methods of major countries. KRPIA New Drug Access Policy Outlook (in comparison with major countries) In the United Kingdom, “a broad and multi-layered ICER criterion is applied, which considers the disease characteristics, clinical utility, payment value, and societal needs,” and in Canada “ICER is a reference for drug pricing negotiations, and the decisive factor is the level of symptom improvement, and the price of the drug is set at the upper limit of the median drug price in 11 major countries. In Germany, “pharmaceutical companies may autonomously set the initial price and negotiate reimbursement and discount rates after 1 year by evaluating its innovativeness and usage,” and Japan also adopts a system that evaluates a drug’s innovativeness and assigns an adjusted price. In consideration of this global trend, KRPIA emphasized that Korea also needs to more flexibly view ICER, etc. to recognize the innovativeness of new drugs. While the pharmaceutical industry welcomes the inclusion of the 'new drug’s innovative value' and 'expanded coverage for severe and rare diseases' in the government's major policies, the industry is emphasizing the need to set a more specific direction. For example, Kay Bae, the Chair of KRPIA stated, “While the government's recognition of the value of new drugs is an encouraging achievement, the government needs to come up with more practical and concrete measures so that new drugs can be supplied to patients quickly and lead to a virtuous R&D cycle in Korea.” The amended system has measures in place that require observation, such as the submission of clinical evidence such as RWD and RWE when renewing RSA contracts. In the long run, the government and the pharmaceutical industry are eye-to-eye in that they believe in the need to recognize the 'innovativeness of new drugs'. Well begun is half done, and the pharmaceutical industry welcomes the government’s launch of the 'Measure to Improve the Drug Pricing System to Reflect the Innovative Value of New Drugs.’ How well the system is applied and supplemented will remain a future task.
- Company
- K-similars won record number of U.S.·Europe nods this year
- by Chon, Seung-Hyun Aug 27, 2024 05:50am
- Korean biopharmaceutical companies have launched the highest number of biosimilars in history this year in the United States and European markets. As Celltrion and Samsung Bioepis won approvals for six biosimilars over eight months in the world's largest market, they topped the previous record of five cases of five years ago. According to sources on August 27th, Celltrion's SteQyma, a biosimilar to Stelara for the treatment of autoimmune diseases, received marketing authorization from the European Commission (EC). It reached the final commercialization step two months after the Committee for Medicinal Products for Human Use (CHMP) of the European Medicines Agency's (EMA) committee issued a positive opinion recommending SteQyma approval at the end of June. Stelara (ustekinumab), developed by Janssen, is a treatment for autoimmune diseases prescribed for plaque psoriasis, psoriatic arthritis, Crohn's disease, and ulcerative colitis. It inhibits the activity of interleukin (IL)-12,23, a type of inflammatory cytokine involved in immune responses. Last year, the global market size for ustekinumab was estimated at US$ 20.4 billion (about KRW 26.5 trillion). The European market was estimated at US$3.1 billion (about KRW 4.0 trillion), taking 15% of the global market. After Samsung Bioepis won European approval for Pyzchiva, a biosimilar to Stelara, in April, Celltrion entered the European market as the second Korean company. In June, Samsung Bioepis obtained a marketing authorization for Pyzchiva. U.S. and European biosimilar approvals obtained by Korean biopharmaceutical companies: (2024) Celltrion won European approval for Omlyclo, a biosimilar to Xolair, and SteQyma, a biosimilar to Stelara in 2024. Samsung Bioepis won European approval for Pyzchiva, a biosimilar to Stelara, and U.S. FDA approval for Opuviz, a biosimilar to Eylea, Pyzchiva, and Epysqli, a biosimilar to Soliris in 2024 (source: respective companies and FSS). Celltrion and Samsung Bioepis won six approvals for biosimilars in the United States and Europe this year. It exceeded the previous record of five approvals in 2019, breaking the highest number of approvals in history. Celltrion received a marketing authroziation for Omlyclo, a biosimilar to Xolair, from the EC in May. Omlyclo is the first Xolair biosimilar to receive the European marketing authorization. Xolair is an antibody biosimilar used to treat allergic asthma, chronic rhinosinusitis with nasal polyps (CRSwNP), and chronic spontaneous urticaria (CSU). Samsung Bioepis won four biosimilar approvals this year in the United States and Europe this year, with three approvals in the United States and one in Europe. In April, Samsung Bioepis won European approval for Pyzchiva, a biosimilar to Stelara. Since May, Samsung Bioepis succeeded in obtaining biosimilar approvals from the U.S. FDA for the past three months. It received approval for Opuviz, a biosimilar to Eylea. Eylea, developed by Regeneron in the United States, is indicated for treating wet age-related macular degeneration (AMD). Eylea generated approximately KRW 13 trillion in global sales last year. After winning FDA approval for Pyzchiva in June, Samsung Bioepis obtained a marketing authorization for its orphan drug Epysqli. Epysqli, developed by Alexion in the United States, is a biosimilar to Soliris. Epysqli won FDA approval as a treatment of paroxysmal nocturnal hemoglobinuria (PNH) and atypical hemolytic uremic syndrome (aHUS). In 2019, Celltrion and Samsung Bioepis received five biosimilar approvals in the United States and Europe. In November 2019, Celltrion received European approval for Remsima SC, a subcutaneous injection formulation of Remicade. Remsima SC is a biosimilar developed by Celltrion by changing the formulation of Remicade from intravenous (IV) to subcutaneous (SC) injection. In 2019, Samsung Bioepis received FDA approval for four biosimilars to Herceptin, Enbrel, Humira, and Lucentis. In January 2019, Ontruzant, a biosimilar to Herceptin, received a marketing authorization in the United States, followed by Eticovo and Hadlima, biosimilars to Enbrel and Humira, in April and July of the same year, respectively. Samsung Bioepis obtained FDA approval for its Byooviz, a biosimilar to Lucentis, in the United States in September 2021. Korean pharmaceutical companies have started actively targeting the global biosimilar market after Celltrion's Remsima received European marketing authorization under the title of 'The world's first antibody biosimilar' in August 2013. Since 2016, Korean biopharmaceutical companies continued to win new approvals yearly in the United States and Europe. Celltrion achieved success, obtaining eight approvals in Europe and six in the United States. The company also entered the European market for Remicade, Mabthera, Herceptin, Avastin, Humira, Xolair, and Stelara. In 2016, Celltrion's Inflectra, a biosimilar to Remicade, became the first to win FDA approval in the United States. In 2018, Truxima and Herzuma received FDA approvals. In September 2022, Celltrion obtained a marketing authorization for Vegzelma, a biosimilar to Avastin, from the FDA, and last year, Yuflyma, a biosimilar to Humira, also received FDA approval. In August last year, Celltrion received a marketing authorization for Zymfentra, a SC formulation of Remsima, as a novel drug. Samsung Bioepis won eight approvals in Europe and six in the United States. In January 2016, Samsung Bioepis began its global market expansion by obtaining approval for its Benepali, a biosimilar to the autoimmune disease treatment Enbrel, in Europe. Afterward, the company received European approval for biosimilars referencing Remicade, Herceptin, Humira, Avastin, Lucentis, Soliris, and Stelara. In April 2017, Samsung Bioepis received the first FDA approval in the United States for its Renflexis, a biosimilar to Remicade. After that, the company entered the U.S. market for biosimilars referencing Herceptin, Enbrel, Humira, Lucentis, Eyela, Stelara, and Soliris.