- LOGIN
- MemberShip
- 2026-04-17 08:54:41
- Policy
- The government invests ₩16 trillion for COVID-19
- by Lee, Jeong-Hwan Mar 03, 2020 06:09am
- The government announced on the 28th that it would invest about ₩16 trillion to finance, tax and banking to solve the problems caused by the spread of COVID-19. From March to June, the check and credit card income deduction rate will be doubled to the previous level, and individual consumption tax will be cut by 70%. VAT payments for self-employed business owners who suffered the damage of COVID-19 with annual sales of less than ₩60 million will be reduced to the level of simplified taxation by the end of next year. The government is also planning to submit additional revision budgets worth ₩6.2 trillion, the similar size of MERS crisis, with the aim of COVID-19 quarantine, diagnosis, treatment, recovery of damaged small business, public welfare and employment stability, and support of the stagnant regional economy. The economic vitality council held by Nam-ki Hong, Deputy Prime Minister & Minister of Economy and Finance included the contents. Nam-ki Hong, Deputy Prime Minister said, “As the spread of infections in the community worsened, the difficulties for the public's lives and the economy became severe and included special measures worth up to ₩16 trillion. It is to support strong overcoming damage and to reinforce consumption, investment and export slowdown”. The government will invest about ₩7 trillion in finance (₩2.8 trillion), tax (₩1.7 trillion), banking (₩2.5 trillion), and ₩9 trillion. in public and financial institutions. If the government adds ₩4 trillion of measures already in place, the government will spend ₩20 trillion. In addition, if the supplementary budget of more than ₩620 trillion is included, over ₩26 trillion will be spent to overcome COVID-19 outbreak. In February, the government revised the tax law in the extraordinary parliament and the income deduction rate of workers' credit card use will be from 15% to 30%, the use of ATM cards and cash receipts from 30% to 60%, and the use of traditional markets and public transportation will raise by 40 → 80% temporarily. In the same period, the individual consumption tax will be reduced by 70% from 5% to 1.5% within the limit of ₩1 million. Compared to the 30% reduction from the second half of 2018 to the end of last year, the cut has more than doubled, and the government expects the tax benefits to reach ₩470 billion. Benefits are doubled by adding 70% reduction in the individual consumption tax, which is applied to converting old cars to new cars for more than 10 years by the end of June. It also reduces tax burdens for small business owners who have been damaged by COVID-19. By the end of next year, VAT payments for small business owners with annual sales of less than ₩60 million will be reduced to the level of simplified taxation. The plan is to keep the simple tax base of ₩48 million in annual sales and keep the VAT burden low. As a result, the government expects that 900,000 individual business owners will receive tax incentives of ₩800 billion for two years, with annual averages of ₩200,000 ~ ₩800,000 per business per person The government subsidizes ₩500,000 in family care expenses until the end of COVID-19 outbreak when parents use family care leave to take care of children under 8 years of age, with the closing of nursery schools according to COVID-19. The job coupon system, which provides 20% of the total remuneration to elderly job participants in the form of local gift certificates, is also temporarily implemented for four months. The participant, however, must receive 30% of the total remuneration as a gift certificate, including any additional payment. It also introduces vacation, culture, tourism and maternity coupons, and a system that refunds 10% of the purchase price of high efficiency home appliances will also be implemented.
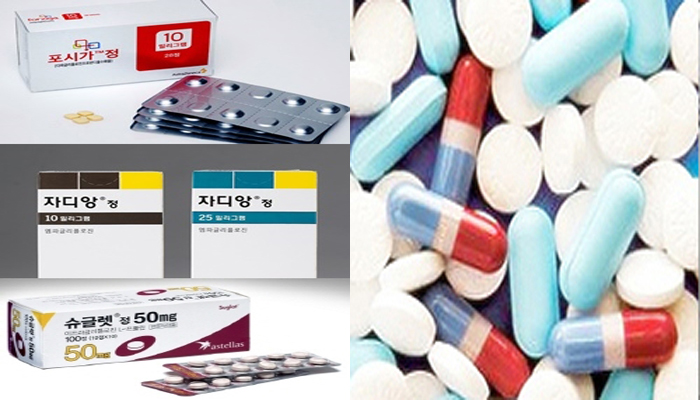
- Antidiabetic prescription trend shifts along CVD-HF option
- by Won, Jong-Hyeok Mar 02, 2020 06:23am
- An analysis research on last five years found oral SGLT2 inhibitor medications and GLP-1 receptor agonist injections have been highly preferred for type 2 diabetes prescription. By expanding an indication to benefit cardiovascular system, these medications have been highly favored by prescribers to treat diabetic patients who experienced myocardial infarction, stroke or heart failure. In the classes of SGLT2 inhibitor tablets and GLP-1 receptor agonist injections, Jardiance (empagliflozin) and Trulicity (dulaglutide) have been prescribed the most, respectively. The latest anti-diabetic treatment guidelines around the world strongly recommending those two products to diabetic patients with complications of cardiovascular disorders seem to be closely related to the recent shift in prescription trend. From top, Foxiga, Jardiance, Suglat An analysis on treatment preference (from 2013 to 2018) of endocrinologist, cardiologist and other physicians associated with type 2 diabetes was published online on Diabetes Care by American Diabetes Association. The detailed data was disclosed on Feb. 10 (https://doi.org/10.2337/dc19-1943). The research team of Professor Chintan V. Dave at Brigham and Women’s Hospital, Harvard Medical School, analyzed the administrative claim data from internal medicine specialists including endocrinologist. In the abstract of the study, the research team explained the shifts in prescription preference followed changes in drug labels and it has been heavily affected by common case of diabetic patients having complications of cardiovascular disease. The team noted a GLP-1 agonist injection with convenience of administering once weekly was highly preferred for an injection with additional option for cardiovascular disease. Change 1. Oral SGLT2 option to focus on additional cardiovascular benefit The detailed data of the study showed 3.4 percent of prescription for type 2 diabetic patients with myocardial infarction, stroke, or heart failure (CVD-HF) has prescribed SGLT2 inhibitor. It also should be highlighted the endocrinologists and cardiologists’ SGLT2 inhibitor preference have been increased by more than 1 percent. Moreover, the prescribers’ preference on drugs within the same class has shown a significant shift. Prescription rate of Invokana (canagliflozin), yet to be released in Korean market, has fallen from 100 percent in 2013 to 24.9 percent in 2018, with a steep drop of 75.1 percent. Whereas prescription rate of Jardiance soared from 13.9 percent to 65.6 percent in the same period and it topped the market. The researchers evaluated the preference on Jardiance has vastly grown as it won an expanded indication based on cardiovascular outcome trial (CVOT) ‘EMPA-REG OUTCOME’ disclosed in December 2016. The drug has confirmed to improve risk of cardiovascular events or death. Although the prescription rate of Jardiance on patients with CVD-HF was increased by 5.3 percent, the rate of Invokana or Forxiga (dapagliflozin) did not show much change. The research team assessed, regarding the prescription preference, “Canagliflozin had to place a black box warning to call attention to risk in amputation related safety issues, whereas the prescription preference rate on Jardiance has shifted drastically as its label changed with confirmed decrease in risk of cardiovascular events.” Change 2. GLP-1 injection options influenced by administration frequency Trulicity The study also found the prescription preference on GLP-1 receptor agonist for patients with CVD-HF has gone up by 3.9 percent. As for single medication, extended release injection with long administration term dominated the market. The first-in-class GLP-1 receptor agonist Victoza (liraglutide) showed high preference rate of 72.4 percent in 2013 as once-daily option, but it was dropped by 32.1 percent in 2018 and landed at 40.3 percent. On the other hand, once-weekly injection Trulicity’s preference rate climbed up by 43.8 percent from 5.0 percent to 48.8 percent in the same period. The trend was consistent in subgroup analysis in patients with CVD-HF. Compared to liraglutide and exenatide’s preference rate increasing by 5.1 percent and 2.1 percent, respectively, dulaglutide’s surge was notable. Released in the market from 2014, Trulicity is the only drug that European Medicines Agency (EMA) and the U.S. Food and Drug Administration (FDA) have indicated as primary and secondary major adverse cardiovascular events (MACE) prevention. The expansion was based on a large-scale CVOT ‘REWIND’ study. The team stated, “Once-weekly extended release injection Trulicity seems to have been preferred over once-daily liraglutide, and Trulicity’s impactful clinical evidence confirming cardiovascular disease benefit also has influenced prescriber’s preference.” Meanwhile in Korea, the growth of GLP-1 receptor agonist prescription volume has been prominent. A market research firm, IMS Health found Trulicity, launched in Korea in June 2016, has made approximately 10.2 billion won in the fourth quarter of 2019, leaping 30 percent from the same quarter the year before. It marked the first time the injection has taken over the market share over 30 percent. The overall sale volume of Trulicity in 2019 reached about 37.5 billion won, about 10 billion won, or about 39 percent jump from the year before. Based on sales volume, the injection’s share of overall antidiabetic injection market grew around 5 percent than the previous year and marked 28.6 percent. Basically, the injection is topping the GLP-1 analogues market, excluding insulin, with 84.6 percent of the market share.
- Company
- GSK speeds up development of COVID-19 vaccine
- by Eo, Yun-Ho Mar 02, 2020 06:22am
- GSK is focusing its capabilities on developing the COVID-19 vaccine GSK recently announced cooperation with China's biotech company Clover, following the Coalition for Epidemic Preparedness Innovations (CEPI). Together, Clover and GSK will jointly develop a protein-based COVID-19 vaccine candidate (COVID-19 S-Trimer). GSK will provide its pandemic adjuvant system to clover for evaluation of S-trimers in the preclinical research phase. Adjuvants are added to some vaccines to boost the immune response and produce a stronger and longer lasting immunity to infection than using the vaccine alone. Clover has China's largest commercial cGMP biopharmaceutical manufacturing capacity, and is expected to rapidly expand and mass produce COVID-19 vaccine production facilities. GSK also provides adjuvant technology to CEPI. CEPI will act as a coordinator between CESK-supported companies and GSK, who want to test their vaccine platform using GSK's adjuvant technology to develop effective COVID-19 vaccines. The agreement was signed between GSK and the University of Queensland, Australia. The University of Queensland began working with the CEPI in January to develop a 'molecular clamp' vaccine platform to enable rapid targeted vaccine production to prevent multiple viral pathogens. The CEPI has decided to extend its support for vaccine platforms to the development of COVID-19 vaccine candidates, and this early stage research is supported by access to GSK adjuvant technology. The announcement also includes four previously announced CEPI research programs in progress with CureVac, Innovio, the University of Queensland, Moderna, and the NIAID, for the development of COVID-19. GSK officials said, "We look forward to helping partners with COVID-19 vaccine candidates by providing their adjuvant technology. Adjuvant can reduce the amount of vaccine protein required per dose, as a result, more vaccine doses can be produced and consequently contributes to the protection of more people".
- Policy
- Remdesivir, Phase III clinical trials are imminent in Korea
- by Lee, Tak-Sun Mar 02, 2020 06:22am
- Gilead has applied for Remdesivir’s IND for COVID-19 patients at the MFDS. In Asian countries, including Korea (excluding China), the study targets 1000 patients. There is no country-specific number of subjects, but it is known to be recruited on a first come, first served basis. The MFDS announced that Gilead submitted a Phase III clinical trial plan for Remdesivir. An official from the MFDS said, "The IND was submitted today and plans to be reviewed for expedited examination". It will be approved as early as this week. Gilead Science said in a press release that it will conduct two Phase III trials to evaluate the efficacy and safety of Remdesivir in adult patients diagnosed with Corona19. Many medical institutions in Asia and many other cases of COVID-19 diagnosis plan to recruit 1000 patients from March. The trial will be conducted in accordance with the prompt review and approval of the US FDA. In China, two clinical trials are underway in Hubei Province. Results are reported in April, with 761 patients in clinical trials. Gilead says the trial evaluates two different durations of Remdesivir. In the first trial, approximately 400 COVID-19 confirmed patients with severe clinical features, will be randomly assigned to the 5- or 10-day Remdesivir group. The primary endpoint in the two trials is the same as the clinical improvement. Remdesivir is a drug that Gilead has already completed in Phase II clinical trials to develop Ebola. It is being used as the first treatment for COVID-19 in the United States, and it is getting attention as a treatment for COVID-19. It is a mechanism that prevents virus replication by binding to RNA, like Kaletra, an AIDS drug currently used in COVID-19 patients in Korea.
- Policy
- Single Hib vaccine Vaxem Hib exits Korean market
- by Lee, Tak-Sun Mar 02, 2020 06:21am
- Apparently, there are now only two single Haemophilus influenzae type b (Hib) vaccines left in the market that prevents cerebromeningitis in children by blocking Hib bacteria. Experts analyze, although a part of National Immunization Program, single Hib vaccine has lost its competitiveness in Korean market against other combination vaccines covering various viral infection. According to Korea’s Ministry of Food and Drug Safety (MFDS), GlaxoSmithKline (GSK) has dropped the license on Vaxem Hib on Feb. 25. It was not a surprise. GSK has suspended supply of Vaxem Hib since August last year. The company stated, “In the process of integrating and maintaining the production line to enhance efficiency in the vaccine business, the company has inevitably decided to suspend supply of Vaxem Hib that has been supplied only to Korean market.” Previously, GSK had suspended supply of Hib vaccine Hiberix in 2016. The license on Hiberix was terminated last August. ` Vaxem Hib was absorbed to GSK when it merged with Novartis Vaccine Division in 2015. But with the latest termination of license, the company is completely pulling out from the Korean Hib single vaccine market. Rather, the global company would concentrate on combined vaccine market with Infanrix IPV/ Hib combination vaccine. Now, LG Chem’s Euhib and Sanofi Pasteur’s Activ are the only Hib vaccines in Korea. Euhib was developed solely with Korean-made technology and received license in August 2010 after eight years of development costing approximately 7.7 billion won. Thanks to Euhib, Korea has a self-sustainable supply of Hib vaccine. Hib vaccine used to be a blockbuster market generating 10 billion won per each item. But Hib vaccine lost its competitiveness as combination vaccine covering Hib virus was designated as a National Immunization Program since 2017. According to last year’s data disclosed by IQVIA, Vaxem Hib, Euhib and Activ made around 700 million won, 500 million won and 64 million won, respectively. Meanwhile, pentavalent vaccine Pentaxim by Sanofi Pasteur has generated 11.9 billion last year. With a single shot, Pentaxim can prevent diphtheria, tetanus, pertussis, poliomyelitis and invasive infection by Hib bacterium. Frequently found in children under five, Hib bacteria could cause invasive infection-induced diseases, such as meningitis, epiglottitis, septic arthritis, pneumonia and cellulitis. Approximately 90 percent of pediatric meningitis is reportedly induced from the bacteria. Accordingly, Korea’s National Immunization Program has included Hib vaccine since March 2013.
- Company
- Hanmi-GC sign MOU for new drug R&D partnership
- by Chon, Seung-Hyun Mar 02, 2020 06:21am
- Hanmi Pharmaceutical and GC Pharma have inked the first new drug R&D partnership deal in the history. Two companies would concentrate their R&D capacity to increase the possibility of successful development of an innovative new drug. On Feb. 27, Hanmi Pharmaceutical and GC Pharma have announced the two have signed MOU on co-developing next generation enzyme replacement therapy (ERT) for rare disease on Feb. 26. At the GC Pharma headquarter, Hanmi Pharmaceutical President and CEO Kwon Se-chang and GC Pharma CEO Huh Eun Chul were convened at MOU signing ceremony along with other major R&D associates of two companies. The MOU promises two companies’ collaboration of candidate medicine exploration to the investigational drug commercialization targeting lysosomal storage disease (LSD). Based on Hanmi Pharmaceutical’s agent patent, GC Pharma is to lend their hands on developing the agent. Containing many hydrolysis enzymes, lysosome is a small organelle in a cell that functions as digestive system. However, excessive storage of lysosome could cause severe metabolic disease leading up to death. Apparently, about 4,000 patients in Korea are suffering from LSD. There are about 50 rare diseases induced by lysosomal storage. Currently, LSD patients are treated with ERT that intravenously injects enzyme made from a patient’s genetic recombination. The point of the MOU is to develop a breakthrough ERT that significantly enhances safety, half-life, administration convenience, and financial burden of the first generation treatment. Two companies aim to result in innovative new drug development by integrating Hanmi Pharmaceutical and GC Pharma’s R&C capacity. The plan is to maximize R&D synergy through sharing material resource, human resource, and R&D collaboration. R&D partnership between Hanmi Pharmaceutical and GC Pharma is a first time. Both companies anticipate raising the potential of new drug development by converging Hanmi Pharmaceutical’s new drug development capacity and GC Pharma’s rare disease treatment development expertise. GC Phrama has a history of developing Hunterase, a treatment for Hunter syndrome, a kind of LSDs. President Kwon Se-chang of Hanmi Pharmaceutical said, “GC Pharma has specialized expertise in rare disease sector, but more importantly it sets out a vision of innovation. We hope to present hopes to patients suffering from rare disease by putting together Hanmi Pharmaceutical’s rich experience in R&D and GC Pharma’s specialized expertise.” GC Pharma CEO Huh Eun Chul stressed, “We are more than thrilled to collaborate with Hanmi Pharmaceutical making new histories in pharmaceutical R&D in Korea. The R&D partnership of two companies with respective strengths would bridge the next generation treatment development in rare disease to an improvement in innovative treatment environment.”
- Opinion
- [Reporter’s View] Fake news deadlier than novel virus
- by An, Kyung-Jin Mar 02, 2020 06:21am
- “Don’t talk to anyone! Don’t touch anything!” These were the catchphrases printed on marketing poster of a movie Contagion, released in September 2011. Starring beloved Hollywood stars like Gwyneth Paltrow and Kate Winslet, the movie focused on an unknown infectious disease creating a global crisis. A woman, who came back from a business trip to Hong Kong, abruptly experiences seizure and dies. Soon after, people who had been in contact with the woman show similar symptoms and die, unfolding dramatic conflicts and chaos. Presumable by the poster’s catchphrase, the infectious disease portrayed in the movie spreads through people by touch. People get infected by handing a credit card back and forth, holding a bus handle, and taking away an empty plate from a restaurant table. In the film, all researchers of World Health Organization (WHO) and the U.S. Centers for Disease Control (CDC) come together to identify the pathogen. The feature film ends with somewhat cliché ending with intelligent scientists on a mission track down the patient zero and research antiviral agent that successfully develops into a vaccine. Nevertheless, the movie has been a hot issue again within social media channels due to the recent outbreak of COVID-19. The plot of the movie resembles a number of actual happenings during the COVID-19 outbreak. The investigated disease source found from animals like bat or pig, people losing mind from the news of severe symptoms reported all over the globe, and of course, the government taking an extreme action to shut down a whole city to stop the fast-spreading disease—the movie almost spoiled the recent outbreak. Despite unknown infectious disease itself, what drives people into extreme fear is fake information and conspiracy. The film realistically illustrates people surrounded by apprehension and insecurity as a freelance journalist publishes blog postings of untested folk remedy and claims of conspiracy among the public agencies. Maybe, the makers of the film wanted to tell the audience the lost trust and relationship between people could endanger the people more than the disease. The nine-year-old movie is getting highlighted again as the portrayed scenes overlap with the actual events taking place in Korea at the moment. Seemingly easing out at first, COVID-19 outbreak has been spreading rapidly since the 31st confirmed case. Centering Daegu and surrounding North Gyeongbuk Province, the number of confirmed cases has soared exponentially. Now the region is filled with fear and desolation. Discriminating terms like ‘Daegu Corona’ or ‘Wuhan of Korea’ are gushing out every day and fake news on confirmed patient’s travel route is spreading through Youtube, KakaoTalk, and online communities in a speed of light. Apparently, there has been a reported case of phishing regarding a confirmed patient. And the public outraged over an attention-seeking Youtuber that screamed “I’m from Wuhan. Get away from me” on a subway, who pretended to be an infected patient. The highly advanced information technology and dependency on social media seem to have worsened the spread of fake news than five years ago with MERS. WHO has recently warned, “Infodemic has reached a severe level,” and started seeking for countermeasure with global IT giants like Facebook, Google and Amazon. ‘Infodemic,’ or short for ‘information pandemic,’ means a chaos caused by inaccurate information and harmful rumor spreading swiftly through mass media and the Internet. Korean government also has declared, an individual disseminating inaccurate information would be charged with strict sanctions. Rumors are more contagious than the novel coronavirus. Sending out fake information without a hint of guilt could paralyze an entire local business, cause delay in disinfection process or even forever scar an innocent individual. To step away from such infodemic, everyone in the community should help out. Amid chaos, people need to close their ears on fake news and know better to block off inaccurate information spreading. Of course, the news media should also remind themselves that their mission is to deliver accurate information, and not focusing on shocking breaking news.
- Policy
- Additional indication of interferon or Kaletra is recognized
- by Kim, Jung-Ju Mar 02, 2020 06:20am
- The government recognizes widespread benefits of antiviral drugs even if it is not approved indication, including interferon, as well as AIDS treatments such as Kaletra for the treatment of The Ministry of Health and Welfare issued a partial amendment of 'Details on the Pharmaceutical Reimbursement Listing Standard and Method' on the 20th and released the relevant standards. The General Principles of COVID-19 coverage are included in the Mers Cronona Virus (MERS-CoV) benefit standard. Therapeutic agents are ▲Interferon preparations ▲Lopinavir + Ritonavir preparations ▲Antimalarial drugs Hydroxychloroquine preparations ▲Hepatitis C treatment Ribavirin preparations ▲Human immunoglobulin G (IVIG) preparations ▲ Antibiotic such as Oseltamivir and Relenza (Zanamivir) for external use. First, Interferon preparations include pegylated interferon and no single administration is recommended. The Lopinavir + Ritonavir formulation such as Kaletra is a treatment for AIDS and Ribavirin is not recommended as a single drug or primary medication. IVIG is IV-Globulin SN injection and can be used for sepsis or acute respiratory distress syndrome. Oseltamivir such as Tamiflu capsules and Zanamivir, such as Relenza, are used in cases where the influenza infection is complicated or strongly suspected. The reimbursement will be applied retroactively starting on January 4th. The Ministry of Health and Welfare plans to review the revised provisions one year from the date of implementation.
- Company
- Panicking with first confirmed COVID-19 in industry
- by Chon, Seung-Hyun Feb 28, 2020 06:03am
- Korean pharmaceutical industry now has a first confirmed case of the 2019 novel coronavirus, COVID-19. Working at a research laboratory without much of external interaction, the infected employee is not expected to create a severe impact. But pharmaceutical companies are reinforcing precautionary measures against disease transmission. More pharmaceutical companies are ordering their employees to work from home and desperately trying to minimize break at work. ◆ Dong-A ST R&D Center employee tested positive, first confirmed case in the industry Dong-A ST R&D Center Pharmaceutical industry source reported on Feb. 27, a female employee working at Dong-A ST’s R&D Center located in city of Yongin, Gyeonggi Province, has been tested positive for COVID-19. Within pharmaceutical industry, she is the first confirmed case of COVID-19. The employee has visited Andong, North Gyeongsang Province, from Feb. 22 to 23. On Feb. 25, the employee received a message that Andong Health Center has confirmed a positive case, where the employee has been. After receiving the relayed message from her, Dong-A ST ordered the employee to self-quarantined herself and had her tested at Giheung-gu Health Center of Yongin, Gyeonggi Province. Apparently, she is not a researcher, but a part-time staff assisting laboratory experiment. Dong-A ST immediately closed down and disinfected the R&D center and its nearby Human Resource Center. All employees at the R&D center were ordered to self-quarantine for two weeks. From 4 p.m. on the same day, Dong-A ST has ordered their headquarter employees to leave offices and work from until Feb. 28. Although the infected employee has not visited the headquarter office, the company has decided to disinfect the entire headquarter building to prevent any further spread of the infection. Sales force of the company has been working from home starting from Feb. 25. Because of the preventive measure, the entire Dong-A ST R&D Center is halted. As the research work can only be done at laboratories, working from home is technically impossible and ongoing research programs would be suspended for a while. Because of one employee at Dong-A ST R&D Center, the whole pharmaceutical company has been stirred. The laboratory assistant apparently had not directly interacted with many other employees at the building with the nature of her work. But Dong-A ST cannot risk affecting other pharmaceutical companies in case other employees at the research center has contracted COVID-19. ◆ Pharmaceutical companies on high alert, infected salesperson would impact the industry Pharmaceutical companies are significantly reinforcing precautionary measures with the first case confirmed within the industry. At the moment, the industry is relieved it was not a salesperson who contracted COVID-19. If it were the case, the impact would have been devastating for the industry. With their nature of visiting tens of healthcare centers a day, sales forces could be a dangers carrier of COVID-19. While the government focuses on blocking secondary and tertiary infection by disclosing and immediately closing down locations a confirmed patient has visited. Some criticizes pharmaceutical companies letting their salespeople go around healthcare institutes is being ignorant about the severity of the risk. For instance, if a salesperson visiting 20 healthcare institutes a day were to have been tested positive, medical professionals, all patients and their families could have been exposed to the risk. The government announces all routes of infected patient, and healthcare institutes and pharmacies visited by an infected salesperson would unavoidably make a loss. A hospital restricted entry of pharmaceutical salesperson Assuming a salesperson has contracted COVID-19, the industry would inevitably get scrutinized for spreading the virus. The infected salesperson would be reprehended for spreading the virus around and damaging healthcare institutes. On Feb. 20, Korean Medical Practitioners Association has issued an official statement to Korea Pharmaceutical and Bio-pharma Manufacturers Association (KPBMA) and other organization to refrain salespeople from visiting hospitals and clinics. With the situation, pharmaceutical companies’ sales divisions are basically open with no business. Started with multinational pharmaceutical companies from last month, working from home has been recommended among Korean companies as well. Pfizer Pharmaceutical Korea, Novartis Korea, MSD Korea, AstraZeneca Korea, Bristol-Myers Squibb Korea, Sanofi-Aventis Korea, AbbVie Korea, Amgen Korea, Gilead Sciences Korea, Mundipharma Korea, Baxter Korea, Janssen Korea, and Bridge Biotherapeutics have decided to have all employees work from home. As for Korean companies, the managements of Hanmi Pharmaceutical, LG Chem, GC Pharma, CJ HealthCare, Dong-A ST, and Dong Wha Pharm have banned their employees to visit healthcare institutes. As COVID-19 has been spreading rapidly since Feb. 19 centering Daegu, North Gyeongsang Province, more companies are considering on having office staffs, not just salespeople, to work from home,. A source from a pharmaceutical company commented, “Because COVID-19 continues to spread exponentially, most of employees have been ordered to cancel external meetings and to work from home for all employee is being considered.” Over the span of 27 days, 505 have been confirmed with COVID-19 infection overnight. The number of total confirmed cases in Korea has soared to 1,766. 13 deaths from the infection have been reported in Korea so far. Most of companies have started to work in flexible or reduced hours. The maximum working hours in Dong-a ST has been reduced to from 10 a.m. to 5 p.m. Chong Kun Dang also segmented commuting time frame for each division to commute in. Other companies have suspended use of company cafeteria or divided employees to have their meals in different time. ◆ Industry fatigued with long-term outbreak, business and performance to take unwanted break Severance Hospital and other general hospitals have completely banned ward visitation to prevent spread of COVID-19 Apparently, the pharmaceutical companies are complaining of their extremely fatigued from the prolonged outbreak. A pharmaceutical company insider noted, “Working from home for a month, business to be done face-to-face has been suspended. By avoiding meetings with vendors and clients as much as possible, the company’s productivity has dropped dramatically.” Beside the low productivity, the companies also have to worry about the steep fall in sales. Pharmaceutical market research firm UBIST found last month’s outpatient prescription volume marked 1.25 trillion won. The figure dipped by 4.4 percent compared to same period last year, and by 4.6 percent compared to the previous month. It contrasts from recent January prescription volumes that have constantly increased over 5 percent every year. The volume in January 2019 was raised by 8.5 percent than January 2018. Januaries in 2016, 2017 and 2018 have surged by 5.4 percent, 7.6 percent and 18.2 percent, respectively, than the previous years. Although the early Lunar Calendar New Years may have affected the prescription volume with less number of working days, majority of experts analyze COVID-19 outbreak is the biggest cause of fall in outpatient prescription volume. The public has been on high alert since the first case of COVID-19 was confirmed on Jan. 20. The outpatient prescription from then was stricken by increasing number of patients avoiding healthcare institutes. The companies are anxious that this month’s sales would be even worse. Except for patients with severe or chronic disease, other patients with mild condition would naturally hesitate to visit healthcare institute. A decrease in number of patients would bring down the drug sales straight. Also, hindered sales activity would cripple Korean pharmaceutical companies highly dependent on generics. Within the same class of drugs, generic sales are swung by sales force. Meanwhile, breakthrough drugs or new drugs without generic and alternative options would hardly get affected from impeded sales activity. Salespeople staying away from healthcare institutes could also drag down the inflow of cash. Usually, the salespeople visit healthcare institutes and pharmacies to settle the drug product payments. But as most of healthcare institutes are obviously shunning the salespeople, collecting bills would also get difficult. A pharmaceutical company insider commented, “The company is deeply stressed about counteracting against the COVID-19 outbreak that doesn’t seem to ease out any time soon. This year’s overall sales would plummet if the situation lingers longer.”
- Policy
- COVID-19 treatment is active in domestic clinical
- by Lee, Tak-Sun Feb 28, 2020 06:02am
- As the number of Korean COVID-19 patients has soared, the development of therapeutics is also active. In addition to multinational pharmaceutical company Gillead, domestic pharmaceutical company Komipharm has applied for clinical trial approval of the pipelines, and Immunemed's antiviral drug was approved for therapeutic use. Gilead said that it will begin a clinical trial to research the efficacy and safety of Remdesivir in adult patients diagnosed with COVID-19. The trial will evaluate two different dosing periods for Remdesivir. In the first trial, approximately 400 COVID-19 patients with severe clinical features, will be randomized into the Remdesivir 5 or 10 day group. In the second trial, approximately 600 patients with moderate COVID-19 will be randomized to the Remdesivir 5-day, 10-day, or standard therapy alone groups. The primary evaluation variable in both trials is the same as the clinical improvement. In multi-national trials, the number of subjects in Korea is unknown. However, it is known that it recruits 1,000 people on a first-come, first-served basis. The MFDS said that IND was received on the same day, and the exact information will be released after approval. The MFDS plans to quickly approve clinical trials. Remdesivir is a nucleotide analog pipeline that has shown extensive antiviral activity in vitro and in vivo animal models of new viral pathogens such as Ebola, MERS and SARS. The symptoms improved with the first patient in COVID-19 in the United States, and is currently undergoing clinical trials in 761 patients in Wuhan, China. Domestic companies also started to develop treatments. Komipharm announced on the 26th that it has submitted IND application to KFDA to approve the clinical trial plan to expand the application of COVID-19 pneumonia from the new drug 'Panaphix', which is under development to suppress cytokines. The company said the drug would treat the pneumonia caused by viral infections quickly and by inhibiting immune cell signaling factors and inhibiting the release of inflammatory cytokines TNF-α, IL-1β and IL-6. The company emphasized that it has been effective in treating pneumonia, especially in animal studies and clinical trials involving 372 patients with other diseases. The trial will include a total of 100 patients, 50 in each cohort. The antiviral drug developed by Immunemed has been approved for emergency use in patients with COVID-19. The MFDS explained to a COVID-19 confirmed patient who was admitted to Seoul National University Hospital on the 21st that they approved the use of the antiviral pipeline 'HzVSF v13' which is being developed by Immunemed. The drug is known to have humanized the virus suppressing factor (VSF). The company has been conducting Phase I clinical trials for influenza diseases since 2018. If there is a therapeutic effect in patients receiving this agent, the possibility of full-scale clinical trials cannot be excluded. The company, however, said it could not tell the question if it had benefited from patient administration. Meanwhile, emergency introduction has not yet been decided on Abigan ( Favipiravir), which is being used as COVID-19 treatment in Japan. In general, the use of the emergency introduction system to import domestic unlicensed items requires a request from the KCDC to the MFDS. The MFDS official explained, however, that no request has been made so far, and it is currently being discussed with the central clinical trial committee.