- LOGIN
- MemberShip
- 2026-04-08 03:35:27
- Policy
- MSD withdraws SGLT2 inhibitors in a row
- by Lee, Hye-Kyung Mar 20, 2024 05:44am
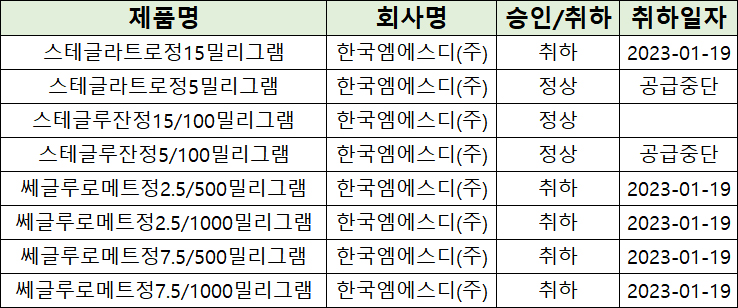
- MSD Korea is facing tough competition in the market for SGLT-2 inhibitors in South Korea. MSD decided that they will discontinue the supply of Steglatro Tab 5 mg and 'Stegluzan Tab 5/100 mg (ertugliflozin plus sitagliptin)' after voluntarily withdrawing 'Steglatro Tab 15 mg (ertugliflozin)' and metformin combination therapy, 'Segluromet,' last year. The list of approved and withdrawn SGLT-2 inhibitors by MSD in South Korea. Except for Stegluzan Tab 15/100 mg, all of MSD According to the Ministry of Food and Drug Safety (MFDS) report on the 15th, supply discontinuation was reported for Steglatro Tab 5 mg and Stegluzan Tab 5/100 mg. Imports of Steglatro Tab 5 mg will end on May 31st and Stegluzan Tab 5/100 mg will end on August 30th, after which these drugs will be discontinued. MSD says, “Due to a reduction in market demand, we will discontinue these drugs after the final import.” And MSD also stated, “The last import date is flexible depending on the manufacturing schedule and planned between mid-May and mid-August.” MSD stated that substitution is possible since other SGLT-class products are already available in the market. They also mentioned that they will notify healthcare professionals before discontinuing sales to ensure appropriate measures can be taken. If MSD voluntarily withdraws two items that they have decided to discontinue importing, Stegluzan Tab 15/100 mg will be their only SGLT-2 inhibitor containing ertugliflozin. However, Stegluzan Tab 15/100 mg has no distribution record after approval, and the company does not intend to distribute. The market for SGLT-2 inhibitors has grown to KRW 170 billion. According to the pharmaceutical market research agency UBIST, Steglatro generated merely KRW 1.6 billion in outpatient prescriptions. Industry analysis suggests that Steglatro lacks a competitive edge compared to Forxiga and Jardiance because its indication is limited to type 2 diabetes and clinical trials are lacking in expanding treatment areas. Changes in prescription performance by SGLT-2 inhibitors. SGLT-2 inhibitors have emerged as treatments for type 2 diabetes and are broadening their use in various diseases targeting the heart and kidney. SGLT-2 inhibitors inhibit the reabsorption of glucose in the kidneys, leading to glucose excretion in the urine. This results in reduced blood sugar, weight loss, kidney function protection, and blood pressure reduction. As of 2022, the items have recorded over KRW 40 billion in outpatient prescriptions individually, including AstraZeneca’s monotherapy 'Forxiga (ingredient: dapagliflozin),' a combination therapy ‘Xigduo,’ and Boehringer Ingelheim's monotherapy Jardiance (ingredient: empagliflozin). AstraZeneca’s drug generated KRW 91.4 billion, approximately KRW 15 billion more than Boehringer Ingelheim’s KRW 76.1 billion.
- Policy
- President Yoon stands on expanding quota to 2,000
- by Lee, Jeong-Hwan Mar 20, 2024 05:44am
- President Yoon Suk Yeol. President Yoon Suk Yeol reaffirmed the government’s plan to increase the medical school enrollment quota to 2,000 and stated, “Korea’s policy related to the number of doctors is not meeting the standards of the current era and actual needs, repeating the history of failure. Doctors’ licenses should not be used as a tool to intimidate Korean citizens and raise anxiety.” President Yoon presided over a Cabinet meeting at the presidential office in Yongsan and stressed, “The medical reform we are facing is our duty for the benefit of citizens, and it is a request from citizens.” And adding, “Korea’s policy related to the number of doctors is not meeting the standards of the current era and actual needs, repeating the history of failure.” “Delaying the expansion will eventually result in greater harm to the Korean citizens,” Yoon said. “Moreover, a much larger expansion will be needed in the future, and social controversies and conflicts surrounding medical reforms will also intensify every year. If citizens must constantly plead with their doctors for healthcare, can we truly say that the country is functioning properly?” In particular, “A stepwise approach or delaying expansion will not save the lives of the citizens nor accomplish medical reform to stop the collapse of regional and essential healthcare,” Yoon explained. “I hope trainee doctors and doctor organizations participate in a discussion about detailed measures to implement medical reform instead of engaging in fights outside of hospitals,” Yoon said. “I have been leading government-public discussions and will continue to hold sessions about medical reform in the form of government-public discussions.” Finally, Yoon mentioned visiting Asan Medical Center in Seoul to meet with hospital officials, pediatric obstetricians, and nurses. “I hope the medical staff who left will return to patients and join these staff,” Yoon said. “I will make a promise as a president to talk face-to-face with citizens about medical reform.”
- Policy
- Essential medical devices will be designated by next year
- by Lee, Hye-Kyung Mar 20, 2024 05:44am
- The Ministry of Food and Drug Safety is considering establishing a system to designate national essential medical devices. More specifically, the government plans to set a specific scope of medical devices that are essential for healthcare, such as rapid antigen test kits and life support devices that were necessary during COVID-19 but were difficult to supply stably through the market’s function alone. Nam-hee Lee, Director-General of Medical Device Safety at MFDS At a briefing with the MFDS’s correspondent journalists briefing, Nam Hee Lee, Director-General of the Pharmaceutical Safety Bureau at MFDS, said, "The need to designate essential medical devices has emerged as with pharmaceuticals amid the COVID-19 crisis. However, no specific scope has been set for the medical devices, and it is necessary to discuss how to designate local or existing licensed medical devices, so we will need to conduct a study first." For pharmaceuticals, the Pharmaceutical Affairs Act defines national essential medicines, and the government has designated and is managing 448 items as national essential medicines as of 2024. "There are frameworks set under the law to manage rare medical devices and medical devices subject to supply interruption reports, but the COVID-19 crisis has revealed global supply chain issues and that we rely on overseas import for many medical devices. In part, we need to localize medical devices, and there are medical devices we have the technology for but are not produced due to profitably issues, so we need to study this." Various areas such as infectious disease treatment and life support devices are emerging as candidates for essential medical devices, and Lee said, "The concept of essential medical devices is expected to be similar to that of the essential medicines. However, since there is no legal definition, we will conduct a research service this year and hope to see visible results by next year.” Another rising issue for MFDS this year is the ‘Digital Medical Products Act,’ which was enacted in January. The Digital Medical Products Act covers not only digital medical devices with digital technologies such as digital sensors and mobile applications, but also digital medical and health support devices that apply digital technologies for medical support and health maintenance and improvement, and digital convergence medicines that combine these devices and medicines. Under the act, a regulatory system for clinical trials, approval, and post-approval management has been established to efficiently and systematically evaluate the safety and efficacy of digital medical products based on digital characteristics such as the use of artificial intelligence and network connectivity. (From the left) Sang Hyun Kim, Director of the Medical Device Management Division, Nam-Hee Lee, Director-General of the Pharmaceutical Safety Bureau, Hong-Mo Sung, Director of the Medical Device Policy Division, Seung-Young Lee, Director of the Innovative and Diagnostic Medical Devices Policy Division Director-General Lee said, “We will prepare sub-statues so that digital medical products can be quickly commercialized. It will be beneficial to the industry if our law can be used to promptly provide devices to the public and will position Korea as a global leader." Last year, the digital therapy devices Somzz’ and Welt's ‘WELT-I’ were designated as the first and second digital therapy devices. However, it took more than 10 months from the approval to actual prescriptions, and it will take time for their reimbursement as the clinical outcome data has not been accumulated in hospitals. Director-General Lee said, “The Ministry of Health and Welfare is thinking about various ways to improve the system, and has first decided to designate products as innovative medical devices to enable integrated review. However, we are devising ways to enter the devices into the insurance system in some form, because reimbursement is possible only after the evaluation results of the product." However, when the next digital therapeutic devices, such as the third and fourth devices, are approved by the end of the year, Lee expected the innovative medical device designations to shorten the time to their prescriptions in the clinic first on a non-reimbursed basis. If it is designated as an innovative medical device, it will be subject to an expedited approval review, which currently takes more than 600 days on average. So I believe the clinical prescription period of the No. 3 product will be shortened further."
- Company
- SK Life Science’s sales rise tenfold in 5 years in the US
- by Chon, Seung-Hyun Mar 20, 2024 05:44am
- SK Biopharmaceuticals' U.S.-based subsidiary posted sales of nearly KRW 500 billion last year. The sales of its epilepsy drug cenobamate increased in the U.S., increasing the company’s sales by over tenfold in 5 years. According to the Financial Supervisory Service on Tuesday, SK Life Science’s sales totaled KRW 49.9 billion last year, up 26.5% from the previous year. SK Life Science is a wholly-owned subsidiary of SK Biopharmaceuticals. It is in charge of the U.S. sales of cenobamate, a new drug developed by SK Biopharmaceuticals. Annual sales of SK Life Science (Unit: KRW 100 million, Data: FSS) SK Life Science’s sales have been showing strong growth, up 83.7% in 2 years since posting KRW 267.2 billion in 2021. Its sales have expanded more than tenfold in five years from a mere KRW 47.5 billion it generated in sales in 2018. This is in part due to the surge in sales of its new epilepsy drug cenobamate in the United States. Xcopri, which contains cenobamate, is an epilepsy drug independently developed by SK Biopharmaceuticals and sold in the United States. It is prescribed for the treatment of partial-onset seizures in adults in the U.S. Its mechanism of action relieves seizure symptoms by simultaneously regulating two targets related to excitatory and inhibitory signaling, which are causes of epilepsy. SK Biopharmaceuticals had independently performed the whole course of Xcopri’s development and commercialization from early development to U.S. Food and Drug Administration (FDA) approval. The company received approval for its cenobamate under the brand name ‘Xcopri’ in November 2019, and has been selling the drug through SK Life Science since May 2020. Last year, cenobamate’s U.S. sales reached KRW 270.8 billion, up 60.1% YoY. In 2021, cenobamate posted sales of KRW 78.2 billion, more than tripling in 2 years. SK Life Science has about 150 medical representatives. Epilepsy is a condition that is treated by a small number of focus specialists in the United States. Due to the small number of target physicians, the company was able to sell the drug directly in the U.S. with such a small sales force. SK Life Science’s marketing center, which consists of sales and marketing personnel, has experts with more than 20 years of experience in successfully launching and selling epilepsy treatments and major central nervous system drugs at Johnson & Johnson, UCB, and other leading companies in the U.S. CNS market. Quarterly sales of Xcopri in the US (Unit: KRW 100 million, Data: FSS) With the rise in cenobamate’s sales, SK Life Science’s parent company has also increased its operating expense support. Last year, SK Life Science's revenue included KRW 220 billion in operating expenses paid by its parent company SK Biopharmaceuticals. SK Biopharmaceuticals earned revenue by supplying SK Life Science with cenobamate. The company pays SK Life Science a certain amount of operating expenses each year, which are reflected in SK Life Science's revenue, according to the company. The company said, "The number of prescriptions in December last year, the 44th month of cenobamate’s launch, was about 26,000, which is about 2.2 times the number of prescriptions made in the 44th month by its competitors. We will work to accelerate growth by increasing the number of monthly prescriptions to over 30,000 this year.”
- Company
- Sales of Tagrisso stay strong, Leclaza rises in lung cancer
- by Mar 20, 2024 05:44am
- In the EGFR-positive non-small cell lung cancer (NSCLC) treatment market, Leclaza’s sales growth stood out amid strong sales of Tagrisso. Tagrisso and Leclaza are both third-generation targeted therapies, and the two were approved as a first-line treatment for lung cancer this year and were given the green light to expand sales. Among first- and second-generation targeted therapies, all drugs other than Giotrif posted sluggish sales last year. According to the market research institution IQVIA on the 20th, Tagrisso generated sales of KRW 111 billion last year, up 4.2% YoY. Tagrisso is a third-generation tyrosine kinase inhibitor (TKI) that was developed by AstraZeneca. EGFR-positive lung cancer treatments are categorized into four types: first-generation EGFR TKIs like AstraZeneca's Iressa (gefitinib) and Roche's Tarceva (erlotinib); second-generation EGFR TKIs like Boheringer Ingelheim's Giotrif and Pfizer's Vizimpro (dacomitinib); and third-generation EGFR TKIs like Leclaza (lasertinib) and AstraZeneca's Tagrisso (osimertinib). Sales of Tagrisso, which exceeded the KRW 100 billion mark in sales in 2020, have remained stagnant for 2 years. After reaching KRW 106.5 billion in 2020, the drug posted similar sales in 2021 and 2022. After working to receive reimbursement for the drug as a first-line treatment for years, AstraZeneca received the reimbursement approval this year as a first-line treatment for EGFR-positive lung cancer, and this is also expected to expand sales. Moreover, Tagrisso is also the only third-generation TKI to be approved for use in early-stage lung cancer. In December last year, Tagrisso was approved as an adjuvant therapy for patients with EGFR exon 19 and exon 21 mutated NSCLC after complete resection. In the phase III ADAURA study, Tagrisso was shown to reduce the risk of death by 51% compared to conventional therapy. Powered by the advantages, Tagrisso’s sales are expected to increase further in the coming years. Leclaza posted sales of KRW 22.6 billion last year, up 40.3% YoY. Leclaza, which was approved in Korea in January 2021, was also granted reimbursement the same year. After posting KRW 4.1 billion in sales in Q2, it posted sales of KRW 16 billion the following year, then surpassed KRW 20 billion last year. Leclaza’s approval as a first-line treatment for EGFR-positive NSCLC in July last year is analyzed to have contributed to the rise in sales. Before the approval, patients had to be confirmed to be T790M-positive through an additional tissue test after using a first- or second-generation EGFR TKI to be eligible to use Leclaza with reimbursement. With all 3 TKIs, including Leclaza and Tagrisso, now covered as first-line treatments, physicians and patients will have a full range of first- and third-generation targeted therapies to choose from. Another benefit of Leclaza is its potential for use in combination with Rybrevant. Combination therapies, including Tagrisso plus platinum-based chemotherapy and Rybrevant plus platinum-based chemotherapy, are currently approved by regulatory authorities abroad for the first-line treatment of lung cancer. Combining the use of Leclaza, which targets EGFR-mutated exon 19 and exon 21 deletion, with Rybrevant, which targets exon 20 deletion, has been attracting attention as a viable targeted therapy combination. Currently, the Leclaza + Rybrevant combination is being reviewed for approval in the US as a first-line treatment for lung cancer. Sales of all first-generation EGFR TKIS other than Giotrif decline During the same period, first- and second-generation TKIs saw sluggish growth. Only Giotrif’s sales showed growth, while all others declined. Giotrif generated KRW 27.2 billion in sales last year, up 5.8% from 2022. Since its launch in Korea in 2014, Giotrif has seen a gradual but steady increase in sales. After surpassing the KRW 10 billion mark in sales for the first time in 2017, Giotrif posted sales of KRW 16.6 billion in 2019, KRW 22 billion in 2021, and KRW 25.7 billion in 2022. Giotrif benefited how third-generation TKIs not being used as first-line treatments. On the other hand, Vizimpro’s sales had been sluggish. Vizimpro, which had been released later in 2020, had increased slightly to KRW 1.4 billion in 2021 and KRW 2.8 billion in 2022, but stagnated at KRW 2.7 billion last year. The weakness of Vizympro is in its side effects. In the ARCHER 1050 study, the Phase III trial that became the grounds for its approval, Vizympro showed better efficacy compared to first-generation TKIs, but also a higher rate of adverse events. 60% of patients in the Vizympro arm required dose adjustments due to adverse events. In general, first-generation TKIs are continuing to show declining sales. First, Iressa posted sales of KRW 12.7 billion last year, down 9.2% YoY. Sales have been on a steady decline since 2020 when sales fell below KRW 20 billion (USD 19.6 billion). Iressa posted KRW 16.7 billion in sales in 2021 and KRW 14 billion in 2022. Iressa is known to be a milder cancer drug with fewer side effects among targeted therapy options. As such, it has been used as a priority in patients with relatively poor health. However, sales have been declining with more treatment options becoming available, with latecomers confirming their efficacy over Iressa. Tarceva’s sales fell 13.8% YoY to KRW 3.1 billion last year. This is the fifth consecutive year the drug saw declining sales after posting KRW 8.2 billion in 2019. Compared to the KRW 17.3 billion it had earned in 2016, Tarceva’s sales have more than halved. Tarceva’s sales are also on a decline due to the emergence of more effective second and third-generation targeted therapies. Tarceva also suffered a setback in 2016 when its maintenance therapy indication was removed for patients with stage IIIA or higher NSCLC. Also, Tarceva was not proven effective in retrospective clinical studies. Therefore, its sales are expected to continue to decline when combined targeted therapies are approved for use in the first line.
- Company
- Professor Yoo discovers specific protein expression in HCC
- by Lee, Tak-Sun Mar 19, 2024 05:45am
- Professor Changhoon Yoo, Department of Oncology, Asan Medical Center On the 15th, CMIC Korea, a contract research organization (CRO), announced that the results of the RENOBATE (IIT, Phase II) trial led by Professor Changhoon Yoo (Department of Oncology, Asan Medical Center) were recently published in the international academic journal, Nature Medicine (IF=82.9). This is the first time the results of a clinical study conducted by a Korean researcher have been published in Nature Medicine. In the RENOBATE 42 patients with hepatocellular carcinoma were administered the immunoconology drug regorafenib in combination with the targeted anticancer drug nivolumab. The study used circulation tumor DNA and single-cell RNA sequencing analysis to identify the characteristics of immune cells expressed in patients who did not respond to the combination therapy. The recent development of immuno-oncology drugs, also known as 3rd generation anticancer drugs, that offer better anticancer effects with less toxicity and side effects, has raised hopes for many cancer patients. However, 3 out of 10 patients with HCC were either not responding to or getting worse with the use of targeted anticancer drug+immune-oncology drug combination therapy. The RENOBATE study found that these patients had more than 2 times more expression of a specific protein called TMEM176A/B. This is very promising news for HCC patients who were unable to see much effect with the use of existing anticancer therapies and raises expectations for further developments. The study, which has raised the profile of Korea’s anti-cancer research, was initiated by Professor Changhoon Yoo in 2019 with his collaborators and was completed 3 and a half years later in late 2022. Even though the study was an investigator-initiated trial, which usually has a small amount of research funding, CMIC Korea, the first clinical contract research organization (CRO) in Korea, took charge of monitoring, data management, and clinical analysis to ensure and collect accurate and ethical clinical research data. According to Professor Yoo, this has helped improve the credibility of his research, which has been recognized by peer review and Nature Medicine. This has also been mentioned in the methodology part of the paper as well. Professor Yoo said, "We will continue on our efforts to support patients suffering from incurable diseases through reliable clinical research."
- Company
- Celltrion rolls out Zymfentra in the U.S.…its 1st new drug
- by Chon, Seung-Hyun Mar 19, 2024 05:44am
- On the 18th, Celltrion announced that it had launched Zymfentra, a subcutaneous (SC) formulation of infliximab, in the United States. Pic of Zymfentra Zymfentra is a subcutaneous injection formulation of Celltrion's infliximab. The company received approval from the U.S. Food and Drug Administration last year for its use as a treatment for patients with ulcerative colitis or Crohn's disease. Zymfentra is currently approved and marketed in Europe under the brand name Remsima SS. The original infliximab product is Remicade. Zymfentra’s wholesale price has been set at $6,181 (for 2 doses, 4 weeks). A Celltrion official said, “Based on Zymfentra’s new drug status, the price of competitors in inflammatory bowel disease, and the characteristics of the U.S. pharma-biotech market, we have set an optimal price for us to carry out an effective sales strategy.” If the company’s patent applications for Zymfentra’s SC formulation and method of administration are granted and registered, the drug will receive patent protection by up to 2040. According to the market research institution IQVIA, the U.S. market for TNF-α inhibitors, which includes infliximab, has been valued at approximately KRW 62.06 trillion in 2022. The IBD market, which is the company’s area of focus for Zymfentra, had been valued at KRW 12.8 trillion. Celltrion aims to raise the prescription rate of Zymfentra to reach at least 10% of its target patient population by the second year, 2025, and establish Zymfentra as a global blockbuster that brings in over KRW 1 trillion in annual sales. Celltrion plans to sell Zymfentra directly through its U.S. subsidiary. A Celltrion official said, “Already, several small and mid-sized payers have recognized Zymfentra’s competitivity and have added the product to their formularies without negotiation, laying the groundwork to expand prescriptions.”
- Opinion
- [Reporter’s View] Let Enhertu’s case guide new listings
- by Eo, Yun-Ho Mar 19, 2024 05:44am
- The reimbursement listing of the advanced ADC anti-cancer drug Enhertu is imminent. The company completed drug pricing negotiations with the National Health Insurance Service at a ‘superfast’ speed when considering the slow progress it had made during previous procedures. At this pace, Enhertu is likely to be reimbursed in April this year. After passing HITA’s Cancer Disease Deliberation Committee last year, Enhertu’s reimbursement agenda was rejected by the Drug Reimbursement Evaluation Committee 8 times and was unable to cross the threshold until February this year. But its pricing negotiation took less than one month. This is both encouraging and unusual. Adding on to the wonder, the ICER threshold that the government had set for Enhertu is likely above KRW 50 million. Due to its superior efficacy, the company had difficulty calculating Enhertu’s cost-effectiveness. This is also the reason why it took a long time for the drug’s reimbursement agenda to be submitted to DREC, because the Economic Evaluation Subcommittee was unable to conclude on cost-effectiveness for a long time. The fact that this good drug with a high threshold passed the DREC review signifies that the government set an ICER threshold to pass the drug. Although there is no documented figure, it is generally accepted that the maximum ICER granted for reimbursement in Korea is KRW 50 million. And even the KRW 50 million threshold is known to have been granted very few times. Raising the ICER threshold has been a long-standing desire of the pharmaceutical industry. In a study published last year in the online edition of the medical journal Springer, "An Industry Survey on Unmet Needs in South Korea's New Drug Listing System," the ICER threshold was the No.1 improvement desired by market access managers in the industry. 93% of the respondents chose so. Improving the ICER threshold also topped the list of benefits the government promised to provide as preferential drug pricing measures for innovative new drugs last year. Enhertu’s case rose to attention amid such a trend. Other factors did take part in the incredibly rapid progress made in its drug price negotiations. When considering the given conditions, the pricing negotiation for Enhertu could not have gone so easily. This is why some analysts have been suggesting that the political pressure had played a role ahead of the general elections. However, regardless of the pace, Enhertu has set a promising example for other companies that need to pass pharmacoeconomic evaluations. Enhertu’s case should not end as a one-time event; rather, it should serve as a milestone for other cases to come.
- Policy
- Liver cancer therapy, ‘Stivarga’ set for 2nd RSA renewal
- by Lee, Tak-Sun Mar 19, 2024 05:44am
- Stivarga. It has been reported that a risk sharing agreement (RSA) has been renewed for the second time for Stivarga (regorafenib; Bayer Korea), a second-line treatment for liver cancer. The news comes two months before the reimbursable RSA contract for Stivarga is set to expire on May 31st. According to industry sources on the 18th, the National Health Insurance Service (NHIS) agreed to renew a RSA with Bayer for Stivarga. Under the RSA, Stivarga is expected to receive reimbursement for five years until May 2029. Stivarga was first listed In June 2016 as reimbursable under RSA for the first time for treating gastrointestinal stromal tumor (GIST). Then, since May 2018, it has been reimbursable through RSA for a second-line treatment for hepatoma. In May 2020, Stivarga successfully received the first contract renewal. The contract duration is four years until May 31st, 2024. At the time of the first renewal, Stivarga’s list price was reduced by 7%. After the price cut in January 2022, the current maximum list price is KRW 303,868. It has not been confirmed whether it will receive a price cut in the list price in this contract renewal. However, it is anticipated that there will be a change in the actual price. Stivarga is a second-line treatment for liver cancer, used after Nexavar, the first-line treatment. Its out-patient prescription amount was KRW 9.3 billion won, down 24% from the previous year. Overall sales of existing targeted anticancer therapies are slowing down as immunotherapy with reimbursement emerged. According to Real World Data, which involved 182 Asian patients (Korea, China, and Taiwan), median Overall Survival (OS) with Stivarga was 16.3 months, an improvement of about 54% compared to the previous Phase 3 clinical study, ‘RESORCE.’
- Company
- New CKD drug Kerendia is reimbursed in Korea
- by Son, Hyung-Min Mar 18, 2024 05:50am
- Sunggyun Kim, Professor at Hallym University Sacred Heart Hospital (Secretary-General, The Korean Society of Nephrology) Whether the reimbursement coverage of Bayer’s Kerendia will help address the unmet needs of patients with chronic kidney disease with type 2 diabetes, is gaining attention. Until now, blood pressure medications and diabetes drugs have been used to treat chronic kidney disease, but the introduction of Kerendia has expanded the treatment options. Supported by its proven clinical efficacy, the drug is expected to be more widely used by patients with chronic kidney disease. On the 15th, Bayer Korea held a press conference to celebrate the launch of Kerendia (finerenone) for chronic kidney disease with type 2 diabetes in Korea. Kerendia is a first-in-class, selective, non-steroidal mineralocorticoid receptor antagonist(MRA) that has a novel mechanism of action that inhibits the overactivation of mineralocorticoid receptors, which can cause inflammation and fibrosis in the kidneys and blood vessels. Overactivation of mineralocorticoid receptors causes inflammation and fibrosis in the kidneys, which can lead to deterioration of kidney function and cardiovascular disease. The drug was granted reimbursement in combination with ACE inhibitors and ARBs for adult patients with type 2 diabetes who have been taking an angiotensin-converting enzyme (ACE) inhibitor or angiotensin receptor blocker (ARB) for at least 4 weeks but ▲have an albumin-to-creatinine ratio (uACR) >300 mg/g or a positive urine dipstick test (1+), or ▲have an estimated glomerular filtration rate (eGFR) of at least 25 but less than 75. Kerendia’s reimbursement approval was based on the reduction in kidney disease progression, cardiovascular benefit, and safety that was demonstrated through the Phase III trials FIDELIO-DKD and FIGARO-DKD. The FIDELIO-DKD study evaluated the safety and efficacy of Kerendia compared with placebo in 13,171 adult patients with type 2 diabetes in 48 countries. Over a median follow-up of 3 years, Kerendia reduced the composite cardiovascular endpoint (cardiovascular death, nonfatal myocardial infarction, and stroke, etc) by 14%, and the renal composite endpoint (occurrence of a sustained decline in eGFR below 15 Ml for at least 4 weeks, chronic dialysis, and kidney transplantation) by 23%. Results were consistent with and without treatment with GLP-1 receptor agonists and SGLT-2 inhibitors at baseline. Kerendia also demonstrated significant risk reduction in the cardiovascular composite endpoint in the FIGARO-DKD study, which included patients with Stage I and II chronic kidney disease. Yong-Ho Lee, Professor of Endocrinology and Metabolism at Severance Hospital (Secretary General, Korean Diabetes Association), said, “We have been using GLP-1 receptor agonists, SGLT-2 inhibitors, and blood pressure medications on chronic disease patients with diabetes, but there always remained a residual risk of chronic kidney disease in the patients. SGLT-2 inhibitors do not completely reduce proteinuria, but Kerendia's efficacy, which was demonstrated through multiple clinical trials, makes the drug an important treatment option for patients with chronic kidney disease.” Sunggyun Kim, Professor at Hallym University Sacred Heart Hospital (Secretary-General, The Korean Society of Nephrology), said, “ The American Diabetes Association guidelines recommend a reduction in uACR of at least 30%, and Kerendia reduced the average uACR by 32% compared to placebo in the first 4 months of treatment. Therefore, the drug can be considered in priority for patients with kidney disease."