- LOGIN
- MemberShip
- 2026-04-10 02:22:17
- Company
- Novartis counterattacks against Celltrion / a patent lawsuit
- by Kim, Jin-Gu Jun 07, 2023 09:47pm
- Novartis is investigating Celltrion for patent infringement of its biosimilar Xolair, asthma, and urticaria treatment. According to the pharmaceutical industry on the 2nd, Novartis filed two claims against Celltrion on the 1st. A proactive trial to confirm the scope of rights is to ask the Intellectual Property Trial and Appeals Board to accurately determine the scope of patent validity to see if the patent holder has been infringed. This case is interpreted as a situation in which Novartis is claiming Celltrion's patent infringement. In other words, Celltrion is claiming that the Xolair biosimilar, which is being developed, has infringed on two pharmaceutical patents it owns. Xolaire's material patent has already expired, and only the formulation patent remains. In Korea, the formulation patent expires in March 2024. Celltrion is developing 'CT-P39' as a Xolair biosimilar. It is evaluated that it is close to commercialization, such as announcing the interim results of the global phase 3 clinical trial in April. It has already applied for product approval in Europe and plans to apply for approval within the year in Korea. Analysts say that if the Intellectual Property Trial and Appeals Board upholds Novartis, the original company, in an active trial to confirm the scope of rights, setbacks in Celltrion's plan to release the Xolair biosimilar in Korea are inevitable. Xolair is a biopharmaceutical developed by Genentech and Novartis. It is used to treat allergic asthma, chronic urticaria, and chronic rhinosinusitis. According to IQVIA, a pharmaceutical market research institute, Xolair's domestic sales in the first quarter were 4.6 billion won. It increased by 34% from 3.4 billion won in the first quarter of last year. Last year, it posted sales of 15.7 billion won in Korea.
- Company
- Kwangdong wins patent lawsuit against Ibrance in reverse
- by Kim, Jin-Gu Jun 07, 2023 05:38am
- Ibrance As Kwangdong Pharmaceutical succeeded in reversing the second trial of the patent lawsuit against Ibrance, it boarded the last train for generic exclusivity. As a result, Ibrance's generic for exclusivity is owned by three companies, including Daewoong Pharmaceutical and Shinpoong Pharmaceutical, which won in the first trial, and Kwangdong Pharmaceutical. According to the pharmaceutical industry on the 3rd, the Patent Court ruled in favor of the plaintiff on the 2nd in the patent trial cancellation lawsuit filed by Kwangdong Pharmaceutical against Pfizer. Kwangdong Pharmaceutical, which was defeated in the first trial, succeeded in reversing in the second trial. A total of five companies have challenged the Ibrance patent. These are Kwangdong Pharmaceutical, Shinpoong Pharmaceutical, Daewoong Pharmaceutical, Boryung, and Samyang Holdings. They requested a passive trial to confirm the scope of rights to the Ibrance crystalline patent, which expires in February 2034. Their plan was to avoid crystalline patents and release generics early in time for substance patents to expire in 2027. In the first trial, the decision was mixed. The Intellectual Property Trial and Appeal Board rendered a victory decision for Daewoong Pharmaceutical and Shinpoong Pharmaceutical, and a defeat decision for Kwangdong Pharmaceutical, Boryung, and Samyang Holdings. With the first trial victory, Daewoong Pharmaceutical and Shinpoong Pharmaceutical acquired Ibrance generic for exclusivity. Kwangdong Pharmaceutical, Boryung, and Samyang Holdings, which lost in the first trial, appealed to the Patent Court. Among the three companies, the ruling of Kwangdong Pharmaceutical came out first. Kwangdong Pharmaceutical barely succeeded in obtaining generic exclusivity through a second trial victory. This is because the period for acquiring generic for exclusivity was about to expire. According to the current regulations, there are three requirements to receive generic for exclusivity. A patent trial must be requested for the first time, this trial or subsequent litigation must be won, and a generic license must be applied for the first time. At this time, one proviso clause is attached to the requirement that the judgment and lawsuit be won. The point is that a generic company must apply for generic exclusivity to the Ministry of Food and Drug Safety and obtain a winning trial decision or ruling within nine months from the date the original company is notified of this fact. Kwangdong Pharmaceutical applied for a generic for the exclusivity of Ibrance on August 29 last year. In early September, it is expected that this fact will be notified to Pfizer. In other words, if Kwangdong Pharmaceutical did not win the case by early this month, even generic for exclusivity would be useless. As Kwangdong Pharmaceutical's second trial proceeded quickly, it succeeded in winning the case in early June, and generic exclusivity was eventually made possible. Kwangdong Pharmaceutical is the only company that has even obtained product approval among patent challengers. Kwangdong Pharmaceutical received approval for Alency as a generic version of Ibrance in March of this year. On the other hand, it is analyzed that Boryung and Samyang Holdings, which appealed in the second trial along with Kwangdong Pharm, are highly unlikely to receive generic for exclusivity. The hearing date for Boryung and Samyang Holdings was set for July. Ibrance is a hormone receptor (HR) positive or human epidermal growth factor receptor 2 (HER2) negative treatment for advanced and metastatic breast cancer. According to IQVIA, a pharmaceutical market research institute, Ibrance posted sales of 56.2 billion won in Korea last year. In the first quarter of this year, it was 12.6 billion won, down 13% from the same period last year.
- Company
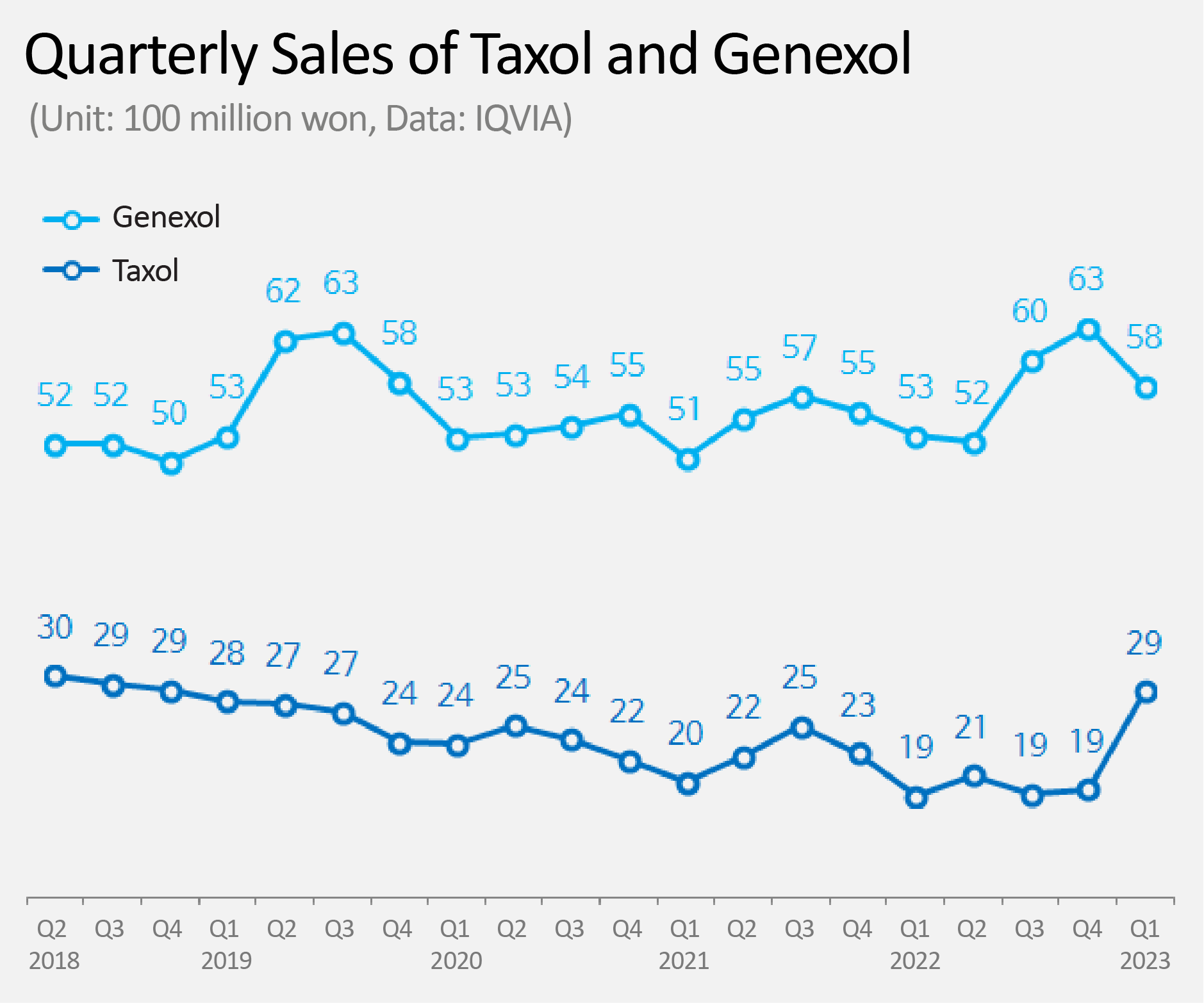
- Sales of anti-cancer drug 'Taxol' increases by 53%
- by Kim, Jin-Gu Jun 07, 2023 05:38am
- Product Photos of Taxol (LT) and Genexol (RT) Sales of the original Paclitaxel anti-cancer drug 'Taxol' increased 53% in one year. This change has seemed to have occurred as Boryung took over domestic sales from the beginning of this year. According to IQVIA, a pharmaceutical market research institute, on the 2nd, Taxol's sales in the first quarter was KRW 2.9 billion. This is a 53% increase from KRW 1.9 billion in the first quarter of last year. Taxol is a paclitaxel-based cytotoxic anti-cancer drug, and is widely used in various types of cancer such as ovarian cancer, breast cancer, lung cancer, and stomach cancer. Despite having been nearly 30 years since it was approved in Korea in 1996, and is still widely used. Until last year, sales of Taxol had been on a steady decline. The downhill started in the second quarter of 2018 from KRW 3 billion, declining to KRW 1.9 billion in quarterly sales last year. The pharmaceutical industry cites Boryung as the reason for Taxol's successful rebound. Boryung has been in charge of sales of Taxol since the first quarter of this year. It is analyzed that Boryung brought rebound in sales of Taxol through its already-established solid sales grounds in the field of anti-cancer drugs. Quarterly Sales of Taxol and Genexol (Unit: 100m million won, Data: IQVIA) Boryung's past actions are quite interesting. From 2008 to 2015, Boryung jointly sold Taxol with BMS for 8 years. From 2016 up to last year, Boryung jointly sold Genexol, a competing product of Taxol, with Samyang Holdings (formerly Samyang Biopharmaceuticals Corp.). From this year onwards, Boryung has taken charge of the sales of Taxol again. While Boryung was in charge of sales of Genexol, Genexol surpassed its original product Taxol and rose to no.1 in the market. For Boryung, a situation unfolded in which it now has to compete with the product that it had put on the top of the market. Genexol, which was previously sold by Boryung, is now being sold by HK inno.N from this year. Genexol's first quarter sales were KRW 5.8 billion, which is a 9% increase from the first quarter of last year, KRW 5.3 billion. HK inno.N has jointly sold Genexol from 2001, when Samyang Holdings first developed the product as a generic Paclitaxel drug, to 2013. The joint sales agreement reunited the two companies after 10 years. The pharmaceutical industry's attention is focused on whether Boryung, which has again taken charge of the sales of Taxol, will be able to catch up with Genexol by maintaining the same upward trend as in the first quarter. For HK inno.N and Samyang Holdings, on the other hand, the key is how firmly they maintain the market share of Genexol.
- Company
- Pfizer's ADC Mylotarg can be prescribed by Big 5
- by Eo, Yun-Ho Jun 07, 2023 05:38am
- Pfizer's new ADC drug, Mylotarg, has settled in a Big 5 general hospital. According to the related industry, Pfizer Korea's acute myeloid leukemia (AML) Mylotarg is a drug for medical institutions such as SMC, SNUH, Seoul St. Mary's Hospital, AMC, Shinchon Severance Hospital, as well as advanced general hospitals such as the National Cancer Center and Hwasun Chonnam National University Hospital. passed the committee. Mylotarg is an antibody-drug conjugate that can be used for the first-line treatment of adult patients with newly diagnosed CD33-positive AML. However, Mylotarg has not yet received insurance benefits. This drug was presented to HIRA in May of last year, but it was judged that the reimbursement standard was not set. Mylotarg, which was approved in Korea in December 2021, is an ADC composed of a CD33-targeting monoclonal antibody and a cytotoxic drug, calicheamicin, and acts on cells expressing the CD33 antigen, which is seen in 90% of all AML patients. This blocks cancer cell growth and induces apoptosis. The approval of Mylotarg was based on a clinical study conducted on 271 newly diagnosed AML patients with no treatment experience before the age of 50 to 70 years. The clinical trial was an open-label, randomized, multicenter phase 3 clinical trial for ALFA-0701. The existing anticancer chemotherapy, daunorubicin or cytarabine combination therapy, and Mylotarg, daunorubicin, and Cytarabine combination therapy were compared and evaluated. As a result, the Milotac + Daunorubicin + Cytarabine combination administration group had a median event-free survival (EFS) of 17.3 months, which was about 7.8 months longer than the Daunorubicin + Cytarabine combination administration group's 9.5 months. seemed It reduced the risk of induction failure, relapse, or death by approximately 44%. The median relapse-free survival (RFS) was 28.0 months for the Milotac + Daunorubicin + Cytarabine combination group and 11.4 months for the Daunorubicin + Cytarabine combination group, showing a significant difference of about 16.6 months. The median overall survival was 27.5 months for the Mylotarg + Daunorubicin + Cytarabine combination group and 21.8 months for the Daunorubicin + Cytarabine combination group, showing no statistically significant difference.
- Company
- 'Saxenda' continues to dominate weight-loss market
- by Kim, Jin-Gu Jun 07, 2023 05:38am
- Product Photos of Saxenda (LT) and Qsymia (RT) 'Saxenda (Liraglutide)' is further strengthening its dominance in the obesity treatment market. In the first quarter, it recorded sales of KRW 15.9 billion, 53% increase from the previous year, more than doubling the gap with the runner-up product, 'Qsymia (Phentermine+Topiramate).' However, it remains to be seen how much longer Saxenda's dominance will last, as release of promising products such as 'Wegovy (Semaglutide)' and 'Mounjaro (Tirzepatide)' are imminent in the obesity treatment market. The pharmaceutical industry predicts that the two products which have proven their marketability in the global market will be released in Korea as early as this year. Saxenda grows 53% in 1 year... more than doubles the gap with Qsymia According to IQVIA, a pharmaceutical market research institute, on the 5th, Novo Nordisk's Saxenda recorded sales of KRW 15.9 billion in the first quarter. This is a 53% increase in 1 year, compared to KRW 10.4 billion in the first quarter of 2022. Saxenda is the world's first obesity treatment approved as a glucagon-like peptide-1 (GLP-1) analog. It has the same ingredients as the type-2 diabetes treatment 'Victoza,' but with different usage and dosage. Saxenda has grown rapidly since its domestic release in the second quarter of 2018. In 2019, the second year after its release, Saxenda dominated the obesity treatment market with sales of KRW 42.6 billion. Saxenda, unlike existing obesity treatments, is not a psychotropic drug, and therefore gained explosive popularity because it is relatively safe and can be taken over a long period. The sales of Saxenda have somewhat slowed down due to the impact of COVID-19 in 2020 and 2021. However, as outdoor activities have become revitalized again from last year, demand for obesity treatments regained its place, and Saxenda's sales soared up to KRW 58.9 billion. Quarterly Sales of Saxenda and Qsymia (Unit: 100 million won, Data: IQVIA) Qsymia, the runner-up product in the market, marked sales of KRW 7.7 billion in the first quarter. Compared to KRW 6.3 billion in the first quarter of last year, sales has increased by 21%. It is analyzed that Qsymia was also affected by the recovery of the obesity treatment market. Qsymia is a combination drug of 'Phentermine' and 'Topiramate,' which Alvogen Korea secured domestic sales right from the US pharmaceutical company Vivus in 2017. Alvogen Korea started domestic sales with Chong Kun Dang at the end of 2019. In addition to the advantage that the content of psychotropic ingredient is relatively low even though it is an oral drug, Chong Kun Dang's sales power generated synergy and quickly penetrated the market despite being a latecomer. However, the gap with Saxenda has widened. Although Qsymia chased Saxenda's sales up to about 90% with KRW 5.9 billion in the first quarter of 2021, the gap is widening again as the obesity treatment market is recovering. In the first quarter of this year, the gap between to two products widened by 2.4 times. Some in the pharmaceutical industry predicts that the gap between the two products will widen further in the future. This is because obesity treatments containing Phentermine and Phendimetrazine, including Qsymia, were included in the 'list of narcotics and drugs of concern for misuse and abuse' as the non-face-to-face treatment pilot project was implemented. The government urged caution in prescibing psychotropic drugs such as Qsymia through non-face-to-face treatment. Saxenda has not been included in the list. In addition, treatments such as Daewoong Pharmaceutical's Dietamin, Korea Prime Pharm's Phendimen, Huons's Hutermin, etc. recorded sales of more than KRW 1 billion in the first quarter. In case of Phendimen, its sales, which recorded just KRW 300 million in the first quarter of last year, increased about 6 times to KRW 1.8 billion in one year, showing remarkable growth. Wegovy and Mounjaro's impending release... Saxenda's domination coming to an end It remains to be seen how much longer Saxenda's dominance will last. Two mega-sized products that have proven their competitiveness in the global market are waiting to be released. In the pharmaceutical industry, the prevailing view is that Saxenda's dominance will come to an end with the advent of next-generation products such as Wegovy and Mounjaro. Product Photos of Wegovy (LT) and Mounjaro (RT) In April, the Ministry of Food and Drug Safety approved Novo Nordisk's Wegovy. Wegovy is a GLP-1 analog, just like Saxenda. Novo Nordisk improved its Saxenda, which was administered daily, to weekly administration. In the US market, in which Wegovy was released earlier, demand for the product soared, with shortages occurring. In particular, due to its popularity, shortage of Ozempic, a diabetes treatment with the same ingredients and usage, has also occurred. Even now, there is still a lack of supply of Wegovy in the US. Due to the circumstances, the official release of Wegovy is being delayed in Korea even after product approval. The pharmaceutical industry predicts that domestic supply will be possible at the end of this year or early next year. The domestic release of Eli Lilly's Mounjaro, which is considered a strong competitor of Wegovy, is also imminent. The Ministry of Food and Drug Safety recently completed a safety and efficacy review on Mounjaro. Completing the review means that the product approval process will soon begin. Mounjaro is a GLP-1 analog, just like Saxenda and Wegovy. After obtaining approval as a type-2 diabetes treatment, Lilly is trying to expand its indications for obesity. In the case of the Mounjaro, in addition to the mechanism acting on the GLP-1 analog, its mechanism also acts on the glucose-dependent insulinotropic polypeptide (GIP). Due to this, Mounjaro's weight loss effect was better than that of Wegovy in each drugs' clinical trials. Lilly also entered a phase 3 clinical trial comparing the effects of Mounjaro and Wegovy on a one-to-one basis.
- Policy
- Voluntary price reduction of Diacomit/Olpadine up to↓40%
- by Lee, Hye-Kyung Jun 05, 2023 05:37am
- The drug prices of Diacomit, a treatment for Dravet syndrome, and Olpadine, a treatment for hereditary tyrosinemia, which is a rare essential drug, fell by up to 40%. The KOEDC said in a press release on the 1st, “In the case of Diacomit and Olpadine, insurance prices were voluntarily reduced on April 1 and May 1, respectively.” As a result, the price of Diacomit per capsule was reduced by about 23% from 19,160 won to 14,700 won. This is due to the fact that overseas wholesalers can reduce transportation costs by importing directly from the manufacturer, and as export certification by the manufacturer becomes possible, tariff exemption is possible under the Korea-EU FTA. Olpadine cut the price by about 40% from 37,386 won per capsule to 22,420 won to adjust for the difference between the cost of import and insurance drug price. As a drug subject to tariff exemption, the insurance price of Enhertu, a breast cancer treatment, was cut from 5.4 million won to 5 million won, and the Alzheimer's drug, Aduhelm, was cut from 3.44 million won to 3.2 million won. The Korea Customs Service imposes a basic tariff rate of 8% when importing medicines from abroad, but no duty is imposed on immune products. The KOEDC took note of this and reduced drug prices through tariff reductions on Enhertu, a breast cancer treatment, and Aduhelm, an Alzheimer's treatment. Enhertu applied for a preliminary review to the Korea Customs Service in June 2022 and was notified of the result in August 2022, and the drug price per vial was reduced from 5.4 million won to 5 million won through tariff reduction. Aduhelm applied for a preliminary review to the Korea Customs Service on March 7, 2023, and was notified of the result on March 20, 2023. Through tariff reduction, the drug price per vial increased from 2.04 million won to 1.9 million won for 170 mg, and 3.44 million won for 300 mg was reduced to 320 million won. In the case of Quinidine, a rare arrhythmia treatment, an attempt was made to import and supply it as an Epic Pharma drug due to Sandoz’s suspension of supply in June 2022, but the cost of the drug was 75 times higher than the existing drug price (50,000 won/bottle), adding to the patient’s burden. The KODC announced that it has been able to supply Nisco Quinidine, which can be imported directly from the manufacturer, by lowering the price to 35,000 won through continuous efforts.
- Policy
- Evrysdi, a tx for Roche's spinal muscular dystrophy, passed
- by Lee, Hye-Kyung Jun 05, 2023 05:37am
- Roche Korea's oral 5q spinal muscular atrophy treatment Evrysdi Dry Syrup has been recognized for its benefits and has crossed the insurance threshold. The HIRA released the results of the '2023 6th Pharmaceutical Reimbursement Evaluation Committee' deliberation on the 1st. Ace Pharma's Megval 50mg and H.I.Pharm's Melpspal 50mg, both of which are Evrysdi and Melphalan Hydrochloride-component multiple myeloma treatments, were all approved for reimbursement. Evrysdi is a liquid formulation that is taken orally once a day and can be applied to patients who have difficulty in spinal treatment. The effect of improving motor function and the safety profile was confirmed in patients with a wide range of disease types, including patients of a wide age range from 2.2 months to 25 months of age and patients with scoliosis-related surgery experience. Since self-administration is possible at home, it reduces not only the additional direct medical costs related to hospitalization and visitation, which occurred during the existing intrathecal injection treatment, but also the burden of indirect medical costs such as study and work interruption, transportation costs, and nursing care, which are accompanied by this, reducing insurance finance It is evaluated that it can produce the effect of reducing the socio-economic burden. In addition, it is a customized prescription according to age and weight, and the liquid formulation corresponding to the recommended dose can be taken once a day. Megval and Melpspal 50mg, whose main ingredient is Melphalan Hydrochloride, have been approved for multiple myeloma treatment. The dosage is 0.4 mg per kg of body weight (16 mg/m2) intravenously over 15 to 20 minutes for adults, and for patients with renal impairment (BUN > 30 mg/dL), reduce the dose by half.
- Policy
- Non-face-to-face demonstration will be alternative relief ?
- by Lee, Jeong-Hwan Jun 05, 2023 05:37am
- While the non-face-to-face treatment pilot project started on the 1st, it is attracting attention that the government will "seek ways to use substitute drugs" as a way to prevent inconvenience caused by the lack of prescription drugs at the pharmacy near the patient after treatment. There is a lot of interest in whether the alternative treatment simplification method can be implemented within the pilot project period, such as exempting the follow-up notification of alternative treatment limited to non-face-to-face treatment. In particular, the government also announced that it will prepare guidelines such as banning automatic allocation of pharmacies after prescribing for non-face-to-face treatment platform management and regulation measures, and to induce improvement in case of problems after monitoring the platform by operating a pilot project advisory group such as the pharmaceutical society. On the 1st, the Ministry of Health and Welfare answered a question about the non-face-to-face treatment operation plan of Rep. Jeon Hye-sook, a member of the National Assembly Health and Welfare Committee. First, the government said that it would use alternative drugs to prevent cases of inconvenience because the medicines prescribed by medical institutions that have performed non-face-to-face treatment are not in pharmacies located near the patient's area. Starting from the pilot project, the method of receiving prescription drugs after the non-face-to-face treatment will be changed from delivery to the patient's (agent)'s direct visit to the pharmacy. The government's will to minimize the situation where the pilot project has a hard time because there is no medicine in the pharmacy through alternative medicine. In order to improve this inconvenience and lack, pharmacists are demanding that non-face-to-face treatment be allowed to 'simplify post-notification of replacement drugs' and 'prescribe ingredients'. However, the Ministry of Health and Welfare did not take a specific position regarding the exemption of non-face-to-face treatment substitutes, the introduction of simpletion, or ingredient name prescriptions. I answered only with the intention of using the alternative medicine system to solve the problem that occurs when non-face-to-face prescription drugs are not in pharmacies. The Ministry of Health and Welfare announced that it has prepared guidelines such as 'prohibition of automatic allocation of pharmacy' on ways to manage and control expedient, illegal, and deviant behavior of non-face-to-to-face treatment platforms. In particular, he said that he would monitor the platform through the operation of a non-face-to-face treatment pilot project advisory group in which health and medical organizations such as the Korean Pharmaceutical Society participate, and would demand improvement if there were any problems. However, the Ministry of Health and Welfare has made it clear that in order to impose legal obligations on platform laws, or to make effective regulations such as data submission requirements and corrective orders, it is necessary to legislate the institutionalization of non-face-to-face treatment. The Ministry of Health and Welfare said it would "promote amendments to the health care law based on the legislation proposed by the National Assembly" regarding platform regulation. The Ministry of Health and Welfare did not provide a specific position or method on the 'electronic prescription' that electronically transfers a prescription issued by a doctor to a patient-designated pharmacy after a non-face-to-face treatment. "The method of sending and delivering prescriptions from medical institutions to pharmacies is to deliver faxes, e-mails, etc. to pharmacies designated by patients," the Ministry of Health and Welfare explained. In addition, during the pilot project period, we reaffirmed our position that it is in principle to implement non-face-to-face treatment with video treatment rather than telephone consultation. He said that the principle of video treatment is to conduct non-face-to-face treatment as a 'means that can communicate with real-time video communication', not just by phone call or voice. "Elderly people who have difficulty making voice calls or do not have smartphones can have non-face-to-face treatment with voice calls," the Ministry of Health and Welfare said. It is read that even if a non-face-to-face patient lies that it is difficult to make a video call or does not have a smartphone, there is no way to specifically isolate or regulate it. When asked if the government plans to support the cost of building a video care system for medical institutions during the pilot period, the Ministry of Health and Welfare said, "there is no separate cost support plan."
- Company
- Interleukin inhibitors Cosentyx·Tremfya busy extending reim
- by Eo, Yun-Ho Jun 05, 2023 05:37am
- News of reimbursement extensions granted for interleukin inhibitors is continuing in Korea. According to industry sources, the scope of reimbursement for Novartis Korea's IL-17A inhibitor 'Cosentyx UnoReady Pen' 300mg/2mL, and Janssen Korea's IL-23 inhibitor ‘Tremfya (guselkumab)' have been extended since last month, and the 1st of this month, respectively. As with the other doses of Cosentyx, its 300mg dose can also be prescribed to patients with moderate-to-severe plaque psoriasis, active and psoriatic arthritis (PsA), adults with active ankylosing spondylitis (AS), adults with severe ankylosing spondylitis, etc. In Korea, Cosentyx is reimbursed for patients with chronic severe plaque psoriasis that lasts for more than 6 months, and ▲10% of the total skin area has been affected, ▲has a PASI 10 or higher, ▲has shown no response to methotrexate (MTX) or cyclosporine when administered for more than 3 months or cannot continue treatment due to side effects, or ▲has shown no response to PUVA or UVB therapy when treated for more than 3 months or cannot continue treatment due to side effects. The reimbursement approval was made based on the MATURE trial. The trial results showed that 95.1%/75.6%/43.9% of patients that received Cosentyx 30mg reached PASI 75/90/100 each, and demonstrated better efficacy over its placebo. In the case of Tremfya, the drug will be granted reimbursement for palmoplantar pustulosis from this month. Under the new reimbursement standards, the drug may be used with insurance benefits from June 1st in patients aged 18 and older with moderate-to-severe palmoplantar pustulosis, ▲whose PPPASI is 12 or higher and has shown no response to therapeutic doses of acitretin, methotrexate, or cyclosporin when administered for more than 3 months or cannot continue treatment due to side effects, or ▲who has shown no response to phototherapy when treated for more than 3 months or cannot continue treatment due to side effects. Palmoplantar pustulosis is characterized by pustular blisters accompanied by erythema on the skin of the palms and soles that turn into brown scales, and then become dry, thick, and cracked. When the condition persists, the patient’s nails may be deformed and even fall out. The condition interferes with the patient's daily life because the condition causes severe itching and pain. As it is difficult to distinguish palmoplantar pustulosis from other skin diseases such as fungal infections, accurate differential analysis between the two is necessary. Cosentyx 300mg was approved on November 1st, 2022 to treat ▲plaque psoriasis patients and ▲psoriatic arthritis (PsA) patients who have accompanying moderate-to-severe plaque psoriasis or have not adequately responded to an anti-TNAα treatment before. Meanwhile, Tremfya was approved as a treatment for adult patients with plague psoriasis in April 2018 and as a treatment for adult patients with palmoplantar pustulosis in May 2019, and the indications applied for insurance benefit in September 2018 and May 2021 respectively.
- Opinion
- [Reporter’s View]Rise of adjuvant therapies amid concerns
- by Eo, Yun-Ho Jun 05, 2023 05:37am
- Although it may not be the same, the concept of administering a drug for ‘prevention’ has been around for a while. Patients who have chronic diseases have been taking drugs to ‘manage’ their condition rather than treat it. Also, some drugs just exist to prevent a condition, like anticoagulants. The same has just recently been applied to anticancer drugs. Companies have been acquiring or seeking indications as adjuvant or neo-adjuvant therapy for early-stage disease with their various new anticancer drugs through various research. Even at the 'American Society of Clinical Oncology 2023 Annual Meeting’ that is being held in Chicago, USA, adjuvant therapy research data are pouring in for 'Keytruda (pembrolizumab)', 'Kisqali ( ribociclib)', 'Herceptin (trastuzumab)' and others. However, concerns have followed the uprise of adjuvant therapy, with the biggest burden being financial. As everyone knows, even after being cured, cancer can frightfully recur. Depending on the type of cancer, some cancers have a recurrence rate that nears 80%. In the current era of high-priced pharmaceuticals, prescribing anticancer drugs that are leading the high-priced trend as adjuvant therapy and granting insurance reimbursement would naturally pose a burden for the health authorities. Another fact is that the benefits of adjuvant therapy are also receiving attention from the academic world. Already, adjuvant therapy has begun to be listed in the guidelines of world-renowned academic societies, with higher and higher grades of recommendation. So this is now the time for all those involved to ponder. Carefully examine the necessity of adjuvant anticancer therapy for each drug, and weigh the practical benefits rather than the indistinct and vague worry about its 'burden.' Treating patients with recurrent cancer may be less cost-effective. Recurrence and metastasis are fatal factors that increase cancer mortality. Since there is no right answer, we have to weigh the good and the bad. It is not possible to leave the piling number of applications for drugs to be used as adjuvant or maintenance therapies unattended. It is not just about the profits and losses. The stakeholders should also consider the specificity of each drug and the patient's situation and find consensus that takes into account Korea’s unique health insurance system and the pharmaceutical industry’s ecosystem.