- LOGIN
- MemberShip
- 2026-04-08 03:35:27
- Company
- Lotte Biologics’ sales amount to KRW 228.6 billion
- by Kim, Jin-Gu Mar 22, 2024 06:09am
- Lotte Biologics. Lotte Biologics recorded sales of KRW 228.6 billion last year. As the Syracuse manufacturing plant in the United States went into full-scale production, sales of KRW 83.1 billion in the first half of the year expanded to KRW 145.6 billion in the second half of the year. The analysis indicates that Lotte Biologics’ Contract development and manufacturing organization (CDMO) business is on the right track. According to the Financial Supervisory Service on the 23rd, Lotte Biologics' sales for last year amounted to KRW 228.6 billion. The company, launched in May 2022, began generating significant revenue last year. Sales for the first quarter were KRW 20.7 billion, KRW 62.3 billion for the second quarter, KRW 89.8 billion for the third quarter, and KRW 55.8 billion for the fourth quarter. The net income for the year amounts to KRW 56.7 billion. In 2022, the company turned around from a loss of KRW 17.7 billion. Lotte Biologics' revenue is expected to derive most of its revenue from its CDMO business in the United States. In May 2022, Lotte Biologics signed a contract with BMS to acquire a biopharmaceutical production plant in Syracuse, U.S., for $160 million (approximately KRW 220 billion). This plant can produce raw materials for antibody medicines at an annual scale of 35,000 liters. The acquisition process was completed as of December 31, 2022. Starting from January 1, last year, the plant commenced full-scale production. Lotte Biologics inherited the existing CDMO contracts as part of acquiring the BMS plant. The company will commit to produce pharmaceuticals manufactured by BMS for the next three years after acquiring the plant. According to this contract, last year, the Syracuse plant reportedly produced BMS's immune checkpoint inhibitors 'Opdivo' and 'Yervoy,' immunosuppressants for kidney transplantation 'NeuroLex,' and multiple myeloma treatment 'Empliciti.' Lotte Biologics’ Syracuse manufacturing plant in the United States. The company began generating significant revenue last year. Securing new clients has remained a challenge for the company. Last year's revenue was mostly derived from inheriting BMS's CDMO contract; thus, there were almost no new orders. Considering the three-year contract period, it is analyzed that the company needs to secure new clients within the next two years to meet the target annual revenue of KRW 1.5 trillion by 2030. Lotte Biologics' focus on global events such as the BIO International Convention, CPhI Global event, and the JP Morgan Healthcare Conference aligns with this strategy. Lotte Biologics has announced its participation in BIO International Convention, which will be held in San Diego in June this year. For the third consecutive year since 2022, the company will operate a booth, emphasizing its distinctive capabilities by showcasing the company’s manufacturing technology, process development services, quality systems, and plans for establishing a domestic mega plant. During last year's conference, the company set up meeting tables and private meeting rooms within the booth, facilitating partnering with over 30 global pharmaceutical companies and small to medium-sized biotech firms interested in biopharmaceuticals CDMO. This year, they plan to continue focusing on securing CDMO contracts with global pharmaceutical companies through meetings. Lotte Biologics aims to achieve sales of KRW 1.5 trillion by 2030. To accomplish this goal, they are securing domestic production facilities, in addition to expanding CDMO business centered around the Syracuse plant in the United States. In June last year, they signed an MOU with the Incheon Metropolitan City and the Incheon Free Economic Zone Authority to construct a mega plant in Songdo, Incheon. By 2030, the company plans to build three mega plants, each with a capacity of 360,000 liters.
- Policy
- Economic benefits paid to doctors to be publicly disclosed
- by Lee, Jeong-Hwan Mar 22, 2024 06:08am
- On the 21st, the government announced the guidelines for the public disclosure of expenditure reports on the legitimate financial benefits paid by pharmaceutical and medical device companies to doctors and pharmacists that will be disclosed in December. The guidelines stipulate the detailed schedule, content, and method for disclosure of the economic benefit expenditure report prepared by companies as of the fiscal year 2023, per the amendments to the Pharmaceutical Affairs Act and the Medical Device Act, made on July 20, 2021. Under the current law, the legally permissible economic benefits that can be provided are the provision of physical products, support for conferences, support for clinical trials, product presentations, post-marketing surveys, cost discounts based on payment terms, and use for performance verification before purchase (medical devices only). When the expenditure report is disclosed, the public will be able to check the medical institution (name, symbol of the medical institution) that received economic benefits from drug and medical device suppliers, information on support for conferences, and the amount of support provided to participants at product presentations. However, under the Enforcement Rules of the Pharmaceutical Affairs Act and the Medical Device Act, the names of recipients such as medical practitioners whose personal information is at risk of exposure and clinical trial information containing confidential business strategies will be de-identified and disclosed in the expenditure report. This is in accordance with Article 44 (2) (3) of the Enforcement Rule of the Pharmaceutical Affairs Act, which stipulates, "If the Minister of Health and Welfare is notified that the details of the expenditure report contain information subject to non-disclosure under Article 9 of the Official Information Disclosure Act, it shall be disclosed after taking necessary measures to ensure that the contents cannot be identified. Kyung-sil Chung, Director Bureau of Healthcare Policy at the Ministry of Health and Welfare (MOHW), said, "The disclosure of the expenditure reports is to create an environment for the industry to self-censor rebates through the disclosure of legitimate economic benefits. We will be receiving related data through the Health Insurance Review and Assessment Service from June this year, and we ask for the companies’ cooperation to ensure accurate information disclosure." The operating guidelines for the expenditure report will be available on the websites of HIRA and other related associations.
- Company
- Antihistamine sales benefit from end of pandemic and endemic
- by Son, Hyung-Min Mar 22, 2024 06:06am
- Last year, sales of major antihistamines showed a clear recovery, exceeding the level it had made before the pandemic. Sales of antihistamines had plummeted in 2020 and 2021 during the COVID-19 pandemic. The analysis is that the increase in sales is likely due to the rise in allergy patients with the lifted mask mandates and the increase in outdoor activity that followed. According to the market research institution IQIVA on the 21st, sales of the antihistamine Xyzal increased 18% YoY to reach KRW 14.4 billion the past year. Xyzal is a third-generation antihistamine developed by UCB Korea and is the leader in the antihistamine market. Antihistamine drugs block histamine, which is involved in allergic reactions such as hives, redness, and itching. In addition to allergic conditions, they are also used to relieve rhinitis, asthma, and nasal congestion. Because antihistamines work on the central nervous system (CNS), they can cause side effects such as drowsiness and sedation. However, third-generation antihistamines are known to cross the blood-brain barrier (BBB) less frequently than first- and second-generation medications, making them less likely to cause drowsiness. Xyzal’s sales decreased by 17.6% to KRW 9.3 billion in 2020 from KRW 11.3 billion in 2019, but increased again to KRW 9.9 billion in 2020 and then to KRW 12.2 billion in 2022. The industry believes sales of antihistamines will continue to grow along with the increase in the number of allergic rhinitis patients in Korea. According to the Korea Disease Control and Prevention Agency, the rate of physicians’ diagnosis of allergic rhinitis increased from 16.8% in 2012 to 21.2% in 2022. The NHIS data also showed that the number of patients diagnosed with allergic rhinitis increased to 6.01 million in 2022 from less than 5 million in 2021 during the COVID-19 pandemic. Other third-generation antihistamines also saw a resurgence in sales with the end of the pandemic. Handok's Allegra generated sales of KRW 8.1 billion last year, up 10.9% YoY. Allegra is a fexofenadine-based antihistamine developed by Sanofi, and Handok holds domestic rights for the drug in Korea. Allegra’s sales were sluggish in 2020 and 2021, KRW 4.8 billion and KRW 5 billion, respectively, but recovered to KRW 7.3 billion in 2022 and surpassed KRW 8 billion last year. Fexonadine, a same-ingredient drug from Hanmi Pharmaceutical, also saw a 21% increase in sales YoY. Sales of Organon Korea’s Aerius, a desloratadine-based third-generation antihistamine, rose 44.4%. The rebound in sales has led to a series of launches of third-generation antihistamines by domestic drugmakers as well. Samjin Pharm and GC Biopharma also launched their fexofenadine-based allergy treatments this month. first-and-second generation antihistamines also show good sales...Sales of Clarityne sluggish In addition to third-generation antihistamines, sales of first- and second-generation antihistamines also grew in general. UCB Korea’s Zyrtec, a second-generation antihistamine, generated sales of KRW 14.3 billion last year, up 14.4% from KRW 12.5 billion in 2022. Zyrtec has been sold exclusively in Korea by the local drug distributor BeoYoung since last year. In Korea, Zyrtec sold 2.628 million tablets last year. Sales of UCB Korea’s Ucerax, a first-generation antihistamine, reached KRW 1.4 billion, up 55.5% YoY. The company had previously considered withdrawing the marketing authorization of its Ucerax due to insufficient supply, but the company succeeded in resuming supply in 2019 with Yuhan Corp taking over domestic distribution. Sales of Hansol Pharm’s Zeromine soft capsules grew 58.3% compared to 2022. On the other hand, sales of Bayer’s Clarityne and Yungin Pharmaceutical’s Plakon dropped sharply. Clarityne posted sales of KRW 40 million last year, down 60% from the KRW 1 billion in 2022. Clarityne is a second-generation antihistamine developed by Merck, and Bayer acquired its marketing rights in 2014 when it acquired Merck's OTC division. Clarityne's sales fell from KRW 1.1 billion in 2019 to KRW 900 million in 2021, and last year, sales fell to less than KRW 100 million. Bayer Korea explained, "The product has been out of stock since Q1 last year due to a shortage of supply that was unable to meet the high demand, which led to the decline in sales.” Plakon’s sales fell 70% YoY and reached KRW 90 million last year. Before COVID-19, Plakon ‘s sales were in the KRW 2 billion range, but sales have continued to decline. Yungin Pharmaceutical withdrew the product from the Korean market by withdrawing its marketing authorization for the drug. Plakon, which was approved in Korea in 1962, is a first-generation antihistamine containing piprinhydrinate and was subject to clinical reevaluations in 2021. Yungin Pharmaceutical did not submit the reevaluation data and received administrative dispositions - a two-month sales suspension in December of the same year. The company voluntarily withdrew the product in February 2022. Accordingly, Plakon was also removed from the drug shortage prevention list in April 2022.
- Company
- Luxturna prescriptions available for rare retinal disease
- by Eo, Yun-Ho Mar 22, 2024 06:06am
- Novartis Korea’s Luxturna (voretigene neparvovec), a therapy used to treat inherited retinal dystrophy (IRD). Prescriptions for the gene therapy ‘Luxturna,’ which targets only a few patients, are becoming available in South Korea. According to industry sources, Novartis Korea’s Luxturna (voretigene neparvovec), a therapy used to treat inherited retinal dystrophy (IRD), has cleared the Drug Committee (DC) of the tertiary general hospitals, including Samsung Medical Center in Seoul, Seoul National University Hospital, and Severance Hospital. Luxturna was first administered at Samsung Medical Center in Seoul among these hospitals. Luxturna, listed for reimbursement last February, restores function by substituting the RPE65 gene mutation, which is one of the causes of IRD, with a normal gene through a single administration. Essentially, it is a disease-modifying treatment. Since Luxturna is a gene therapy, the number of hospitals prescribing it may not increase significantly. Hospitals that can prescribe Luxturna should screen patients to ensure they receive benefits, given the limited number of patients. Phase 3 clinical trials, including patients with confirmed biallelic RPE65 mutations-associated IRD, demonstrated the efficacy of Luxturna. The clinical results revealed that the patients treated with Luxturna had significantly improved functional vision after one year compared to the comparator group. The 'Multi-Luminance Mobility Test (MLMT) average score, which evaluates the ability to pass various obstacles of different heights in everyday walking environments, was used as the primary endpoint at the one-year treatment assessment. The Luxturna treatment group's score increased by 1.8 points, 1.6 points higher than the control group's 0.2-point increase. Initially, Luxturna applied for reimbursement in September 2021, but no progress was made. In March last year, it was considered for review by the Drug Reimbursement Evaluation Committee (DREC) of the Health Insurance Review and Assessment Service (HIRA) but failed to establish a reimbursement criterion. After improving its documents, Luxturna completed negotiations for drug pricing in December and was listed for reimbursement in February.
- Policy
- Enhertu’s price falls to KRW 70 mil range with reimb
- by Lee, Tak-Sun Mar 22, 2024 06:06am
- The list price of the petitioned anti-cancer drug Enhertu was reportedly set at KRW 1.4 million per vial. This is significantly cheaper than the non-reimbursed price of KRW 2.3 million. In addition, the National Health Insurance Service (NHIS) will apply a risk-sharing agreement (RSA) system for the reimbursement of the drug, so the actual price of the drug is expected to be lower. According to industry sources on the 21st, Enhertu’s price is going to be set at an upper limit of about KRW 1.4 million per vial. The price had been reportedly applied to the drug when it passed HIRA’s Drug Reimbursement Evaluation Committee review in February. In other words, the NHIS seems to have been negotiating around the price set by DREC. This may explain why the negotiations began in late February and were completed in just a month. Considering that an average weight patient requires 3 vials per treatment cycle (21 days), the drug’s non-reimbursed price is about KRW 7 million, and the listed reimbursed price is KRW 4.2 million. In terms of annual treatment costs, the non-reimbursed price is about KRW 120 million and the list price is about KRW 71.4 million. The actual price is likely to be lower with the application of the RSA system. The expenditure cap type and refund type will be applied to Enhertu. Enhertu’s reimbursement listing will be officially notified after being reported to the Health Insurance Policy Deliberation Committee's meeting scheduled for next week. Enhertu is an antibody-drug conjugate (ADC) anticancer drug that is highly effective against HER2-positive breast cancer, which accounts for around half of all metastatic breast cancers. It is also used for HER2-positive gastric or gastroesophageal junction (GEJ) adenocarcinoma. In particular, the drug demonstrated a better effect over existing drugs in clinical trials. Compared to chemotherapy, Enhertu reduced the risk of disease progression or death by 50%. The overall survival (OS) was 23.4 months with Enhertu, 6.6 months longer than the 16.8 months of the control group. The superior effect prompted the call for fast-track registration of Enhertu in the medical field. The petition was signed by 50,000 people and reported to the National Assembly.
- Policy
- National Essential Medicines to be categorized into 4 groups
- by Lee, Hye-Kyung Mar 21, 2024 05:53am
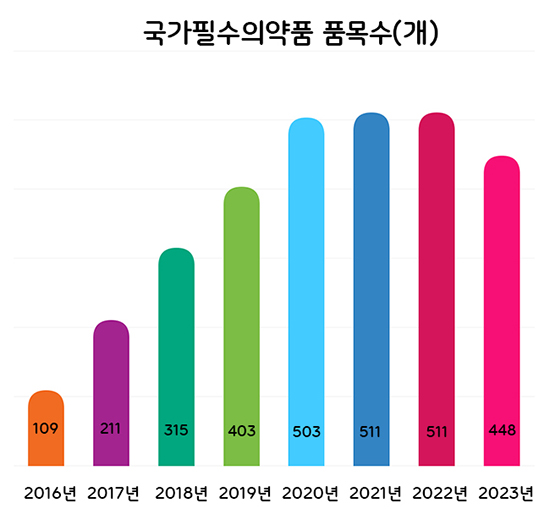
- The Ministry of Food and Drug Safety (MFDS) plans to categorize national essential drugs into 4 groups and conduct regular level assessments to better manage the drugs. According to the 'Research Service to Prepare a Measure to Classify and Manage the National Essential Medicine ‘ that was recently announced by MFDS, the authorities will discuss improvements and revisions to the classification criteria used to set the supply stability grade of national essential drugs by May. The four-level classification plan was made in response to the need to establish a differentiated management system for already designated national essential drugs, which was pointed out in the 2nd Comprehensive Plan for Stable Supply of National Essential Drugs in 2022. Through the project last year, the MFDS prepared four-level classification criteria for national essential drugs: ▲drugs that require prior stockpiling, ▲drugs that require immediate supply in case of a crisis, ▲ drugs that require constant monitoring of the supply situation and promotion of localization, and ▲drugs that require continuous attention. The classifications are based on the impact on public health, urgency of use, and potential availability of supply. Number of National Essential Medicines by Year Based on the research service results, the MFDS will finalize the classification and management methods of already designated national essential medicines and prepare feasibility studies and designations for ingredients and formulations that request new designations. For already designated national essential drugs, the MFDS plans to apply the 4-level classification system and conduct regular level management. For this, the research will identify and revise improvements to the classification system. The proposed list of ingredients and formulations that are applied to the standards and improvements prepared through the research will be reviewed by the National Essential Medicine Subcommittee. Ingredients and formulations requesting new designations this year will be reviewed for healthcare necessity and supply stability based on the evaluation criteria that had been determined through the Council for the Safe Supply of National Essential Medicines that was in November last year. The MFDS explained, "We need to continue to designate drugs that are essential for healthcare but are difficult to be supplied stably through market functions alone as national essential medicines and update the list of already designated national essential drugs. The final ingredients and formulations will be designated after resolution by the Council for the Safe Supply of National Essential Medicines Meanwhile, the number of national essential medicines has steadily increased from 109 in 2016 to 511 in 2022, and a total of 448 ingredients and formulations have been designated through new and de-designations last year.
- Company
- "Envlo shows superior effects in diabetic kidney disease"
- by Son, Hyung-Min Mar 21, 2024 05:52am
- Daewoong Pharmaceutical Daewoong Pharmaceutical announced on the 19th that Envlo (ingredient: enavogliflozin), an SGLT-2 class diabetes treatment from the company’s drug discovery, demonstrated superior effects in patients with diabetes compared to dapagliflozin. Based on the clinical results, Envlo has shown superior effects in all four indexes, including ▲glycosylated hemoglobin, ▲fasting glucose, ▲urine glucose-to-creatinine ratio (UGCR), and ▲homeostasis model assessment-insulin resistance (HOMA-IR), compared to dapagliflozin, another SGLT-2 inhibitor. The clinical study evaluated changes in glycosylated hemoglobin and fasting plasma glucose in 470 patients with type 2 diabetes. Based on their kidney functions, the patients were assigned to two groups and were given either Envlo or dapagliflozin for 24 weeks. The study enrolled patients with type 2 diabetes patients who had inadequate glycemic control despite undergoing a combination of metformin and another antihyperglycemic agent or a combination therapy of metformin and gemigliptin (DPP-4 inhibitor). The clinical results demonstrated that Envlo reduced fasting plasma glucose more significantly than dapagliflozin. The Envlo group had fasting plasma glucose of 26.65 mg/dl at week 6 and 28.54 mg/dl at week 24, while the dapagliflozin group had fasting plasma glucose of only 21.54 mg/dl at week 6 and 23.52 mg/dl at week 24. Envlo, a new Korea-made drug, has a superior effect in reducing ‘glucose’ than a global pharmaceutical drug…The results from the study of diabetic kidney disease patients. A significant reduction in glycosylated hemoglobin in the Envlo group was observed, with a decrease of 0.76% at week 6 and 0.94% at week 24. This indicates a nearly 1% reduction in glycosylated hemoglobin over 24 weeks. Dapagliflozin resulted in a reduction of 0.66% at week 6 and 0.77% by 24 weeks. Among the clinical result indexes, the ‘urine glucose-to-creatinine ratio’ (UGCR) particularly stands out. The UGCR measures the ‘glucose’ secretion in urine. The patients administered with Envlo had UGCR of approximately 55g/g from week 6 to week 24. This result is similar to the level seen in diabetic patients with normal kidney function. Daewoong Pharmaceutical anticipates that the new Korea-made drug Envlo will provide an outstanding alternative for diabetic patients with kidney disorders. Currently, patients who have been taking Forxiga are facing finding a new drug because the global pharmaceutical company announced the discontinuation of Forxiga. “It is of particular interest that we provide clinical evidence when diabetic patients require a new drug prescription,” Daewoong Pharmaceutical’s CEO Chang-jae Lee said. “As the clinical result was published in a globally recognized SCIE research paper, we plan to obtain additional evidence to prove Envlo as an alternative solution,” Lee added. The study was published in the SCIE-listed international research paper, ’Cardiovascular Diabetology.’ It was titled ‘Efficacy and safety of enavogliflozin vs. dapagliflozin as add-on therapy in patients with type 2 diabetes mellitus based on renal function: a pooled analysis of two randomized controlled trials.’
- Company
- Haleon Korea declares full independence from GSK
- by Eo, Yun-Ho Mar 21, 2024 05:52am
- The consumer healthcare company Haleon has declared its full independence from GSK. Haleon Korea, the Korean subsidiary of Haleon, announced on the 20th that it has completed the official registration of its business and is embarking on a new journey. The company had been completely spun off from GSK in July 2022 to become a 100% consumer healthcare specialty company. In Korea, the company has been preparing for the change for the past year and a half, including changing the packaging of its products, and has finally finalized the process and has officially launched as Haleon Korea in March 2024. Haleon Korea has been selling 12 leading brands in Korea, including ▲Centrum (multivitamin brand), ▲Sensodyne (toothpaste for sensitive teeth), ▲Parodontax (toothpaste for gum health), ▲Polident (denture cleaning and adhesive brand), ▲Teraflu (cold relief brand), ▲Otrivine (nasal congestion relief brand), and ▲Driclor (hyperhidrosis management brand). Dong-Woo Shin, CEO of Haleon Korea, said, “We will continue to introduce scientifically verified products every year to empower consumers' self-care capabilities and race for the development of the Korean consumer healthcare market.” Haleon was launched in 2015 and 2020 as the combined consumer healthcare business of Novartis and Pfizer, and then GSK, respectively. The name Haleon is a combination of health and strength, inspired by the old English word "Hale," meaning health, and the Greek word "Leon," which is associated with strength.
- Company
- Fintepla gets orphan drug designation for Dravet syndrome
- by Eo, Yun-Ho Mar 21, 2024 05:52am
- UCB 'Fintepla' has been designated as an orphan drug in South Korea for its indication for Dravet syndrome. The Ministry of Food and Drug Safety (MFDS) has announced this in the posting of the Orphan Drug Designation. UCB’s Fintepla (fenfluramine) initially acquired FDA approval for the treatment of rare epilepsy of infancy in 2020. Additionally, in 2022, it added an indication for Lennox-Gastaut syndrome (LGS). Fenfluramine, banned in the United States in 1997, suppresses appetite by increasing serotonin content in the brain. It was discontinued because overuse of the drug led to the stimulation of the central nervous system, damaged heart valves, schizophrenic symptoms, and even death. Fenfluramine is currently prescribed as a low-dose and liquid form for oral administration. Randomized, double-blind, placebo-and comparator-controlled Phase 3 clinical trial, including children and adolescents aged 2 to 18, demonstrated efficacy of fenfluramine. The primary endpoint was a comparison of the monthly convulsive seizure frequency (MCSF). MCSF comparison results show that the frequency of convulsive seizure for a month in the fenfluramine group reduced by 54.0% compared to the placebo. In the fenfluramine group, 54% had a clinically significant reduction in MCSF, compared to 5% in the placebo group. The fenfluramine group had a longer median seizure-free interval than the placebo group (22 days vs. 13 days). The percentage of patients who were seizure-free after an episode was 12% in the fenfluramine group and 0% in the placebo group. Patients treated with fenfluramine had a significant reduction in convulsive seizures compared to the patients treated with a placebo. Notably, the fenfluramine group had a reduction in seizures within three to four weeks and maintained treatment effects over 14 to 15 weeks. Dravet syndrome is a rare disease characterized by tonic-clonic seizures or myoclonic seizures, which cause muscles in the body to contract and relax, during infancy. It has been reported that SCN1A gene mutations are a common cause of the disease.
- Policy
- Gov’t raises price of Harmonilan due to unstable supply
- by Lee, Tak-Sun Mar 21, 2024 05:52am
- Enteral nutrition products Harmonilan(left) and Encover(right) The insurance ceiling price of the enteral nutrition formula Harmonilan (B Braun Korea), which the company has been experiencing difficulties in ensuring supply and demand recently, is expected to rise from April. The company had continuously been applying to register the drug as a ‘drug shortage prevention drug' and preserve production costs due to its low profitability, to no avail. This is why criticism arose on how the government has deferred taking measures until now and only set out to adjust the price ceiling after the supply and demand instability intensified. Harmonilan’s actual transaction price had been cut 2 years ago According to industry sources on the 20th, the company has completed negotiations to raise the ceiling price of Harmonilan with the government, and the new drug price is expected to be applied next month after being reported to the Health Insurance Policy Deliberation Committee. Encover and Harmonilan are the two products that dominate the enteral nutrition market. Enteral nutrition products are given to patients who have difficulty getting nourished through food. The product is directly into the digestive tract through a tube. They are essential for patients with eating disorders. However, enteral nutrients such as Harmonilan and Encover are often in short supply depending on import situations, which has caused a lot of frustration among patients. This is why healthcare providers and patients have long requested such enteral nutrition products be designated as a drug shortage prevention drug, but health authorities have been unwilling to do so. Pharmaceutical companies have also requested the designation. Because they are low-margin products, the companies explained that they would need to raise the price of the drugs to conserve their cost and keep up with the high demand. However, the price of these enteral nutrition products has fallen in the past due to actual transaction price cuts and the Price-Volume Agreement system. The price of Harmonilan 200ml was lowered from KRW 2,291 to KRW 2,282 in January 2022 as part of actual transaction price cuts. In the case of Encover, the price of the 200ml product had fallen 2.9% in October 2022 under the PVA system. The company had applied for price adjustments at the time, but the price had not been increased then. Such price reductions are regarded as a major cause of supply instability, as they decrease profitability. Therefore, the price increase granted for Harmonilan this time is regarded as one example of the government's inconsistent drug price adjustment policy. Drug that require price increases are also subject to price cuts The government has been postponing the implementation of the actual transaction price cut plan, which had been scheduled to take effect in January. This in part is interpreted as the government’s effort to to prevent cases like Harmonilan, as drugs with unstable supply and demand may be subject to price reductions. According to government officials, it is likely that the price reduction will not be implemented in April either. The postponement has also halved the health insurance financial savings the government sought to earn through the drug price reduction. The government seems to be in a dilemma, caught between its recent move to broadly raise drug prices of drugs with unstable supply and measures previously prepared to cut the prices of existing drugs. The industry is calling for a complementary measure to prevent price cuts not only for existing drug shortage prevention drugs but also for drugs that are likely to have unstable supply and have difficulty continuously treating patients. An industry official said, "The list of non-eligible items should be expanded after close examination when selecting subjects for actual transaction price cuts and PVA price cuts reductions. We need to get rid of the absurd situation where a drug subject to a price reduction suddenly becomes a drug subject to a price increase.”