- LOGIN
- MemberShip
- 2026-04-07 06:20:58
- Company
- Meningitis B vaccine Bexsero is released in Korea
- by Moon, sung-ho Jul 25, 2024 05:51am
- Competition in the 'meningococcal' vaccine market, which is mainly vaccinated in pediatric clinics, has recently been reignited. Although its domestic market is worth less than KRW 10 billion, the emergence of next-generation vaccines is expected to spark new competition among multinational pharmaceutical companies. #This is because GSK, the market leader, has launched Bexsero (meningococcal serogroup B vaccine), a next-generation vaccine. Professor Hyunmi Kang (Department of Pediatrics, St. Mary's Hospital, Seoul) explained the clinical implications of the introduction of Bexsero at an event held by GSK on the 16th. Meningococcal meningitis is a statutory Class 2 infectious disease with a fatality rate of approximately 10-14%. It affects 500,000 patients worldwide each year. The main symptoms include headache, fever, neck stiffness, vomiting, and decreased consciousness, and is often accompanied by petechiae or purpura fulminans. 11 to 19% of recovered patients may suffer from sequelae such as hearing loss, cognitive impairment, and neurological disorders, making it an important infection to prevent. In particular, as meningococcal disease is transmitted person-to-person by respiratory droplets or secretions, vaccination is recommended for those who are about to enter a group setting. For example, new recruits and college students who will be living in dormitories may want to consider meningococcal vaccination. Other recommended populations for meningococcal vaccination include travelers and residents of meningococcal endemic areas, such as Africa, and pilgrimage travelers to Mecca, Saudi Arabia. Typical serogroups of meningococci that cause invasive meningococcal infections in humans include A, B, C, W, X, and Y. The most predominant meningococcal serogroup in Korea among these is serogroup B. From 2010 to 2016, the proportion of Meningitis B cases identified in Korea was 28%, but from 2017 to 2020, the rate rose significantly to 78%. In this scene, GSK launched Bexsero, a vaccine that prevents meningococcal serogroup B, in the Korean market 2 years after its approval in 2022. Professor Kang assessed that Bexsero can play a significant role in addressing the unmet need in the field as it prevents meningococcal serogroup B. "Globally, meningococcal infections are most prevalent in infants under one year of age compared to other age groups,” explained Professor Kang. “It causes bacterial meningitis and sepsis, and one to two out of 10 survivors also experience brain damage, hearing loss, and limb loss.” "The prevalence of meningococcal serogroups varies across countries and time periods, so it is not easy to predict. In Korea, serogroup B meningococcal infection cases have increased in recent years, increasing the need for its prevention.” In Korea, GSK is leading the meningococcal vaccine market. The vaccines, which are non-reimbursed, cost KRW 150,000. The first quadrivalent meningococcal vaccine in Korea, GSK’s Menveo has been dominating the market with sales of KRW 5.2 billion based on IQVIA last year. Sanofi's Menactra is also available, but the vaccine only generated KRW 500 million in sales during the same period. In addition, Sanofi received domestic approval for Menquadfi Inj (meningococcal (A, C, Y, W) tetanus toxoid-conjugate vaccine) earlier this year. "In countries such as the United Kingdom, Portugal, and Canada, the importance of preventing disease through immunization has been emphasized due to the high prevalence of meningococcal B," said Joon Bang, Director of Medical Affairs at GSK Korea. "The predominance of meningococcal B in Korea has made it necessary for us to introduce a vaccine to prevent infections caused by meningococcal B.” "We are pleased to be able to contribute to the prevention of meningococcal disease caused by serogroup B, which accounts for a high proportion of meningococcal disease in Korea with the launch of Bexsero. Together with Menveo, the company now owns a vaccine portfolio that can protect against a wide range of serogroups."
- Company
- 'Padcev+Keytruda' combination therapy is set to land in KOR
- by Eo, Yun-Ho Jul 25, 2024 05:51am
- Product photo of Padcev.The combination therapy of 'Padcev+Keytruda,' which is expected to bring a paradigm shift to bladder cancer treatment, will soon land in South Korea. The Ministry of Food and Drug Safety (MFDS) is reviewing the expansion of indication for Astellas Korea's Padcev (enfortumab), an antibody-drug conjugate (ADC), in combination with Keytruda (pembrolizumab), a PD-1 inhibitor that is used in immunotherapy for cancer, as a first-line treatment of urothelial cancer locally advanced or metastatic urothelial carcinoma (la/mUC). The official approval is expected soon. The combination therapy of these drugs for urothelial cancer gained attention after the presentation of its Phase 3 EV-302/KEYNOTE-A39 study results at the 2023 congress of the European Society for Medical Oncology (ESMO Congress 2023), held in October last year, and received a standing ovation. Based on the clinical results, patients treated with Padcev combination had a median progression-free survival (PFS) of 12.5 months, a primary endpoint of the study, indicating a significant improvement compared to the 6.3 months in patients treated with chemotherapy for cancer. Another primary endpoint of the study was median overall survival (OS). The patients had a median OS of 31.5 months, a twofold extension compared to the placebo. Cisplantin-eligible patients treated with Padcev combination had an OS of 31.5 months compared to 18.4 months in placebo-treated group, reducing the risk of death to 47%. Cisplantin-ineligible patients had not reached the median value, whereas the placebo-treated group had an OS of 12.7 months, reducing the risk of death to 57%. In clinical settings, 'Padcev+Keytruda' combination therapy is being considered as a replacement for a first-line treatment GemCis therapy, which has been used as the standard therapy for 30 years. Meanwhile, Padcev was approved in South Korea in March 2023 as a monotherapy for patients with locally advanced or metastatic urothelial cancer who had previous experience with platinum-containing chemotherapy or PD-1 or PD-L1 inhibitors. Astellas has applied for the inclusion of this therapy in reimbursement listing, and it has passed the Cancer Disease Review Committee of the Health Insurance Review and Assessment Service (HIRA).
- Opinion
- [Reporter's View] Keeping up with the Chinese bio industry
- by Son, Hyung-Min Jul 24, 2024 05:51am
- The research and development (R&D) capabilities of Chinese pharmaceutical companies are going from strength to strength year after year. Last year, the immuno-oncology drug Loqtorzi, developed by Junshi Biosciences, was approved in the United States. It became the first Chinese immuno-oncology drug to be approved by the U.S. Food and Drug Administration (FDA). While Chinese anti-PD-L1/PD-1-targeted immuno-oncology drugs have been approved within China, this is the first time they have crossed the FDA approval threshold. This year, BeiGene’s immuno-oncology drug Tevimbra was approved by the FDA. Another Chinese drugmaker, Innovent, is also seeking FDA approval for its own immuno-oncology drug sintilimab in partnership with Eli Lilly. The domestic pharma and biotech industry has also launched a number of immuno-oncology drugs, but most of them are still in the pre-Phase II stage. This is in contrast to Chinese companies, which have completed Phase III trials and are closer to global commercialization. China is leading the way in global commercialization not only of immuno-oncology drugs but also of next-generation therapies such as targeted anti-cancer drugs, gene therapies, and nucleic acid therapeutics. On the other hand, Korea has not been able to make a significant impression in the global market outside of biosimilars. In fact, the number of global new drug approvals by Korean pharmaceutical companies is incomparable, far less than that of Chinese pharmaceutical companies. The number of global new drug approvals by Chinese pharmaceutical companies in countries other than the US has also been steadily increasing, receiving approvals for 44 new drugs in 2020, 40 in 2022, and 14 last year. In the United States, 11 Chinese-made drugs have been approved in the past 3 years. In the case of Korean pharmaceutical companies, only 8 new drugs have been approved so far. In terms of clinical trials, China's share grew by 6.1% from 2019 to 2023, while Korea's share grew by only 0.3% during the same period. Government support has played a major role in raising the Chinese pharmaceutical industry's R&D capabilities. The Chinese government has deisgnated the pharma-bio industry as its economic growth engine and has shortened the approval time for drugs by innovating the CFDA’s review system. Examples include priority review drugs, expanding the number of Centers for Drug Evaluation (CDEs), and accepting overseas clinical data. As a result, China's clinical and new drug review time has been reduced by about one-third compared to before 2015. The expansion of China’s insurance reimbursement that increased the number of drugs on the reimbursement list has also worked in favor of Chinese pharmaceutical companies. The Chinese government continues to expand insurance reimbursement coverage of new drugs, and the revenues are being channeled into R&D investments. Korea, on the other hand, has been less successful in supporting R&D. The Yoon Suk-Yeol's government pledged to support bio R&D from the beginning of his administration, pledging to ‘establish a pharma-bio control tower’ to integrate, foster, and support the pharma-bio industry. However, to date, cooperation between the government and the industry has not been active, as the control tower, the Biohealth Innovation Committee, has not been fully operationalized, and regulations restricting the pharmaceutical industry, such as the drug price reduction system and reimbursement adequacy reevaluations, continue to increase. In order to create new global drugs, Korea needs to establish specific regulations and support systems for innovative drug development. It is illogical to expect good drugs to be made while investing less. Only more investment can lay the foundation for the development and launch of new global medicines. The government has budgeted KRW 2.9 trillion for fundamental R&D next year, which is the largest amount ever allocated. It is this reporter’s hope that this will spur the biotech industry to invest in challenging research areas and develop innovative new drugs.
- Policy
- Gvt commences 'Machine Learning AI Project' for new drug dev
- by Lee, Jeong-Hwan Jul 24, 2024 05:51am
- The government will actively pursue a project for faster new drug discovery. It will fund KRW 34.8 billion over 5 years, from 2024 to 2028, and conduct a project utilizing federated learning of new drug discovery data owned by pharmaceutical companies, university hospitals, research institutes, and corporations. The Ministry of Health and Welfare (MOHW) and Ministry of Science and ICT (MSIT) announced on July 23rd that 26 projects have been selected for the 'Machine Learning Ledger Orchestration for Drug Discovery (K-MELLODY)' project. The MOHW and MSIT are jointly promoting the MELLODDY project, which aims to reduce the cost and time for new drug discovery using a federated learning-based AI model that can safely protect data while exploring. Federated learning is a decentralized learning approach that enables machine learning to be trained internally on data from multiple locations, such as private and institutes, without directly sharing. Analyzed data are sent to a central server. The government has commenced this project and designated the Korea Pharmaceutical and Bio-Pharma Manufacturers Association (KPBMA) to run the project. The project group has selected 26 subprojects and research institutes for each project through a public survey and evaluation. The selected subprojects are categorized into three fields: ▲Establishing Federated Drug Discovery (FDD) platform, ▲Utilizing new drug discovery data and quality management, and ▲Developing AI solution. First, for 'Establishing Federated Drug Discovery (FDD) platform,' project has been selected to establish FDD platform to enable machine learning safely without sharing data between institutes and keeping privacy. As a supervision agency, Evident Inc. has been chosen. Also, 20 institutes have been selected as supervision agencies for AI-driven drug discovery. They will share new data from the fields of drug metabolism·toxicity testing and pharmacoepidemiology. Pharmaceutical companies, universities·hospitals, research centers, and corporations are included. Eight pharmaceutical companies have been selected, including Daewoong Pharmaceutical, Dong-wha Pharm, Samjin Pharm, Yuhan Pharm, Jeil Pharm, Hanmi Pharm, Huons, and JW Pharmaceutical. Universities·hospitals include Gachon University, Catholic University of Korea, Kyungpook National University, Seoul National University, and Seoul National University Hospital. Participating research centers·foundations include Daegu Gyeongbuk Advanced Medical Industry Promotion Foundation, KRIBB, Institut Pasteur Korea, and KRICT. Selected corporations include CIMPLRX and Apace. For 'developing AI solution,' five institutes, including GIST, Mogam Institute for Biomedical Research, AIGEN Sciences, Chonbuk National University Industrial Cooperation Foundation, and KAIST. They will develop the 'Federated ADMET Model (FAM)' to aid in drug candidate discovery, using experimental data from all stages of new drug discovery. The MOHW and MSIT will allocate KRW 34.8 billion to these selected projects over a five year period, from 2024 to 2028. They anticipate establishing an AI-driven new drug discovery infrastructure by collaborating with the government, pharmaceutical companies, research centers, and universities. "By developing machine learning with a federated learning approach that safely shares and uses data, previously challenging large-scale data analysis and use will be enabled, accelerating drug discovery," Ko Hyeongu, MOHW's Advanced Healthcare Support Officer, said. Ko said, "We will strengthen the data usage system and support AI‧Data R&D. We will also strive to lead future healthcare and pharmaceutical innovation and promote citizen's welfare." "The federated learning approach will enable the safe utilization of high-quality drug discovery data accumulated from multiple institutes to develop an AI-driven drug discovery platform and machine-learning solution," Kwon Hyeonjun, MSIT's R&D Policy Director, said. "We will support the R&D of digital bio-health businesses, which converges digital technology, and creating value so that South Korea emerges as a hub for cutting-edge biotechnology."
- Policy
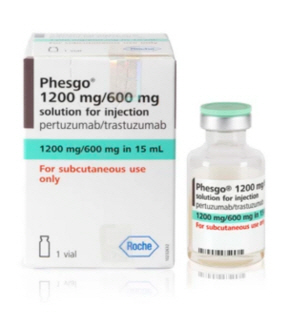
- Roche’s Phesgo is applied RSA for reimb in Korea
- by Lee, Tak-Sun Jul 24, 2024 05:51am
- Korea’s insurance authorities are introducing various measures to reduce drug costs. The government is applying risk-sharing agreements to precalculation drugs and requiring submission of follow-up data for drugs that are exempt from sumitting pharmacoeconomic evaluation data. The drugs subject to the authorities’ measures are Phesgo SC Inj (pertuzumab/trastuzumab) and Ilaris Solution (canakinumab). Phesgo is a biobetter that was developed as a fixed-dose subcutaneous injection formulation of the intravenous injected Herceptin and Perjeta to improve dosing convenience and reduce treatment time for breast cancer patients. Since it is approved as an improved biologic, its insurance price was automatically set at 110% of the upper limit of its targeted products. As a result, Phesgo SC Inj 600/600mg will be listed at KRW 3,490,410, and Phesgo SC Inj 1200/600mg at KRW 5,914,418. According to IQVIA, sales of Herceptin and Perjeta in Korea amounted to KRW 56.5 billion and KRW 111.3 billion, respectively, last year. As an improved version of the two drugs, Phesgo is also expected to record high sales. However, its impact on insurance finances is also expected to be significant. This is why the insurance authorities decided to apply RSA to Phesgo and save insurance finances. The decision was influenced by the fact that Phesgo’s development target, Perjeta, is currently reimbursed through RSA. Like Perjeta, Phesgo is applied the refund-type RSA, under which the company refunds a certain percentage of the claims. "Considering that Phesgo’s target product is an RSA drug, we decided to apply RSA to Phesgo through negotiations with the National Health Insurance Service," explained the insurance authorities. “The product has improved the administration route of the target product, which is convenient for patients to administer, and we expect it to bring financial savings when it replaces the target product." Phesgo passed the Health Insurance Review and Assessment Service’s Cancer Disease Review Committee in August last year, but it took more than a year for it to be listed for reimbursement, the delay which can be explained by the cost-saving measure. Ilaris, which is being reimbursed after 9 years of approval in Korea, also has a number of safeguards in place to reduce costs. It is applied to the refund-type and expenditure cap-type RSA, and conditional follow-up is also required as a PE exemption drug. Its list price is KRW 11,029,469 Ilaris is a rare disease drug for which there are no alternatives and is being publicly reimbursed in over 3 of the A8 countries, allowing the drug to omit submission of pharmacoeconomic evaluation data. However, HIRA’s panel determined that the drug needs to be followed up in the future, including reevaluation of clinical effectiveness and cost-effectiveness, given the uncertainty of its improvement in clinical utility and high cost. Therefore, HIRA imposed a condition on the pharmaceutical company to conduct a prospective clinical study (observation period of 2 years for each patient and submission of observation data and results in 1-year increments) and submit clinical utility and cost-effectiveness data at the end of the RSA term. For example, the pharmaceutical company is required to conduct relevant prospective clinical studies, in an objective, cross-sectional survey format, with at least 2 years of observation per patient, and submit observations and results on a yearly basis. For the NOMID (neonatal onset multisystemic inflammatory disease)/CINCA (chronic infantile neurocutaneous joint syndrome) indication, the company needs to submit efficacy comparison data with existing treatments and cost-effectiveness data at the end of the RSA term. In addition, details such as plans to conduct prospective clinical studies for follow-up management and measures to be taken in case of failure to conduct such studies are required in the RSA. "In the future, drugs that are exempted from economic evaluation, but whose clinical usefulness is unclear, will be thoroughly reevaluated through follow-up management measures," explained a HIRA official.
- Company
- Samsung Bioepis’ Soliris biosimilar is approved in the U.S.
- by Chon, Seung-Hyun Jul 24, 2024 05:51am
- View of the Samsung Bioepis building [데일리팜=천승현 기자] 삼성바이오에피스는 미국 식품의약품국(FDA)으로부터 희귀질환치료제 ‘에피스클리’의 품목허가를 획득했다고 23일 밝혔다. 에피스클리는 미국 알렉시온이 개발한 솔리리스의 바이오시밀러 제품이다. 에피스클리는 발작성 야간 혈색소뇨증, 비정형 용혈성 요독 증후군의 치료제로 FDA 승인을 받았다. 삼성바이오에피스는 2019년 7월부터 2021년 10월까지 발작성 야간 혈색소뇨증 환자들을 대상으로 임상 3상을 통해 에피스클리와 오리지널 의약품 간 비교 연구를 수행했다. 유관 학술대회 발표를 통해 임상의학적 동등성을 입증했다. 삼성바이오에피스는 지난해 7월 에피스클리를 유럽에 출시했고 독일, 이탈리아 솔리리스 바이오시밀러 시장 점유율 1위 등의 성과를 내고 있다. 국내에서는 지난 4월부터 삼성바이오에피스가 직접 에피스클리를 판매하고 있다. 삼성바이오에피스는 오리지널 의약품의 약가 대비 절반 수준으로 솔리리스 바이오시밀러를 출시했다. 삼성바이오에피스는 이번 허가로 미국 시장에서 총 8개 바이오시밀러 제품을 승인받았다. 미국에서도 자가면역질환 치료제, 항암제, 안과질환 치료제에 이어 혈액·신장질환 치료제 분야까지 치료 영역을 확대했다. 삼성바이오에피스 고한승 사장은 “미국에서도 희귀질환 치료제를 승인받아 글로벌 수준의 R&D 역량을 인정받았다”며 “에피스클리는 바이오시밀러의 사회적 가치를 극대화할 수 있는 제품으로서 세계 최대 의약품 시장인 미국에서도 그 가치를 실현하기 위해 지속 노력하겠다”고 전했다.
- Company
- Reimb applied for new HIV drug 'Vocabria+Rekambys'
- by Eo, Yun-Ho Jul 24, 2024 05:50am
- Long-acting HIV treatment 'Vocabria+Rekambys' combination therapy aims to be listed for insurance reimbursement after receiving approval in South Korea two years ago. Industry sources said that GSK Korea and Janssen Korea have applied for reimbursement for the combination therapy of Vocabria (cabotegravir) and Rekambys (rilpivirine), which each company owns. GSK will be responsible for the overall reimbursement process. In February 2022, two drugs were approved by the Ministry of Food and Drug Safety (MFDS) as a combination therapy for the treatment of adult patients with HIV-1 infection who are virologically suppressed with no history of treatment failure and with no known or suspected drug tolerance to either cabotegravir or rilpivirine. In South Korea, Vocabria +Rekambys combination therapy was approved as injection therapy with intervals of monthly or bimonthly administration. The advantage of this combination therapy is its convenience. Previously, patients had to take a tablet formulation drug once daily for conventional HIV treatments. However, with the marketing approval of these two injectables, the treatment frequency has been reduced to monthly or bimonthly as an intramuscular injection, resulting in high patient satisfaction and reduced patient burden. These two drugs were initially developed as oral formulations and later developed as injectables. As a long-acting injection therapy, these drugs cannot cure HIV injection but target white blood cells, helping to lower and maintain AIDS virus replication. The combination therapy was approved in Europe in December 2020 after a clinical trial demonstrated its efficacy and safety in a patient group treated once every 4 weeks or once every 8 weeks. The most common adverse reactions observed in a group treated with Vocabria+Rekambys combination therapy include injection site reaction, headache, fever, nausea, fatigue, general malaise, and muscle aches. Consequently, it remains to be seen whether the government agency will approve the combination therapy of these two drugs for their advantage in convenience and whether the therapy will later become listed for reimbursement.
- Company
- Will 'Zejula' be reimbursed for HRD-positive ovarian cancer?
- by Eo, Yun-Ho Jul 23, 2024 05:48am
- Product photo of Takeda Pharmaceuticals Korea Whether the insurance reimbursement criteria for 'Zejula,' a PARP inhibitor, will include 'HRD-positive' is gaining attention. Sources said that Takeda Pharmaceuticals Korea is negotiating the price with the National Health Insurance Service (NHIS) for the Poly ADP-ribose Polymerase (PARP) inhibitor, Zejula, which is used to treat ovarian cancer. The drug price negotiation is intended to expand reimbursement for the drug. Zejula is currently reimbursed for use as maintenance therapy in patients with BRCA mutation ovarian cancer who have responded to first-line treatment with platinum-based chemotherapy. The company aims to add the condition of homologous recombination repair deficiency (HRD)-positive for Zejula during the negotiations. For the treatment of ovarian cancer, Zejula was the first PARP inhibitor to demonstrate efficacy by involving HRD-positive patients in clinical studies. However, the reimbursement was limited to BRCA mutation. Since then, the company has gathered significant data on Zejula in HRD-positive patients. In a phase 3 PRIMA follow-up study, predicted variables related to progression-free (PFS) in Zejula-treated patient group were analyzed. Based on the results, predicted variables for long-term PFS after the use of Zejula for first-line maintenance therapy were found to include having a BRCA mutation or HRD, FIGO staging, the primary site of tumor, and the number of non-target lesions with conditions. Patients treated with Zejula who have BRCA mutation or are HRD-positive are highly likely to maintain PFS for over two years. Patients who are tested positive for BRCA2 gene mutation or HRD had a higher odds ratio (OR) compared to other subgroup patients. In the PRIME study result, enrolling Asians (Chinese), a significant clinical treatment effect with Zejula for patients tested positive for BRCA mutation or HRD have been confirmed. In the study, Zejula-treated patients who tested positive for HRD had an ORR of 61.3%, higher than the 29.4% ORR in the placebo group. Jae Weon Kim, Professor in the Department of Obstetrics & Gynecology at Seoul National University Hospital, said, "For ovarian cancer patients who have remaining lesions, antitumor activation by PARP inhibitors is a crucial factor in delaying the progression of the disease and prolonging PFS in patients. The findings related to long-term benefits of Zejula, including improved prognosis, are encouraging."
- Company
- Opdivo approved as 1st-line Tx for urothelial cell carcinoma
- by Hwang, Byung-woo Jul 23, 2024 05:48am
- Pic of Opdivo Ono Pharma Korea and BMS Korea announced on the 22nd that Opdivo (nivolumab) has been additionally approved by the Ministry of Food and Drug Safety as a first-line treatment for urothelial cell carcinoma (UCC). The new indication is for the first-line treatment of unresectable or metastatic urothelial cell carcinoma in combination with cisplatin and gemcitabine. Patients with unresectable or metastatic urothelial cell carcinoma who have not received prior therapy are eligible to receive the combination treatment. The approval was based on the results of the Phase III CheckMate-901 study, which compared the use of Opdivo isplatin+gemcitabine combination followed by Opdivo monotherapy with the use of cisplatin+gemcitabine combination. In the study, the Opdivo combination arm demonstrated statistically significant and clinically meaningful improvements in the primary efficacy endpoints of overall survival (OS) and progression-free survival (PFS) as assessed by a blinded independent central review committee (BICR). The safety profile of Opdivo therapy in this study was consistent with the established safety profiles of the agents in previous trials and there was no new safety information. Opdivo was first approved in August 2017 as monotherapy for the treatment of patients with locally advanced or metastatic urothelial cell carcinoma whose disease has progressed after platinum-based chemotherapy. In February 2022, the drug was additionally approved as postoperative adjuvant therapy for patients with muscle-invasive bladder cancer (MIBC) at high risk of recurrence after radical resection.
- Company
- "Trodelvy gives new hope for triple-negative breast cancer"
- by Son, Hyung-Min Jul 23, 2024 05:48am
- Triple-negative breast cancer refers to a breast cancer that is negative for estrogen receptor (ER), progesterone receptor (PR), and human epidermal growth factor receptor 2 (HER2). It is a rare cancer that accounts for about 12-15% of all breast cancer. This cancer tends to be aggressive and has a high risk of metastasis and recurrence, leading to poor diagnosis. However, patients with this type of cancer have relied on cytotoxic anticancer medicines, the first-generation cancer treatment. They were unable to benefit from the latest cancer treatments, such as antihormone therapies and targeted cancer therapies, because all the receptors that could be targeted were negative. Trodelvy, a Trop-2 protein targeting antibody-drug conjugate (ADC), has opened a road for a new treatment for patients with metastatic triple-negative breast cancer. Trop-2 protein is a cell membrane antigen that is highly expressed in breast cancer and overexpressed in over 90% of triple-negative breast cancer. Trodelvy binds with Trop-2 protein and releases cytotoxic agents inside the cancer cells. It has the advantage of minimizing damage to healthy cells while maintaining the beneficial effects of targeted cancer therapies and cytotoxic anticancer medicines. In the multinational phase 3 clinical study, Trodelvy significantly improved overall survival (OS), progression-free survival (PFS), and objective response rate (ORR). In May, Trodelvy obtained approval from the Ministry of Food and Drug Safety (MFDS) and launched in South Korea. (From the left) Dr. Sohn, Joo Hyuk, Professor in the Department of Internal Medicine at Yonsei Cancer Hospital·Dr. Aditya Bardia, Professor in the Department of Medicine at UCLA Health Jonsson Comprehensive Cancer Center. Daily Pharm met with Dr. Aditya Bardia, the first author of 'ASCENT' clinical study and Director of Translational Research Integration and Professor in the Department of Medicine at UCLA Health Jonsson Comprehensive Cancer Center, and Dr. Sohn, Joo Hyuk, Professor in the Department of Internal Medicine at Yonsei Cancer Hospital and Korean Cancer Study Group (KCSG)'s Head of Breast Cancer Committee, to look at recent therapy trends for triple-negative breast cancer and clinical use of Trodelvy. Q. For a long time, there has been a high demand for new treatment options for triple-negative breast cancer. What is the reason for the difficulty in developing treatments? [Dr. Bardia] Triple-negative breast cancer shows the most aggressive characteristic among breast cancers, and it occurs in relatively young patients. Cytotoxic anticancer medicines are mainly used for the treatment because it is difficult to use targeted therapies due to potential therapeutic targets such as ER, PR, and HER are all negative. The problem is anticancer agents do not work efficiently in patients with metastatic cancer. Such difficulties contributed to high unmet needs for new drugs among breast cancers. [Dr. Sohn] Triple-negative breast cancer can be categorized as a diagnosis of exclusion. It refers to all breast cancer types that do not test positive for hormone receptor or HER2. Within the type, subtypes also exist. Therefore, the cancer does not exhibit consistent characteristics, posing difficulty in developing a generalized drug to be used for triple-negative breast cancer. The number of patients is relatively small, and a biologically verified target has not been identified; therefore, a treatment has not been developed. The disease itself shows aggressiveness, and patients tend to have poor diagnoses because anticancer chemotherapy that was developed decades ago is used. Q. What are the advantages Trodelvy for treating triple-negative breast cancer, where new drug development has been slow? Dr. Sohn, Joo Hyuk, Professor in the Department of Internal Medicine at Yonsei Cancer Hospital. [Dr. Bardia] Triple-negative breast cancer is negative for three receptors, ER, PR, and HER, but it does not entail that there are no receptors. Trodelvy is a treatment that targets Trop-2 protein, which is known to be overexpressed in over 90% of triple-negative breast cancer. Trodelvy can target cancer cells more efficiently than conventional standard therapies, showing superior effects. It has been designed to affect less on healthy cells, lowering toxicity and side effects that patients experience. [Dr. Sohn] Targeted therapies to date have used oncogenes that affect the progression and expansion of cancer as a target. However, Trop-2, which Trodelvy targets, is only a receptor expressed on the cell surface. Trodelvy is the first medicine to demonstrate the concept that a therapy targeting receptor expressed on cancer cell surface instead of an oncogene can be used to destroy cancer cells. In other words, it is meaningful that we have confirmed that identifying a receptor expressed on a cancer cell surface can lead to the development of a therapy. Q. According to the phase 3 ASCENT study, Trodelvy improved OS, PFS, and ORR statistically significantly compared to anticancer chemotherapies. What do these results indicate? [Dr. Bardia] Trodelvy had twofold longer OS than anticancer chemotherapy, extending patients' life expectancy. It also improved symptoms that affected patients' quality of life due to cancer, such as pain, thereby improving the quality of life during survival. The result gives patients hope to live a longer and better quality of life. [Dr. Sohn] The last drug to demonstrate the benefit of OS in triple-negative breast cancer was Halaven, released 10-20 years ago. New drugs, such as immunotherapy for cancer and PARP inhibitors, have been introduced. However, they are designed to target particular patient groups, such as PD-L1-positive patients and BRCA-mutation patients. Other than these, cytotoxic anticancer medicines are the only treatment that can be used in all patient groups with triple-negative breast cancer. Therefore, the recent data showing extended OS is historically meaningful. Patients are the only ones who can understand the differences in the number of months. Q. Has there been any change in clinical settings, including breast cancer treatment guidelines, since Trodelvy was introduced? Dr. Aditya Bardia, Professor in the Department of Medicine at UCLA Health Jonsson Comprehensive Cancer Center.[Dr. Bardia] The breast cancer guidelines of the National Comprehensive Cancer Network (NCCN) and the European Society for Medical Oncology (ESMO) foremost recommend Trodelvy as a second-line treatment for metastatic triple-negative breast cancer. In particular, Trodelvy has received a score of 5 on the ESMO-MCBS, a value-evaluation tool for anticancer medicines. Out of 37 approved treatments for metastatic breast cancer, there have been only two medicines that received a score of 5 (Trodelvy and HER+/HER2- medicine, Ribociclib). [Dr. Sohn] Previously, the effect of standard therapies for the treatment of triple-negative breast cancer was not good. When a first-line anticancer treatment had failed, the survival expectancy was only 7 months. It is a great hope to introduce a new treatment with verified survival effects to patients. Patients are welcoming treatments even if they are yet non-reimbursable. Until now, clinical settings for triple-negative breast cancer have been challenging, but this medication is likely to resolve a long-standing issue. Q. What resources are most needed to improve treatment settings for triple-negative breast cancer in South Korea? [Dr. Bardia] What we need most is to introduce the most effective new drugs regardless of the situation. We hope to enhance patients' survival duration and quality of life. I know that due to non-reimbursement, patients have limited access to Trodelvy in South Korea. I hope that National Health Insurance will be quickly applied and that access to Trodelvy in combination with other medicines will be granted. [Dr. Sohn] Patients who have private insurance or have financial well-being always choose Trodelvy. Considering the clinical data, it is a must. However, it is unfortunate that patients cannot use the medicine for economic reasons despite of the clinical evidence. I hope that healthcare financing will be allocated to those in need so that no patients are left without access to life-saving medicines. By reducing unnecessary medical spending and increasing resources for essential healthcare, South Korea can become a society that provides hope and easy access to new drugs for desperate patients. Q. What do you envision as the future approach to treating metastatic triple-negative breast cancer? [Dr. Bardia] I was once interested in AKT and PI3CA mutations and conducted two accounts of phase 3 clinical studies. However, I did not achieve fruitful outcomes. We guessed that because triple-negative breast cancer is further characterized into many subtypes, targeting AKT and PI3K3CA mutations was not effective for triple-negative breast cancer. Currently, treatments that target genetic mutations are limited to PARP inhibitors, which are offered as a first-line or second-line treatment to patients with BRCA mutations. [Dr. Sohn] Since recent studies to find new targets to treat triple-negative breast cancer are failing to demonstrate effectiveness, the remaining solution is likely to be the development of medicines targeting oncogene addiction. I heard that 350 ADCs are currently under development. New drug development is anticipated through studies about antibodies targeting cell surface receptors, cytotoxic agents, and linkers. I hope there will be more research related to combination therapies of ADCs and cytotoxic anticancer agents.