- LOGIN
- MemberShip
- 2026-04-09 14:41:31
- Policy
- MOHW-Roche have difficulty discussing resupply of Madopar
- by Lee, Jeong-Hwan Aug 28, 2023 05:21am
- The Ministry of Health and Welfare seems to be having difficulty bringing in Madopar, a Parkinson's disease drug that had withdrawn from the domestic market, back to Korea. The MOHW had previously announced that it would make efforts to resupply Madopar in Korea. Roche Korea is known to have refused to supply Madopar Tab to Korea, citing how additional administrative procedures are required to resupply Madopar and that significant expenses are required to produce quantities for export to Korea. On the 25th, the MOHW responded, "We will find a way to solve the patient's difficulties, including resupply," to a written inquiry submitted by Rep Eun-mi Kang, a member of the Justice Party and the National Assembly's Health and Welfare Committee regarding the supply of Madopar. According to MOHW, the authorities had listened to the patient's difficulties through an interview with the Korean Parkinson's Disease Association. At the same time, the Ministry of Health and Welfare also listened to the company's difficulties through a meeting with Roche Korea and suggested measures for Madopar’s resupply, such as guidance on drug price increase procedures. However, Roche Korea’s position is that it is difficult to resupply the drug in Korea because the company has to obtain documents necessary for new domestic approval from its Italian manufacturing plant and the manufacturing facility has been demanding a considerable price as the cost for restoring manufacturing facilities to produce the quantities for export. As a result, Myung-In Pharm's Myungdopar Tab, a generic version of Madopar, is being used instead of Madopar in the field. The MOHW said, "To address the need for Madopar in Korea, we will hold in-depth discussions with the pharmaceutical company the Ministry of Food and Drug Safety, and the Korean Parkinson's Disease Association to seek a way to resolve the difficulties of patients, including whether to resupply."
- Company
- Price negotiation period for Spinraza and Evrysdi extended
- by Eo, Yun-Ho Aug 28, 2023 05:21am
- The companies of the spinal muscular atrophy (SMA) treatments ‘Spinraza’ and ‘Evrysdi’ failed to conclude drug pricing negotiations for their drugs within the deadline and began extended negotiations. As a result, Spinraza reimbursement extension and Evrysdi’s new listing will only be possible in October at the earliest. According to industry sources, the reason for the delay in negotiations for the two drugs is known to be the differences in opinions between the companies and the authorities on the Expenditure Cap that should be set, which is a type of Risk Sharing Agreement (RSA) scheme that is applied to drugs eligible for reimbursement through the pharmacoeconomic evaluation exemption system. In other words, the companies were unable to agree on the cap that should be applied, in consideration of factors such as the expected amount of use and the number of patients. Discussions on reimbursement and reimbursement extension of SMA treatments had seemingly gained speed in line with the movement that pressed for the abolition of Spinraza’s reimbursement suspension standard, but discussions were prolonged after a certain group requested a wider coverage than what the government and pharmaceutical companies expected. At the time, the government announced plans to proceed with discussions on Evrysdi’s listing after agreeing on the reimbursement extension standards for Spinraza. Both drugs were finally put on the final table for negotiations, but the news on the conclusion of the negotiations can only be expected in September. Meanwhile, SMA treatments currently reimbursed in Korea include Spinraza and Novartis Korea's one-shot treatment, 'Zolgensma (Onasemnogene abeparvovec-xioi))'. Spinraza is administered directly into the spinal fluid, and Zolgensma is administered intravenously. Evrysdi, which is attempting new listing, is an oral drug that reduces the social and economic burden patients and their caregivers face due to injections, such as school/work interruption, transportation costs, and nursing care It also has the advantage of being able to reduce drug costs for infant patients as it offers customized prescriptions by age and weight.
- Company
- Trulicity's share reaches 99.8%
- by Kim, Jin-Gu Aug 28, 2023 05:21am
- Trulicity Trulicity, a GLP-1 analog class diabetes treatment, once again set the record for highest sales. However, it is unclear whether this upward trend will continue in the second half. This is because it is said that some disruptions have occurred in domestic supply due to a shortage of global distribution supplies. Ozempic and Rybelsus, which are considered strong opponents, are waiting for release. In the pharmaceutical industry, if the two drugs are added in earnest, it is expected that the diabetes treatment market, which is dominated by Trulicity, will fluctuate greatly. According to IQVIA, a pharmaceutical market research institute, on the 26th, Trulicity's sales in the second quarter were 18 billion won. Compared to the second quarter of last year, it increased by 24.8% in one year. Trulicity is a GLP-1 analog class diabetes treatment. GLP-1 analogs are drugs developed using the GLP-1 hormone involved in blood sugar control in the body. The GLP-1 hormone promotes insulin secretion right after a meal to drop blood sugar, and when blood sugar drops below a certain level, it reduces insulin secretion to help prevent hypoglycemia. It has been 7 years since it was released in Korea, but it is still growing rapidly. Trulicity exceeded 10 billion won in quarterly sales in the third quarter of 2019 and exceeded 15 billion won in sales in the second quarter of last year. After that, it stagnated in the fourth quarter of last year and the first quarter of this year, but set a new record again in the second quarter. Even though it was released as the latest among the drugs in the same class, it won the competition with Victoza, Lyxumia, and Byetta, and in 2017, it virtually solidified its solo system. As of the second quarter of this year, Trulicity's market share reached 99.8%. Shortage of domestic supply and notice of the emergence of competitive drugs. In June, Eli Lilly notified its partner Boryeong that Trulicity would be difficult to supply in Korea. It is said that the clinical site has been struggling with the supply of Trulicity since last July. In this regard, Eli Lilly's position is that there is a disruption in supply due to the increase in global demand. On the other hand, some in the pharmaceutical industry argue that Trulicity's production has decreased relatively as Eli Lilly focused on producing Mounjaro, a next-generation drug, at the headquarters level. The timing of the release of competing drugs is also considered a variable. In the pharmaceutical industry, Novo Nordisk's Ozempic and Rybelsus are considered strong competitors to Trulicity. Ozempic is a long-acting, once-a-week injection of the same type as Trulicity, and Rybelsus is an oral drug. Rybelsus is taken once daily. Novo Nordisk received licenses for Ozempic in April and Rybelsus in May, respectively. It is said that Ozempic has already overtaken Trulicity in the global market. However, the official launch of Ozempic in Korea has been temporarily suspended due to a lack of global supply.
- Company
- Discussion on expanding coverage for Leclaza begins
- by Eo, Yun-Ho Aug 28, 2023 05:21am
- Attention is focused on expanding insurance coverage for the first-line treatment of the anticancer drug Leclaza. According to related industries, Yuhan Corporation's 3rd generation EGFR TKI Leclaza will be scheduled for the HIRA on the 30th. The specific indication is 'primary treatment for EGFR active non-small cell lung cancer'. Leclaza obtained approval from the Ministry of Food and Drug Safety for the indication in June. If it is presented to the review committee, the evaluation discussion will proceed within two months of approval. This drug passed the review committee about a month after January 2021, when it was first approved as a second-line treatment, and was listed in July of the same year. As a domestic new drug, it is showing rapid speed. Considering that it usually takes 4 to 5 months for an anti-cancer drug to be submitted to the cancer screening after applying for benefits, this is a considerable speed. Leclaza confirmed a statistically significant improvement in progression-free survival in first-line non-small cell lung cancer therapy through LASER301 in a phase 3 study. As a result of the analysis of the primary endpoint PFS in this study, the Lazertinib treatment group improved PFS statistically with 20.6 months in the Leclaza administration group and 9.7 months in the control Iressa administration group. Yuhan Corporation is operating an Early Access Program that provides free drugs to patients until they can receive health insurance benefits. The program will run from the date of each IRB approval until the time of Lazertinib's reimbursement standard expansion. In Korea, EGFR TKIs such as AstraZeneca's Iressa and Roche's Tarceva, second-generation drugs Giotrif and Vizimpro, and AstraZeneca's Tagrisso, like Leclaza, are currently prescribed in Korea.
- Company
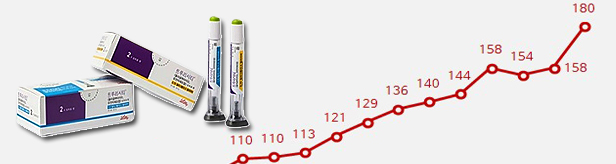
- New drug Shingrix leads shingles vaccine mkt
- by Chon, Seung-Hyun Aug 28, 2023 05:21am
- GlaxoSmithKline's shingles vaccine 'Shingrix' took the lead in Korea’s market just 7 months after its release. With its powerful shingles prevention effect, its sales surpassed that of its competitors. SkYZoster, a homegrown vaccine, had taken the lead in the market for the first time at the end of last year but was overtaken by Shingrix in just 2 quarters. According to the market research institution IQVIA on the 26th, the market for preventive vaccines for shingles was KRW 45.4 billion in 1H last year, which was a 123.3% increase from the KRW 20.4 billion in 1H last year. The shingles vaccine market grew 147.0% YoY to record KRW 21.9 billion in Q1 this year, then again rose 124.1% to KRW 23.5 billion. The shingles vaccine market, which had only been KRW 10.5 billion in Q4 last year, saw a more than twofold increase in a single quarter. This abrupt market expansion is related to the introduction of the new vaccine Shingrix. Shingrix, which started being vaccinated in December last year, showed off its presence by occupying 28.9% of the market with sales of 6 billion won in Q1 this year. In Q2, the drug quickly rose to lead the shingles vaccine market with sales of KRW 11.1 billion. Shingrix's share of the shingles vaccine market in Q2 reached 47.4%. In other words, within 7 months of its launch, the vaccine has occupied about half of the entire market. The greatest benefit of Shignrix is its strong shingles prevention effect. In a Phase III clinical trial (ZOE-50) that was conducted on adults aged over 50 years of age, Shingrix showed a 97.2% efficacy compared to the non-vaccinated group at 3.2 years of follow-up, and in another Phase III clinical trial (ZOE-70) conducted on adults aged 70 years and above, Shingrix showed an 89.8% efficacy at 3.7 years of follow up. This is superior to the 5% protection in adults aged over 50 years of age and 41% in adults aged 70 years and above demonstrated with the use of Zostavax. The prevention rate of SKYZoster is also known to be similar to Zostavax. Also, Shingrix’s safety profile was confirmed through 5 clinical trials that were conducted on immunocompromised patients aged 18 years and older. Based on such evidence, patients who received autologous hematopoietic stem cell transplantation or those with solid cancer, blood cancer, or solid organ transplant patients who have an increased risk of shingles are also eligible to receive vaccination with Shingrix. At the time of its release, Shingrix's significantly higher price than existing vaccines was pointed to as an obstacle to the vaccine’s early settlement into the market. vaccines. The price of Shingrix, which is administered two times in total, is set at around KRW 500,000 to 600,000. This is more than twice as high as the existing vaccine, which costs KRW 150,000 to 200,000. However, Shingrix rapidly increased its market share in the market thanks to its superior efficacy despite the high price. Also, the sales support from 2 domestic pharmaceutical companies contributed to Shingrix’s rapid market penetration. GSK started domestic sales of Shingrix in partnership with two companies, GC Biopharma and Kwangdong Pharmaceutical. Although other existing shingles vaccines like SKYZoster and Zostavax also saw a sales increase from last year, their market share declined due to Shingrix. SK Bioscience’s SKYZoster’s sales had risen 83.2% YoY to record KRW 16.7 billion in 1H this year. SKYZoster led the shingles market in Q1 with sales of KRW 9.5 billion, which is a 151.5% YoY increase. SKYZoster’s sales continued to rise in Q2 to record KRW 6.5 billion, a 31.1% YoY increase, it lost its lead to Shingrix at the time. Sky Zoster is a live attenuated vaccine for the prevention of shingles that was developed by SK Bioscience with its proprietary technology. The vaccine has proven its non-inferiority compared to its competitor (Zostavax) in adults aged 50 or older in 8 domestic clinical institutions. In October 2017, SK Bioscience obtained approval for SKYZoster from the Ministry of Food and Drug Safety for 'preventing shingles in adults over the age of 50'. Before its approval, MSD's Zostavax was the only vaccine on the market. SKYZoster’s sales surpassed Zostavax for the first time since its release in Q4 year and took the lead. It maintained the lead for two consecutive quarters until Q1 this year. However, due to Shingrix's rapid rise, the vaccine lost its lead after 2 quarters. Zostavax’s sales in 1H this year was KRW 11.9 billion, up 2.6% YoY. Zostavax’s sales in Q1 this year rose by 18.9% YoY to reach KRW 6 billion but then fell 10.1% to KRW 5.8 billion in Q2. Zostavax lost its lead to SKYZoster in Q4 last year and then fell to third place with the introduction of Shingrix. Zostavax only occupied 24.8% of the market in Q2.
- Company
- Hanmi Pharm starts P1T on its next-gen immuno-oncology drug
- by Chon, Seung-Hyun Aug 25, 2023 06:06am
- On the 22nd, Hanmi Pharm announced that it has recently received approval to initiate a domestic Phase I trial for its next-generation immuno-oncology drug ‘‘BH3120(PD-L1/4-1BB BsAb)’ from the Ministry of Food and Drug Safety. In May, the company had previously received approval from the US FDA for the same drug candidate after submitting an Investigational New Drug Application. BH3120, which is be codeveloped by Hanmi Pharm and Beijing Hanmi Pharm, is a new drug candidate that applies the company’s, bispecific antibody platform technology ‘Pentambody’ that allows for one antibody to bind to two targets at once. The drug offers the benefit of providing both immunotherapy (that activates immune cells) and targeted therapy (that specifically targets cancer cells). BH3120 is an immunoglobulin G (IgG) like dual antibody designed that has biased binding affinities against PD-L1 and 4-1BB. The different binding affinities that were designed based on various studies are expected to induce better efficacy and safety. BH3120 has a dual antibody mechanism of action that targets both 4-1BB and the PD-L1 expressed on the surface of cancer cells that specifically elicit antitumor activities in the tumor microenvironment (TME), and has shown a potent antitumor effect by activating immune cells within cancerous tissues. In addition to BH3120’s benefits as monotherapy, Hanmi Pharm also confirmed the drug’s strong synergistic effect as combined therapy, as its use in combination with a PD-L1 inhibitor resulted in the disappearance of cancerous tissue. No toxicity or immune system-related side effects were observed with BH3120 in the safety study that was conducted on primates and was confirmed to have a superior safety profile compared to same-class competitors that are currently being developed. The company presented its study results at the American Association for Cancer Research (AACR) Annual meeting that was held in April this year. The results showed that a clear decoupling of immune activity between cancer and normal tissues was observed, signifying the possibility of the development of a new immuno-oncology drug that minimizes the side effects of existing immuno-oncology drugs Based on the preclinical study results, the company plans to start a global Phase I trial on BH3120 in the US and Korea within this year. An official from Hanmi Pharm said, “BH3120 is the first project that uses the next-generation bispecific antibody platform technology ‘Pentambody’ for a global clinical trial. We believe ‘Pentambody’ will follow Hanmi’s proprietary ‘Lapscovery’ technology and continue to create future value for the company.
- Policy
- Are there any countermeasures against Rx of hair loss pills?
- by Lee, Jeong-Hwan Aug 25, 2023 06:06am
- The Health and Welfare Committee of the National Assembly will start reviewing legislation to institutionalize non-face-to-face treatment, excluding the prescription restriction bill for high-risk non-insured drugs such as hair loss treatment, which is being prepared by Rep. Jeon Hye-sook, and the public e-prescription legislation proposed by Rep. However, in the process of institutionalizing non-face-to-face treatment, the issue of prescription of high-risk non-reimbursed drugs and the need for regulatory and management measures for electronic prescriptions to be issued after non-face-to-face treatment are expected to be discussed together. Whether the Ministry of Health and Welfare can clearly come up with measures to solve the problem of abnormal prescriptions and dispensing that can be mass-produced when non-face-to-face treatment is enacted, or the collapse of the healthcare ecosystem such as hospitals, clinics, and pharmacies, will determine the legislative success or failure. On the 24th, the Welfare Committee began reviewing five previously proposed amendments to the Medical Act related to the institutionalization of non-face-to-face treatment and the implementation plan submitted by the Ministry of Health and Welfare. The screening, which began at 9:30 a.m., is expected to continue until lunchtime. The non-face-to-face treatment bill submitted by the Ministry of Health and Welfare is the main point of the pilot project currently being implemented. It is a content that allows non-face-to-face treatment mainly for returning patients and clinics and allows first visits only in special cases such as residents of medically underserved areas, people with mobility difficulties, and people with disabilities. Prescription drug delivery is allowed only in special cases by designating it as a home recipient, and basically, it is required to visit a pharmacy and receive face-to-face medication instruction. However, the Ministry of Health and Welfare submitted an improved opinion on the regulation of non-face-to-face treatment platforms such as Dr. Now. First of all, platforms that want to run a non-face-to-face treatment brokerage business must go through a reporting process that requires government repairs. In the case of non-reporting or false reporting, a fine of not more than 3 million won was imposed. According to the Ministry of Health and Welfare, a report requiring repair is a report system operated with a relaxed permit. It is not simply a report that meets formal requirements, but it must meet practical requirements such as human standards and material standards. The report review by the Ministry of Health and Welfare also includes not only a formal review but also a substantive review on whether or not the statutory requirements or legislative purpose are met. The view of multiple welfare committee members is that the key to reviewing the non-face-to-face treatment bill will be whether the Ministry of Health and Welfare has prepared specific countermeasures for the side effects that occurred during the pilot project. The purpose is that legislation may be difficult if it is not clear how to manage the ratio of non-face-to-face treatment, which is focused on prescriptions for non-insured high-risk drugs such as hair loss treatments, morning-after pills, and acne medications. In addition, it is expected that the Ministry of Health and Welfare will have to come up with solutions for electronic prescriptions issued after non-face-to-face treatment, such as managing sensitive patient information and preventing concerns about forgery. In particular, the range of non-face-to-face treatment patients allowed in the pilot project stage is also a legislative issue. The current pilot project permits all diseases other than those requiring a prescription for narcotic drugs or medicines concerned with misuse or abuse during non-face-to-face treatment other than first visits. For this reason, there are criticisms that the side effects of non-face-to-face treatment, such as hair loss pills, morning-after pills, and acne medicine, are accelerating, such as the concentration of prescriptions in some treatment categories or disease groups. Some welfare committee members are expressing the opinion that legislating such a pilot project as it is is too broad. It is the opinion that non-face-to-face treatment should be allowed locally only for residents of medically vulnerable areas where non-face-to-face treatment is unavoidable due to poor mobility or lack of medical institutions and pharmacies in the living radius. Cho Won-joon, the senior expert on the matter, said, "We examine the non-face-to-face treatment bill submitted by the Ministry of Health and Welfare, but how to manage non-consensual and irrational non-face-to-face treatment prescription behavior, such as non-insured hair loss prescriptions that have not yet been proposed or are omitted from the agenda, and how to manage e-prescriptions. The position of the Ministry of Health and Welfare will determine the success or failure of the legislation,” he explained. Commissioner Cho Won-joon explained, "The Ministry of Health and Welfare must clearly present a policy to operate the system so that non-face-to-face treatment does not produce side effects that threaten the safety of the public and patients and destroy the health care ecosystem."
- Policy
- Bayer completes pricing negotiations for Jivi Inj in KOR
- by Lee, Tak-Sun Aug 25, 2023 06:06am
- Bayer’s hemophilia A treatment Bayer’s s hemophilia A treatment ‘Jivi Inj (damoctocog alfa pegol)’ has come one step near to receiving reimbursement in Korea. On the other hand, reimbursement discussions for Lilly Korea’s ‘Retevmo (selpercatinib)’ have returned to square one after the company failed to complete pricing negotiations for its RET targeted anticancer drug with the NHIS. In addition, Teva-Handok started pricing negotiations for its antibody drug for asthma, ‘Cinqair (reslizumab)’ with the NHIS. According to the industry on the 24th, the NHIS had updated the drug pricing negotiation status of new drugs on its webpage recently. The drug pricing negotiations for Jivi Inj were found to have been completed. In May, the drug was approved for reimbursement under the condition that it accepts a price below the amount evaluated by the Drug Reimbursement Evaluation Committee of the Health Insurance Review and Assessment Service. Jivi Inj then initiated and completed drug pricing negotiations with the NHIS. However, due to the timing of the agreement, the drug will be listed for reimbursement the month after the next, rather than next month. Jivi Inj is indicated for use in previously treated adults and adolescents (12 years of age and older) with hemophilia A (congenital Factor VIII deficiency) as an on-demand treatment and control of bleeding episodes, for the perioperative management of bleeding, and routine prophylaxis to reduce the frequency of bleeding episodes. Another drug, Lilly’s Retevmo Cap, had failed to reach an agreement after completing pricing negotiations. Retevmo passed the DREC review in May but was unable to close the gap in opinion with the NHIS during pricing negotiations without extending the negotiation period. As such, the company would need to restart the reimbursement application process from scratch for reimbursement in Korea. Retevmo is indicated for the treatment of ▲RET-mutated medullary thyroid cancer and ▲ RET fusion benign thyroid cancer. Although the number of patients is small, the drug is used in RET-mutated patients with poor prognosis. No drug for RET mutation has been approved for reimbursement in Korea yet. Teva-Handok newly started pricing negotiations for the reimbursement of its antibody drug for asthma, ‘Cinqair Inj.’ Cinqair passed DREC review in July. The drug is used to treat severe eosinophilic asthma. As an interleukin-5 (IL-5) inhibitor, it has a new mechanism of action that reduces the levels of blood eosinophils, a type of white blood cell that is involved in the development of asthma exacerbation. Roche failed to reach an agreement within the deadline for its spinal muscular atrophy (SMA) treatment 'Everysdi Dry Syrup (risdiplam)', which has been under negotiations since last June and extended its negotiation period.
- Policy
- Reduction in the scope of platform regulation & re-examinati
- by Lee, Jeong-Hwan Aug 25, 2023 06:06am
- Following the decision to postpone the non-face-to-face treatment institutionalization bill again by the National Assembly Health and Welfare Committee on the 24th, it is evaluated that the number of issues to be resolved for legislation has increased. At the same time, the need to reduce the scope of non-face-to-face treatment patients compared to the current pilot project was raised, and criticism followed that there was no mechanism to regulate illegal and expedient actions by mediation platform companies. Furthermore, as a result of the pilot project that has been in effect since last June, it is still difficult to punish without distinguishing first-time and returning patients in non-face-to-face treatment, and it is a shame for violations of the law that exceeds 30% of non-face-to-face treatment and dispensing rates allowed by medical institutions and pharmacies. Members of the National Assembly’s subcommittee point out that there is not even a countermeasure against losing. On this day, the non-face-to-face treatment legislative review was in the direction of a collision between the Ministry of Health and Welfare's request to regulate expedients on some platforms through prompt institutionalization and the National Assembly's criticism of the Ministry of Health and Welfare's countermeasures against many side effects that may occur in the enactment of non-face-to-face treatment legislation. It is said that it has been done. Opposition members of the welfare committee who participated in the review are of the opinion that the Ministry of Health and Welfare has not come up with a proper legislative bill to address problems that threaten public health and cause confusion in the health and medical ecosystem. Although the Ministry of Health and Welfare submitted a government bill to the National Assembly that went through the process of amending and improving the provisions with the Welfare Committee expert committee, criticism continued that it failed to come up with legal provisions to solve even the problems that occurred during the pilot project. The basics of the welfare committee members are that there are no measures on how to classify first-time and returning patients when institutionalizing, what punishments to be imposed in case of violation, and how to regulate the problem of implementing non-face-to-face treatment exceeding the allowable rate (up to 30% for pilot projects). negative opinion.
- Company
- Company Roche and Drug Keytruda will lead industry in 5 yrs
- by Kim, Jin-Gu Aug 25, 2023 06:06am
- A projection has been made that Roche will become the top global-grossing pharmaceutical company 5 years from now in 2028. Evaluate, the global pharmaceutical market analysis institution that released such projections, also predicted that Keytruda would take over Humira’s throne and become the No. 1 grossing drug among pharmaceuticals. On the 24th, KoreaBIO cited Evaluate’s data and predicted the top-grossing pharmaceutical companies and drugs for 2023. According to KoreaBIo’s projections, biopharmaceutical companies that have been producing results will be in the top ranks this year. The top-grossing company 5 years from now will be Roche, supported by the sales of its immuno-oncology drug ‘Tecentriq,’ multiple sclerosis treatment ‘Ocrevus’ and macular degeneration treatment disease drug ‘Vabysmo.’ No.2 and No.3 projections were the U.S. companies Merck and AbbVie, respectively. However, the difference in revenue between the top 3 companies is expected to be small. No. 4 was expected to be Johnson & Johnson. Top 10 Grossing ETC Pharmaceutical Companies in 2028 Pfizer, which ranked No.1 with unrivaled sales last year was thought to fall to 5th place. In addition, Novartis, AstraZeneca, Sanofi, Novo Nordisk, and Eli Lilly were expected to make the top 10 sales list. Compared to the current status, Novo Nordisk and Eli Lilly are new to the top 10 list in 2028, taking the place of BMS and GSK. The expectation was that sales of diabetes and obesity drugs that the companies had recently released would surge, leading to a rise in the rankings. The immuno-oncology drug Keytruda is expected to take over the top spot in terms of global sales. In particular, it is predicted that Keytruda will rise to No. 1 this year, surpassing Humira, which had held the lead until now. In addition to Keytruda, immuno-oncology drugs such as BMS's Opdivo, Roche’s Tecentriq, and AstraZeneca's Imfinzi are also expected to make the Top 10. If the current upward trend continues, the total sales of immuno-oncology drugs are expected to surge from $40 billion in 2022 to $71 billion in 2028. In addition, Sanofi’s atopic dermatitis treatment ‘Dupixent,’ Eli Lilly’s antidiabetic treatment ‘Mounjaro,’ Johnson & Johnson’s multiple myeloma treatment ‘Darzalex,’ AbbVie’s autoimmune disorder treatment ‘Skyrizi,’ Gilead Science’s HIV treatment ‘Biktarvy,’ Merck’s cervical cancer vaccine ‘Gardasil,’ and Vertex Pharmaceutical’s ‘Trikafta’ are also expected to make the Top 10 in 2028. Top 10 Selling Drug Projections in 2028