- LOGIN
- MemberShip
- 2026-04-12 12:52:57
- Company
- Benefit of Pfizer's Cibinqo is applied
- by Eo, Yun-Ho Apr 26, 2022 06:12am
- Another JAK inhibitor aims to enter the atopic dermatitis insurance benefit right. According to related industries, Pfizer Korea recently submitted an application for benefits of Cibinqo, a new drug for JAK1. As Lilly's Olumiant and AbbVie's Rinvoq are expected to be applied in May. Cibinqo was approved in Korea in November last year when the listing process of Olumiant and Rinvoq was underway. There was an observation that Cibinqo would also hurry to apply for registration and proceed with the registration process together, but the timing of Pfizer's application was later than expected. This drug acts as a mechanism to control the levels of IL 4, 13, 31, and 22 and TSLP on the thymus substrate, which are involved in the pathophysiological characteristics of atopic dermatitis. The permitted indication is the treatment of symptomatic atopic dermatitis in moderate cases of adults and adolescents aged 12 or older. Cibinqo proved its effectiveness through phase 3 clinical studies such as JADEMONO-1, MONO-2, and COMPARE. In the 12th week, the eczema severity evaluation index was lowered by more than 70%, and the treatment effect was proved in indicators such as relieving itching within two weeks of treatment. Among them, the JADE Mono-1 study randomly assigned and analyzed oral Cibinqo 100 mg, Cibinqo 200 mg, or placebo administration groups once a day for 12 weeks for patients with severe-moderate atopic dermatitis over the age of 12. In the Cibinqo 200m group, 63% of patients achieved a 75% improvement in Eczema Severity Index (EASI) at 12 weeks of treatment, compared to 12% in the placebo group, and 90% improvement in Eczema Severity Index (EASI-90) at 39%.
- Company
- RET targeted therapy Retevmo opens era of precision medicine
- by Apr 26, 2022 06:12am
- Lilly Korea’s RET targeted therapy Retevmo(selpercatinib) has landed in Korea. The drug is expected to become a new treatment option in RET fusion-positive patients with non-small-cell lung cancer and thyroid cancer. Lilly Korea held an online press conference to celebrate the approval of Retevmo on the 25th. Professor Min Hee Hong of Oncology at Yonsei Cancer Center and Professor Won Gu Kim of Endocrinology and Metabolism at Asan Medical Center attended the virtual event to explain the significance of Retevmo’s approval. Retevmo was approved by the Ministry of Food and Drug Safety in March last year as a treatment for RET fusion-positive NSCLC and thyroid cancer patients. More specifically, the drug is indicated for the treatment of▲ adult patients with metastatic RET fusion-positive non-small cell lung cancer (NSCLC); ▲adults and pediatric patients 12 years of age or older with advanced or metastatic RET-mutated medullary thyroid cancer who require systemic therapy; and ▲ adult patients who are refractory to radioactive iodine therapy and who have prior sorafenib and/or lenvatinib treatment, with advanced or metastatic RET-fusion benign thyroid cancer who require systemic therapy. At the press conference, Professor Hong said, “Lung cancer patients with RET mutations are twice more likely to experience CNS metastasis, but had to be treated with chemotherapy, which is less effective and more prone to toxicity due to the lack of RET targeted therapies. Retevmo demonstrated significant response in the LIBRETTO-001 trial, as well as an 82% ORR in patients with CNS metastasis. 23% of these patients achieved complete response.” Kim added, “Although the survival rate for thyroid cancer is known to be high patients with RET point mutated medullary thyroid cancer have a poor prognosis and low survival rate. Also, patients with thyroid cancer who are refractory to radioactive iodine therapy have a low survival rate and life expectancy. Retevmo has shown a 79% and 69% ORR in the abovementioned patient groups, opening a new era of precision medicine.” RET mutations occur in 2 to 6 % of all NSCLC cases and are more often found in adenocarcinomas and younger patients under 60 years of age and non-smokers. In NSCLC, RET fusions occur more than RET mutations. In thyroid cancer, RET fusions are reported in up to 40% of the cases. RET fusions can be identified through rest such as NGS, FISH, RT-PCR, etc. Among the tests, NGS is known to be the most effective diagnostic method for identifying RET fusions. However, it takes around one month to receive results after NGS testing, and as ‘Keytruda,’ a reimbursed immunotherapy option already exists for NSCLC in the first line, it is highly likely that Retevmo will not be selected as a first-line treatment in these patients. Professor Hong said, “As RET fusion is not a common mutation, we cannot idly just wait for the test results, therefore it is likely that we will use immunotherapies first. However, using immunotherapies before Retevmo may reduce the response rate to Retevmo, this is why it is important for us to receive the NGS results as quickly as possible.”
- Company
- President-elect Yoon visits SK Bioscience HQ
- by Kim, Jin-Gu Apr 26, 2022 06:12am
- President-elect Suk-yeol Yoon visited SK Bioscience headquarters in Seongnam, Gyeonggi-do, and promised “We will spare no effort in the development of vaccines so no one can say they could not develop vaccines due to lack of money.” Transition team chair Cheol-soo Ahn and members of the Social Welfare subcommittee of the transition team accompanied Yoon to the site, and Tae-won Chey, Chairman of SK Group, and Jae-Yong Ahn, CEO of SK Bioscience were also present at the site. According to SK Bioscience, president-elect Yoon observed the entire vaccine R&D process, from the extraction of animal cells used for vaccine development and production to culturing, fermentation, purification, and analysis. Yoon said, “I will visit more R&D sites in the future. Korea’s economy, security, and health all depend on this.” Yoon said, “In my administration, I plan to spare no support for companies that develop vaccines and treatments for the pandemic, including SK Bioscience. If companies ask for loosening of some regulations in the R&D process, I will actively review it and make sure that the companies are not inconvenienced." Jae-Yong Ahn, CEO of SK Bioscience, said, “We need steady support from the government to continue on our hard-earned vaccine development capabilities." Transition team chair Cheol-soo Ahn said, “The new virus is an important national crisis that requires continued management. Using this as an opportunity, it is the new administration’s task to create a data-driven disease control and prevention system. To SK Bioscience, transition team chair Cheol-soo Ahn said,” The NA had always deferred making decisions on vaccine-related budgets as a low priority agenda, but the new administration’s goal is to secure vaccine sovereignty in Korea by investing in vaccine development so Korea could gain vaccine development capabilities.” On the morning of the same day, SK Bioscience had announced that its own synthetic antigen COVID-19 vaccine candidate’GBP510’ had shown superior immune response to its comparator in a Phase III clinical trial. After securing safety data on GBP510 within the month, the company plans to apply for domestic and overseas approvals for its drug as a vaccine used for COVID-19 prevention after basic vaccination .
- Company
- Cancer immunotherapy are being applied to expand benefits
- by Eo, Yun-Ho Apr 26, 2022 06:11am
- Tecentriq will apply primary therapy benefits for liver and non-small cell lung cancer from May According to related industries, Roche Tecentriq will be applied to liver cancer and non-small cell lung cancer indicators from May as Merck and Pfizer Bavencio (Avelumab) passed the Cancer Drugs Benefit Appraisal Committee. The benefit standards were set as the first solo maintenance therapy in adult patients with local progressive or metastatic urinary tract epithelial cell cancer that did not progress in platinum-based chemotherapy treatment. If benefit adequacy is recognized by the HIRA's Drug Benefit Evaluation Committee and drug price negotiations are concluded with the NHIS, it will be eligible for insurance benefits. In the case of Tecentriq, benefits will be applied in the first monotherapy if it is a metastatic non-small cell lung cancer with positive PD-L1 expression and no EGFR or ALK mutation from the 1st of next month. It succeeded in applying liver cancer benefits for the first time among immuno-cancer drugs. Combination therapy of Tecentriq and Avastin (Bevacizumab) can be given first-line benefit if both 3 or higher, Child-Pugh class A grade, and ECOG PS) 0 to 1 point are satisfied among patients with advanced hepatocellular carcinoma that cannot be operated or treated locally. MSD's Keytruda has expanded its standards for primary therapy for non-small cell lung cancer since last month. Benefits are also applied to patients with recurrent or refractory Hodgkin lymphoma and children aged 2 or older who have failed at least two previous treatments, patients with progressive (stage 4) non-small cell lung cancer with positive PD-L1 expression and no EGFR or ALK mutation, patients with metastatic non-flat non-small cell lung cancer without EGFR or ALK mutation, patients with metastatic squamous non-small cell lung cancer, and if autologous hematopoietic stem cell transplantation fails, or autologous stem cell transplantation is not a treatment option. Benefits are also applied to patients with recurrent or refractory Hodgkin lymphoma and children aged 2 or older who have failed at least two previous treatments, patients with progressive (stage 4) non-small cell lung cancer with positive PD-L1 expression and no EGFR or ALK mutation, patients with metastatic non-flat non-small cell lung cancer without EGFR or ALK mutation (combined with chemotherapy), patients with metastatic squamous non-small cell lung cancer (combined with anticancer chemotherapy), and if autologous hematopoietic stem cell transplantation fails, or autologous stem cell transplantation is not a treatment option.
- Policy
- Phase III for development of Alecensa & Keytruda
- by Lee, Hye-Kyung Apr 26, 2022 06:11am
- Roche Korea and MSD Korea are accelerating clinical trials to develop combination therapy for non-small cell lung cancer treatments. On the 22nd, the MFDS approved phase 3 clinical trials of MSD's Keytruda and Keytruda SC and Roche's Alecensa. All of these pharmaceutical companies begin clinical trials to confirm the safety and effectiveness of combination therapy using two or more drugs together. Alecensa received domestic permission for the treatment of patients with ALK-positive local progressive or metastatic non-small cell lung cancer. Phase 3 clinical trials will be conducted at Seoul National University Bundang Hospital, Asan Medical Center, and Seoul National University Hospital to evaluate the efficacy and safety of various treatments in the cohort of patients with stage 3 non-small cell lung cancer. Clinical trials are conducted to utilize multi-drug therapy along with comparative studies of Keytruda SC and Keytruda IV. In March, it was decided to expand insurance benefits as the primary treatment for non-small cell lung cancer in Korea and apply Keytruda's insurance benefits to recurrent or refractory typical Hodgkin lymphoma. This phase 3 clinical trial will be conducted at Korea University Guro Hospital, Chungnam National University Hospital, and Hwasun Chonnam National University Hospital to compare pharmacokinetics and safety of Keytruda SC administered with platinum-based two-drug chemotherapy in the primary treatment of metastatic squamous or non-squamous cell lung cancer patients. With the recent increase in targeted anticancer drugs that can be used to treat non-small cell lung cancer, the development of combination therapy using more than one drug is in full swing.
- Policy
- Exclude drugs that have increased due to COVID-19 from PVA
- by Lee, Tak-Sun Apr 25, 2022 06:08am
- It is reported that the pharmaceutical industry plans to officially propose to the government to exclude respiratory treatments that have increased their use due to the increase in COVID-19 patients from PVA. It is asked to reflect that the explosive increase in the use of the drug is inevitable due to the increase in the number of patients with infectious diseases, and that it has tried to meet the supply according to the government's policy. According to the industry on the 24th, the KPBMA will listen to the opinions of its members and suggest that respiratory treatments, which have increased in use due to the treatment of COVID-19 patients, should be excluded from PVA. An association official said, "As the number of home-based patients increased in the aftermath of Omicron, demand for respiratory treatments increased significantly, and in response to the government's request, we operated the factory two to three times more than usual to produce drugs. We plan to request that the public-private consultative body, which will be held at the end of this month, be excluded from the PVA" The NHIS revised in December 2020 to correct the use of drugs that have been confirmed to be used to treat infectious diseases when negotiating PVA. Correction was possible only when a temporary increase in use was confirmed among drugs designated by the head of the KDCA that stable securing and supply were needed to prevent and treat infectious diseases. The pharmaceutical industry is asking for the revised guidelines to be greatly expanded and reflected in this respiratory treatment. An official from the pharmaceutical industry stressed, "It is unreasonable and unfair to cut the drug price by targeting PVA for drugs that have exploded in use for a short period of time in the aftermath of Omicron." PVA is a system that adjusts drug prices as usage increases after drugs are listed to manage financial uncertainty, and if they exceed a certain level of expected claims agreed with the NHIS or a certain level of claims in the previous year, drug prices will be reduced by up to 10%.
- Company
- JAKi options added to Dupixent for atopic dermatitis
- by Eo, Yun-Ho Apr 25, 2022 06:07am
- Two new treatment options were added to the field of atopic dermatitis that had only Dupixent as an option. The new options are both JAK inhibitors. The Ministry of Health and Welfare had issued a pre-administrative notice on the ‘Details on the standard and methods for applying long-term care benefits (pharmaceuticals)’ and announced it will expand the scope of reimbursement for two types of JAK inhibitors – Lilly Korea’s ‘Olumiant (baricitinib),’ and Abbvie Korea’s ‘Rinvoq (upadacitinib)’ – in atopic dermatitis. With the approval, JAK inhibitors have entered the field of atopic dermatitis in just one year since applying for reimbursement (for Olumiant). By reimbursement standards, the drugs may be used for the treatment of patients with severe chronic atopic dermatitis whose condition has lasted for over 3 years and whose symptom is not adequately controlled despite the use of topical treatment (corticosteroids and/or calcineurin inhibitors) and does not respond to over 3 months of systemic immunosuppressants (cyclosporine or methotrexate) (which is measured by a 50% or more reduction of EASI) or is not eligible for their use due to side effects, and whose EASI score is 23 or higher before starting treatment. As both drugs are relatively cheaper than Dupixent, the drugs are expected to become major options in the field of atopic dermatitis in the future. Dupixent selectively inhibits IL-4, and IL-13, which are known as key cytokines that cause atopic dermatitis. The drug is highly effective as it targets specific cytokines, but may not be as effective in some patients. On the other hand, JAK inhibitors including Rinvoq are involved in a relatively broader set of cytokines. The cytokines deliver signals through various pathways including the JAK-STAT pathway by binding to their receptors on the cell surface, and JAK inhibitors target the JAK enzyme that orders proteins that play a key role in Immune-inflammation regulation. The differences in MOA also bring other differences. The biologic agent Dupixent is an injection type of drug whereas JAK inhibitors are small-molecule drugs that can be taken orally. Oral drugs like Rinvoq can be a good alternative that can increase convenience for patients who have trouble receiving regular injections in clinics. Meanwhile, the insurance authorities are discussing extending reimbursement of Dupixent to pediatric and adolescent patients with atopic dermatitis.
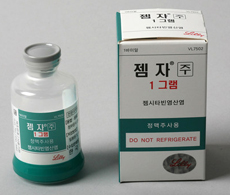
- Policy
- Boryung’s Gemzar switches to domestic production
- by Lee, Hye-Kyung Apr 25, 2022 06:07am
- In 2 years since Boryung Pharmaceutical acquired the domestic rights for Lilly’s anticancer treatment ‘Gemzar (gemcitabine HCl)’ in Korea, the company switched all its items to domestic productions. According to the Ministry of Food and Drug Safety, Boryung Pharmaceutical withdrew its import license for ‘Gemzar’ on the 21st, and switched the name of its domestic generic ‘Boryung Gemcitabine HCl Injection’ to the original name, ‘Gemzar,’ The company had co-promoted Lilly Korea’s Gemzar since 2014 and signed an agreement to transfer and acquire the rights of Gemzar, an anticancer treatment, in the local market with Eli Lilly in May 2020. Under the agreement, Boryung Pharmaceutical acquired the total rights of Gemzar in Korea including its sales and marketing authorization rights from the US company. Boryung and Lilly had been co-promoting Gemzar in Korea since 2015 Gemzar is indicated as a mono- or combination therapy in the first- and second-line treatment for pancreatic cancer, non-small cell lung cancer, bladder cancer, breast cancer, ovarian cancer, and biliary tract cancer. According to the market research firm IQIVA, Gemzar had sold ₩9.5 billion in 2016, which increased by around ₩3 billion in 5 years to record ₩12.44 billion in 2020. Gemzar’s sales account for 2-3% of Boryung’s total pharmaceutical sales every year, and sales are expected to increase due to cost reduction with the company converting all its imported products to domestic productions. Since 2019, Boryung had started driving the anticancer drug business and tripled its existing anticancer drug production facilities with the completion of its Yesan plant. Boryung plans to accelerate its anticancer drug business by introducing new products and expanding co-promotion products. Last year, the company had signed another contract to acquire and transfer assets for the schizophrenia treatment Zyprexa (olanzapine) with Lilly. Zyprexa had raised ₩14 billion in the domestic olanzapine market last year and is the No.1 prescribed product that holds a 50% share of the market. The agreement was made as part of Boryung’s LBA strategy it announced while conducting a capital increase of ₩98.5 billion in July last year. In addition to Gemzar and Zyprexa, Boryung plans to expand its portfolio by acquiring the domestic marketing approval of off-patent anticancer drugs that raise around ₩15 billion every year. Currently, the company is selecting candidates for digestive cancer, women's cancer, blood cancer, and lung cancer, and will also work to individual develop new anticancer drugs in the long term to strengthen its anticancer drug lineup.
- Policy
- Chong Kun Dang salt modified Entresto will be released soon
- by Lee, Hye-Kyung Apr 25, 2022 06:07am
- The market launch of Sacubitril/Valsartan Calcium, developed by Chong Kun Dang is imminent. According to the pharmaceutical industry on the 21st, Chong Kun Dang filed an application with the MFDS for permission for the drug developed by changing Valsartan Sodium, the main ingredient of Novartis' chronic heart failure treatment Entresto. This drug is likely to be a candidate substance called CKD-349, which was completed in June and October last year by conducting phase 1 clinical trials at Chungnam National University Hospital and H Plus Yangji Hospital, respectively. CKD-349 was tested with Entresto as a control drug. Entresto is the first double-inhibitor ARNI-based treatment that combines ARBI Valsartan and Sacubitril that inhibits Neprilsysin, and is currently recommended as a standard treatment in domestic and foreign heart failure treatment guidelines. As Entresto, which was approved for items in April 2016, was officially released in October 2017 after being listed, 13 domestic companies, including Hanmi Pharmaceutical and Chong Kun Dang, filed for patent judgment last year. On December 23, 2020, the Korean Intellectual Property Tribunal sided with generic companies by making a judgment on the establishment of the claim in a passive confirmation of the scope of rights of Entresto's crystalline patents filed against Novartis by 13 companies, including Hanmi Pharmaceutical and Chong Kun Dang. Entresto is protected by a total of five patents, including salt patents expiring in November 2026, use patent expiring in July 2027, crystalline patent expiring in September 2027, composition patent expiring in November 2028, and composition patents expiring in January 2029. If domestic companies succeed in targeting patents for use, they will be able to launch generics for Entresto early, which will end PMS on April 13. Entresto's outpatient prescription amounted to 32.3 billion won last year, up 37.3% from 23.5 billion won last year. This is the first achievement in four years since its release in October 2017.
- Company
- COVID-19 Tx Lagevrio lands in tertiary hospitals in Korea
- by Eo, Yun-Ho Apr 25, 2022 06:07am
- The oral COVID-19 treatment ‘Lagevrio’ has officially landed for prescriptions at general hospitals in Korea. According to industry sources, MSD Korea’s Lagevrio (molnupiravir) has passed the drug committees of tertiary hospitals including the Seoul National University Hospital in Korea. With the approval, the drug may be prescribed for in-hospital dispensing at tertiary hospitals and for in-hospital and out-hospital dispensing at general hospitals. Lagevrio, which is directly supplied by the Korea Disease Control and Prevention Agency through direct contracts, has been prescribed at dispensed at frontline convalescent hospitals, medical institutions, and designated pharmacies until now. The World Health Organization had issued conditional recommendations for the use of Lagevrio for patients at very high risk of hospitalization such as ▲those who haven’t received vaccinations for COVID-19, ▲those severely immunocompromised due to immunotherapy, etc., and ▲those with chronic diseases such as diabetes and published this revised COVID-19 treatment guideline in the British Medical Journal in March. Lagevrio is a ribonucleoside analog that is inserted in the place of the normal RNA needed in the viral replication process to induce lethal mutagenesis. As the first oral antiviral to be included in the WHO’s COVID-19 treatment guideline, the metanalysis of 6 clinical trials on Lagevrio in 4,827 patients showed that the drug reduced the rate of hospitalization by 36% compared to its comparator and showed a 3.4 day faster symptom improvement. Also, an interim and full analysis of a Phase III trial on Lagevrio showed that the drug demonstrated a reduction in the risk of hospitalization and death in outpatients with mild-to-moderate COVID-19 who were at risk of progressing to severe disease. In the planned interim analysis at 29 days after treatment, 14.1% (53/377) of the randomized patients had been hospitalized or died (8 cases) in the placebo group, whereas 7.3% (28/385) of the randomized patients were hospitalized with no deaths recorded in the Lagevrio group. The absolute risk reduction between the Lagevrio and placebo group was 6.8%, in which Lagevrio reduced the risk of hospitalization or death by around 50% by Day 29. Meanwhile, Lagevrio was approved for use in the UK as the first oral antiviral treatment for COVID-19 in November last year and then received emergency use authorization from the US Food and Drug Administration in December of the same year. MSD has signed supply agreements for the drug in 30 countries including Korea, the US, the UK, Germany, Australia, and Japan, and has been supplying Lagevrio to the countries.