- LOGIN
- MemberShip
- 2026-04-07 06:20:58
- Policy
- Daewoong secures generic Xigduo through a transfer
- by Lee, Tak-Sun Jul 29, 2024 05:48am
- Daewoong Pharmaceutical has secured a generic version of the diabetes treatment Xigduo XR Tab (metformin hydrochloride/dapagliflozin propanediol hydrate) through a transfer and acquisition deal. The drug will be covered by reimbursement and introduced to the market in August. With the addition of generic Xigduo XR Tab, Daewoong Pharmaceutical now completes the product lineup, as its generic Forxiga 10 mg (dapagliflozin propanediol hydrate) is listed for reimbursement this month. According to industry sources on July 26th, Daewoong Pharmaceutical's Forxilomet XR Tab 10/1000mg, Forxilomet XR Tab 10/500mg, and Forxilomet XR Tab 5/500mg will be listed for reimbursement starting in August. These products' upper-limit prices are KRW 435, KRW 402, and KRW 342, respectively. Daewoong Pharmaceutical acquired these products already on the reimbursement listing through a transfer and acquisition deal. Daewoong Pharmaceutical acquired products from Jinyang Pharm and Huons. As a result, these products are manufactured by different contract manufacturing organization (CMO) sites. Kukje Pharm manufactures Forxilomet XR Tab 5/500mg and Forxilomet XR Tab 10/500mg, and Samik Pharmaceutical manufactures Forxilomet XR Tab 10/1000mg. Dongkoo Bio&Pharma manufactures Daewoong Pharmaceutical's monotherapy Forxilo tab 5mg and 10mg. Most pharmaceutical companies have listed their products immediately after the substance patent expiration on April 7th last year, but Daewoong Pharmaceutical was unable to do so. At the time, Daewoong was in a joint-sales agreement with the original company, AstraZeneca, to sell Forxiga and Xigduo. From 2018 to the end of last year, Daewoong Pharmaceutical was jointly selling Forxiga and Xigduo. As Forxiga is set to discontinue sales in South Korea, it was canceled for approval and deleted from the reimbursement listing. HK inno.N has been jointly selling Xigduo with AstraZeneca under a joint sales agreement since January. The ceiling price of Xigduo has been reduced by mandate since July 20th, after the introduction of generics. It was reduced after a year and three months after the generic entry. Although delayed, the price of Xigduo was reduced after AstraZeneca withdrew from filing a suit to cancel the mandatory reduction. Following a price reduction, sales reduction is inevitable. Therefore, AstraZeneca and HK inno.N as a team will likely put more effort into marketing. As a result, they are expected to compete fiercely against Daewoong Pharmaceutical. Although Boryung and Hanmi Pharm are now leading the generic market for dapagliflozin, Daewoong Pharmaceutical's entry will likely intensify competition among companies vying for the top spot. Companies aiming to dominate the market, including HK inno.N, which gained Forxiga's indication, are set to begin.
- Policy
- P3T for new insomnia drug will be conducted in KOR
- by Lee, Hye-Kyung Jul 29, 2024 05:47am
- New drug candidate for insomnia The Swiss biotechnology company Idorsia will conduct a Phase III clinical trial in Korea for ACT-541468 (daridorexant), its new drug candidate for insomnia. The Ministry of Food and Drug Safety (MFDS) approved Nxera Pharma Korea's application for a "multicenter, randomized, double-blind, placebo-controlled, parallel-group, Phase III clinical trial to evaluate the efficacy and safety of ACT-541468 (daridorexant) in adults and older adults with insomnia disorders” on the 26th. Daridorexant was previously approved by the U.S. FDA on January 10, 2022, and the European Commission on May 3, 2022, for the treatment of adult patients with insomnia who have difficulty initiating and maintaining sleep. In the second half of 2022 alone, the company reported sales of approximately KRW 100 billion in the U.S. and Europe. The FDA approval was based on an extensive clinical program involving 1,854 adult insomnia patients across 18 countries worldwide. Unlike existing therapies, daridorexant treats insomnia by blocking the binding of orexin, which promotes wakefulness, thereby inhibiting excessive wakefulness. This reduces the inconvenience of daytime sleepiness in daily life. Insomnia is a prevalent condition that affects up to 10% of all adults worldwide. It is defined as a symptom of hyperarousal that is characterized by dissatisfaction with sleep and its negative impact on daytime functioning. A person is classified as having insomnia if he or she is dissatisfied with their sleep and experiences difficulty falling asleep or staying asleep at night at least 3 times per week for at least 3 months. Daridorexant induces sleep by reducing arousal without altering the rate of sleep cycles. The recommended dose consists of a 50 mg tablet taken orally within 30 minutes before bedtime in the evening. However, in certain cases, such as patients with moderate hepatic impairment or those taking CYP3A4 inhibitors, a 25 mg dose may be recommended.
- Company
- Samsung Bioepis’ 3mth operating profit exceeds last year's
- by Chon, Seung-Hyun Jul 26, 2024 05:47am
- Samsung Bioepis posted the largest Q2 revenue in its history. The company recorded twice as much revenue as in its previous record, and its operating income surpassed what it earned in the entire year last year. This is due to the large influx of milestones, receiving a series of approvals for biosimilars in the U.S. and Europe. According to Samsung BioLogics on the 25th, Samsung Bioepis posted revenue of KRW 529.9 billion in Q2, up 107.1% from the KRW 259.9 billion a year earlier, and an operating profit of KRW 257.1 billion, up more than six times from the KRW 41.9 billion a year earlier. Both revenue and operating profit are the largest ever. Samsung Bioepis' Q2 revenue was 89.2% higher than the company’s previous record of KRW 288.9 billion it had made in Q4 last year. Operating profit more than doubled from the KRW 101.5 billion it had recorded in Q3 2021. In just 3 months, the company posted more operating profit than the KRW 205.4 billion it earned in the entire year last year. As a result, the company’s operating profit margin in Q2 reached 48.5%. Samsung Bioepis Smooth sales of biosimilars overseas, and new biosimilar approvals in the U.S. and Europe brought in large milestones. Samsung Bioepis received approval for a total of 3 biosimilars in Europe and the U.S. in Q2 this year. Last month, Samsung Bioepis received marketing authorization from the European Commission for the Stelara biosimilar Pyzchiva. Stelara is an autoimmune disease treatment developed by Janssen that is prescribed for plaque psoriasis, psoriatic arthritis, Crohn's disease, and ulcerative colitis. It inhibits the activity of interleukin (IL)-12,23, a class of inflammatory cytokines involved in the immune response. The drug has made annual global sales of KRW 14 trillion. Samsung Bioepis received marketing authorization from the U.S. Food and Drug Administration (FDA) to sell Pyzchiva in the U.S. In May, Samsung Bioepis received approval from the U.S. Food and Drug Administration (FDA) for Opuviz, a biosimilar of the macular degeneration treatment Eylea. Eylea, which was developed by Regeneron, has indications for wet (neovascular) age-related macular degeneration. Eylea generated global sales of approximately KRW 13 trillion last year. Sandoz has been marketing the newly approved Stelara biosimilar in the U.S. and Europe. The Eylea biosimilar is marketed by Biogen. The new approvals in Europe and the U.S. have led to an influx of milestone payments from its partners. Considering how Samsung Bioepis' Q2 sales and operating income increased by KRW 249.8 billion and KRW 219 billion from the previous quarter, respectively, the milestone inflow in Q2 is expected to have been around KRW 200 billion. View of the Samsung Bioepis building Sales of Samsung Bioepis' existing biosimilar products also continued to grow steadily. Samsung Bioepis has received approval for 8 biosimilars each in Europe and the U.S. In Europe, Samsung Bioepis has biosimilars of Enbrel, Remicade, Humira, Herceptin, Avastin, Lucentis, Soliris, Stelara. In the U.S., the company has neared commercialization of its Remicade, Herceptin, Enbrel, Humira, Lucentis, Eylea, Stelara, and Soliris biosimilars. Samsung Bioepis' licensed biosimilars are marketed by Biogen, Organon, and Sandoz in the global market. Biogen is in charge of 3 biosimilars of autoimmune disease treatments in Europe, including Enbrel, Remicade, and Humira. Biogen also markets Samsung Bioepis' Lucentis biosimilar in the U.S. market. Organon markets 3 biosimilars of Enbrel, Remicade, and Humira in the global market outside of Europe and South Korea. Organon also markets 3 autoimmune disease treatments in the U.S. and 2 anticancer drugs, Herceptin and Avastin biosimilars, outside of Europe and Korea.
- Company
- "Active viral hepatitis treatment can prevent carcinoma"
- by Son, Hyung-Min Jul 26, 2024 05:47am
- Maria Buti, Chair of Public Health at the European Association for the Study of the Liver (EASL) "Most individuals affected by viral hepatitis do not have symptoms. They may not be aware of their infection. When individuals realize that they have abnormalities in their body, liver disease has already progressed. It is important to identify and treat patients early in their disease progression to prevent complications related to hepatocellular carcinoma and liver disease." During a meeting with Daily Pharm, Maria Buti, Chair of Public Health at the European Association for the Study of the Liver (EASL), emphasized the importance of early treatment for hepatitis B and hepatitis C. Hepatitis B is caused by hepatitis B virus (HBV). It can cause infection, advanced liver injury, and chronic liver disease. Hepatitis B is implicated in the cause of hepatocellular carcinoma by over 60%. Recently, there has been a discussion about maximizing the preventative effect of hepatocellular carcinoma by early treatment of hepatitis B. Vemlidy is indicated for treatment. Compared to the conventional use of TDF (tenofovir disoproxil fumarate), such as Viread, Vemlidy has been shown to reduce the occurrence of hepatocellular carcinoma by half. Hepatitis C is identified as contributing to the occurrence of hepatocellular carcinoma in 10-15% of the patients. However, hepatitis C is now curable with the introduction of Epclusa. There are various genotypes of hepatitis C, and Epclusa is highly effective in most patients, regardless of their genotypes. Buti emphasized the timely use of treatments to prevent hepatocellular carcinoma now that effective new drugs are available. Vemlidy has been shown to be effective in preventing hepatocellular carcinoma Because Hepatitis B is incurable, individuals must take medicines their whole lives, but treatments for managing the virus are available in the market. Vemlidy, a type of TAF (tenofovir alafenamide), Vemlidy, and Baraclude are used to treat hepattiis B. Currently, the drugs used in clinical practice are known to suppress viral load significantly. These treatments can suppress the virus to an undetectable level in tests, even though the virus continues to attempt replication. "Antiviral drugs such as Vemlidy, Viread, and Baraclude can help prevent liver decompensation in patients," Buti said. "However, ensuring the safety of drugs is increasingly important due to their long-term use." Vemlidy is also suitable for use in pregnant women and has the advantage of not requiring dose adjustment based on the patient's renal function status. The 8-year follow-up clinical trials, 'Studies 108 & 110,' for Vemlidy conducted on both treatment-naive and treatment-experienced patients, confirmed 5-year viral suppression rates and liver cancer prevention effects. Out of the 1,298 patients involved in the study, there were no instances of decompensated hepatocirrhosis reported in the Vemlidy-treated group. Moreover, during the 8-year period, there were 21 cases (1.6%) of hepatocellular carcinoma in the Vemlidy-treated group. There were also no documented cases of drug resistance among patients treated with Vemlidy. In a study evaluating the safety of Vemlidy in patients with severe kidney dysfunction and kidney diseases, no additional adverse reactions were observed. "If active treatment with drugs like Vemlidy is initiated early during hepatitis B infection, favorable outcomes can be achieved," Buti said. "Additionally, it has been shown that Vemlidy can prevent the occurrence of hepatocellular carcinoma and other complications. Currently, treatment trends focus on the early use of TAF-based drugs such as Vemlidy." Buti added, "Korean treatment guidelines, such as those from the Korean Association for the Study of the Liver, have stricter restrictions on the use of drugs compared to European guidelines. In Europe, the criteria for initiating treatment, such as HBV DNA levels or liver function tests, are relatively lower, allowing for earlier treatment initiation." Buti said, "To effectively treat hepatitis B, starting treatment as early as possible for as many patients as possible is crucial. It is necessary to compare different guidelines to broaden the scope of treatment." Hepatitis C, without vaccines…drugs can be used for prevention Hepatitis C has a lower risk of hepatocellular carcinoma compared to hepatitis B, but if left untreated, it can lead to liver cancer or severe liver disease. Hepatitis C is considered curable if detected early and treated with drugs on time. Epclusa has made it possible to treat hepatitis C regardless of genotype or liver cirrhosis status. Buti emphasized, "Hepatitis C has various genotypes, but Epclusa is effective regardless of genotype. Epclusa can be administered orally once daily, making treatment easier for patients." Patient screening is important for Hepatitis B since it's curable, according to Buti. "Spain was the first country in the world to achieve hepatitis C elimination. A key factor in this success was the micro-elimination strategy. While definitions of high-risk groups may vary by country, in Spain, high-risk groups with high hepatitis C prevalence, such as injection drug users and patients with mental illnesses, were required to undergo screening whenever they visited the emergency room," Buti said. In Spain, individuals have participated in screening for hepatitis C, which has led to significant achievements. "Since there is no vaccine for hepatitis C, treatment is used as a preventive measure. From a public health perspective, this is a critical concept," Buti said. "Treating infected individuals to block further virus spread is a good approach to prevention." "When a patient is diagnosed with hepatitis C through screening, it is crucial to provide timely information about appropriate treatments, such as antiviral drugs, and ensure that the treatment is pursued. A diagnosis without follow-up treatment is meaningless. Therefore, linking diagnosis to treatment is crucial for achieving hepatitis C elimination," Buti added. "To prevent viral liver infection from progressing and patients suffering from side effects, early diagnosis of patients is crucial. The only way is to do a screening. This applies to both hepatitis B and hepatitis C," Buti emphasized.
- Policy
- KDCA "Will secure budget for domestic mRNA vaccine dev."
- by Lee, Jeong-Hwan Jul 26, 2024 05:47am
- The Korea Disease Control and Prevention Agency (KDCA) plans to secure national budget for developing domestic mRNA vaccines. The Korea Disease Control and Prevention Agency (KDCA) announced that they will focus on securing national budget to develop Korea-made mRNA vaccines and secure platforms, supporting up to phase 3 trials. Its vision is to secure domestic mRNA technology based on Japan's case. Japan designated companies that could develop domestic vaccines during the COVID-19 pandemic from 2020 to 2023 and invested KRW 930 billion in the entire process from the beginning of the development, clinical trials, and production. The KDCA has submitted this plan to the Health and Welfare Committee of the National Assembly on July 24th. The KDCA explained that they set a government-wide policy to develop mRNA vaccines and secure platforms. They are in the process of allocating the relevant budget. They also planned this project to allocate a sufficient budget by continuing to communicate with fields, including experts, conferences with companies, and interviews. In particular, the KDCA emphasized that they prioritize securing a multi-KRW 100 billion budget so that they could support up to phase 3 trials by referencing Japan's domestic mRNA vaccine development case. The agency aims to support companies by acquiring budgets based on Japan's KRW 930 billion investment in the development. Additionally, they stated that they are continuing to hold meetings with the Ministry of the Interior and Safety to set up a support team for mRNA vaccine development. Furthermore, they stated that discussions and communications, including conferences with companies and interviews, to establish a system for technology collaboration. The KDCA said, "We will work to secure a sufficient budget with an aim to complete mRNA vaccine development." They added, "As the budget allocation is in progress and the project details are being discussed, we will quickly organize a supporting team within the second half of the year." "When the project takes off, we will officially organize an operating committee with public-private experts and company officials," the KDCA added. "It will allow companies to share their difficulties in their field. We will also supply various technology support packages for vaccine development to research agencies such as the National Institute of Infectious Diseases."
- Policy
- Will Rinvoq’s pediatric indication be expanded in KOR?
- by Lee, Hye-Kyung Jul 26, 2024 05:47am
- AbbVie Korea will conduct a clinical trial in Korea to expand the indication of its atopic dermatitis drug Rinvoq (upadacitinib) to children aged 2 to 12 years old. On the 24th, the Ministry of Food and Drug Safety (MFDS) approved AbbVie’s application to initiate a Phase III, open-label, blinded trial (START UP) to compare the safety and efficacy of Dupixent (dupilumab) vs RInvoq in children aged 2 to 12 years with moderate-to-severe atopic dermatitis. The trial will be conducted at Gangnam Sacred Heart Hospital, Chung-Ang University Hospital, Konkuk University Medical Center, Korea University Ansan Hospital, Soonchunhyang University Hospital Bucheon, and Seoul National University Hospital. RInvoq can currently be administered once a day to adolescents aged 12 to 17 years weighing 40 kilograms or more. The MFDS-approved label does not recommended for adolescents under 40 kilograms because it has not been studied in the patient population. Other JAK inhibitors used for atopic patients include Eli Lilly’s ‘Olumiant (baricitinib)’ and Pfizer’s ‘Cibinqo (abrocitinib).’ While both are reimbursed for adult patients, only Rinvoq and Cibinqo (for patients 12 years of age and older) and Dupixent (for patients 6 years of age and older) have been available for pediatric and adolescent patients since April of last year. Starting next month, Dupixent will also be reimbursed for infants and children with severe atopic dermatitis aged 6 months and older. In particular, Dupixent is the only pediatric atopic dermatitis treatment that confirmed symptom control and a consistent safety profile in infants, children, and adolescents with moderate-to-severe atopic dermatitis aged 6 months and older through a Phase III clinical study. If the company can generate significant clinical data in children and adolescents aged 2 to 12 years through the new trial, this will allow pediatric atopic dermatitis patients to have broader treatment options. The dosing regimen differs for the two drugs. While Rinvoq is an extended-release tablet that can be taken orally once a day, Dupixent is a subcutaneous injection that is injected into the fatty layer under the skin. Meanwhile, Korean companies are also intent on developing atopic dermatitis drugs in Korea. LG Chem changed the indication o itsf LC510255, a new drug candidate for autoimmune diseases, to atopic dermatitis in the second phase of domestic clinical trials. JW Pharmaceuticals transferred the technology of its atopic drug candidate JW1601 to LEO Pharma in Denmark, but the contract was terminated after the candidate failed to meet the primary endpoint in the initial results of the global Phase IIa/b clinical trial.
- Company
- Mitsubishi accepts DREC results for Uplizna...nears reimb
- by Eo, Yun-Ho Jul 26, 2024 05:47am
- Mitsubishi Tanabe Pharma’s twice-yearly neuromyelitis optica spectrum disorder (NMOSD) drug Uplizna is headed for reimbursement listing in Korea. According to industry sources, Mitsubishi Tanabe Pharma’s Korea accepted the ‘below the evaluated amount’ condition set by the Health Insurance Review and Assessment Service's Drug Reimbursement Evaluation Committee for the reimbursement of Uplizna (inebilizumab), a treatment used to treat adult patients with for neuromyelitis optica spectrum disorder (NMOSD) who are positive for anti-Aquaporin-4 (AQP4) antibodies. As a result, the company will enter pricing negotiations with the National Health Insurance Service in the near future. Uplizna is administered at an initial 300 mg dose, followed by an additional 300 mg dose 2 weeks later, and then every 6 months thereafter from the date of the initial dose. NMOSD occurs when AQP4 autoantibodies, a disease-specific biomarker produced by B cells, bind to AQP4, a target antigen present on glial cells in the central nervous system, and activate the immune responses, causing nerve damage. Uplizna is an anti-CD19 human monoclonal antibody that selectively binds to CD19, a B cell-specific surface antigen, depleting B cells that produce AQP4 antibodies, thereby preventing disease relapse. The safety and efficacy of Uplizna were demonstrated in the N-MOmentum study, which evaluated the use of Uplizna monotherapy in 230 patients without the use of concomitant immunosuppressive agents. Study results showed that 89% of patients treated with Uplizna did not experience a relapse during 197 days of follow-up, resulting in a 77.3% reduction in the risk of relapse compared to placebo. Safety evaluations Uplizna also showed comparable rates of adverse events to the placebo group. Also, in an extension study, Uplizna continued to reduce the risk of relapse for at least 4 years, with an 87.7% relapse-free rate. In terms of long-term safety profile, Uplizna was generally well tolerated, with no increase in infection rates due to B-cell depletion. NMOSD is a serious autoimmune disease in which most patients experience persistent relapses and incomplete recovery, resulting in accumulated nerve damage that can cause vision loss, gait disturbances, and even death from respiratory failure.
- Company
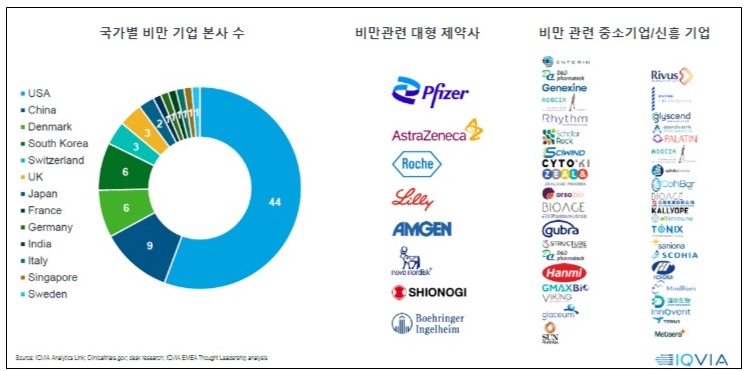
- Guidelines for new obesity drugs must be established
- by Moon, sung-ho Jul 26, 2024 05:46am
- Obesity is projected to impact around 2 billion individuals by 2035. Including overweight, about 4 billion individuals will be affected by obesity, making it a significant global healthcare issue. Following this trend, pharmaceutical and biotech industry is focusing on developing next-generation treatments for obesity. Glucagon-like peptide 1 (GLP-1) drugs, including Saxenda, Wegovy, and Zepbound, showed outstanding weight loss effects in clinical trials. As a result, companies are developing GLP-1-based therapeutics. The number of companies developing obesity drugs by country; big pharmas developing obesity drugs; small businesses and new companies developing obesity drugs. Recently, these drugs have been found to provide cardiovascular benefits and aid weight loss effects. As a result, their expanded use has been gathering attention in clinical settings. The analysis suggests that it's time to consider using these drugs in clinical settings in South Korea. According to drug market research company IQVIA on July 20th, there are 79 obesity drug pipelines worldwide, from preclinical to launched products. Pharmaceutical and biotech companies have developed over 148 products. GLP-1 drugs account for 39% of the pipelines. This suggests that pharmaceutical and biotech companies have begun developing drugs in this class as latecomers after witnessing the success of Saxenda, Wegovy, and Zepbound. 79 obesity drug pipelines worldwide, from preclinical to launched products, and 148 products from pharmaceutical and biotech companies have been developed. The analysis indicates that major companies that have begun developing obesity drugs employ two major development strategies. They either develop a drug as 'monotherapy' based on their differentiation strategy, or consider potential expansion for treating obesity, type 2 diabetes, cardiovascular diseases, and metabolic dysfunction-associated steatohepatitis (MASH) as part of their portfolio strategy. As part of differentiation therapy, monotherapy is being developed to achieve the ▲Highest weight loss rate, ▲Improved safety, and ▲Chronic disease management with oral formulation. For instance, Pfizer and Viking Therapeutics are developing candidate products. Under the portfolio strategy, the market leaders, such as Novo Nordisk (Wegovy) and Lily (Zepbound), and latecomers, aim to expand indications. Lately, the development of 'oral formulation drugs' has gained attention. These drugs are expected to shift the paradigm of a market dominated by injectables. Global companies, Pfizer and Viking Therapeutics, and domestic companies, including Ildong Pharmaceutical and D&D Pharmatech, have started to develop them. Novo Nordisk and Lily, the market-leading companies that already have injectables, have proprietary pipelines. However, Wegovy and Zepbound, which dominate the global market for obesity, have unresolved issue of weight loss rebound. IQVIA Korea's Marketing & Sales Director Kang-Bok Lee said, "Most obesity pipelines at the clinical stage are being developed as oral formulations." Lee added, "However, there are still discussions about oral obesity drugs. Along with the convenience, we must consider whether these drugs are suitable for chronic and maintenance management, and whether their cost and supply network outweigh any remaining issue." Lee added that "There are questions about whether it can have similar efficacy compared to injectables, as well as concerns about tolerability." Lee expressed optimism about oral drug development, saying, "Recently, Viking's oral drug, VK2735, had no clinically significant gastrointestinal side effects compared to placebo, with most being mild." Reflecting on the global trend for obesity drug development, new GLP-1 drugs, such as Wegovy and Zepbound, will likely be introduced to South Korea. According to IQVIA, the market for obesity drugs is rapidly growing after the launch of Wegovy. The worldwide market size in 2023 totaled US$11 billion (about KRW 15.3 trillion), driven by Wegovy. Wegovy contributed 72% of the total US$11 billion-worth market in 2023. In contrast, in South Korea, the release of Wegovy has been delayed due to an issue with 'securing stock' after obtaining marketing approval. As a result, Saxenda (Novo Nordisk) and Qsymia (Alvogen Korea) have taken 60% of the market share, dominating the market. Sources said that the release of Wegovy is set to be released in the Korean market, making it the ninth country globally. A professor from the Department of Endocrinology at an unnamed University Hospital, who is also an executive member of the Korean Society for the Study of Obesity, said, "Following Japan, China has also approved Wegovy. Since an official launch date has not yet been set, it is difficult to guarantee the timing of the release." He analyzed, "This appears to reflect the position of the domestic market within the global market." "Even if it is released, it seems that obesity drugs will be used entirely as non-reimbursable in the domestic market," He added. "While there has been some progress in recognizing obesity as a disease, reimbursement coverage, especially concerning domestic insurance finances, will not be easy." As a result, the pharmaceutical and biotech industries have proposed that, considering the potential of the drug market due to the rising prevalence of obesity, there is a need to accelerate discussions on disease recognition, enhancements in social awareness, and the establishment of clinical practice guidelines and insurance. At the same time, domestic pharmaceutical and biotech companies developing treatments may need to focus on differentiation strategies from competing products, improving efficacy such as preventing weight regain after discontinuation and establishing a stable supply chain to meet global demand. IQVIA Korea's Director Kang-Bok Lee said, "In the past two years, global spending on obesity has increased rapidly with new drugs, and by 2030, more than 15 new items are expected to enter the market, making the next-generation obesity treatment market much more competitive." Lee also said, "Improving educational programs for healthcare professionals to raise awareness of obesity treatment and integrating it into chronic disease management would be an ideal approach." Lee also added, "Currently, even though obesity treatments are approved by the Ministry of Food and Drug Safety (MFDS), there are no cases where these drugs are covered by reimbursement. In the U.S., Medicare (Part D) will now cover Wegovy for some patients with a history of heart disease, as announced by the Centers for Medicare & Medicaid Services (CMS)." Lee added, "In the future, obesity drugs will be divided into reimbursed and out-of-pocket markets, so it is necessary to consider establishing reimbursement criteria for patients with severe obesity or accompanying diseases. We must develop and distribute comprehensive obesity treatment guidelines through collaboration with the medical community and organizations."
- Company
- Antibiotic prescriptions had surged with the pandemic
- by Kim, Jin-Gu Jul 25, 2024 05:51am
- The amount of outpatient antibiotic prescriptions in Korea had changed dramatically during the COVID-19 pandemic. In 2020 and 2021, early stages of the pandemic, antibiotic prescriptions dropped sharply but then surged in 2022. In contrast, the proportion of cephalosporin and quinolone antibiotic prescriptions, which are more powerful than other antibiotics, increased in 2020 and 2021 and then decreased in 2022. The Ministry of Health and Welfare released the "Healthcare Quality Statistics" that contained the above findings on the 24th. The MOHW releases this data annually for comparative statistics among OECD countries. One of the statistical items examines outpatient antibiotic prescriptions in primary care clinics. According to the statistics, in 2022, outpatient prescriptions of local antibiotics, not systemic antibiotics amounted to 21.3DDD per 1000 inhabitants in primary healthcare centers in Korea. Defined Daily Dosage (DDD) is a unit used to measure drug consumption and refers to the average maintenance dose that an adult weighing 70 kg should take per day. Total outpatient antibiotics prescribed for systemic use (Source: Healthcare Quality Statistics 2022) The number of antibiotic prescriptions in Korea has been steadily decreasing since 2016 when it reached 26.9 DDD. Especially during the pandemic, the number dropped below 20.0 DDD in 2020-2021. It is analyzed that as people refrained from outdoor activities due to social distancing measures, respiratory infections decreased, reducing antibiotic prescriptions. In 2022, when the social distancing measures were eased, antibiotic prescriptions made a rebound. From 16.0 DDDs in 2021 to 21.3 DDDs in 2022, the amount increased 33% in 1 year. In contrast, prescriptions for cephalosporin and quinolone antibiotics, which are broader and more potent than other antibiotics, increased significantly early in the pandemic and then decreased near the endemic. Among all outpatient antibiotic prescriptions, the percentage of cephalosporin and quinolone antibiotics increased from 39.5% in 2019 to 43.6% in 2020, then to 44.8% in 2021. In 2022, their share decreased slightly to 43.1%. Percentage of cephalosporin and quinolone antibiotics prescribed (Healthcare Quality Statistics 2022) The prescription and use of antibiotics require management due to resistance issues and is considered one of the important areas to monitor through national antimicrobial resistance management policies. The OECD Healthcare Quality Statistics also includes two indicators related to antibiotics: the total number of outpatient antibiotics prescribed for systemic use and the proportion of cephalosporin/quinolone antibiotics prescribed. The MOHW explained, "The total volume of outpatient antibiotic prescriptions decreased by 34% in 10 years since 2011 due to the strengthening of antibiotic management policies and increased public awareness. However, the proportion of cephalosporin/quinolone antibiotics prescribed for systemic use has continued to increase over the decade and remains high at 43% as of 2022."
- Policy
- Samjin and Korea Pharma’s first generics are reimb in KOR
- by Lee, Tak-Sun Jul 25, 2024 05:51am
- Samjin Pharmaceutical and Korea Pharma have launched first generics in Korea. Samjin Pharmaceutical will launch the first generic of Sanofi's atrial fibrillation drug Multaq Tab, while Korea Pharma will launch the first generic of Pfizer's antidepressant Pristiq ER Tab. According to industry sources, Samjin Pharmaceutical's ‘Samjin Dron Tab as (dronedarone)’ will be listed at KRW 808 per tablet in Korea. The drug is indicated for the management of atrial fibrillation (AF) in patients in sinus rhythm with a history of paroxysmal or persistent AF to reduce the risk of hospitalization. Adults may take 1 tablet (dronedarone 400 mg) twice daily with breakfast and dinner. The original generic version of dronedarone is Sanofi's Multaq Tab. It was approved in Korea in February 2010. The product patent for Multaq expired in 2022. According to UBIST, its last year's outpatient prescriptions amounted to KRW 10.9 billion, exceeding KRW 10 billion for the first time last year. As Samjin already owns a high market share in the field of cardiovascular disease withPlatless (clopidogrel) among others, Samjin Dron Tab is expected to settle in the market in the short term. In particular, sales growth will further accelerate as there are no generic competitors in the market. Korea Pharma will launch the first generic version of Pfizer's Pristiq 100mg. Pristiq’s generics are already available in the market. In 2020, 4 domestic pharmaceutical companies entered the market with salt-modified drugs that evaded the patents. Therefore, the currently available follow-on drugs have different ingredients from the original product. Whereas Pfizer's Pristiq contains desvenlafaxine succinate monohydrate, the salt-modified drugs contain desvenlafaxine benzoate or desvenlafaxine. Korea Pharma’s ‘Pharma Desvenlafaxine ER Tab. 100mg’ is a first generic that uses the same salt as the original. Desvenlafaxine is a serotonin-norepinephrine reuptake inhibitor (SNRI), which is characterized by its low risk of drug interactions and a low risk of side effects such as hypertension and sexual dysfunction. Last year, Pristiq generated sales of KRW 7.2 billion according to IQVIA. Its sales have been declining with newer generics gradually gaining influence in the market. A representative from Korea Pharma said, "Pharma Desvenlafaxine ER Tab is a competitive product that was first approved with the same salt formulation as the original Pristiq ER Tab. We plan to quickly land and expand our market share through active sales activities." Samjin Pharmaceutical and Hankook Pharma's products received an insurance price of 59.5% of the original’s insurance ceiling price instead of 53.55% because the two companies have listed the first and only first generics. As a result, Samjin Dron Tab was listed at KRW 808 and Pharma Desvenlafaxine ER Tab at KRW 742 in Korea. Their original versions, Multaq Tab and Pristiq ER Tab are priced at KRW 1,357 and KRW 1,247, respectively.