- LOGIN
- MemberShip
- 2026-04-09 14:41:29
- Policy
- Cough Syrup and Synatura Syrup complete PVA negotiations
- by Lee, Tak-Sun Aug 08, 2023 05:30am
- Respiratory drugs such as Cough Syrup and Synatrua Syrup were found to have been added as subjects for drug pricing negotiations under the Price-Volume Agreement (PVA) this year. The use of the items had increased significantly last year due to the sharp increase in confirmed COVID-19 cases last year. According to industry sources on the 7th, the National Health Insurance Service added 134 items and updated the list of drugs that completed drug pricing negotiations as ‘Type C PVA drugs’ recently. Previously, the number of items subject to PVA Type C negotiations this time was known to be 138, and a similar number of items had been added to the list as those that completed negotiations as of July 31st. Among the added items, antitussive expectorants, antibiotics, and antivirals stand out as the use of the drugs was presumed to have increased due to the increase in confirmed COVID-19 cases last year. Specifically, the drugs include Yuhan Corp’s ‘Cough Syrup,’ Daewon Pharmaceutical’s ‘Codaewon Forte Syrup,’ Ahngook Pharm’s ‘Synatrua Syrup,’ Hanmi Pharm’s ‘Cossac-L Tab,’ Hanwha Pharma’s ‘Muteran Cap. 200mg,’ Korea United Pharm’s ‘Kalomin Tab And Kalomin S Tab.’ According to UBIST, outpatient prescriptions of the Cough Syrup had increased 111% YoY to record ‘KRW 26.7 billion’ last year. Prescriptions of Codaewon Forte Syrup had also increased 88% YoY to reach KRW 21.6 billion, and Synatrua Syrup by 96% to record KRW 35 billion. Prescriptions of Cossac-L Tab increased by 121% to reach KRW 5.6 billion. Prescriptions of major items increased by around 100%, and the dominant analysis in the industry is that this is because prescriptions for respiratory drugs, such as antitussive expectorants and antibiotics increased significantly with the rise of confirmed COVID-19 patients last year. Drugs are subject to drug pricing negotiations as Type C of the PVA if its claims amount of a drug increases by over 60%, or by 10% but the increased amount exceeds KRW 5 billion. Therefore, drugs whose claims have increased by around 100% due to the influence of COVID-19 were highly likely to be included in the PVA negotiations and subject to price cuts. Regarding the issue, the pharmacuetical industry insisted that drugs whose use increased due to COVID-19 should be excluded from PVA negotiations, and the health authorities partially accepted this claim and decided to adjust the use amount of drugs related to COVID-19. For example, the government prepared a correction formula that excludes the 10 months last year when the number of COVID-19 patients surged and converts the claims amount of the 2 remaining months into 12 months to compare with the claims amount of the previous year. As a result, the use amount of 36 items of the same product group was corrected and completed negotiations with the NHIS. In addition to respiratory drugs, blockbuster drugs used to treat chronic conditions such as Boryung’s ‘Dukarb Tab,’ Hanmi Pharm’s ‘Rosuzet Tab,’ Chong Kun Dang’s ‘Telminuvo Tab,’ and AstraZeneca’s ‘Xigduo XR Tab’ were also found to have completed negotiations this time.
- Company
- Sales of the flu tx market increased due to the endemic
- by Chon, Seung-Hyun Aug 07, 2023 05:23am
- In the first half of the year, the outpatient prescription market for flu treatment exceeded 10 billion won in four years. As the number of flu patients surged after the end of the Corona 19 pandemic, the flu treatment market, which had disappeared over the past three years, expanded significantly. According to UBIST, a drug research institute on the 5th, the outpatient prescription amount for influenza (flu) treatment in the first half of last year was recorded at 11.5 billion won. It fell short of 100 million won in the first half of last year but soared more than 200 times in one year. Quarterly outpatient Rx amount of flu treatment (unit: 100 million won, source: UBIST) In the first and second quarters of last year, the flu treatment market size was only 40 million won and 4 million won, respectively. It rose sharply to 5.1 billion won in the first and second quarters of this year. It is the first time in four years since 2019 that the prescription of flu treatment in the first half of the year exceeded 10 billion won. After recording 8.2 billion won in prescriptions in the first quarter of 2020, the flu treatment market virtually disappeared by the third quarter of last year. After the spread of COVID-19 in earnest, the outbreak of infectious diseases has significantly decreased due to strengthened personal hygiene management such as washing hands and wearing masks. According to the KDCA, the number of suspected flu patients per 1,000 outpatients recorded at 6.3 in the 9th week, the first week of March 2020, and has never exceeded 5 until August of last year. This means that there has not been a single outbreak of the flu in 2 years and 6 months. Influenza tx recorded 8.2 billion won in prescriptions in the first quarter of 2020 and virtually disappeared by the third quarter of last year, but started to rebound with 10.4 billion won in the fourth quarter of last year. From the beginning of this year to last June, the number of flu patients exceeded the epidemic standard. The number of suspected influenza patients per 1,000 outpatients dropped from 52.5 in the first week to 11.7 in March. However, since April, the number of suspected flu cases has risen again. For six consecutive weeks from the 17th week of the fourth week of April to the 22nd week of the last week of May, the number of suspected influenza patients per 1,000 outpatients was more than 20. It is a level that significantly exceeds the flu prevalence standard of 4.9 set by the KDCA. In the first half of last year, the prescription market for oseltamivir ingredients was limited to 40 million won, but in the first half of this year, it expanded 191 times to 7.9 billion won. Oseltamivir is the main active ingredient in Tamiflu. The size of the prescription market for Oseltamivir never exceeded 100 million won until the third quarter of last year after recording 6.6 billion won in the first quarter of 2020. However, it succeeded in rebounding to 8.5 billion won in the fourth quarter of last year and continued its growth this year. In the first half of last year, prescriptions for Tamiflu were only 30 million won, but this increased to 5.7 billion won in one year.
- Policy
- Paxlovid and Dong-A’s Diosmin Powder were approved in July
- by Lee, Hye-Kyung Aug 07, 2023 05:23am
- The number of prescription drug approvals decreased significantly in July. The approvals, which had continuously increased from 70 in May to 93 in June, had fallen to record 29 in July. However, the drugs approved were nevertheless as significant as Pfizer Korea’s COVID-19 treatment ‘Paxlovid (nirmatrelvir, ritonavir)’, which had first been introduced to Korea through the EUA (emergency use authorization) during the COVID-19 outbreak, received formal approval as a new drug in Korea, and the Takeda-developed Celltrion-owned ‘Alo Gliptinpio Tab’ was approved for export. Drugs Approved in 2023 In July, a total of 76 drugs were approved by the Ministry of Food and Drug Safety. Compared to the previous month, the ETC approvals were reduced by 64, and OTC approvals increased to 47 and exceeded the number of ETC approvals. ◆OTC drugs== A total of 47 over-the-counter drugs were approved (registered) in July. Among those, 3 were data submission drugs(incrementally modified drugs, IMDs), which are modified versions of existing drugs that underwent safety and efficacy reviews due to changes in ingredient, salt base, or dosage form. OTCs approved in July KyungDong Pharma’s Gnal-N Ace Soft Cap (IMD, approved on July 12) Gnal-N is the first product that made KyungDong Pharm, a pharmaceutical company that had previously been focused on clinical areas, familiar to the public. The Gnal-N series was first released in 2009 with the ibuprofen combo ‘Gnal-N Tab,’ followed by ‘Gnal-N Q Tab', ‘Gnal-N Nose Soft Cap, ‘Gnal-N Cold Soft Cap,’ ‘Gnal-N Cough Soft Cap,’ ‘Gnal-N Nose Plus Soft Cap.’ The company received additional approval for ‘Gnal-N Ace Soft Cap’ which contains a combination of acetaminophen and riboflavin on July 12, and increased its Gnal-N product line-up to 13. Gnal-N Ace Soft Cap is indicated for headache, toothache, pain after tooth extraction, sore throat, ear pain, joint pain, neuralgia, back pain, muscle pain, shoulder pain (stiff shoulder), bruise pain, fracture pain, sprain pain, menstrual pain. analgesia of traumatic pain, chills, and fever. Dong-A Pharmaceutical’s Dong-A Pharm Diosmin Suspension Powder (IMD, approved on July 18) Dong-A Pharmaceutical will soon be releasing its approved ‘Dong-A Pharm Diosmin Suspension Powder’ that offers improved convenience in intake to pharmacies in Korea. Each packet of the drug contains diosmin 600mg that can be dissolved into water for intake. It is indicated to improve symptoms related to venous insufficiency (leg heaviness, pain), as supplementary treatment for disorders caused by capillary fragility, and treatment of symptoms related to hemorrhoids. It will be released to pharmacies as a box with 10 packets. Meanwhile, latecomers such as Dong-A Pharmaceutical, Hanmi Pharm, Chodang Pharm, and Samjin Pharm have increased their presence in the diosmin market by releasing high-dose oral hemorrhoids treatments one after another. Dong-A Pharmaceutical's annual sales of Diomax Tab doubled from KRW 500 million in 2020 to KRW 1 billion in 2021, based on IQVIA data. Last year, Diomax posted sales of KRW 1.1 billion, occupying 48% of the market the high-dose (600mg) oral hemorrhoids treatment market. ETCs approved in July ◆ETC=Two new prescription drugs were approved in July. 14 data submission drugs and 13 generics and others were also approved the same month. Celltrion’s ‘Celltrion Alo Gliptinpio Tab 25·15mg, 25·30mg (for export, Approved on July 6 and 7) Celltrion received approval for Celltrion Alo Gliptinpio Tab 25·15mg, and 25·30mg for export on July 6 and 7, respectively. The drug is a new combination made by the company using its rights for ‘Nesina' and 'Actos', which it acquired from the Japanese pharmaceutical company Takeda Pharmaceuticals. Takeda Pharmaceuticals had been directly selling Nesina in Korea until 2020, but in December 2020, it sold all rights, including sales rights and patents, of 12 ETC drug brands and 6 OTC drugs in 9 Asia-Pacific countries to Celltrion Pharm to adjust the size of its debt. I did. Alogliptin and pioglitazone can help patients control blood sugar levels, and patients with Type 2 diabetes can take them to control blood sugar along with diet and exercise. Since last year, Celltrion Pharm has been strengthening its product lineup receiving approval for drugs to export overseas. Sanofi-Aventis Korea’s Enjaymo Inj (new drug, Approved on July 12) Sanofi’s ‘Enjaymo Inj,’ which is indicated to treat adults with cold agglutinin disease (CAD), was also recently approved in Korea. The drug is used to treat the breakdown of red blood cells (hemolysis) in adults with cold agglutinin disease (CAD), which is a form of autoimmune hemolytic anemia (AIHA), CAD is a rare type of autoimmune hemolytic anemia caused when antibodies called cold agglutinins bind to the surface of red blood cells. When cold agglutinins bind to the surface of red blood cells, the body's immune system mistakenly attacks and destroys healthy red blood cells. Pfizer Korea’s Paxlovid Tab (new drug, Approved on July 14) Pfizer Korea’s ‘Paxlovid,’ which was first introduced in Korea through the emergency use authorization (EAU) track during the spread of COVID-19, received official approval in Korea. Paxlovid was granted EAU by the MFDS on December 27, 2021, the same year the number of confirmed and seriously ill patients with COVID-19 surged and the Omicron variant spread, raising the need for an oral treatment for COVID-19. It has been formally approved as a prescription drug 1 year and 7 months after its introduction to Korea. Patients who take Paxlovid take two tablets at the same time. Nirmatrelvir blocks protease (3CL protease) to prevent the production of proteins necessary for viral replication, thereby inhibiting the proliferation of the virus, and ritonavir inhibits the enzyme (CYP3A4) that breaks down nirmatrelvir, extending its duration of effect. After a careful review of the results of the Phase III clinical trial (therapeutic confirmatory trial) conducted on adult patients, the drug was officially approved in Korea in accordance with the 'Pharmaceutical Affairs Act,’ The EAU for Paxlovid, which has allowed patients to use the drug free of charge, will remain in place as is.
- Company
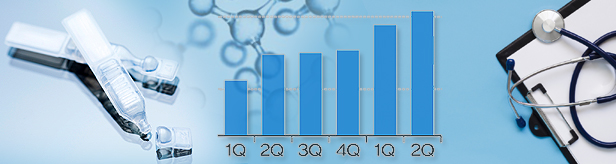
- Prescription of HA eye drops record KRW 166 bil in 1H
- by Kim, Jin-Gu Aug 07, 2023 05:23am
- The prescription market for hyaluronic acid eye drops grew to record the largest scale ever ahead of the reimbursement reevaluations set for the end of this year. In 1H alone, the market grew exceeded KRW 160 billion in prescriptions this year. The number of companies with half-year prescriptions that exceed KRW 10 billion increased from 5 in 1H last year to 7. The pharmaceutical companies that will inevitably suffer huge losses due to the expected reimbursement re-evaluations are preparing to jointly respond to the government's measures. Prescription of HA eye drops exceed KRW 160 bil in 1H, 7 companies exceed KRW 10 bil According to the market research institution UBIST, outpatient prescription of HA eye drops in 1H this year was KRW 16.6 billion. This is a 16% increase from 1H last year. This market shrunk somewhat in 2020 due to the spread of COVID-19 but has been rapidly expanding since then. Quarterly HA eye drop prescriptions(Unit: KRW 100 mil, Data: UBIST) By quarterly prescriptions, HA eye drops exceeded KRW 70 billion in Q2 last year and KRW 80 billion in Q1 this year. At this rate, the total market size may increase to exceed KRW 300 billion this year for the first time this year. The prescription performance of major companies in the market has also increased significantly. The prescription performance of Optus Pharm, which owns the Tearin series, increased by 26% from KRW 14 billion in 1H last year to KRW 17.7 billion in 1H this year. In the case of Taejoon Pharm, which owns New Hyalyuni, Hyalyuni, and New Hyaldrops, its sales increased by 21% from KRW 12.6 billion to KRW 15.3 billion. Sales at Daewoo Pharm, which owns Hyalsan, rose 36% from KRW 10.6 billion to KRW 14.4 billion. As a single product, Hyalsan recorded the highest prescription performance in 1H this year. The number of companies whos half year sales exceeded KRW 10 billion increased from 5 in 1H last year to 7 in 1H this year. As of 1H this year, Optus Pharm, Taejoon Pharm, Daewoo Pharm and SamChunDang Pharm, Huons Meditech, Hanmi Pharm, Kukje Pharm recorded prescriptions of more than KRW 10 billion. Changes in HA eye drop prescriptions in H1 (Unit: KRW 100 mil, Data: UBIST) ‘Protect the KRW 300 bil annual market’...Pharma companies jointly prepare to respond to gov't reimbursement reevaluations However, whether the companies may continue their growth next year remains unknown, because the government included HA eye drops as a subject for reimbursement reevaluations. The government has selected 8 ingredients, including HA eye drops, as targets for reimbursement revelations this year. These include rebamipide, limaprost oxiracetam, acetyl Locartinie hydrochloride, loxoprofen sodium, levosulpiride, epinastine hydrochloride, and HA eye drops Among them, oxiracetam and acetyl L-carnitine hydrochloride are expected to fail to pass the reimbursement revelations as they failed to pass the clinical reevaluations conducted by the Ministry of Food and Drug Safety. Among the 8 ingredients, HA eye drops have the largest scale in terms of number of items and market size. 46 pharmaceutical companies have been selling 77 HA eye drop products in 1H this year. The government has already received related data from each pharmaceutical company and started the practical review. The outline of HA eye drops’ adequacy of reimbursement will be revealed next month at the earliest. After holding an objection period for the pharmaceutical industry, the final result of the reimbursement reevaluation will be determined by the end of this year. From the industry’s perspective, the companies will inevitably experience serious aftermath if the government concludes that the drug lacks grounds to be determined adequate for reimbursement. This is why the companies have been jointly preparing a response to the reevaluation results. About 10 companies, centered around Optus Pharm, have been concentrating on developing a countermeasure with large law firms in Korea. As in the case of choline alfoscerate, there is a possibility that the companies will file a class action lawsuit against the government. If they do file a class action lawsuit, the suit will likely be subject to the drug expenditure refund and redemption law that passed the National Assembly this year. The law, which will be enforced on November 20th, allows for the recovery or refund of drug costs that were already paid according to the outcome of the lawsuit if a pharmaceutical company delays government actions such as drug price cuts by applying for a suspension of execution along with an administrative lawsuit. Some companies have also started to develop alternatives in preparation for the market withdrawal of HA acid eye drops. In June of last year, Samil Pharmaceutical and Kukje Pharm received permission for Reba-K eye drops and Reba-i eye drops that contain rebamipide to treat dry eye syndrome. Daewoo Pharm is also known to be developing an eye drop product with the same ingredient.
- Policy
- Sotyktu, the world's first TYK2I, received domestic approval
- by Lee, Hye-Kyung Aug 04, 2023 05:38am
- The Ministry of Food and Drug Safety (Minister Oh Yoo-Kyung) approved BMS Korea's new plaque psoriasis drug 'Sotyktu 6mg' on the 3rd. Psoriasis is a chronic skin disease caused by immune abnormalities, and plaque psoriasis is the most common form. Sotyktu treats psoriasis by selectively inhibiting 'tyrosine kinase-2 (TYK2)' receptors related to inflammation and immune response by oral administration. The drug was approved by the US FDA in 2022 for use in adult patients with moderate to severe psoriasis who require systemic therapy or phototherapy. Sotyktu is the world's first and only TYK2 inhibitor approved for use and is the first oral drug available for moderate to severe psoriasis in 10 years. It is based on the comparative phase 3 POETYK PSO-1 and POETYK PSO-2 studies. This drug confirmed improved efficacy at 16 and 24 weeks after administration compared to the placebo and Otezla, and the clinical efficacy was found to last up to 52 weeks. This drug is expected to provide a new treatment opportunity for adult patients with moderate to severe plaque psoriasis who require phototherapy or systemic treatment. The Ministry of Food and Drug Safety said it would continue to do its best to expand treatment opportunities for patients by promptly supplying treatments whose safety and efficacy have been sufficiently confirmed based on regulatory science expertise.
- Policy
- Cancer immunotherapy Jemperli passes DREC review
- by Kim, Jung-Ju Aug 04, 2023 05:38am
- Jemperli GSK Korea’s immno-oncology latecomer drug ‘Jemperli (dostarlimab)’ has passed its first step to reimbursement in Korea. On the other hand, Koselugo (selumetinib), AstraZeneca’s new drug for pediatric patients with neurofibromatosis type 1 (NF1), received a redicussion decision, warning of a bumpy journey ahead. The Health Insurance Review and Assessment Service held its 8th 2023 Drug Reimbursement Evaluation Committee meeting and deliberated on the adequacy of reimbursement of 6 new drugs and made the decisions above. New drugs that were deliberated and deemed eligible for reimbursement include Jemperli and Ono Pharma Korea’s BRAF inhibitor for colorectal cancer, ‘Bratovi Capsule 75mg (encorafenib)’, and Roche Korea’s treatment for adult patients with neuromyelitis optica spectrum disorder (NMOSD), ‘Enspryng Prefilled Syringe (satralizumab)'. Among the drugs, Jemperli is the first drug in its class to be approved to treat adult patients with primary advanced or recurrent endometrial cancer that is mismatch repair deficient (dMMR) or microsatellite instability-high (MSI-H) that has progressed on or following a prior platinum-containing regimen. BMS Korea’s ‘Zeposia Capsule 0.92mg‘ and ‘Zeposia Capsule Starter Pack 0.23mg/0.46mg (ozanimod hydrochloride) received a ‘conditional approval,’ allowing the drugs to proceed on to drug pricing negotiations with the National Health Insurance Service if the company accepts a price below the evaluated amount. These new drugs are indicated to treat moderate-to-severe ulcerative colitis (UC). AstraZeneca Korea’s Koselugo Capsule 10,25mg (selumetinib hydrogen sulfate) received a rediscussion decision. The drug is indicated to treat NF1 accompanied by plexiform neurofibroma, Koselugo had received a non-reimbursement decision by DREC in March last year, but attention had been rising on the possibility of it passing DREC review this time as the company immediately submitted supplementary data thereafter in May and a risk-sharing proposal this time. Although DREC did not decide to non-reimburse the drug this time, the industry predicts that it would take some time for Koselugo to receive reimbursement as DREC has temporarily postponed making a reimbursement decision on the drug. On the other hand, BMS Korea’s Reblozyl Inj 25,75mg (luspatercept) received a non-reimbursement decision. Reblozyl was designated an orphan drug in 2021 in Korea and is used to treat ▲ myelodysplastic syndromes with ring sideroblasts (MDS-RS) or with myelodysplastic/myeloproliferative neoplasm with ring sideroblasts and thrombocytosis (MDS/MPN-RS-T) and ▲anemia in adult patients with beta-thalassemia who require regular red blood cell (RBC) transfusions.
- Company
- Prescriptions of pseudoephedrine rise 35% in 1H
- by Chon, Seung-Hyun Aug 04, 2023 05:38am
- The prescription market for ‘pseudoephedrine,’ which continues to show an unstable supply and demand in the market, has expanded significantly. Despite the end of the pandemic, demand for the drug has increased significantly due to the increase in flu and cold patients. Accordingly, the need to raise the insurance price ceiling for the low-priced drug is being raised to increase production. According to the market research institution UBIST, outpatient prescriptions of the single-agent pseudoephedrine had been KRW 3.3 billion, up 35.3% YoY. Prescriptions of pseudoephedrine had increased 20.5% YoY to reach KRW 1.6 billion in Q1, and then 53.1% YoY to record KRW 1.7 billion in Q2 this year. Pseudoephedrine is used for common colds, paranasal sinusitis, upper respiratory allergies, etc. It is a prescription drug that is widely prescribed to cold patients. Quarterly outpatient prescriptions of single-agent pseudoephedrine (Unit: KRW 0.1 billion, Data:UBIST) Although the market size for pseudoephedrine had not been large, its demand has surged recently. Quarterly prescriptions of single-agent pseudoephedrine had been below KRW 1 billion from Q2 2020 to Q3 2021. However, after 7 quarters in Q4 2021, prescriptions exceeded KRW 1 billion and then rose to KRW 1.7 billion in Q4 last year. By Q2 this year, pseudoephedrine prescriptions had increased 149.9% from Q2 2020. In 2020 and 2021 the pseudoephedrine market was directly affected by COVID-19 and caused the market contraction. The number of patients with infectious diseases such as flu and colds had decreased sharply after the spread of COVID-19 due to strengthened personal hygiene management such as washing hands and wearing masks, and shrinking related treatment markets. However, the demand for pseudoephedrine increased significantly with as many as hundreds of thousands of confirmed COVID-19 cases pouring out every day from the end of 2021. The recent increase in pseudoephedrine prescriptions seems to be not only due to confirmed COVID-19 patients but also due to the increase in cold and flu patients. This year, the number of flu patients had continued to exceed the epidemic standard until June. According to the Korea Disease Control and Prevention Agency, the number of suspected influenza patients per 1,000 outpatients dropped from 52.5 in the first week of 2023 to 11.7 in March. However, the number of suspected flu cases rose again from April. For 6 consecutive weeks from the 17th week (fourth week of April) to the 22nd week last week of May), the number of suspected influenza patients per 1,000 outpatients continued to be over 20. This greatly exceeds the flu epidemic standard of 4.9 set by the KDCA. The supply shortage of pseudoephedrine is being prolonged. Its demand has soared in a short period, but due to limited production, supply is not keeping up with the demand. The Korean Pharmaceutical Association is working with the Korea Pharmaceutical Distribution Association and related pharmaceutical companies to ensure an even supply of single-agent pseudoephedrine. The KPA has been receiving online orders from pharmacies for Samil Pharma’s ‘Sudafed Tab’ or Kolon Pharmaceutical’s ‘Cosue.’ Pharmacies can order one of the two, and the KPA has been supplying one or two packer vials (500 tablets/vial) of the drug to each pharmacy. The low price of pseudoephedrine is another reason why companies cannot easily increase production of pseudoephedrine. According to the Health Insurance Review and Assessment Service, seven single-agent pseudoephedrine 60mg are listed for reimbursement in Korea, and their insurance price ceiling is a mere KRW 23. Of the 7, 6 products were listed at KRW 23 and 1 at KRW 20. If the supply shortage continues, the industry suggested that there may be a need to raise its drug price like acetaminophen. The Ministry of Health and Welfare raised the insurance price ceiling of 18 acetaminophen 650mg items by up to 76.5% in December last year. The insurance price limit for 650mg acetaminophen, which was only KRW 43 to 51 before then, was raised to KRW 90. The government made an unprecedented decision to raise the price of all acetaminophen products unilateraly when pharmaceutical companies expressed reluctance to increase production due to the drug’s poor cost structure. However, it is a temporary increase that will be adjusted to KRW 70 from December this year. Pharmaceutical companies had promised to increase production of acetaminophen in line with the price hike.
- Company
- Antibiotic resistance is serious
- by Jung, Sae-Im Aug 04, 2023 05:37am
- Antibiotic resistance has been a global issue for several years. In 2016, the British government predicted that 700,000 people worldwide would die from antibiotic-resistant bacteria every year and that the death toll would reach 10 million by 2050 if no special measures were taken. In 2015, the World Health Organization (WHO) presented a 'global action plan' to prevent the occurrence and spread of resistant bacteria transcending borders and urged countries to prepare countermeasures and international cooperation. Carbapenem-type antibiotics, which have the most extensive antibacterial activity, are called the 'last choice', and the number of infections with strains resistant to these antibiotics is rapidly increasing. According to the Korea Centers for Disease Control and Prevention, the number of cases of carbapenem-resistant Enterobacteriaceae (CRE), which was classified as a second-grade infectious disease last year, increased by 31% from the previous year to 30,548. Compared to 2017, when there were 5717, this is a more than five-fold increase in five years. Activities of the Korean Society for Antimicrobial Therapy also focus on antibiotic management to reduce resistance. It is the institutionalization of so-called 'antibiotic stewardship' that uses antibiotics appropriately. As of 2020, antibiotic use in Korea was the 4th highest among 29 OECD countries. As a result of investigating the adequacy of antibiotic prescriptions in 75 domestic hospitals, more than 25% were evaluated as inappropriate. Kim Shin-woo, chairman of the Korean Society for Antimicrobial Therapy Kim Shin-woo, chairman of the Korean Society for Antimicrobial Therapy (Department of Infectious Diseases, Kyungpook National University Hospital), said in a recent interview with Dailypharm, “Antibiotic resistance has become a situation that cannot be resolved through infection control anymore.” There is a need for systematic institutionalization of education,” he stressed. He then asked for appropriate support to quickly introduce new drugs that have emerged as carbapenem alternatives such as Zerbaxa at an appropriate price and to develop domestically produced new drugs that can be supplied in Korea. - Cases of infection with carbapenem-resistant Enterobacteriaceae are rapidly increasing. How much do you feel on-site? = The level of feeling on the spot is serious. The problems typically pointed out in Gram-negative bacteria resistance are resistance in Pseudomonas aeruginosa, Acinetobacter resistance, and Carbapenem resistance. Among them, Pseudomonas aeruginosa and Acinetobacter mainly cause problems in hospital environments, but carbapenem resistance has a greater ripple effect on the community than these and is more serious because resistance is developed in intestinal bacteria. CRE is a case where doctors do not respond even if they use carbapenem antibiotics, which are called 'last bastions', and colonization can lead to a real infection. If the CRE ratio in a person's bacteria increases, it causes various infections such as sepsis, which cannot be treated even with carbapenem. However, there is no monitoring, tracking, or reporting on this, so there is nothing that can be done even if the number of deaths increases. - Antibiotic resistance is a problem that has been pointed out for a long time. Why was the response delayed? = I think the biggest cause is not knowing the seriousness of antibiotic resistance. About 35,000 patients have died from COVID-19 in Korea, but it is not well-known that many people die every day from antibiotic resistance. There is also a famous prediction that by 2050, we will be in a post-antibiotic era, an era in which antibiotics are ineffective, with more deaths from antibiotic resistance than from cancer. Awareness of the term antibiotic stewardship is also very low. Medical institutions should be moved through continuous publicity and awareness improvement. -The Korean Society of Antimicrobial Therapy is citing institutionalization of the Antimicrobial Stewardship Program (ASP) as its top priority goal in the mid-to-long term. Why is Antibiotic Stewardship Institutionalization Necessary? =Antibiotic stewardship is to reduce the misuse and abuse of antibiotics and lower the rate of resistant bacteria. When antibiotic resistance develops, infection control and antibiotic stewardship to prevent the spread of resistant bacteria and the spread of legally infectious diseases must go hand in hand. As in Korea, there is no need to reduce the use of antibiotics in a situation where fee-for-service, high access to medical care, and an increase in the number of elderly people exist. It is easy to use antibiotics even for colds and to use for 3 days for 1 or 2 weeks. Therefore, an organization that monitors antibiotic use and gives feedback is essential. - Specifically, how to operate ASP =The Korean government is making phase 1 and phase 2 measures (national antibiotic resistance management measures) for antibiotic resistance. Institutionalization of antibiotic stewardship is needed in earnest. If there is no system, execution will not follow. A system must be in place so that medical staff can accept feedback on why antibiotics must be managed and used sparingly, and why antibiotics are used a lot. In general, an ASP team consists of a multidisciplinary team led by an infectious disease physician, a pediatric infection specialist, and a pharmacist specializing in infection, together with other departments (information computing team, clinical microbiology, medical quality control, infection control room, etc.). In developed countries where the system is being operated first, nurses are often included in the number of dedicated personnel. In overseas cases, large hospitals use a system that monitors antibiotics prescribed by doctors and supports prescriptions, checks the contents of prescriptions electronically, controls them when necessary, and gives feedback. -Zerbaxa is a new drug that can reduce the use of carbapenem antibiotics. It seems important to use new antibiotics well. = Recently developed antibiotics are trying to prove their therapeutic effect on pneumonia such as complicated urinary tract infection, complicated intra-abdominal infection, ventilator pneumonia, and hospital-acquired pneumonia. Zerbaxa has won these indications. If Zerbaxa can be used regardless of whether or not ESBL is produced, the use of carbapenems can be reduced. However, if the cost of using a new drug is high, carbapenem, which costs relatively less, will be used. Of course, if the use of Dr. Jeo increases, there are concerns about tolerance. Because Dr. Jeo has not yet been widely used, information on tolerance is lacking.
- Policy
- Animal drugs can be manufactured at human drug factories
- by Kang, Shin-Kook Aug 03, 2023 05:35am
- The manufacture of veterinary drugs are now allowed at manufacturing facilities that manufacture human drugs. The Ministry of Agriculture, Food and Rural Affairs recently announced the legislation of an amendment to the ‘Decree on the Facility Standards for Veterinary Pharmacies and Manufacturers, Importers, and Retailers of Veterinary Drugs’ and will be collecting industry opinion until September 4th. According to the amendment, in order to minimize the impact on the existing industry, the products that can be manufactured at human drug manufacturing facilities are limited to those for companion animals. In addition, among the active pharmaceutical ingredients that have been approved for human use in Korea, those that have not been approved for animal use before August 31, 2023, can be manufactured until the expiry date. However, 22 ingredients that have been approved for both human and veterinary use are allowed for use in manufacturing drugs for companion animals. The amendment will take effect immediately after promulgation. The Ministry of Agriculture and Food and Rural Affairs said, "We seek to ease the burden of redundant investment that is required for the establishment of a separate manufacturing facility for veterinary drugs by enabling human drug manufacturers to produce drugs for companion animals using existing facilities." The goal of the amendment is to develop and produce high-value-added veterinary drugs and grow the domestic veterinary drug industry qualitatively and quantitatively through coexistence with existing veterinary drug manufacturers.”
- Product
- Discontinued muscle painkillers in heavy demand
- by Kim JiEun Aug 03, 2023 05:35am
- The aftermath of some pharmaceutical companies’ tricking pharmacies into believing ‘there is not much stock available for order’ An ironic situation has occurred where a drug that pharmaceutical companies have given up manufacture and sales of due to the inability to demonstrate its effect is in heavy demand at pharmacies. On the 1st, in drug online malls used by pharmacists, many muscle pain relievers that are fixed-dose combinations of chlorzoxazone 250mg + acetaminophen 300mg, are out of stock, or can only be ordered in small quantities. The issue arose after it was recently confirmed that pharmaceutical companies that manufacture and distribute related products voluntarily withdrew their products or did not apply for renewal of the product licenses. The stock shortage occurred because some pharmacies are trying to stockpile the drug to sell within the expiration date as the pharmaceutical industry as well as the pharmacist society believe the drug will be naturally removed from the market. As pharmaceutical companies have voluntarily withdrawn or did not apply for renewal within the deadline, pharmacies can only sell those in stock until the expiry date of the currently distributed products. According to pharmacists, most of the muscle pain relievers that contain chlorzoxazone 250mg + acetaminophen 300mg are used at pharmacies and the combination is one of the steady sellers at pharmacies. As a result, some pharmacies trying to stock as much as possible and sell the product until possible. In addition, the fact that some pharmaceutical companies implemented a sales policy of “imminent closing” had also partially affected the out-of-stock phenomenon of related products. In the case of pharmaceutical company A, immediately after applying for voluntary withdrawal of related products, it sent a text message to pharmacists using its online mall to encourage them to order related products. A sales representative of the pharmaceutical company encouraged pharmacies to buy the product, sending the message that “manufacture will be discontinued and the current stock is our last. Those in stock can be sold until the expiration date. Therefore, it would be good to order as much as possible.” It is known that the number of orders that can be ordered per pharmacy has been limited to 300 for each specific time, but orders poured in in a very short period of time. Some pharmacists said it was regrettable that over-the-counter products with certain ingredients, which are in constant demand in pharmacies, are being removed. It seems necessary to look at the overall situation to see if the system is being excessively applied or whether related pharmaceutical companies are being complacent.” A pharmacist in Seoul said, “Most pharmacies have one or two products with the combination, and the demand for them is quite high, it is unfortunate that relevant products are being withdrawn from the market. It seems that the related pharmaceutical companies need to look at the overall situation to see if they are being complacent.”