- LOGIN
- MemberShip
- 2026-04-03 10:38:09
- Company
- Hanmi partners with MSD for next-gen IL-2 analog development
- by Cha, Jihyun May 20, 2025 05:58am
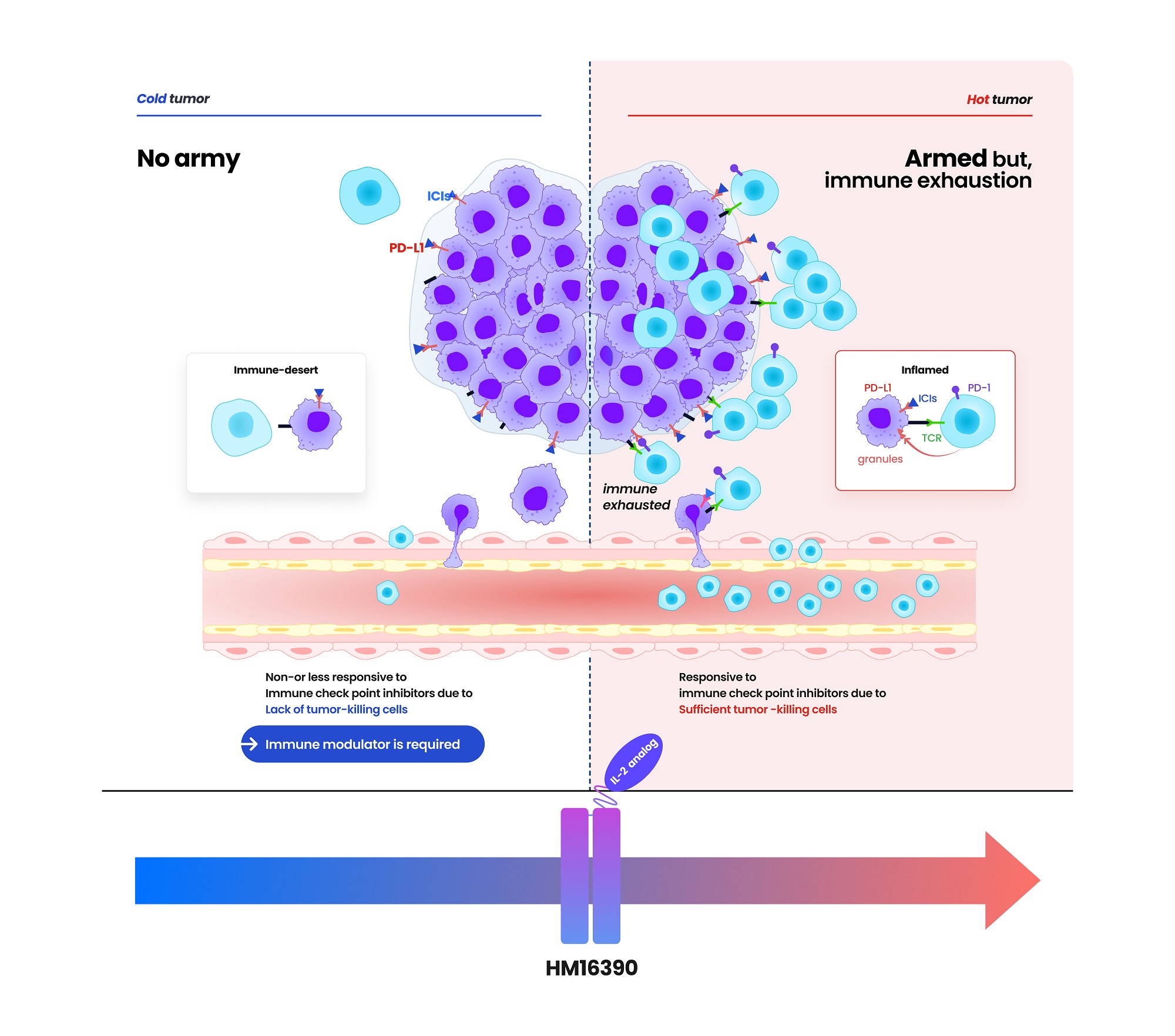
- Hanmi Pharmaceutical (CEO: Jae-Hyun Park) announced on the 19th that it has signed a clinical trial collaboration and supply agreement with U.S. Merck (MSD) to evaluate the combination therapy of its LAPS IL-2 analog 'HM16390' and MSD's anti-PD-1 immunotherapy 'Keytruda' (pembrolizumab). Hanmi Pharmaceutical will sponsor and oversee the Phase I clinical trial to evaluate the safety and efficacy of the combination therapy of HM16390 and Keytruda. MSD will supply Keytruda for the clinical trial. HM16390 is a next-generation IL-2 variant designed with a differentiated strategy that regulates the differentiation and proliferation of immune cells. HM16390 is designed to maximize antitumor effects by increasing the number of tumor-infiltrating lymphocytes that respond to immune checkpoint inhibitors in the tumor microenvironment through a mechanism that induces T cell proliferation and activation, thereby converting cold tumors with low immunogenicity into hot tumors with high immunogenicity. Currently approved recombinant IL-2 therapy 'Proleukin' is recommended for limited use due to side effect issues. Additionally, most IL-2 analogues in development focus on regulating the binding affinity of the IL-2 beta receptor, but this approach has shown limitations in terms of safety, according to Hanmi Pharmaceutical. Reducing the binding affinity of the IL-2 beta receptor decreases side effects such as vascular leak syndrome, but this also reduces anticancer effects. Conversely, increasing the binding affinity of the IL-2 beta receptor and eliminating binding with the IL-2 alpha receptor enhances anticancer effects, but this increases the risk of severe side effects such as cytokine release syndrome. To overcome these limitations, Hanmi Pharmaceutical has introduced a differentiated development strategy for HM16390. Unlike existing IL-2 candidates, HM16390 precisely regulates the binding affinity of the IL-2 alpha receptor, thereby ensuring safety while maximizing the efficacy of the drug. The company expects that this approach will maintain anticancer effects while minimizing serious side effects. (Data: Hanmi Pharmaceutical) HM16390 is an immunotherapy drug that maximizes the efficacy, safety, and durability by applying Hanmi Pharmaceutical's proprietary sustained-release platform technology, LAPSCOVERY. It is currently being developed as a sustained-release therapy that can be administered once per treatment cycle via subcutaneous injection (SC). Hanmi Pharmaceutical is developing HM16390 for use as a monotherapy and in combination with other immunotherapy agents for various solid tumors, and is currently conducting a global Phase I clinical trial. Dr. Jong Chul Park, Professor at the Massachusetts General Hospital (MGH) Head and Neck Cancer Center, Harvard Medical School, and the principal investigator for the Phase I clinical trial of HM16390 in Korea and the United States, said, “Through collaboration with MSD, we anticipate that the combination therapy of HM16390 and Keytruda will improve treatment outcomes for patients with advanced or metastatic solid tumors, and expect significant results in the future.” Young Su Noh, Director of Hanmi's ONCO Clinical Team, said, “Hanmi Pharmaceutical possesses a differentiated pipeline in the field of oncology, particularly in immunotherapy. We plan to sequentially showcase our research achievements through various academic conferences this year.”
- Company
- 'Oxlumo' for primary hyperoxaluria expected to be available
- by Eo, Yun-Ho May 19, 2025 05:56am
- The primary hyperoxaluria treatment, 'Oxlumo,' is expected to be commercialized in South Korea. According to sources, the Ministry of Food and Drug Safety (MFDS) reviewing Oxlumo (lumasiran) for approval. The MFDS granted 'Global Innovative products on Fast Track (GIFT)' designation to Oxlumo last year and orphan drug status in October of the same year. Oxlumo is an RNAi therapy for primary hyperoxaluria type 1 (PH1), a rare kidney disease, that was approved by the U.S. Food and Drug Administration (FDA), and the European Medicines Agency (EMA) in 2020. RNAi is one of the gene therapies considered a next-generation new drug technology, and it provides an advantage for specifically targeting human genes that cause diseases. PH1 is a rare disease in which the liver produces excessive oxalate. It causes the accumulation of oxalate crystals or calcium oxalate in the liver and urinary system. When the disease continues, the kidneys are damaged, requiring kidney dialysis. A treatment option for PH1 became available with the approval of Oxlumo in 2020. Oxlumo is an RNAi therapy targeting hydroxyacid oxidase 1 (HAO1), coding the oxalate-producing glycolate oxidase (GO) enzyme. It works by suppressing HAO1 and reducing GO production, ultimately reducing oxalate levels. Meanwhile, the efficacy of Oxlumo was found in a Phase 3 study involving 39 PH1 patients aged six years or older. Patients treated with Oxlumo had 65.4% lower oxalate levels in urine compared to the placebo group. Furthermore, 84% of patients treated with Oxlumo had oxalate levels close to normal. 52% of the group had recovered to the normal range.
- Opinion
- [Reporter's View] Strengthening GMP for sterile drugs
- by Kim, Jin-Gu May 19, 2025 05:56am
- The government has reaffirmed its existing position on strengthening GMP standards for sterile finished drug products in December. The Ministry of Food and Drug Safety recently met with sterile drug factory managers and stated that there will be no postponement of the enforcement of the “Regulations on Drug Manufacturing and Quality Control” in December. Under these regulations, facilities producing sterile drug products must implement the following measures: ▲Establish and implement a systematic contamination control strategy for the manufacture of sterile drug products; ▲Develop individual good manufacturing practices (GMP) for advanced biopharmaceuticals; ▲Clarify the details of the specific dosage forms, evaluation procedures, and methods for determining compliance with the good manufacturing practices (GMP). The MFDS has been preparing for the implementation of these measures since joining the Pharmaceutical Inspection Co-operation Scheme (PIC/S) in 2014. The MFDS believed that it was necessary to strengthen GMP standards for sterile products in line with international standards. To this end, the MFDS has provided the pharmaceutical industry with sufficient time. When revising relevant regulations in 2023, the MFDS required manufacturers of sterile finished drug products to replace outdated equipment within 2 years after the revision of the notice, and manufacturers of sterile APIs to do so within three years. Additional grace periods were granted for certain provisions. However, as the deadline for implementing the new regulations approaches, an increasing number of pharmaceutical companies are refusing to replace their outdated equipment. Instead of halting production of sterile drug products, they plan to shift production to contract manufacturing facilities. This is because replacing outdated equipment could cost tens of billions to hundreds of billions of won, depending on the facility. At first glance, this seems as if aseptic drug manufacturers are considering production halts to save equipment replacement costs. Additionally, given the sufficient timeframe provided, it seems that they have not been reacting for the past two years and only decided to halt production as the deadline for stricter regulations approaches. However, the story of sterile drug manufacturing plants is different. Their common claim is that the productivity of sterile drug manufacturing itself has become too low, regardless of the need to replace outdated equipment. In a situation where profits are almost nonexistent due to excessively low drug prices, they argue that they have no choice but to halt production, as additional costs of up to hundreds of billions of won would be required for replacements. For this reason, the pharmaceutical industry requested support for facility investment costs and improvements to the drug price structure during several recent meetings with the MFDS. However, the MFDS has drawn a line, saying that it will not provide investment support as it has already provided a sufficient grace period. Discussions on pricing sterile drugs have also stalled. If this situation continues, there are concerns that the supply shortage of sterile drug products will worsen by the end of the year. Already this year, 22 cases of injection supply discontinuations or shortages have been reported. It is known that about 10 sterile drug factories are seriously considering discontinuing production. If they discontinue their product supply by the end of the year, there are concerns that it will lead to a large-scale supply shortage. I agree with the policy direction that GMP should be strengthened to meet international standards. However, before doing so, we must first address the fundamental problem of low productivity. The MFDS must not dismiss the demands of sterile drug product manufacturers as “lack of preparation.” Unless the fundamental problems of excessively low drug prices and the resulting low productivity are resolved, it will be difficult to encourage sterile drug product manufacturers to participate, no matter how long the grace period is.
- Policy
- GSK starts domestic trial on its B7-H3-targeted ADC
- by Lee, Hye-Kyung May 19, 2025 05:56am
- GSK's antibody-drug conjugate (ADC) drug candidate 'GSK5764227,' designated as a breakthrough therapy by the US FDA last year, will begin clinical trials in Korea. On the 16th, the Ministry of Food and Drug Safety approved GSK's application for a Phase I clinical trial to evaluate the safety, tolerability, pharmacokinetics, and clinical activity of GSK5764227 in patients with advanced solid tumors. This Phase I trial will be conducted at three hospitals: Samsung Medical Center, Seoul National University Hospital, and Severance Hospital. GSK5764227 is a B7-H3-targeted antibody-drug conjugate (ADC) intended as an investigational new drug (IND) for adult patients with advanced small cell lung cancer (SCLC) whose disease has progressed after platinum-based chemotherapy, and for adult patients with relapsed or refractory osteosarcoma who have progressed on at least two prior lines of therapy. The FDA designates breakthrough therapies to expedite the development and review process for drugs that show significant improvement over existing treatments for serious conditions. GSK acquired exclusive worldwide rights for GSK5764227 from Chinese pharmaceutical company Hansoh Pharma earlier this year, excluding China, Hong Kong, Macau, and Taiwan. GSK will progress with its global clinical development and commercialization. GSK5764227 is composed of a fully human anti-B7-H3 monoclonal antibody covalently linked to a topoisomerase inhibitor payload (TOPOi). The FDA designated GSK5764227 as a breakthrough therapy for small cell lung cancer last year and added a bone cancer indication earlier this year. Additionally, GSK574227 received Priority Medicines (PRIME) designation from the European Medicines Agency (EMA) last year. Meanwhile, Hansoh Pharma is conducting Phase I, II, and III clinical trials of HS-20093 for lung cancer, sarcoma, head and neck cancer, and other solid tumors in China.
- Policy
- New drugs Bimzelx and Adempas reimbursed from June
- by Lee, Jeong-Hwan May 19, 2025 05:55am
- UCB Pharma Korea's Bimzelx and Bayer Korea's Adempas will be reimbursed by national health insurance starting on the first of next month. The Ministry of Health and Welfare announced an amendment to the Details on the standards for application of drug reimbursement (drugs). Bimzelx is a monoclonal antibody drug that targets both interleukin-17A and 17F and is used to treat moderate-to-severe plaque psoriasis. It can be used for patients who have not responded adequately to existing biological agents or those who have poor tolerability. Specifically, patients with chronic severe plaque psoriasis (adults aged 18 years or older) who have had the condition for six months or longer must meet the following criteria. Patients with plaque psoriasis covering 10% or more of their total body surface area, with a PASI (Psoriasis Area and Severity Index) score of 10 or higher, who have been treated with MTX (Methotrexate) or Cyclosporine for 3 months or longer but have not responded or cannot continue treatment due to side effects are eligible for Bimzelx’s use with reimbursement. However, patients who are contraindicated for both photochemotherapy (PUVA) and medium-wave ultraviolet B (UVB) therapy are only eligible if they have received MTX (Methotrexate) or Cyclosporine for at least 3 months with no response, or if treatment cannot be continued due to side effects. In addition, reimbursement is also available for patients with plaque psoriasis covering more than 10% of the total skin surface area who have been treated with photochemotherapy (PUVA) or medium-wave ultraviolet (UVB) therapy for more than 3 months but have not responded or cannot continue treatment due to side effects. Bayer Korea's Adempas Tab (riociguat) will also be newly covered by reimbursement. Adempas is an oral soluble guanylate cyclase (sGC) stimulator used for the treatment of pulmonary arterial hypertension (PAH) and chronic thromboembolic pulmonary hypertension (CTEPH). It reduces pulmonary vascular resistance by dilating blood vessels, improving exercise capacity, and delaying functional decline. It is indicated for patients diagnosed with pulmonary arterial hypertension (WHO Group I) corresponding to WHO functional classification Stages II–III, who have insufficient response to ERA and/or PDE-5 inhibitor pulmonary arterial hypertension medications, or who are contraindicated for both ERA and PDE-5 inhibitor pulmonary arterial hypertension medications. Among the approved indications, reimbursement is recognized in accordance with the general principles for pulmonary arterial hypertension drugs in the “Details” section when administered for pulmonary arterial hypertension.
- Company
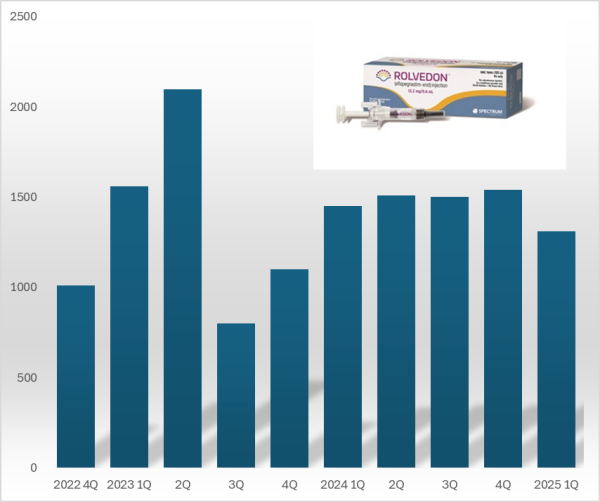
- Hanmi 'Rolvedon' reports US sales gain ₩18B in Q1
- by Chon, Seung-Hyun May 19, 2025 05:55am
- 'Rolvedon,' a treatment for neutropenia that Hanmi Pharmaceutical licensed out, continues to be popular in the U.S. market. Although the growth trend has stalled due to price reduction in the U.S., Rolvedon recorded over US$ 10 million in sales for six consecutive years. Rolvedon's cumulative sales amounted to KRW 200 billion in two years and six months since the drug launched in the U.S. market. According to the reports by Assertio Holdings on May 16, Rolvedon's sales for Q1 amounted to US$13.10 million (KRW 18 billion). However, it decreased by 9.7% from US$14.50 million in Q1 and 14.9% from US$15.40 million in the previous quarter. The company explains that despite slightly reduced sales due to Rolvedon's price reduction, increased sales partially recovered lost sales from the price reduction. Rolvedon Rolvedon is a new biopharmaceutical that Hanmi Pharmaceutical licensed to Spectrum in 2012. It is administered to cancer patients receiving myelosuppressive chemotherapy for the treatment or prevention of neutropenia. It belongs to the 'G-CSF' (granulocyte colony-stimulating factor) class, which stimulates granulocytes to increase neutrophil counts, showing a mechanism of action similar to Amgen's blockbuster drug Neulasta (pegfilgrastim). In South Korea, Rolvedon received new drug approval from the Ministry of Food and Drug Safety in March 2021 under the product name 'Rolontis.' Spectrum was acquired by Assertio Holdings, a pharmaceutical company specializing in central nervous system disorders·pain·inflammation, in April 2023. Assertio Holdings is a pharmaceutical company specializing in developing CNS and inflammation treatments. The company has products such as the non-steroidal anti-inflammatory drug Indocin and buccal dissolving film Sympazan. The company has succeeded in strengthening its oncology pipeline by acquiring Spectrum. Assertio Holdings is responsible for clinical development, regulatory approval, manufacturing, and commercialization of Rolvedon and Poziotinib worldwide, excluding South Korea·China·Japan. Rolvedon recorded its first U.S. sales of US$ 10.1 million in the fourth quarter of 2022, generating revenue in the United States. In December 2022, Rolvedon was included in the prevention·treatment options guideline for febrile neutropenia of the U.S. National Comprehensive Cancer Network (NCCN). Rolvedon posted sales of US$ 15.6 million in the first quarter and US$ 21.0 million in the second quarters of 2023, but declined to US$ 8.0 million in the third quarter. It rebounded to US$ 11.0 million in the fourth quarter of 2023, surpassing US$ 10.0 million in sales for six consecutive quarters through the first quarter of this year. Rolvedon's cumulative U.S. sales reached US$ 138.8 million (KRW 194 billion). At the San Antonio Breast Cancer Symposium (SABCS 2024) held in the United States last December, Assertio Holdings released the Phase 1 clinical trial results of the same-day administration of Rolvedon. Existing neutropenia treatments, such as Neulasta, can only be administered 24 hours after chemotherapy. Administering a neutropenia treatment on the same day offers the advantage of potentially reducing patients' hospital stays. The clinical trial was conducted in 59 breast cancer patients, administering Rolvedon 30 minutes after chemotherapy to evaluate tolerability and safety. The trial found that the average time to neutrophil count recovery with Rolvedon was 1.8 days. Regarding safety, the adverse reactions observed with Rolvedon were similar to those reported in previous clinical studies. Assertio Holdings said, "Rolvedon's sales exceeded our internal forecasts despite securing inventory in the fourth quarter to support first-quarter sales growth," adding, "We expect Rolvedon's sales to continue rising due to strong ongoing demand."
- Company
- Ebglyss may be prescribed in general hospitals in Korea
- by Eo, Yun-Ho May 16, 2025 06:21am
- Pic of Ebglyss The new drug Ebglyss for atopic dermatitis may be prescribed in general hospitals in Korea. According to industry sources, Lilly Korea's interleukin (IL)-13 inhibitor Ebglyss (lebrikizumab) has passed the Drug Committees (DCs) of 9 medical institutions nationwide, including tertiary hospitals like Asan Medical Center and Sinchon Severance Hospital, as well as Seoul National University Bundang Hospital. Accordingly, if Ebglyss is successfully listed for insurance reimbursement, the drug is expected to quickly lead to prescriptions. Lilly accepted a price below the evaluated amount (below the weighted average price of substitute drugs) presented by the Drug Reimbursement Evaluation Committee of the Health Insurance Review and Assessment Service in February and is currently negotiating Ebglyss’s drug price with the National Health Insurance Service. If listed, there will be 7 treatment options available for atopic dermatitis in Korea: biological agents (injectables) “Dupixent (dupilumab)” and “Adtralza (tralokinumab);” and JAK inhibitors (oral) “Rinvoq (upadacitinib),” “Civinqo (abrocitinib),” and “Olumiant (baricitinib).” The health authorities have recently been considering whether to allow JAK inhibitors to be used in cases where patients do not respond adequately to existing treatments (biological agents) or have poor tolerability, which is expected to further intensify market competition. If approved, Ebglyss will immediately benefit from the regulatory changes. The drug was approved by the Ministry of Food and Drug Safety in August 2024 for the treatment of moderate-to-severe atopic dermatitis in adults and adolescents 12 years of age and older (weighing at least 40 kilograms) who are inadequately controlled by topical treatments or for whom such treatments are not recommended. Ebglyss demonstrated its clinical efficacy and safety profile in a pivotal Phase III clinical trial. Patients who achieve a clinical response after 16 weeks of treatment can thereafter receive a maintenance dose (250 mg) every 4 weeks, making it a useful first-line treatment option for patients with atopic dermatitis in Korea. The clinical studies on which the license was based are the Phase III ADvocate-1, ADvocate-2, and ADhere trials. The trials evaluated the clinical efficacy and safety of Ebglyss in 1062 adults and adolescents with moderate-to-severe atopic dermatitis. In ADvocate-1 and ADvocate-2, which evaluated Ebglyss as a monotherapy, Ebglyss improved outcomes, with 58.8% and 52.1% (16.2% and 18.1%, respectively in the placebo arm) achieving Eczema Area and Severity Index (EASI) 75; and 38.3% and 30.7% (9% and 9.5%, respectively in the placebo arm) achieving EASI 90 during the induction period (weeks 0-16) compared to placebo. Also, after one year of maintenance therapy (Week 52), 81.7% of the Ebglyss arm achieved EASI 75 (vs. 66.4% in the placebo arm) and 66.4% achieved EASI 90 (vs. 41.9% in the placebo arm), demonstrating significant symptom improvement in the long term.
- Policy
- Trodelvy and new drugs reached an agreement with the NHIS
- by Lee, Tak-Sun May 16, 2025 06:21am
- Product photo of Trodelvy Three new drugs, including the triple-negative breast cancer (TNBC) treatment Trodelvy (sacituzumab govitecan), have completed negotiations with the National Health Insurance Service (NHIS) and are about to be included in the reimbursement list. These drugs passed the Health Insurance Review and Assessment Service (HIRA)'s Drug Reimbursement Evaluation Committee (DREC) in February and were undergoing negotiations with the NHIS. According to industry sources on May 15, the NHIS posted on its website that the companies of the TNBC drug 'Trodelvy,' the pulmonary hypertension drug 'Adempas Tab (riociguat, Bayer Korea),' and the plaque psoriasis drug 'Bimzelx Autoinjector (bimekizumab, UCB Korea)' have agreed on negotiation deals. During the 2nd DREC meeting on February 6, these drugs were acknowledged for their reimbursement appropriateness. The DREC granted reimbursement appropriateness for Trodevly's TNBC indication and Adempas' pulmonary hypertension among its efficacy·effectiveness. The efficacy·effectiveness of Adempas in chronic thromboembolic pulmonary hypertension (CTEPH) was not acknowledged for reimbursement appropriateness. For Bimzelx, the company received a decision that Bimzelx would meet the appropriateness of reimbursement if the company accepted an amount below the standard. After that, UCB accepted the condition and proceeded to negotiations with the NHIS. The company may have undergone negotiations with the NHIS for drug pricing and estimated claim amount, ultimately reaching an agreement. Among these drugs, Trodelvy received a decision of new drug innovativeness. It gained attention for becoming the first case to clear the DREC review, with its price being measured based on their ICER values. The Korea Alliance of Patients Organization (hereafter, Patients Organization) is asking for a quick reimbursement process since Trodelvy is the third-line treatment for adult patients with unresectable locally advanced or metastatic triple-negative breast cancer (mTNBC). The Patients Organization said, "Trodelvy is a high-cost drug, costing approximately KRW 1,500-2,000 per one cycle and some hundreds of millions of KRW yearly." They added, "Patients cannot continue treatment due to high-cost non-reimbursed drug costs and inevitably give up on receiving treatments." As these companies reached drug pricing negotiations with the NHIS, the drugs will be reviewed by the Health Insurance Policy Deliberation Committee. The drugs have higher chances of being included in the reimbursement list next month (June).
- InterView
- Hemlibra shows long-term efficacy and safety in hemophilia
- by Kim, Jin-Gu May 16, 2025 06:19am
- “The hemophilia patients’ only desire is to lead a normal life like everyone else. Hemlibra (emicizumab) has been found to be effective in preventing bleeding and safe in long-term follow-up studies. Moreover, it demonstrates clear bleeding prevention effects even during high-intensity exercise, significantly helping patients lead normal lives.” Dr. Steven Pipe, Professor of Pediatric Hematology-Oncology at the University of Michigan C. S. Mott Children's Hospital in the United States, said so while participating at the 'HAVEN Symposium' held on the 9th at the Sofitel Ambassador Seoul Hotel in Songpa-gu, Seoul. Dr. Pipe visited Korea to present the results of long-term administration of Hemlibra in patients with hemophilia A. He led the 'HAVEN3' and 'HAVEN4’ trials on Hemlibra. HAVEN3 is a study involving 151 hemophilia A patients who received emicizumab at a dose of 1.5mg/kg weekly or 3mg/kg every two weeks. HAVEN4 is a study involving 40 patients with hemophilia A who received emicizumab 6 mg every four weeks. Five years of follow-up data on the 191 patients showed that the annual bleeding rate (ABR) was 2.0 during the initial treatment period (weeks 1–24). At the long-term treatment stage (217–240 weeks), the annual bleeding rate fell to 0.8 episodes. Joint bleeds, a common complication in hemophilia A patients, also decreased in the long-term follow-up. The annual joint bleeding rate (AJBR) at the 217–240-week mark was 0.9 episodes. The proportion of patients who did not experience any bleeding during Hemlibra treatment increased from 62.2% at weeks 1–24 to 78.8% at weeks 217–240. Only 1 patient discontinued treatment over the five-year period. This case was a mild adverse reaction, and no association with the drug was identified. Twelve patients experienced inadequate bleeding control, and these patients continued treatment with an increased weekly dose of 3 mg/kg. Dr. Pipe highlighted the bleeding prevention effect of Hemlibra during various sports and physical activities. Similar to healthy individuals, he explained that there is little concern about bleeding even with high-intensity physical activity. The long-term follow-up results also showed that the annual bleeding rate (ABR) during sports and physical activities remained low at 0.91. Dr. Pipe said, “What hemophilia patients want most is ‘zero bleeding.’ They want to live their daily lives without any bleeding. Especially, they want to engage in high-intensity physical activities, including intense exercise, without worrying about bleeding.” In this sense, Hemlibra showed long-term efficacy as a preventive therapy. For example, at our hospital, 80% of patients diagnosed with hemophilia in childhood are currently receiving Hemlibra, and the drug shows definite bleeding prevention effects even during high-intensity exercise.” He also explained that the fact that Hemlibra maintains a higher level of clotting factor concentration for a longer period compared to existing treatments contributes to improving the patients' quality of life. Dr. Pipe said, “With previous medications, it was difficult to maintain consistent concentrations throughout the day, so patients had to take additional doses before intense exercise, which was inconvenient. In contrast, Hemlibra maintains consistent concentration levels, allowing patients to live their daily lives without such inconveniences.” Dr. Pipe plans to expand research on the long-term effects of Hemlibra in infants. He is conducting a long-term observational study (HAVEN 7) on the joint damage prevention effects of Hemlibra prophylaxis in 55 infants under one year of age with severe hemophilia A who have not developed antibodies to Hemlibra. R. Pipe stated, “Based on the results so far, infants receiving Hemlibra also exhibit a low annual bleeding rate. Even when bleeding occurs, it typically presents with traumatic bleeding patterns similar to those observed in infants of the same age. This is why we anticipate that Hemlibra will continue to demonstrate high bleeding prevention efficacy in the long term.”
- Policy
- CDDC to discuss partial reimb of anticancer drug combos
- by Lee, Tak-Sun May 16, 2025 06:18am
- The Health Insurance Review and Assessment Service announced today (14th) that it held a Cancer Disease Review Committee meeting and discussed the list of drugs eligible for partial reimbursement as anticancer drug combination therapies, as it had recently announced. The reimbursement standard for anticancer drugs will be revised starting next month based on the list made today. The details discussed at the meeting are expected to be disclosed when the reimbursement standard for anticancer drugs is announced. The Health Insurance Review and Assessment Service (President Jung-Gu Kang) announced that it had deliberated on the “Reimbursement Standards for Drugs Used for Cancer Patients” at the 4th Cancer Disease Deliberation Committee meeting in 2025. The details discussed on that day are follow-up measures to the revision of the general principles regarding “the scope and cost burden of drugs prescribed and administered to cancer patients among severely ill patients as drugs determined and announced by the Health Insurance Review and Assessment Service in accordance with Article 5, Paragraph 4 of the Rules on National Health Insurance Medical Care Benefits” among the details on the application criteria and methods for medical care benefits. At the meeting, the committee discussed the list of combination therapies eligible for reimbursement coverage, taking into account the indication and opinions from academic societies, with the aim of reducing confusion in clinical practice and enhancing predictability when applying the detailed guidelines to existing anticancer therapies and combination therapies with other anticancer drugs. The revised list will take effect on June 1. A total of 54 combination therapies were discussed, and 35 will be included in the revised guidelines. Therapies exceeding the approved indication were excluded. This decision was made as a minimum safety measure against the inappropriate use of combination therapies, such as those exceeding approved indications. The authorities explained that in the future, when academic societies submit requests for combination therapies related to this matter, the Cancer Disease Review Committee will review them and update the list of eligible therapies.