- LOGIN
- MemberShip
- 2026-04-17 08:54:41
- Chronic illness was fatal for COVID-19 infected patients
- by park-sangjun Mar 04, 2020 06:28am
- The number of deaths in COVID-19 in Korea increased to 28, with fatal underlying disease. Much attention should be paid, especially if having heart disease and diabetes. Investigating the characteristics of 28 deaths (as of March 3, 00:00) announced by the KCDC, most of the unhealthy patients, such as decreased immune function in old age, died shortly after infection. First, the average age of the dead was 68.7 years. Exceptionally, younger 40-year-olds and 35-year-olds were included on the list of deaths, which included mental and severe liver disease, respectively. Medical staff are weighing the likelihood that their immune function was not normal in that a 40-year-old Korean male was young but had mental illness. As they died at home, more precise epidemiological investigations are needed. The 35-year-old man, who was a Mongolian, came to Korea for transplantation due to severe liver disease. He was diagnosed with transplantation due to severe symptoms such as ascites and jaundice. He died of infection with COVID-19 during palliative care. In particular, the survey reported a number of deaths from mental illness. Of the 28 deaths, eight were mentally ill (29%). Medical staff are looking for the cause in hospitals vulnerable to infection control and in patients with compromised immune function. Myung-don Oh, professor of the Infectious Diseases Central Clinical Committee, said, “All of the dead were mentally ill patients who were admitted to Cheongdo Daenam Hospital and being sedentary, and this is a very vulnerable environment for infection” and evaluated, “Infection of one person led to mass infection and low immunity patients seemed to have died”. "In addition to the lack of immunity, I think the patient is generally in a bad condition. The muscle mass is also estimated to be low, and there are a number of comorbidities, and this complex factor may have increased mortality", he said. According to the KCDC, most of them developed fever shortly after infection and died of pneumonia as they worsened. In addition, the chronic deaths were included in the list of deaths. The survey found that 36 percent of 28 patients had high blood pressure and diabetes, and some also had strokes and dementia. There were three patients with kidney disease (chronic renal failure or kidney transplant) and two patients with cancer who died shortly after infection. Ji-hwan Bang, a professor of infection medicine at SNU hospital, said that in many cases, patients with chronic diseases are unmanaged, and kidney disease patients (implants) or cancer patients are likely to have died due to poor immune function". According to this analysis, the KCDC orders thorough control of patients with chronic diseases with chronic illnesses. The KMA also recommends social distance campaigns as a way to prevent the conversion of national infections. Among these, children and healthy adults may be mildly infected with COVID-19, However, in case of an emergency, and it is recommended to pay attention to hygiene management in order to prevent an infection in case of emergency. On the other hand, according to the KCDC, there were confirmed patients in children and adolescents, but there are no deaths. There are 34 confirmed children aged 0-9 years, and 204, 10-19 years old.
- Company
- BMS-Celgene merger restructures Korean office
- by Eo, Yun-Ho Mar 04, 2020 06:27am
- The basic frame of Bristol-Myers Squibb (BMS) and Celgene’s merger company has been unveiled. Recently, BMS presented a newly formed division and appointed the head of Business Unit for the merger. The Commercial Business Unit, including marketing and sales, would undergo the biggest change. BMS has decided to integrate BMS’ existing Oncology division and Celgene’s Hematology division based on the latest reorganization decision. As a result, various anticancer immunotherapies, including Celgene’s star product Revlimid (lenalidomide), multiple myeloma pipeline and BMS’ Yervoy (ipilomumab) and Opdivo (nivolumab), would be promoted under a single Business Unit. The merged Oncology and Hematology Business Unit would be led by Senior Direct Lee Seunghun, who used to lead BMS’ Oncology Unit. Medical Affairs division has been integrated as well. Similar to Marketing and Sales divisions, individual account executives would take over multiple myeloma, lymphoma and anticancer treatment under the single roof of Medical Affairs division. As for the heads of Market Access division and Corporate Affairs division, Executive Director Yeo Dongho (a former Market Access manager at Celgene) and Senior Director Park Kye-hyun were appointed, respectively. Government Affair division and other business support divisions would maintain the existing system. The newly merged BMS-Celgene company would house the Region Office within ‘China & Asia Cluster Office,’ located in Singapore. The company’s China & Asia Cluster Market has welcomed Jesus Acebillo, originally from Novartis, as a new CEO.
- Company
- Whan In has signed domestic monopoly rights of 'Cariprazine'
- by Jung, Hye-Jin Mar 04, 2020 06:27am
- Whan In Pharm (CEO Won-bum Lee) announced on the 2nd that it has signed an exclusive contract to introduce Cariprazine, a schizophrenic drug from Gedeon Richter, Hungary. Under this contract, Whan In Pharm is responsible for commercializing, distributing and selling products such as domestic clinical trials and product approval of Cariprazine. Gedeon Richter will supply to Whan In. Cariprazine is a once-daily oral medication for schizophrenia, currently approved and released in 15 countries including the United States and Europe. The U.S. product name is 'Vraylar' and the European product name is 'Reagila'. Cariprazine is sold by Allergan in the United States as a treatment for adult type 1 bipolar disorder and schizophrenia. In Europe, it is approved for the treatment of adult schizophrenia and is sold by Recordati and Gedeon Richter. Won-bum Lee, CEO of Whan In Pharm, said, “We will build a partnership with Gedeon Richter to further strengthen neurology and Psychiatry influences of Whan In Pharm, and we are pleased to offer new treatment options to Korean medical staff and patients through Cariprazine”.
- Product
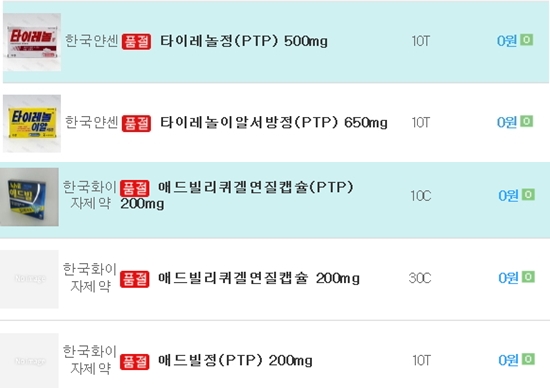
- Panic buying Tylenol-Advil in fear of COVID-19 fake news
- by Jung, Heung-Jun Mar 04, 2020 06:27am
- Related drugs are experiencing shortage in online pharmacy shops Tylenol and Advil are out of stock as their demands have skyrocketed due to online fake news encouraging panic buying of OTC drugs and claiming the Korean government’s containment of COVID-19 has failed. On Mar. 2, a multiple online pharmacies have posted notice on shortage of Tylenol, Advil and Aspirin. Apparently, the circulating ‘story’ is supposedly a chat between graduates of Seoul National University College of Medicine, which claims the outbreak has entered the ‘community infection’ phase that cannot be contained even with utmost care, and the prognosis of individual would heavily depend on their immune system. The made-up story also talked about concerning stories of how visiting hospital has gotten impossible once infected, and contracting the virus at selected testing sites. The story advised to hoard non-steroidal anti-inflammatory drug, antibiotics and antitussive, expectorant and mucoactive agents, such as aspirin, Advil and Tylenol. It also stressed about acquiring emergency food supply as self-quarantined patients would have to stay at home for over two weeks. The participants in the fake chat also argued seniors and individuals with cardiovascular or respiratory conditions are considered high-risk group. The people in the story assumed and hyped the fear of the public institutes and transportations shutting down. A pharmacist from Songpa-gu, Seoul, wondered “There has been a surge of customers buying specifically Tylenol and aspirins. It seems like inaccurate information has been circulating on Youtube and blogs.” Some pharmacies started securing stocks of the said products with abruptly heightened demands, and pharmacists are conflicted among themselves. Another pharmacist in Gyeonggi province commented, “Some pharmacists were worried about stocking up the drugs in the group chat. Basically they are encouraging panic buying, but as others reprimanded them the group chat was split.” The pharmacist emphasized, “It seems like fake news is kept on regenerated and disseminated. Professional pharmacists should not align themselves with it.” Another pharmacist in Gyeonggi province complained of the fake news spreading through social media as many of senior patients suddenly started looking for aspirin. Pharmacists from other regions have agreed on the problem of spreading inaccurate information as many of their customers were asking for aspirin, Tylenol and antitussive, expectorant and mucoactive agents. A few of pharmacy chains have sent out text messages to member pharmacies to inform purchase limit of Tylenol, and suggested purchasing other options with the same agent.
- Company
- In the aftermath of the COVID-19, the BA test was disrupted
- by Chon, Seung-Hyun Mar 04, 2020 06:27am
- COVID-19 infections have also influenced the generic development strategies of pharmaceutical companies. Due to the risk of infection with COVID-19, the bioequivalence task of medical institutions has been disrupted. This is an embarrassing situation for pharmaceutical companies that are conducting bioequivalence tests to reduce drug prices ahead of the reform of the generic drug price system. They are concerned whether the new generics' approval schedule may be delayed or the authorization for drug price maintenance will affect the generic bioequivalence test. According to the industry on the 1st, H Plus Yangji Hospital recently announced the suspension of bioequivalence testing due to the spread of COVID-19 to pharmaceutical companies and analysis agencies. Yangji Hospital has decided to suspend face-to-face testing related to bioequivalence tests, including first-dose monitoring, screening, monitoring, and initiation meeting, by March 15. Tests that have been completed on stage I will proceed normally. Although there were no COVID-19 confirmed patients at the hospital, the company decided to minimize face-to-face work to prevent infection. Yangji Hospital said, “We have made a lot of efforts to conduct a safe test in accordance with the spread of COVID-19, but decided to suspend the work on our own because it is too dangerous”. Yangji Hospital is a medical institution where the largest number of generic bioequivalence tests are conducted in Korea. Last year, 190 of 259 approved bioequivalence studies were performed at Yangji Hospital. Three out of four bioequivalence studies are in charge of Yangji Hospital. Yangji Hospital performed 116 of the 178 approved bioequivalence studies in 2018, accounting for 65.2%. Many medical institutions, except Yangji Hospital, are reportedly conducting bioequivalence testing, such as medication and blood collection. However, if the spread of COVID-19 accelerates in the future, the possibility of business disruption is raised. As a result, pharmaceutical companies are inevitable on generic development strategies. In particular, the development of new generics in preparation for the reform of the drug price system and the strategy of licensed generics may also be affected. The revised drug price plan, which is scheduled to be implemented in July, has the key point that the company must maintain the upper limit of 53.55% compared to the original before the expiration of the patent only when both the bioequivalence test and DMF. Whenever one requirement is not met, the upper limit drops by 15%. In the case of registered generics, the upper limit can be maintained at 53.55% if the bioequivalence test and DMF requirements are met within three years. In the case of new generic drugs, if they do not carry out direct bioequivalence tests, they will receive lower prices than before. The prolonged discontinuation of bioequivalence testing could be a variable in the plan to get higher prices for new generics. There is also same situation with licensed generics. Pharmaceutical companies can maintain their existing high prices only after conducting bioequivalence tests and judging suitability prior to July 2023 for generics licensed under preconditioning. Indeed, pharmaceutical companies are aggressively undertaking bioactivity testing. Approval Status of Monthly Bioequivalence Test Plan (Unit: Case, Source: the Ministry of Food and Drug Safety) According to the Ministry of Food and Drug Safety, 26 bioequivalence plans approved last month were up 85.7% year-on-year. A total of 44 bioequivalence plans were approved in January and February this year, 13 more than 31 January and February last year. A total of 91 bioequivalence plans were approved in the first half of last year, and increased significantly to 168 in the second half of last year. Licensed generics are actively tested. It is a bioequivalence test for the purpose of changing a manufacturer to produce generics approved by a consignment directly in its own manufacturing facility. If the biopharmaceutical test is conducted with generics produced through the drug product research and the equivalent result is obtained, the drug price reduction can be avoided through the change permit. Some pharmaceutical companies are considering launching bioequivalence trials for as many as tens of generics. The delay in the schedule of bioequivalence due to the spread of COVID-19 is a burden. An official of a pharmaceutical company said, “If the COVID-19 situation is prolonged, and the long-term interruption of bioequivalence testing will affect the generic development strategy and in addition to generics, it is also a concern to conduct normal clinical trials of new or improved drugs”.
- Policy
- Finalized gradual drug pricing in effect from July
- by Kim, Jung-Ju Mar 04, 2020 06:27am
- The government finalized the so-called ‘gradual drug pricing system revision’ that differentiates reimbursed generic pricing based on individual bioequivalence test. The approval qualification and pricing linkage, such as conducting individual bioequivalence test and using registered substance with DMF, would come in effect from July 1. While the weighted pricing benefit for incrementally modified drug (IMD) would be maintained as the pharmaceutical industry has demanded. Korea’s Ministry of Health and Welfare (MOHW) has recently finalized and published the partially revised Pharmaceutical Decision and Adjustment Standard the authority reissued preliminary announcement on last Jan. 28. From the first version unveiled last July 2, the revision took seven months to set the seal on the final version. ◆ Drug pricing calculation standard (including eye drop): The key amendments of the finalized revision are not too far from the version revealed last July. Depending on running either individual or joint bioequivalence test, reimbursed generic pricing would be reduced ‘gradually’ by 15 percent, and ‘cut-off line’ would also limit the number of generics with same agents. In detail, the qualifications (submitting individual bioequivalence test data or clinical data, and using registered drug substance) for differentiated pricing have been set down. Limiting 19 or less registered generics in same class, the pricing of the generics would be reduced gradually based on the qualification. Qualifying all, one or none of the conditions would price the generic by 53.55 percent, 45.52 percent and 38.69 percent of the original’s price, respectively. When the 20th generic in the same class is listed, the ‘cut-off line’ system would automatically price the generic at 85 percent of either the lowest maximum price in the class or 38.69 percent of the original’s price. As for eye drop, the applicant product would follow the already-listed eye drop’s pricing calculation, based on whether it is a single-use or multi-use product. Such changes would take in effect from coming July 1. However the pricing standard for eye drop would be enforced immediately. ◆ Weighted pricing benefit: The IMD weighted pricing benefit demanded by the industry would be maintained. Lump-sum changes in benefit for synthetics and biologics, extended period of weighted pricing and other details contained in the first version were kept. The weighted pricing benefit for IMD would be maintained until another IMD or a single or combination drug with same administration method, ingredients and formulation is listed. This includes incrementally modified combination drug. However, the benefit would be not granted to a combination drug, if there are two or more companies supplying single or combination drug, a part of the combination drug, with identical administration method, ingredients and formulation. And a drug listed after the weighted pricing benefit period has expired would not receive the benefit. The pricing benefit for synthetics (chemical) and biologics are unchanged from last year’s version. The benefit-granted period and number of company set individually would be amended and unified. Basically, the weighted pricing benefit for synthetics and biologics would last for a year, and it would be maintained up to two years as long as the number of company stays under three. And the ministry stipulated Drug Reimbursement Evaluation Committee (DREC) to deliberate adjustment of weighted pricing rate and benefit period up to two years upon request by pharmaceutical company. The said revisions would come in effect from next Jan. 1, except for the pricing benefit granted to an applicant that submitted both individual bioequivalence test data and proof of using substance with DMF would also come in effect from July 1 along with other changes.
- Company
- Eisai’s low-dose Pariet passed by general hospital DCs
- by Eo, Yun-Ho Mar 03, 2020 06:10am
- Low-dose Pariet is landing on general hospital prescription code. Pharmaceutical industry source reported Drug Committees in Severance Hospital, Samsung Medical Center, and Korea University Guro Hospital cleared proton pump inhibitor (PPI) Pariet 5 mg tablet (rabeprazole sodium), which received National Health Insurance reimbursement in last September. The newly launched Pariet 5 mg tablet has the lowest dose of the active ingredient among all PPI and rabeprazole medications available in Korea. Pariet 5 mg tablet, the only rabeprazole medication indicated for patients taking low-dose aspirin in Korea, differentiates itself from high-dose PPI medication as a lowest-dose PPI medication. The drug was released in Korea following the first global release in Japan. And by signing a co-marketing contract with Yuhan for marketing and sales in Korea, Eisai Korea is aiming to expand PPI market dominance. The current co-marketing deal is limited to low-dose (5 mg) tablet of Pariet, and the two companies are in discussion to expand the contract to cover all doses (10 mg and 20 mg) of the Pariet brand. The efficacy and safety of Pariet 5 mg tablet was confirmed through randomized, double-blind, multicenter PLANETARIUM study. Over 24 weeks, 472 patients receiving low-dose aspirin for cardiovascular or cerebrovascular protection, but also has a history of peptic ulcers, were divided into three groups each taking Pariet 5 mg, Pariet 10 mg, and mucosal protective agent teprenone 50 mg. As a result, Pariet 5 mg, Pariet 10 mg, and teprenone 50 mg patient groups showed cumulative recurrence rates of 2.8 percent, 1.4 percent, and 21.7 percent, respectively. It confirmed meaningful treatment effect in Pariet patient groups.
- Opinion
- [Reporter’s View] Irony in group-listing competing drugs
- by Eo, Yun-Ho Mar 03, 2020 06:10am
- Inducing competition between pharmaceutical companies could lessen the financial exhaustion, but National Health Insurance (NHI) reimbursement listing procedure would get delayed. Such is an irony we witness during high-cost new drug listing or reimbursement expansion procedures in Korea. The government could take an advantage of price competition between companies as drugs are expensive. Under the NHI system, savings could open up more opportunities. And more savings could affect coverage enhancement. But the issue is timing. It would be preferable to review reimbursement for all same-class drugs at once, but it is hardly feasible. Typically ranging from six months to over a year, the gap in timing of reimbursement listing application could be vastly dispersed. Regardless of the actual date of application submission and other delaying factors, the timing gap is crucial, because there are patients desperately waiting it. And the issue worsens usually with anticancer therapies. The government wishes to review drugs with significant financial impact all together, such as a CDK4/ 6 targeted therapy applying for reimbursement as a combination therapy with PD-1 or PD-L1 inhibiting immunotherapy Faslodex. It is obvious and somewhat understandable. A medication, a part of combination therapy, could fail to get listed for reimbursement and a pharmaceutical company could have been unenthusiastic about the government-proposed ‘trade-off (a policy to have the negotiating pharmaceutical company to lower pricing of off-patent drug and to compensate it with coverage on new drug).’ What has passed is past, and now is different. At least the tug of war between the government and companies should start properly. Whether it be Cancer Deliberation Committee or drug pricing negotiation, the government should be respectful and at least get ready to go. Pharmaceutical companies, who have applied first, are biting their nails as follow-on drugs competent to compete are emerging. COVID-19 is surely an urgent matter, but how could we let down cancer patients? Because there is no answer, a line should be drawn instead. We cannot wait for those sluggish drugs. Rather, the same-class group reimbursement listing review should rule out those latecomers.
- Policy
- The supply cost of NIP quadrivalent flu vaccine is absurd
- by Lee, Tak-Sun Mar 03, 2020 06:09am
- Manufacturers are opposing the government's pricing policy ahead of the Quadrivalent flu Vaccine (NIP) program. Damages will be inevitable by setting a budget without considering production costs more expensive than trivalent vaccines. In particular, pharmaceutical companies are complaining that they have to take losses because medical institutions purchase them at a uniform price without distinguishing between NIP vaccine and general vaccine. Therefore, it is argued that, at least, the PPS should make a bulk purchase of NIP vaccines. According to the pharmaceutical industry on the 2nd, 14 million people including 6month old~12 year olds, pregnant women, and 65-year-olds can receive quadrivalent flu vaccines as NIP for free as early as September this year. Previously, only trivalent vaccines were applied for NIP. quadrivalent flu vaccines have the advantage of being able to prevent four types of influenza viruses, such as two types of A and two types of B. The Ministry of Health and Welfare is said to have set the supply price of the quadrivalent flu vaccine for around ₩9,000 this year. This is 60-70% of the current market price (₩14,000 ~ ₩15,000). Manufacturers are protesting. An official of the related company said, “The price of tetravalent vaccines set by the Ministry of Health and Welfare is about 5% higher than the trivalent influenza vaccine subject to free vaccination in 2019, and this decision is not considered at all because the production cost is more than 25% higher than the trivalent vaccine”. Moreover, it is opposed to spending much less than the huge development investment cost. Hospitals can buy them in bulk all together, As a result, inventory losses were left to pharmaceutical companies. The problem is that the unit price of the NIP vaccine also determines the price of the general vaccine. Generally, the products supplied by the country from the Public Procurement Service are collectively purchased and distributed to the relevant institutions. However, influenza vaccines are purchased and distributed only for products aged 65 or older As a result, vaccines for children aged 6 months to 12 years and pregnant women, who are eligible for free vaccination, are purchased by the hospital and charged after receiving the vaccine. Therefore, the hospital purchases the flu vaccine at a uniform price without discriminating between NIP and general vaccines. This suggests that manufacturers and suppliers have limited room for pricing and supplying general vaccines. If the supply price of tetravalent vaccine is set at ₩9,000 according to the government's budget, the supply price is lower than in previous years, and the loss of the manufacturer is inevitable. Another official in the industry said, “Especially, manufacturers should take stocks of free vaccines that have not been inoculated after supply at the cost of loss, like general vaccines”. The official said, "The low price policy of this NIP business will undermine the development of pharmaceutical companies that invest R&D of billions of dollars, and domestic manufacturers have been contributing to improving vaccine self-sufficiency in line with the government's efforts to establish vaccine sovereignty". The manufacturers of tetravalent vaccines argue that the rational price of the free vaccination market needs to be reasonable, and that the PPS should consider bulk purchasing to relieve inventory burdens. An industry official said, "At least NIP vaccination price and general vaccination price should be made in a structure that can be dualized".
- Policy
- MOHW to speed up anticancer therapy reimbursement review
- by Lee, Jeong-Hwan Mar 03, 2020 06:09am
- The Korean health authority is to expedite the reimbursement review procedure for the latest anticancer therapies including Imfinzi, Bavencio, and Tecentriq. The authority is also reviewing data submitted by Opdivo and Keytruda for their additional indications. On Feb. 28, Ministry of Health and Welfare (MOHW) Pharmaceutical Benefits Division answered with the details on paper as National Assembly Health and Welfare Committee’s Lawmaker Kim Soon-rye (United Future Party) questioned about COVID-19 status report. Some patients taking a drastic measure of consuming canine dewormer fenbendazole as an anticancer treatment seems to have influenced the government accelerating the reimbursement listing and indication expanding procedure for anticancer treatments. MOHW official stated non-small cell lung cancer-targeting Imfinzi has completed negotiating with National Health Insurance Service (NHIS) and is waiting for deliberation by Health Insurance Policy Deliberation Committee (HIPDC). Cancer Deliberation Committee has recently passed Merkel cell carcinoma (MCC) treatment Bavencio, which now awaits follow-up procedure with Drug Reimbursement Evaluation Committee (DREC). Non-small cell lung cancer-targeting Tecentriq plus caboplatin plus etoposide combination therapy has been cleared by Cancer Deliberation Committee and now is in process of financial impact analysis. The reimbursement expansion reviews for Opdivo and Keytruda are still in review. MOHW explained the immunotherapies would be indicated as mono- and combination therapy for various cancer kinds. The ministry also clarified it would maintain the basic principle of primarily providing healthcare reimbursement to drugs with confirmed clinical efficacy and cost-effectiveness. MOHW official noted, “The ministry would set down reasonable reimbursement standard that enhances benefit to cancer patients and predictability of National Health Insurance expenditure.”