- LOGIN
- MemberShip
- 2026-04-10 17:06:18
- Company
- Patent extension system for drugs to be reorganized…
- by Kim, Jin-Gu Mar 22, 2023 05:46am
- A plan to reorganize the patent term extension system of biopharmaceuticals in a manner similar to those of the US and Europe is being reviewed. In the pharmaceutical industry, it is predicted that if the reorganization plan passes the National Assembly, the patent term of some original drugs will be reduced to about one year. According to the pharmaceutical industry, the Korean Intellectual Property Office recently prepared an amendment to the Patent Act to reform the system for the patent term of drugs on the 21st. The amendment is expected to be submitted to the National Assembly in the form of legislative acts. The main point of the amendment is to set the upper limit on the remaining (valid) patent term to 14 or 15 years from the time a new drug receives approval. The pharmaceutical industry's attention is focused on how much the original drug's patent term will be shortened. In this regard, the US and Europe have systems that limit the effective patent term. This method allows the patent to be effective for up to 14 or 15 years from the time the drug has been approved. For example, even if a pharmaceutical company 'A' has been granted approval for a patent for 20+5 years (basic patent term + extended period), the entire patent term is shortened 'up to 15 years from the time of approval' by setting an upper limit. On the other hand, Korea does not have a separate limit on the patent term that is applied from the time of approval. In the current system, there are only regulations that limit the extension of the patent term to a maximum of 5 years, but no upper limit on the effective patent term. As a result, the Korean patent term of pharmaceuticals is longer than the US and Europe, with the same product and same patent. Taking Pfizer’s ALK-targeted anti-cancer drug Xalkori (ingredient: crizotinib) as an example, its remaining patent term is limited to 14 years after approval by the FDA in the US. Under the upper limit, only 1 year and 6 months (547 days) of the patent term extension was recognized for Xalkori in the United States. The situation is similar in Europe. Europe has an upper limit of 15 years from the date of initial marketing authorisation. As a result, only 2 year and 2 months (799 days) of the patent term extension was recognized for Xalkori in Europe. Since there is no separte regulation that states the upper limit in Korea, the extended patent term of Pfizer's Xalkori was fully recognized. The extended patent term approved for Xalkori in Korea is 2 years and 10 months (1034 days), which is about 8 months longer that that of Europe and about 16 months longer than that of US. If the amendment passes the National Assembly, the effective patent term of the original drug will be shortened to about 1 year. In other words, the timing of generic release in Korea will be accelerated. The key is whether the amendment will be applied only to newly introduced drugs or to existing drugs as well. If the amendment is applied to existing drugs, quite a number of drugs will be affected by the new system. According to the Korean Intellectual Property Office, as of the end of 2021, 360 drugs still have effective patent term remaining in Korea. In terms of number of patents, there are 612. Among them, 60 drugs (83 patent rights) are expected to become subject to the new system, having 'effective patent term of 15 years or more.' While the Korean Intellectual Property Office is reportedly considering the 14-year regulation, the US model, the pharmaceutical industry predicts that the effective patent term for 40 to 50 drugs may be shortened if the amendment is applied to existing drugs as well.
- Opinion
- [Reporter's view] PVA improvement plan & domestic drugs
- by Lee, Tak-Sun Mar 21, 2023 05:58am
- There is also concern about the proposal for improving PVA through research service. Last year, researchers (Bae Seung -jin, Ewha Womans University, etc.), who studied PVA's performance evaluation and improvement plan, ordered the screening management of drugs with high financial impacts. As a result, the selection criteria for the usage type 'Ka' were proposed to expand by adding 5 billion won and a 10% increase in the existing claims. Drugs with low financial impacts, such as a plan to raise the exclusion of negotiations from 2 billion won to 3 billion to 5 billion won, are required to ease management. The proposal for improvement will be discussed in May and institutionalized from January next year. In summary, the system improvement is expected to be promoted in the direction of a high -financial reduction rate, low drug reduction rate, and lowering the cut-rate. In the industry, there are many voices in favor of the system improvement direction. It is efficient to differentiate the upper limit according to the number of claims. However, there is a concern that the type of new drug will be added to the 'negotiations', and the burden will be increased to multinational pharmaceutical companies or domestic new drug development companies. In particular, domestic new drugs have been concerned about the improvement plan as the pharmaceutical industry has insisted on easing the PVA in order to ease development and encouragement. As the government uses biotechnology as a next-generation growth engine and declines domestic new drugs, it is also necessary to support the drug price. Domestic new drugs enter the low price without preferential treatment from the time of listing, and if the drug price is increased due to active sales and marketing, the new drug development company, which spends large-scale costs, ' If it was, only regret will remain. Although the government cannot discriminate against domestic and overseas new drugs, the domestic new drug preferential policy is inevitable to protect and foster our industry. Therefore, from May, we should focus on preparing more sophisticated measures in consideration of concerns about the domestic pharmaceutical industry.
- Policy
- Janssen's CAR-T treatment Carvykti was approved
- by Lee, Hye-Kyung Mar 21, 2023 05:57am
- The Ministry of Food and Drug Safety (Director Oh Yu-kyung) announced that it has granted CARVYKTI, a CAR-T treatment agent, a rare drug of Jansen, a rare drug. CAR-T cells are called chimera because they modify the surface receptor to better recognize and attack cancer cells. Carvykti is an anticancer drug that injects the T cell into the patient's body after putting genetic information that can recognize the B cell mature antigen (BCMA) into the patient's immune cells (T cells). B-cell maturity antigen (BCMA) is an ideal target for transplanted cell carcinoma (multiple myeloma) because it is selectively expressed during transformed cell differentiation and is not expressed in other major organs. Carvykti has previously provided new treatment opportunities for patients with recurrence or dissatisfaction with at least four treatments, including Proteaums, immunomodulatory preparations, and anti-CD38 antibodies. The MFDS scientifically reviewed and evaluated the quality, safety and effectiveness, manufacturing, and quality control criteria of the drug in accordance with the screening criteria prescribed in the Act on the Safety and Support Act of Advanced Regenerative Medical and Advanced Bio. Carvykti is a long-term tracking survey under Article 30 of the Advanced Regeneration Bio Act and must be tracked over the case of more than 15 years from the date of administration. The MFDS said, "We will do our best to ensure that the therapeutic drugs that are fully confirmed with safety and effectiveness based on regulatory science are quickly supplied."
- Policy
- Which Vemlidy IMD among the 5 will rise to the top?
- by Lee, Tak-Sun Mar 21, 2023 05:57am
- Although failing to receive reimbursement in advance, many Vemlidy IMDs are awaiting entry into the Korean market. All the drugs are incrementally modified drugs that were developed with different salt formations. According to industry sources on the 20th, 5 more companies are waiting to enter the market with Vemlidy latecomers. These companies follow the earlier entrants, such as Dong-A ST, which received reimbursement approval in January, and Chong Kun Dang and Daewoong Pharmaceuticals, which received reimbursement approvals in February. Vemlidy (tenofovir ala fenamide) is an upgraded version of Gilead Science’s previous chronic hepatitis B treatment, ‘Viread (tenofovir).’ Vemlidy has been evaluated to have improved tolerance and nephrotoxicity over Viread. Same-ingredient Vemlidy generics are unable to enter the market because the drug’s hemi-fumarate patent is set to last until August 15, 2032, However, domestic companies have succeeded in releasing drugs in the market by avoiding the patent by changing the sodium ingredient of the drug. Starting with Dong-A ST’s release of ‘Vemlia (tenofovir alafenamide citrate)’ with reimbursement, Chong Kun Dang followed in February by releasing Tenofobell-A (tenofovir alafenamide succinate) with reimbursement and Daewoong Pharmaceutical also released Vemliver (tenofovir alafenamide hemitartrate) with reimbursement. Their ingredients show that all use different salts with Vemlidy. Donga ST, Chong Kun Dang, and Daewoong Pharmaceutical each developed their own salt-modified drugs. 2 more types of salt-modified drugs are set to follow. Jeil Pharmaceutical Tecavir-D (tenofovir alafenamide) was approved in the same month as those by Chong Kun Dang and Daewoong Pharmaceutical but was not granted reimbursement. On its reason, a company official said, “reimbursement had been delayed due to disruptions in the supply of the drug’s API,” he explained. With the normalization of the supply, it is highly likely that our product will also be released with reimbursement.” Also, 4 tenofovir alafenamide hemimalte products were approved on March 15th and are set to be released with reimbursement in June. The drugs are manufactured and supplied by Samjin Pharm but will be marketed by Bukwang Pharm, which has strong marketing strengths in the hepatitis B treatment market. Also, Dongkook Pharmaceutical, Hutecs Korea Pharmaceutical, and Samil Pharm will also receive products from Samjin and enter the market with individually approved products. As of now, a total of 5 salt-modified Vemlidy IMDs have been released to the market. The fact that so many salt-modified drugs were developed and released for one product shows the interest large companies have in this market. Large companies tend to develop and sell their own products rather than receive drugs upon consignment from other companies. This is why, large pharmaceutical companies that gross large sales, such as Donga ST, Chong Kun Dang, Daewoong Pharmaceutical, and Jeil Pharmaceutical, dedicated and developed their own salt-modified drugs to preoccupy the market. On the other hand, since it is difficult for small and medium-sized companies to develop drugs independently due to development costs, Samjin is known to have addressed the problem through joint development with other companies under the 1+3 regulation set for consigned bioequivalence tests. Therefore, which salt-modified product will be leading the market is gaining attention.
- Company
- Pediatric indication becomes key to sales in Korea
- by Moon, sung-ho Mar 21, 2023 05:56am
- The treatment market for pediatric and adolescent patients have grown rapidly last year. In particular, injections for pediatric and adolescent patients have gained much popularity and raised sales in reimbursed and non-reimbursed markets in hospitals and clinics. The transition from the COVID-19 pandemic to an endemic had increased focus on injections for obesity and growth, which led to great popularity. If the pediatric ‘non-reimbursed’ market had achieved great growth last year, this year, the market for reimbursed treatment for 'severe' pediatric conditions is expected to grow rapidly because major treatments for pediatric patients are attempting reimbursement in the first half of this year. First, Saxenda (liraglutide), the leader in the domestic obesity treatment market, saw rapid growth in sales last year with its expansion to the pediatric indication. #According to the market research institution IQIVA, Novo Nordisk’s Saxenda raised sales of KRW 58.9 billion last year, which is a 62.7% YoY increase. Saxenda’s sales had previously fallen for 2 consecutive years. From KRW 42.6 billion in 2019, sales had fallen to KRW 36.8 billion in 2020, then to KRW 36.2 billion in 2021. However, with COVID-19’s transition to an endemic, its sales had again surged and dominated Korea’s obesity treatment market. Saxenda’s sole lead in the market is expected to continue until new obesity treatments like Wegovy (semaglutide, Novo Nordisk), and Mounjaro (tirzepatide, Lilly) are released to the market. Saxenda’s rapid growth in sales is in line with its indication extension to children and adolescents last year. In December 2021, Saxenda’s indication was extended to treat adolescent patients aged 12 to 18 years with a BMI that corresponds to 30 kg/m2 for adults and weighs over 60 kg in Korea. In other words, the indication expansion to pediatric adolescent patients maximized sales in obesity and growth clinics and hospitals in Korea. The same goes for the growth hormone market. The growth hormone injection market, which had previously consisted of products from domestic pharmaceutical companies, has also grown into a large non-reimbursed treatment market and was evaluated to be sized at KRW 250 billion won last year with rising sales from growth clinics at front-line hospitals and clinics. If the pediatric ‘non-reimbursed’ market had achieved great growth last year, reimbursement extensions of major treatments for major pediatric conditions are expected this year. Key products include Dupixent (dupilumab) and Hemlibra (emicizumab). Both drugs received recognition for their need to extend reimbursement by the Health Insurance Review and Assessment Service’s Drug Reimbursement Evaluation Committee and are undergoing drug pricing negotiations with the National Health Insurance Service. If the pricing negotiations between the NHIS and pharmaceutical companies progress smoothly, the drugs will be able to receive reimbursement extensions within the first half of the year. In the case of Dupixent, if reimbursement extensions are finally made to cover children and adolescents with atopic dermatitis in May, its sales are expected to continue to grow as in the previous year. In the case of Dupixent, sales increased 35% YoY from the KRW 77.2 billion in the previous year to reach KRW 104, but experts predict there is still room for additional growth. The same goes for JW Pharmaceutical’s Hemlibra. Hemlibra is currently only reimbursed to treat patients who show resistance and have antibodies to existing treatments. As most of the Type A hemophilia patients are ‘non-antibody’ patients, this means that only a very few patients are receiving reimbursement for Hemlibra. This is why parents of hemophilia A patients had been continuously raising demand for Hemlibra's reimbursement extension. According to the 2019 Hemophilia White Paper, over 90% of the 1,746 patients with hemophilia A in Korea, 1,589 patients, are non-antibody patients. Drug pricing negotiations are underway after HIRA made the decision to extend reimbursement, and if the negotiations are completed within the first half of the year, non-antibody patients will be able to use Hemlibra with reimbursement as well. Meanwhile, the financial authorities expect the health insurance claims amount to increase rapidly if Hemlibra’s reimbursement is extended to cover non-antibody patients. According to IQVIA, Hemlibra’s sales last year increased 5% YoY from the KRW 7.2 billion to reach KRW 7.6 billion last year. In other words, if reimbursement is extended to cover non-antibody patients, the drug may grow to become a large blockbuster product that brings in millions of won every year. Moreover, the pharmaceutical industry predicts that the drug will become a 'dark horse' in Korea’s hemophilia treatment market which is currently led by GC Pharma if Hemlibra’s reimbursement is extended to cover non-antibody patients. A health insurance official said, “In the case of hemophilia, around 10% are antibody patients, and 90% non-antibody patients. Therefore, if reimbursement for Hemlibra is extended to cover non-antibody patients, the drug’s sales will naturally increase significantly.”
- Company
- Increasing male infertility
- by Eo, Yun-Ho Mar 21, 2023 05:56am
- As male infertility increases, interest in follicle-stimulating hormone preparations is increasing. So far, women's infertility treatment has been more active than male infertility treatment. 93.4%of the cost of infertility, which was carried out by 2021, was used for female infertility patients, and only 6.6%were used for male infertility patients. Infertility can occur in both men and women due to deficiency of sperm formation due to male sperm, mobility, and low pre -stimulating hormonal glands, or ovarian function of women, closure of fallopian tubes, and endometriosis. According to the Social Society of Society of Propaganda (SRS), the cause of infertility is about 40% of male factors. Looking at the number of infertility patients in domestic men, it can be seen that about 43%increased from 6,2468 to 89,350 in five years from 2017 to 2021. On the other hand, female infertility patients increased by about 11% over five years. The reason for the recent increase in male patients who are diagnosed with infertility is that the perception that there may be a cause of infertility is expanded compared to the previous one. Kim Soo -woong, a professor of urology medicine at Seoul National University Hospital, said, “Infight is that infertility is a problem of all couples, and interest in the cause and treatment of male infertility seems to be increasing. "I need a treatment option and research on continuous male infertility treatment is needed." Infertility treatments, which are used in general, have a genital-stimulating hormone. The genital stimulating hormone components include Follicle Stimulating Hormone, Human Menopausal Gonadotropin (HMG), and Human Chorionic Gonadotropin preparations. Among them, the FHS formulation is a hormone that is synthesized and secreted in the pituitary gland, which is not only involved in producing eggs in the ovaries of women but also acts as a hormone that produces sperm in the testicles of men, thereby improving the reproductive function of infertility patients. Among the FSH preparations used in Korea, 'Puregon' is the only man who has an indication of the deficiency of sperm formation caused by men's low -stimulating hormonal glands. PureGon confirmed the effectiveness of the combination of HCG, which was conducted with HCG, which was a lowly stimulating hormonal sexual dysfunction that failed to treat infertility with HCG monotherapy. According to the guidelines of the Endocrinology Society, FSH+HCG combination therapy is essential for men who are reduced in the genitals of the central central central central central central central central central central central central cavity in the treatment of reproductive ability of men. Professor Kim said, “The infertility treatment environment is far from effective infertility through male infertility diagnosis and treatment, and it is increasing the burden on infertility treatment for women. "Everybody needs to be infertile and treated in an appropriate time regardless of gender." World Health Organization (WHO) defines as infertility a couple with a normal marital relationship without contraception who are not pregnant for a year. As of 2021, the number of male and female patients diagnosed with infertility in Korea was about 250,000, up 9% from 2019, and nearly 10,000 people per year for nearly five years. Recently, the city of Seoul also announced a plan to expand infertility support to solve the problem of low outflows.
- Policy
- Erleada applied for health insurance in April
- by Lee, Tak-Sun Mar 20, 2023 05:51am
- As this drug is an upgraded version of prostate cancer treatment, which is increasing every year, many patients are expected to benefit if health insurance benefits are applied. On the 16th, the HIRA foretold the reimbursement standards for anticancer drugs, which include the establishment of new reimbursements for ADT and combination therapy for Erleada's hormone-responsive metastatic prostate cancer (mHSPC) patients. The effective date is the 1st of the following month. According to the announced reimbursement criteria, Erleada is covered as first-line therapy, block therapy (ADT), and combination therapy for mHSPC patients. ADT is an androgen deprivation therapy, limited to orchiectomy, LHRH agonist + 1st generation anti-androgen, and LHRH antagonist. In addition, LHRH agonist + 1st generation anti-androgen among ADT therapy will not be recognized when administered in combination with new hormone therapy drugs. The HIRA is a drug that Erleada has approved for use in combination with ADT for the treatment of patients with hormone-responsive metastatic prostate cancer (mHSPC). As a result of reviewing with reference to textbooks, guidelines, and clinical papers, the therapy is mentioned in textbooks, and the NCCN Guide It is stated that it is recommended as category 1 in the line, [Ⅰ, A] in the ESMO guidelines, and ESMO-MCBS score 4. In phase 3 clinical trial (TITAN study) for patients with hormone-responsive metastatic prostate cancer (mHSPC), as a result of comparing the therapy with the control ADT monotherapy, median overall survival (mOS) NR vs. mPFS2 NR vs. 52.2 months, confirmed by PSA progression, etc. He explained the justification for reimbursement, saying that clinical usefulness was confirmed at 44 months. Erleada passed the HIRA in February last year and the committee in December and has been negotiating with the NHIS since January.
- Company
- Pharmaceutical exports continue to be sluggish...
- by Kim, Jin-Gu Mar 20, 2023 05:51am
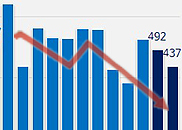
- Domestic drugs are continuing to show sluggish sales overseas. During the prolonged COVID-19 crisis, Korea enjoyed an uprise in pharmaceutical exports, but due to the nearing endemic, exports are returning to the pre-COVID-19 level. In particular, the sharp drop in exports of COVID-19 vaccines manufactured in Korea affected the decline in Korea’s overall export performance. According to the Korea Customs Service on the 17th, Korea's pharmaceutical export amounted to USD 928.49 million (approximately KRW 1.22 trillion, based on the won-dollar exchange rate of 1313.00 won). This is the lowest amount recorded in January and February alone over the past three years. It is also 16.5% lower than the USD 1.12 billion made in January and February of last year and 34.0% lower than the USD 1.47 billion made in January and February 2021. Domestic pharmaceutical exports had increased significantly during the prolonged COVID-19 period. Monthly exports prior to COVID-19 were around USD 300 million, however, after COVID-19 started to spread and become a pandemic in March, exports nearly doubled to average around USD 600 million per month. In December 2021, monthly exports soared to exceed USD 1 billion. The increase in confirmed COVID-19 patients increased the demand for medical care, and this is analyzed to have led to the increase in the export of domestic drugs. Also, from the end of 2021, exports of Moderna and Novavax vaccines that were manufactured in Korea began in earnest and contributed to the increase in exports. However, the slowdown in the spread of COVID-19 in the second half of last year then led to a decrease in the monthly exports of domestic drugs. In particular, this decrease in domestic pharmaceutical exports started in earnest in Q4 last year. Monthly drug exports, which consistently remained over USD 500 million until September last year, fell further to USD 428.51 million in October, then to USD 393.33 million in November. Although exports briefly increased to USD 525.97 million in December last year, the amount fell back to USD 400 million range entering this year. The industry believes that the decline in COVID-19 vaccine exports affected the decline in total exports. Overseas exports of domestic vaccines had soared from December 2021 to the first half of last year. Vaccine exports, which ranged between USD 10 million and USD 20 million a month before then, increased to exceed USD 270 million in December 2021, and then steadily maintained exports of over USD 100 million every month. However, after falling to USD 60 million in July last year, the amount has since returned to the standard level.
- Policy
- Lansoprazole+antacid combo awaits entry into market
- by Lee, Tak-Sun Mar 20, 2023 05:51am
- A new combination is awaiting entry into the Proton pump inhibitor (PPI)+ antacids combination market. This time, the new entrant is a combination of lansoprazole and precipitated calcium carbonate. Ever since Chong Kun Dang achieved outstanding results in the market, new combinations have been pouring out every year in the market. With already over 70 products in the market, whether the newly released product may settle in the market is gaining attention. According to industry sources on the 19th, the lansoprazole 30mg + precipitated calcium carbonate 600mg combination that was approved in Korea in January is expected to be listed for reimbursement in April. 6 companies including Guju Pharm, Myungmoon Pharm, Yu & Life Science, Hana Pharm, and Union Korea Pharm had received approval for related products. The drugs are being manufactured by Yu & Life Science. Four PPI ingredients are now available for use in combination with antacids - omeprazole, esomeprazole, rabeprazole, and lansoprazole. The antacids used with the PPIs are sodium hydrogen carbonate, precipitated calcium carbonate, and magnesium hydroxide. PPI+antacids are characterized by the shorter time required to take effect. The drugs combine antacids with PPI, which is weak against stomach acid, to complement the shortcoming and provide a rapid effect. Chong Kun Dang’s Eso Duo is a representative product in the market. Based on UBIST, Eso Duo recorded outpatient prescription sales of KRW 16.3 billion last year, which was a 10% YoY decrease from the previous year. This is analyzed to be due to the immense inflow of PPI+antacids in the market. 25 esomeprazole+ sodium hydrogen carbonate products are currently available in the market, and over 70 PPI+antacids are approved in the market. In this context, attention is focused on whether the latecomer lansoprazole+precipitated calcium carbonate will be able to overcome the fierce competition and settle in the market. However, in terms of price, the new entrant is expected to have competitivity over previous combination drugs due to its relatively cheaper price. The combination drug awaiting entry contains the high-strength lansoprazole (30mg, ceiling price KRW 877), and the ingredient’s price is relatively lower than the high-strength esomeprazole (40mg, ceiling price KRW 1,078), or the high-strength rabeprazole (20mg, ceiling price KRW 1,069). The price of the combination drugs is calculated based on the price of the single ingredient, the ceiling price for lansoprazole+precipitated calcium carbonate is expected to be lower than those set for other high-strength PPI combination drugs. However, whether a lower price will lead to an increase in market prescriptions remain unknown. There is also an analysis that the low price may instead become a burden on the profitability of the pharmaceutical companies.
- Company
- Leclaza costs 88 billion won for phase 3 in 3 years
- by Chon, Seung-Hyun Mar 20, 2023 05:51am
- Last year, Yuhan invested a total of 88 billion won in phase 3 clinical trial of Leclaza, a new anti-cancer drug. Yuhan Corporation is challenging the first-line treatment approval based on the efficacy and safety of Leclaza confirmed in clinical trials. According to the Financial Supervisory Service on the 20th, as of the end of last year, Yuhan Corporation's development costs reflected as intangible assets totaled 104.8 billion won. It increased by 33.1 billion won in one year from 71.7 billion won at the end of 2021. In 2019, the Financial Supervisory Service set a standard that accounting assets can be treated only when there is technical feasibility of R&D tasks such as new drugs. The Financial Supervisory Service suggested that R&D costs can be turned into assets by initiating phase 3 clinical trials for new drugs and approval of phase 1 clinical trials for biosimilars. Generics can be capitalized after the BA test is approved. Yuhan's R&D intangible assets are R&D costs invested in Leclaza and eight IMDs. Among them, Leclaza's development cost intangible assets amounted to 88 billion won. A total of KRW 88 billion was used for Leclaza's phase 3 clinical trial. Leclaza accounted for 84.0% of Yuhan's development cost intangible assets. Leclaza is a non-small cell lung cancer treatment approved as the 31st new drug developed in Korea in January 2021. Patients with locally advanced or metastatic non-small cell lung cancer who developed T790M resistance after administration of first- and second-generation epidermal growth factor receptor (EGFR) tyrosine kinase inhibitors (TKIs) are eligible for the treatment. It acts as a mechanism to inhibit the proliferation and growth of lung cancer cells by interfering with the signal transduction involved in the growth of lung cancer cells. Leclaza recognized development costs of 32.6 billion won as intangible assets for the first time in the fourth quarter of 2020. As phase 3 clinical trials began in earnest, development costs were reflected as intangible assets. Leclaza's development cost intangible assets increased to 61.4 billion won at the end of 2021, and 26.6 billion won was added last year. As the cost of phase 3 clinical trials for Lecraza increased, the development cost that Yuhan Corp. reflected as an asset exceeded 100 billion won for the first time. Yuhan is challenging the first-line treatment approval based on the results of Leclaza's phase 3 clinical trial. On the 17th, Yuhan Corporation applied for approval to use Lexraza as a first-line treatment for patients with EGFR exon 19 deletion or exon 21 (L858R) substitution mutations in locally advanced or metastatic non-small cell lung cancer. Leclaza demonstrated superior safety and efficacy compared to existing treatments in phase 3 clinical trial (LASER301) conducted on 393 patients with active EGFR mutation-positive locally advanced or metastatic non-small cell lung cancer who had not previously received treatment. The clinical results were recently unveiled at the Asian Congress of the European Society for Oncology held in Singapore. If Leclaza's use as a first-line treatment expands, sales are projected to soar. Although Leclaza is used on a limited basis as a second-line treatment, it is evaluated that it has successfully settled in the market. According to IQVIA, a pharmaceutical research institute, Leclaza recorded sales of 16.1 billion won last year. It increased by 4 times from 4.1 billion last year. Leclaza entered the prescription market in earnest in July 2021 with its listing on the health insurance benefit list. Sales of KRW 1.5 billion and KRW 2.6 billion occurred in the third and fourth quarters of 2021, respectively. Leclaza sold 3.2 billion won and 3.7 billion won in the first and second quarters of last year, respectively, and expanded to 4.6 billion won and 4.5 billion won in the third and fourth quarters. Leclaza recorded cumulative sales of 20.2 billion won for a year and a half after its release. Leclaza has already broken the sales record for new anti-cancer drugs developed in Korea. Domestically developed anti-cancer drugs approved prior to Leclaza include Ilyang Pharm's Supect, Dongwha Pharm's Milican, Chong Kun Dang's Camtobell, Samsung Pharm's Riavax, and Hanmi Pharm's Olita. None of these products exceeded 10 billion won in annual sales.