- LOGIN
- MemberShip
- 2026-04-09 21:05:43
- Policy
- HIRA declines NHIS and KPBMA's request
- by Lee, Tak-Sun Jul 14, 2023 05:39am
- The National Health Insurance Service and the Korea Pharmaceutical and Bio-Pharma Manufacturers Association have requested to participate in the Drug Reimbursement Evaluation Committee, but the Health Insurance Review and Assessment Service announced plans to amend its operational regulations without reflecting their requests. The NHIS has been strongly pushing to take part in the Drug Reimbursement Evaluation Committee since earlier this year, which drew attention to the recent amendment of the operational regulations. According to HIRA on the 13th, the amendment to the operational regulations for the Drug Reimbursement Evaluation Committee will be officially announced soon and will incorporate the contents pre-announced last month. The pre-announced contents of the amendment included increasing the committee's member pool from 100 to 105 and increasing the number of experts recommended by academic societies from 65 to 70. HIRA will reorganize the specific departments for specialized experts among those recommended by the Korean Medical Association by removing the Korean Society of Gastrointestinal Cancer Research and adding 4 specialized departments including the Korean Orthopedic Association, the Korean Society of Coloproctology, the Korean Society of Medical Oncology, and the Korean Society of Surgical Oncology, HIRA made a pre-announcement of its administrative decision on the 21st of last month and has collected opinions until the 27th of the same month. After the announcement, the NHIS immediately offered to take part in the Drug Reimbursement Evaluation Committee. The NHIS has been emphasizing the need for their participation in the committee to enable smooth operation of tasks related to drug reimbursement.since earlier this year. While NHIS participated in the 3rd Drug Reimbursement Evaluation Committee, they were not given voting rights due to a conflict of interest and are now no longer involved in the committee. In March, Sang-Il Lee, Executive Director for Benefit at NHIS, argued for NHIS’s participation in the Drug Benefit Evaluation Committee during a briefing with the Korea Special Press Association. He said, "NHIS’s participation in the Drug Reimbursement Evaluation Committee is necessary to ensure the appropriateness of reimbursement, secure consistency of financial impacts when listing new drugs, and serve as an organic link between the evaluation and negotiation conducted on the uncertainty of new drugs under the risk-sharing agreement. We will further discuss this matter with relevant organizations, such as MOHW and HIRA.” Subsequently, the National Health Insurance Trade Union and the Korea Alliance of Patients Organization also supported the participation of NHIS in the Drug Reimbursement Evaluation Committee. The Korea Alliance of Patients Organization submitted a letter of opinion in favor of NHIS’s participation when the amendment to the operating regulations for the Drug Reimbursement Evaluation Committee had been pre-announced. However, HIRA showed a negative stance toward NHIS’s participation in the Drug Reimbursement Evaluation Committee. Mi-Young Yoo, Head of the Department of Drug Management at HIRA stated during a briefing in March that “As an insurer, NHIS directly negotiates with pharmaceutical companies on the ceiling price for new drugs, etc. Concerns may arise about fairness and objectivity if the negotiating party participates in the decision-making process,” essentially rejecting their opinions. In addition to the NHIS, the Korea Pharmaceutical and Bio-Pharma Manufacturers Association also submitted their opinion for its participation in the Drug Reimbursement Evaluation Committee during the pre-announcement period, with the aim of ensuring transparency in the process of reimbursement decisions. However, their opinions to take part in the Drug Reimbursement Evaluation Committee were not accepted. The official announcement is expected to align with the pre-announced contents. Thus, NHIS and KPBMA representatives will not be part of the 9th Drug Reimbursement Evaluation Committee that is set to commence in October.
- Policy
- Approval of phase 3 for Retatrutide
- by Lee, Hye-Kyung Jul 14, 2023 05:38am
- Eli Lilly's anti-obesity drug Ly3437943 is undergoing phase 3 clinical trials in Korea. The Ministry of Food and Drug Safety recently approved a phase 3 clinical trial to study the efficacy and safety of once-weekly administration of Retatrutide in obese or overweight type 2 diabetic subjects. Seoul National University Hospital, Severance Hospital, Ulsan University Hospital, Hanyang University Guri Hospital, Korea University Ansan Hospital, and Yeungnam University Hospital participate in the phase 3 clinical trial conducted worldwide. Retatrutide is a triple agonist of Lilly's GIP/GLP-1/glucagon (GCG) receptor, which is famous as an obesity treatment following Mounjaro. Mounjaro was clinically confirmed to have a 22.5% weight loss effect when administered at the highest dose of 15mg/0.5ml for 72 weeks in obese patients without diabetes. approved as an adjuvant in therapy. The indication for obesity treatment plans to receive approval from the FDA within the next year. On the other hand, Retatrutide showed a weight loss effect of 24.2% after 48 weeks in the 12mg group, the highest dose group, in phase 2 clinical trial conducted on 281 adults aged 18 to 75 years at the recently opened ADA. It is evaluated that the combination of glucagon receptor action with glucose-dependent insulinotropic polypeptide (GIP) and glucagon-like peptide-1 (GLP-1) receptor action led to the weight loss effect of Retatrutide. GLP-1 was developed as a diabetes treatment because it promotes insulin secretion and lowers blood sugar levels, but as the side weight loss effect was confirmed, it is being used as an obesity treatment. Meanwhile, representative drugs such as GLP-1 agonists include Novo Nordisk's Saxenda and Wegovy.
- Policy
- HIRA is reviewing reimb of Korea’s first donepezil 3mg
- by Lee, Tak-Sun Jul 14, 2023 05:38am
- Donepezil Powder released by Hyundai Pharmaceutical last year The 3mg donepezil tablet that was developed by Hyundai Pharmaceutical is being reviewed by the health authorities for reimbursement in Korea. No other tablet formulation of donepezil 3mg currently exists in Korea’s reimbursement list. Therefore, the Health Insurance Review and Assessment Service is seeking for the most appropriate development target product to determine its price. Hyundai Pharmaceutical received approval for Hipezil Tab 3mg (Donepezil hydrochloride monohydrate) on the 28th of last month. Hipezil Tab 3mg is indicated to slow the progression of symptoms of dementia associated with Alzheimer's disease and is the first 3mg donepezil formulation introduced to the market. Although the 3mg dose is being used abroad, Hyundai is the first in Korea to receive approval for the specific dose. Currently, 5mg, 10mg, and 23mg formulations of donepezil are listed for reimbursement in Korea. The 3mg donepezil tablet has a specific use. Its dosing schedule indicates that an initial dose of 3mg once daily can be used for the purpose of reducing gastrointestinal adverse reactions. However, the use of the 3mg dose is conditional and limited to 1-2 weeks. Also, the dosage for underweight female patients aged 85 or older is not allowed to exceed 5mg, therefore are allowed the use of the 3mg formulation. Hyundai Pharmaceutical is thought to have adopted the strategy of receiving approval for Hipezil Tab 3mg to target a less attended spot in the highly competitive donepezil market. Hyundai Pharmaceutical has also released a powder-type formulation of donepezil, Hipezil Powder, that offers better convenience in intake. In the domestic donepezil market, competition among pharmaceutical companies is so fierce that 121 10mg tablets are on the reimbursement list. According to HIRA, the amount of claims submitted for dementia (ATC code N06D) in Korea amounted to KRW 414.9 billion in 2022, up 58.4% compared to 2020. The market is growing due to the increasing number of dementia patients and the normalization of hospital visits by the elderly with COVID-19 becoming endemic. Therefore, whether Hyundai Pharmaceutical’s strategy will show an effect is receiving industrywide attention.
- Policy
- ‘Refer to Japan’s half-price policy for generic's price'
- by Lee, Tak-Sun Jul 13, 2023 05:35am
- The Health Insurance Research Institute, a research institution under the National Health Insurance Service, stressed the need to refer to Japan’s generic drug price reduction policies in its recently published ‘Issue & View June 2023.’ Hyun-Ok Lee, Associate Research Fellow of the Health Insurance Research Institute, and her fellow researchers published an article containing the need above in the June issue of Issue & View under the title of 'Recent Changes in Japan's Drug Pricing System and Implications'. The authors argued that it is necessary to refer to and reflect on Japan’s generic drug pricing system in the paper’s conclusion and implications. The research team said, “Korea’s elderly population continues to increase, and the country is expected to enter a super-aged society by 2025. Therefore, various reforms need to be made to Korea’s current pricing system in preparation for a super-aged society, and we should refer to Japan's revised drug pricing system in the process.” As its background, the paper pointed to the rise in drug expenditures that followed the rise in multiple chronic diseases caused by population aging. The medical expenditures in Korea’s national health insurance from KRW 43.6 trillion in 2010 to KRW 86 trillion in 2019, and health insurance drug expenditures increased from KRW 12.7 trillion in 2010 to KRW 19.4 trillion in 2019. In other words, drug expenditures accounted for 24.1% of the total medical expense in 2019. “In Korea, an equal price policy between original and generic drugs was implemented in 2012. Based on the system, the upper limit of insurance listing price for generic drugs was set at 59% of the initial original drug price in the first year, then at 53.55% from the second year. In Japan, generic drug prices are set at 50% of the original drug price, and generic drugs that are listed thereafter are set at the same price as the lowest price of previously listed generics.” Japan also has a drug price system where if 10 or more generic drugs are listed, their price is set at 40% of the original price, and at 90% of the lowest price when 20 or more are listed. Unlike Korea, Japan implemented a claim addition system to encourage the use of generic drugs. Japan's first generic drug price (50%) is lower than that of Korea (53.55%). The method of lowering drug prices according to the number of generics introduced has also been introduced in Korea, but the difference is that Japan applies the standard from 10 or more, and Korea from 20 or more. The authors explained, "Korea has conducted drug price reevaluations for already-listed drugs in 2012 and unilaterally adjusted drug prices in 2012, but has not been regularly conducting drug price reevaluations thereafter. However, Korea has a post-marketing Price-Volume Agreement system in place that lowers prices as a drug’s use volume increases. “On the other hand, Japan has been conducting an annual drug price survey on all items since 2020 to adjust its drug prices to reflect actual market prices. Korea should also refer to Japan’s drug price adjustment policy that reflects actual transaction price and generic price decision method to improve Korea’s drug pricing policy in the future, ahead of the surge in pharmaceutical expenses that may occur in line with the rapidly aging population.” In other words, the paper stressed the need to periodically make price adjustments to generic drugs in Korea. The paper is expected to serve as a basis for raising the legitimacy of generic drug price cuts at a time when the government has recently started measures to improve the drug price cut investigation system and reform the generic drug price system.
- Company
- "Cibinqo secures solid data in atopic dermatitis"
- by Jung, Sae-Im Jul 13, 2023 05:35am
- 'Cibinqo (abrocitinib),' the third JAK inhibitor to be introduced for atopic dermatitis in Korea, will be reimbursed starting this month. Healthcare professionals have expressed high expectations for Cibinqo, as the drug has accumulated robust data in treating severe atopic dermatitis. On the 11th, the Korean pharmaceutical company Hanmi Pharmaceutical held a press conference to celebrate the reimbursement of Cibinqo, an oral atopic dermatitis treatment, at the Seoul Dragon City Hotel in Yongsan-gu, Seoul. Professor Yong-Hyun Jang of the Department of Dermatology at Kyungpook National University Hospital explained the background and significance of Cinbinqo's reimbursement for atopic dermatitis in Korea. Professor Yong-Hyun Jang of the Department of Dermatology at Kyungpook National University Hospital is presenting at the press conference celebrating the reimbursement and listing of Cibinqo Cibinqo is a Janus kinase 1 (JAK1) inhibitor that was approved by the Ministry of Food and Drug Safety in November 2021. It is also the 4th JAK inhibitor in Korea and 2nd JAK class treatment to be introduced by Pfizer. Unlike Xeljanz, which is used only for ulcerative colitis, Cibinqo can also be used for the treatment of severe atopic dermatitis. Cibinqo was granted reimbursement this month. The 50mg, 100mg, and 200mg doses of Cibinqo were listed at KRW 11,087, 17,739, and 25,942 respectively. The ceiling price is known to be set at 88% of the calculated average price of its substitutes. With the listing of Cibinqo, patients with severe atopic dermatitis now have a much wider range of new treatment options. In addition to the biological agent ‘Dupixent’ that was first introduced, 3 JAK inhibitors – the already-reimbursed Olumiant (baricitinib) and Rinvoq (upadacitinib), and Cibinqo – are now available for use in Korea. Among these treatments, Dupixent, Rinvoq, and Cibinqo can be used in adolescent patients aged 12 and above. Although being a latecomer, Cibinqo received recognition for having acquired solid data in atopic dermatitis from various clinical trials. 6 Phase III clinical trials, including JADE DARE, JADE COMPARE, and JADE TEEN, have been conducted. Cibinqo was the first drug that attempted to directly compare (head-to-head) its efficacy and safety with the biological agent Dupixent, and Cinbinqo was observed for the possibility of deterioration upon dose reduction or discontinuation of treatment after initial response, and evaluated on its ability to recover from the reaction. By confirming the drug’s effect on patients that showed no response to Dupixent, the company accumulated grounds for patients to switch to Cibinqo. The rapid improvement of itch, which was observed in various clinical trials of Cibinqo, is expected to significantly improve patient’s quality of life. Jang explained, "Itch is a very important symptom in atopic dermatitis. It causes patients to scratch their skin, weakens the skin barrier, and exacerbates inflammation as a secondary reaction. Cibinqo showed a rapid and significant improvement in itch from the day after initial administration, increasing patients' quality of life. Also, more patients reached the target response rate for the Eczema Area and Severity Index (EASI). We believe Cibinqo will benefit patients who are suffering from severe itch." Cibinqo has also demonstrated additional benefits in patients who did not respond to Dupixent. The JADE EXTEND study investigated the effect of switching to Cibinqo from Dupixent in patients who received Dupixent in the JADE COMPARE study. The patients were further divided into those that responded to Dupixent and those that didn’t respond to Dupixent. In the arm that did not respond to Dupixent, 80% achieved EASI-75 (75% improvement based on the Eczema Area and Severity Index). Also, 77% achieved an improvement of 4 points or more on the Peak Pruritus Numerical Rating Scale (PP-NRS). This is expected to serve as evidence when companies of JAK inhibitors apply for reimbursement extensions to cover patients switching from Dupixent. Jang stated, "Unlike other JAK inhibitors, Cibinqo holds significance as it conducted various studies solely in the field of atopic dermatitis, which is significant. With the basis for switching treatments established, we expect reimbursement extensions to follow in the near future. We are currently collecting various opinions in academia as well.”
- Policy
- Will the number of generics decrease?
- by Lee, Tak-Sun Jul 13, 2023 05:35am
- Analysts say that the government has begun strengthening the standard for cascading drug pricing in order to reduce the number of generics. A plan to reduce the existing list of 20 items, which is currently used as the standard for lowering the upper limit of new items, to 10 seems promising. However, the industry points out that there is a limit to reducing the number of large generics even if the tiered drug pricing standard is strengthened. Rather, it is predicted that it will only encourage development. The Ministry of Health and Welfare recently entrusted a research service to Kongju University professor Kim Dong-sook's team on the subject of 'preparation of measures to improve the drug price system for generic drugs'. In this study, the appropriateness of 20 products, which is the standard for the number of products to which differential price is applied, will be addressed. The 20 baselines were introduced in 2020 with the reorganization of the drug pricing system. If a generic enters the market with more than 20 identical products already listed, the upper limit will be set at 85% of the lower price between the lowest price of the same product and 38.69%. Unfortunately, the NHIS-affiliated Health Insurance Research Institute recently introduced the Japanese generic drug pricing system through a publication in the June 2023 issue and View, arguing that it needs to be reflected in the domestic system. In particular, when the number of generics is 10 or more, the generic price is calculated at 40% of the original price, and when there are 20 or more, the system is set at 90% of the lowest price. There was an atmosphere that the current baseline of 20 needs to be reduced to 10 like Japan. It is known that the Ministry of Health and Welfare, NHIS, and HIRA have also mentioned the plan to have a strict baseline for generic drug price cuts. Accordingly, the industry is of the opinion that it is not possible to further strengthen the cascading drug pricing system by creating a clear basis through research services. However, it is pointed out that there is a limit to reducing the number of large generics no matter how low the baseline for cascading drug pricing is. The cascading drug pricing system will be applied from the month after the first generic is listed. Therefore, in the first month, there is no penalty even if the number of entries exceeds 20. In the case of Forxiga's generic 10mg, which served as an opportunity for insurance authorities to pursue a plan to reduce the number of generics, 57 were listed in the first month of April alone. Most of the generic companies tried to register their products according to the patent expiry schedule to avoid the cascading drug pricing system. For the next two months, only one of Forxiga's generic 10mg was listed. Industry analysts say that generics with a large market size, such as Forxiga's generic, are highly likely to be listed on a large scale in the first month regardless of the cascading drug pricing baseline. The same is true for Januvia generics scheduled to be listed in September. Therefore, in Korea where generic competition is fierce, even if the price cut baseline is reduced from 20 to 10, the number of large generics will not change significantly. Rather, it is pointed out that generic companies can only waste social costs by trying to develop without asking regardless of expected performance in order not to be late for entering the insurance coverage. An official from the pharmaceutical industry explained, "There is a possibility that generic companies will flock to patent challenges at once to get reimbursed early." However, if the baseline is set from 20 to 10, the effect of not increasing the number of listed generics will be more significant. However, it is also pointed out that there is a limit to reducing the total number of generics. Another official in the pharmaceutical industry said, “Although Japan has generally reduced the price of generic drugs, it is different from Korea in that it is simultaneously promoting generic incentives, such as actively granting incentives to doctors and pharmacists for generic prescriptions.” He criticized, "Our insurance authorities are only concentrating on lowering the price of generic drugs and seem to have no interest in encouraging their use."
- Company
- Schizophrenia drug Invega Hafyera lands in general hospitals
- by Eo, Yun-Ho Jul 13, 2023 05:35am
- The long-acting formulation of the schizophrenia drug ‘Invega’ has landed in general hospitals in Korea. According to industry sources, Invega Hafyera 1092mg/1560mg (paliperidone palmitate), Janssen Korea’s extended-release injectable suspension for patients with schizophrenia that is injected every 6 months, has passed the drug committees of medical institutions in Korea, including Seoul Asan Medical Center. Janssen has been continuously working to create a prescription environment since it was listed for reimbursement in May. Invega Hafyera was approved by the Ministry of Food and Drug Safety in September 2022. The 1-month extended-release injectable formulation of the same drug, Invega Sustenna, had been approved in July 2010 and granted reimbursement in November 2015. With reimbursement, the drug is covered for all patients with schizophrenia in Korea, including first-episode patients. The 3-month extended-release injectable formulation, Invega Trinza, was approved in June 2016 and is being reimbursed from September 2016. Invega Hafyera is indicated for use in patients who have been adequately treated with: Invega Sustenna (once-a-month extended-release injectable suspension) for at least 4 months; or Invega Trinza (every-three-month extended-release injectable suspension) for at least one three-month cycle. Patients who wish to switch to Invega Hafyera from Invega Sustenna 156mg receive Invega Hafyera 1092mg, and those switching from Invega Sustenna 234mg receive Invega Hafyera 1560mg as an initial dose. In the case of Invega Trinza 546mg and 819mg, patients may switch to Invega HafyeraTM 1092mg, and 1560mg, respectively. The safety and tolerance of the every-six-month injectable Invega Hafyera were confirmed through the PSY3015 study. Se Hyun Kim, Professor of Psychiatry at Seoul National University Hospital, said, “Invega Hafyera can offer a broader range of benefits to patients who have seen a stable effect with Invega Sustenna or Invega Trinza. The new formulation offers an opportunity to lower the barrier to injectable treatments for schizophrenia patients in Korea.” Kim added, “The long-acting injectable formulation allows continuous treatment with its stable medication adherence and improved convenience, and can provide benefits such as returning to society and regaining confidence for patients with schizophrenia.” In schizophrenia, the risk of worsening or relapsing symptoms is high with low medication adherence. The long-acting injectable formulations offer a benefit in that aspect as it improves medication adherence over oral formulations, reduce medical cost and relapse or rehospitalization rates, improve symptoms and social function of the patients (when switching to a long-acting injectable formulation), and offer convent administration. The treatment guidelines for schizophrenia in Korea allow long-acting injectable formulations to be administered to patients in all patients, from early to chronic stages.
- Policy
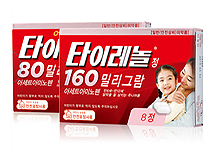
- 2 Tylenol products stopped production will be decided in 2Q
- by Lee, Jeong-Hwan Jul 13, 2023 05:35am
- In the second half of this year, the Ministry of Health and Welfare plans to discuss the cancellation of the safety household medicine designation and the designation of additional alternative medicines for two Tylenol items, which have stopped production due to the sale of Janssen Korea's Hyangnam plant. On the 11th, the Ministry of Health and Welfare Ministry of Health and Welfare's Pharmaceutical Affairs Policy Division announced through a press briefing on media reports that the response to Tylenol, a safe and household medicine that has been suspended for more than a year, was insufficient. Some items, such as Tylenol 80mg for children and Tylenol 160mg, were withdrawn in March 2022 due to the relocation of the Tylenol manufacturer's factory overseas. A media company reported a critical article claiming that the government was neglecting the two items even though they had not been supplied for a year and four months due to a halt in production. In response, the Ministry of Health and Welfare explained that there is a considerable amount of inventory that has already been produced and that there are items that are trying to re-permit after relocating the factory, so they are examining the situation in consideration of the use of inventory and the possibility of re-permission. In particular, it also announced that it would make a decision within the second half of the year on the cancellation of the designation of safety and emergency medicines for two items that have been discontinued and the designation of additional alternative medicines. Regarding the suspension of supply, the Pharmaceutical Affairs Policy Division said, “It is not that the supply to convenience stores has stopped because the stock that has already been produced is being distributed.” The Pharmacy Policy Division continued, "Tylenol tablets for children 80mg and Tylenol tablets 160mg are alternative items such as children's Ibuprofen syrup and children's Tylenol suspension." "We will quickly discuss and decide on the need to cancel the designation of safe household medicines for the two items that have been discontinued and the need to additionally designate alternative drugs. According to the safety and household medicine system, the designation decision is made in consideration of the ingredients, side effects, content, dosage form, awareness, and convenience of purchase of the item," the Pharmaceutical Affairs Policy Division said.
- Company
- Samsung Biologics, cumulative order amt exceeded 2 trillion
- by Hwang, Jin-joon Jul 11, 2023 05:41am
- Panoramic view of Samsung BiologicsSamsung Biologics announced on the 10th that it has signed a main contract for biopharmaceutical consignment manufacturing (CMO) worth 511.1 billion won from global pharmaceutical company Novartis. Through this contract, the cumulative amount of orders received this year increased to 2.3387 trillion won. This contract with Novartis was signed one year after the letter of intent (LOI) of 100 billion won in June last year. Samsung Biologics and Novartis signed a main contract with a size that is five times larger than the previous LOI. Samsung Biologics signed a CMO contract worth 1.2 trillion won with global pharmaceutical company Pfizer on the 4th before signing a large-scale contract with Novartis. The amount of orders received from Pfizer and Novartis alone amounts to 1.7 trillion won. This is similar to last year's total order amount of 1.7835 trillion won. Samsung Biologics is increasing large-scale, long-term contracts while securing major global big pharma customers. It has secured 13 of the top 20 global pharmas as customers. Samsung Biologics is strengthening its ability to win orders based on its competitiveness in the global No. 1 production capacity (CAPA), speed, and quality. Samsung Biologics started with the 30,000-liter plant 1 in 2011 and expanded the 154,000-liter plant 2 in 2013. In 2015, a third plant with a capacity of 180,000 liters was built. Starting in 2020, construction began on the 240,000-liter class 4 plant, the world's largest single plant, and began full operation last month. The total production capacity is 604,000 liters. Samsung Biologics started construction of its 5th plant in April to preemptively respond to the market's demand for biopharmaceutical CMO. Completion is targeted for April 2025. When completed, the total production capacity will be 784,000 liters. Through process innovation, Samsung Biologics has shortened the technology transfer period, which is essential for biopharmaceutical production, to three months, half of the industry average. Process optimization was carried out through a specialized technology transfer team. The deployment success rate of Samsung Biologics is over 98%. A batch refers to a biopharmaceutical production unit. The cumulative number of regulatory approvals was 231. Samsung Biologics is strengthening its capabilities to respond to the demand for next-generation drugs such as antibody-drug conjugates (ADCs). In April of this year, it invested in Araris Biotech, an ADC treatment technology development company, through the Life Science Fund. It also plans to have an ADC production facility by 2024. Samsung Biologics plans to actively respond to the blockbuster drug market, such as Alzheimer's treatment. Based on the world's largest production capacity, it plans to expand orders by focusing on products that require mass production and products that newly expand indications. It plans to win orders for the 5th plant targeting Alzheimer's treatment, which has a lot of unmet demand. An official from Samsung Biologics said, "We have expanded our global bases, mainly in North America, to narrow the physical distance with our customers and provide prompt services." "We opened a sales office in New Jersey in March of this year following San Francisco in October 2020 to provide services to global customers," he said.
- Company
- ‘Leclaza will be free as first-line Tx until reimbursement'
- by Kim, Jin-Gu Jul 11, 2023 05:41am
- Wook-Je Cho, CEO of Yuhan Corp announced that it will be providing its anticancer drug ‘Leclaza (lasertinib)’ for free as a first-line treatment until its reimbursement is extended to cover first-line treatment. Cho announced the company’s launch of the Early Access Program (EAP) at a press conference it had held on the 10th at the Seoul Plaza Hotel to celebrate Leclaza’s approval as a first-line treatment in Korea. Leclaza is Korea's 31st homegrown novel drug that was approved for the treatment of NSCLC in January 2021. At the time of its approval, the drug was approved as a second-line treatment for patients with locally advanced or metastatic NSCLC who developed resistance after being previously treated with 1st generation or 2nd generation EGFR-TKIs. Last month, the company received additional approval for Leclaza as a ‘first-line treatment for non-small-cell lung cancer. Cho said, “The company will return a certain proportion of its profits to support the treatment of lung cancer. We will allow free access to Leclaza (as a first-line treatment) for patients suffering from lung cancer until the drug is applied reimbursement." In other words, the company will be providing Leclaza for free as a first-line treatment to all patients that wish to use the drug in the first line until the use is covered by reimbursement. Cho emphasized, “Regardless of the amount, all patients who need and want Leclaza will be able to benefit from our EAP.” Also, Cho dismissed the concerns that the EAP could violate fair competition with its competitors, saying "We feel confident because we have no alternative purpose." “Many patients with EGFR-positive NSCLC have been filing one petition after another through the National Assembly, the Ministry of Health and Welfare, and the President's Office. Many patients cannot afford to use Leclaza because the drug costs KRW 100 million a year without insurance reimbursement. After the drug was approved as a first-line treatment, we decided that it was right to provide its benefit faster to more patients.” “Carrying on the spirit of our founder, Dr.Ilhan New, we will be giving back our profits to society through the EAP. Regarding issues of compliance that have been voiced, we feel confident in providing EAP because we have no alternative purpose.”