- LOGIN
- MemberShip
- 2026-04-03 10:38:09
- Company
- AZ 'Imfinzi' leads the paradigm shift in cholangiocarcinoma
- by Whang, byung-woo May 16, 2025 06:18am
- "Introduction of Imfinzi in cholangiocarcinoma treatment can be seen as a critical advance. That a new therapy offering the possibility of long-term survival has appeared after 12 years is highly encouraging." As new treatment options for cholangiocarcinoma are introduced, a paradigm shift is said to be brought to this area, which was previously neglected and poorly developed. Although it is too early to be certain of long-term survival in South Korea, Dr. Yoo says that it could be a game-changer since new treatment options can benefit patient. Dr. Changhoon Yoo, Professor in the Department of Oncology at Asan Medical Center in SeoulDr. Changhoon Yoo, Professor in the Department of Oncology at Asan Medical Center in Seoul, who has expertise in this field, shared limitations in the treatment setting of cholangiocarcinoma and possible improvements. The prevalence of cholangiocarcinoma is known to be higher in Asia regions, including South Korea, China, and Taiwan, compared to Western countries. However, patients are often in advanced stages when diagnosed due to the challenging early diagnosis. It is one of the cancers that is difficult to reach a curative intent. Dr. Yoo explained, "Cholangiocarcinoma has a low prevalence due to its high mortality, resulting in a low cumulative patient number relative to its incidence. Currently, only about 20–30% of cholangiocarcinoma patients are eligible for surgery, and the remaining approximately 70% must rely on drug treatments such as chemotherapy or immunotherapy." While liver fluke infection was the leading cause of cholangiocarcinoma in the past, new factors such as fatty liver have emerged due to the westernization of dietary habits. 임핀지(더발루맙)After Imfinzi (durvalumab) received approval from the Ministry of Food and Drug Safety in November 2022, it is continuously expanding its influence. Dr. Yoo said, "Although less than three years have passed since the indication approval and it is therefore difficult to confirm long-term survival rates, in clinical practice the proportion of patients showing improvement has increased compared with before," adding, "Considering that patients who received Imfinzi combination therapy early in the 2021 clinical study still have favorable outcomes, Imfinzi can be seen as providing benefits to patients." In particular, Dr. Yoo focused on Imfinzi's side effects and safety. Dr. Yoo explained, "Most side effects are caused by the cytotoxic chemotherapy agents used in combination, and there are almost no issues attributable to Imfinzi. It rarely causes patients to struggle or reduces clinical efficacy, making it a medication that is of considerable help not only to patients but also to healthcare providers." "Korean subgroup analysis data on Imfinzi demonstrates long-term survival effects" Another reason why Imfinzi combination therapy is gaining attention in cholangiocarcinoma is that overall survival (OS) was shown to be higher in Korean patients. According to the study results, the two-year survival rate in the Korean patient group receiving the Imfinzi combination therapy was 38.5%, more than twice the 14.1% observed in the group that received chemotherapy. Furthermore, the survival rate at 36 months was 21.0% in the Imfinzi combination group, more than double the 8.8% in the chemotherapy group. Dr. Yoo analyzed, "In my opinion, I consider the Korean subgroup analysis data from the TOPAZ-1 study very encouraging. These results reflect the rapid accessibility and thorough patient management within the healthcare system." In cholangiocarcinoma, where inflammation or adverse reactions often occur during anticancer treatment, leading to treatment interruptions and repeated hospital admissions and discharges, continuous cancer treatment itself is challenging. According to Dr. Yoo, it is particularly common for treatment to be paused for a month or two due to inflammation, worsening the disease, and cholangitis can occur even when the cancer itself is not progressing. Therefore, high accessibility to treatment is critical in cholangiocarcinoma. Dr. Yoo said, "In Korea, if inflammation or jaundice occurs, patients can quickly visit a hospital, receive a procedure, and recover, thereby immune checkpoint inhibitors can be administered continuously," and added, "Compared to countries with less-established healthcare systems, our accessibility and level of care are higher, so I believe the effectiveness of immune checkpoint inhibitors can be more pronounced." Dr. Yoo also said, "Cholangiocarcinoma is indeed a challenging disease, but prognosis has improved recently and long-term survival cases are increasingly common," and added, "To secure approval or reimbursement for new drugs, one side's opinion is not enough. It is also necessary for patients and healthcare professionals to raise their voices together." Reimbursement discussions remaining for Imfinzi combination Therapy… "The standard criteria application should be avoided" However, the Imfinzi combination therapy is only reimbursed for the chemotherapy, and the cost barrier remains high. Currently, Imfinzi's reimbursement criteria were established in November of last year. Following AstraZeneca Korea's application for the cost‐effectiveness track, the Health Insurance Review and Assessment Service (HIRA)'s Economic Evaluation Committee is expected to discuss Imfinzi's cost‐effectiveness this month. According to industry sources, this month's Economic Evaluation Committee will review the cost‐effectiveness of Imfinzi+gem-cis combination therapy as a first‐line treatment for locally advanced or metastatic cholangiocarcinoma. It will be forwarded to the Drug Reimbursement Evaluation Committee if it passes the Economic Evaluation Committee. In this regard, Dr. Yoo noted that, for the sake of patient access, the standard criteria should be avoided. For example, in hepatocellular carcinoma, the standard treatment, sorafenib, is not particularly low‐cost, so its price could not be matched when a new drug emerged. Still, it is disadvantageous for a new drug to meet such price benchmarks for rare diseases or those where drug development has lagged. Dr. Yoo said, "When nanoliposomal irinotecan was introduced as a second‐line treatment for pancreatic cancer, it also faced challenges in economic evaluation when compared with 5‐FU." And added, "Likewise, I do not think conducting a straightforward economic comparison between existing cholangiocarcinoma drugs, whose patents have expired and thus are inexpensive, and an innovative new drug developed after a decade is appropriate." Dr. Yoo also described cholangiocarcinoma as 'the lung cancer of the gastrointestinal cancer family,' emphasizing the importance of precision medicine in new drug development. "Although cholangiocarcinoma has one of the poorest prognoses among gastrointestinal cancers, I am interested in the possibility of developing targeted therapies based on genetic analysis of specific biomarkers," Dr. Yoo added, "Approximately 4–5% of cholangiocarcinoma patients carry specific gene mutations, making this a cancer type with high potential for precision‐medicine application, and research is underway." Finally, Dr. Yoo urged, "Support for precision medicine and targeted‐therapy development is urgently needed to broaden patient treatment opportunities." And, "I hope that cholangiocarcinoma patients will not lose hope and will actively pursue their treatments."
- Policy
- Gov't begins patient advocacy service for medical incidents
- by Lee, Jeong-Hwan May 15, 2025 06:24am
- The Korean Ministry of Health and Welfare (MOHW)The Ministry of Health and Welfare (MOHW) will start implementing the 'patient advocacy service,' which provides a legal representative matching service for mediating disputes to strengthen the rights of patients who are victims of medical incidents, effective this month (May). The service supports the conflict between a patient and a medical institute·healthcare provider. The MOHW will initiate the program on the 16th by appointing approximately 50 lawyers. The patient advocacy service will include patients who are victims of medical incidents during surgical operations and prescription·drug preparation errors that occur in hospitals. During the meeting with the Korea Special Press Association on May 14, Minjung Kwon, Director of MOHW's Healthcare Institution Policy Division, stated this. Medical incident patient advocacy service is related to the MOHW's 'Act on Special Cases Concerning Medical Accident,' which was part of the essential healthcare package. The 'Special Act for Handling Medical Accident' exempts criminal punishment of doctors when accidents occur during medical practice in essential healthcare departments. Accordingly, patient·citizen organizations shared criticism that the act invades the patient's rights while maximizing doctors' benefits. The MOHW has established a patient advocacy service for medical incidents to safeguard patient rights. To carry out this plan, the MOHW has completed the selection of patient advocates, focusing on lawyers with experience in medical malpractice litigation, among those who are both lawyers and hold medical licenses, and individuals with expertise and experience in medical accidents. The patient advocacy service is activated·operated during the dispute mediation stage before or instead of proceeding to court when a medical accident occurs. Patients who wish to use the advocate system must apply to the Korea Medical Dispute Mediation and Arbitration Agency. The MOHW plans to implement the patient advocate system as a provisional program while preparing the legal basis for it in the future. Director Kwon explained, "Many lawyers applied for the patient advocate service," and added, "We aimed to select around 50 people, but received more applications. After the appointment ceremony on the 16th and the training course, we plan to start the service at the end of May." "This is a program in which patients who wish to apply for medical accident mediation can apply to the advocate system, receive assistance, and proceed with mediation together," Director Kwon added, "The advocacy service is not intended for patients who want to file law suits. If mediation is activated through the advocate system, reducing cases that proceed to law suits will be possible." Lastly, Director Kwon said, "The term for a patient advocate is two years. After two years, performance will be evaluated, and reappointment will be made if no special circumstances exist." And added, "Many lawyers are interested in public service and have applied for this system. We intend to encourage advocates' participation and develop these various medical dispute mediation and assessment systems together."
- Opinion
- [Reporter's View] Korea’s vaccination space: ups and downs
- by Eo, Yun-Ho May 15, 2025 06:23am
- Vaccines are at the forefront of localized pharmaceuticals in Korea. Leading domestic companies such as GC Biopharma and SK Bioscience are now responsible for vaccine production and supplying preventive vaccines for various diseases, including influenza. The commercialization of so-called “premium homegrown vaccines,” such as pneumococcal protein vaccines, cervical cancer vaccines, and shingles vaccines, is also progressing steadily and is set to be completed soon. However, despite the improvement in vaccine competitiveness, the vaccination fee cartel among doctors in South Korea remains unchanged. For example, if the purchase price of a vaccine (the price at which doctors buy vaccines from pharmaceutical companies) is KRW 100,000, the implicitly accepted appropriate inoculation fee among doctors is around KRW 200,000. However, after some time passes, some local clinics lower the vaccination fee to as low as KRW 150,000 in an attempt to sell more at lower prices. Some even abandon their margins altogether, offering even lower prices through promotional events. These clinics then face criticism from neighboring doctors. They are labeled as traitors who betrayed their colleagues for their own gain. Such controversies can escalate into disputes between different medical specialties. The medical society of the relevant specialty may launch a campaign claiming that people should be vaccinated by specialists in their respective field. The interesting point lies in the margins. For vaccines, the difference between the inoculation fee and the purchase price, minus approximately 30% tax, constitutes the doctors' income. If they receive the “appropriate price” they claim—KRW 200,000 per vaccination—the actual income may vary depending on tax reporting, but amounts to roughly 70,000 won. For some workers, this is equivalent to a day's wage. Even if they receive KRW 150,000, approximately KRW 35,000 margin remains. This is by no means a small amount. Private practitioners claim that after including vaccination fees and labor costs, nothing remains. Meanwhile, during the flu season, they engage in fierce competition to secure supplies of flu vaccines, where the purchase price is KRW 10,000, and an approximately KRW 12,000 margin remains per inoculation. Private practice doctors are self-employed. This is a fact. Therefore, it is understandable that they want a high income that commensurates with the effort they put into obtaining their medical license. Additionally, since vaccine prices are not fixed, they have the right to set their own prices. However, they should refrain from claiming that twice the purchase price is the “appropriate price” and form a cartel. I earnestly hope they will finally “recognize” that the era when doctors' words were unquestioningly accepted is now over.
- Company
- Doctors ‘Reimb too slow for new drugs in Korea’
- by Eo, Yun-Ho May 15, 2025 06:23am
- Most doctors were found to believe that the speed of reimbursement for new drugs in Korea is too slow. The Korean Research-based Pharmaceutical Industry Association (KRPIA) released the results of a survey of 100 domestic medical professionals on the 14th. In January, the global polling agency Ipsos Research surveyed domestic clinical experts from various medical departments to ask their opinions on access to new drugs. According to survey results, all medical professionals unanimously answered that the period from the Ministry of Food and Drug Safety approval to health insurance reimbursement listing is “long,” with 74% stating it is “too long.” Regarding the appropriate period from approval to health insurance listing, 81% of medical professionals answered “up to 10 months,” with 41% deeming “within 6 months” as appropriate. As of 2022, it takes an average of 608 days (approximately 20 months) for innovative new drugs to be approved by the MFDS and listed for health insurance reimbursement in Korea. This is twice the appropriate period cited by most medical professionals (10 months) and significantly longer than in major overseas countries such as Germany (281 days), Japan (301 days), and France (311 days) during the same period. Furthermore, experts directly treating patients in the clinical settings anticipate that the swift and widespread introduction of innovative new drugs will provide substantial benefits for patient care. Eighty-three percent of medical professionals expected that “if drugs already in common use overseas are covered by health insurance in Korea, patient treatment outcomes will improve significantly.” A large proportion of medical professionals (85%) responded that “even for drugs already covered by health insurance, if reimbursement standards are eased to enable early or wider use, patient treatment outcomes will improve significantly.” In addition, 95% of medical professionals urged the MOHW to introduce a “fast-track listing procedure or system” for health insurance coverage, similar to the MFDS's Global Innovate Products on Fast Track (GIFT) system, which shortens the drug approval review period for severe or life-threatening diseases by up to 75%. Medical professionals who participated in the survey also expressed concerns about Korea's low access to new drugs. Ninety-four percent of medical professionals pointed out that “Korea's access to new drugs is lower than overseas,” and 97% answered that “the government must set appropriate and reasonable drug prices to prevent the ‘Korea passing’ phenomenon, where multinational pharmaceutical companies give up the launch of innovative new drugs in Korea due to domestic regulations on pharmaceuticals.” Seventy-six percent of medical professionals were concerned that the proportion of new drug expenditures (13.5%) in total domestic drug expenditures is 60% lower than the OECD average (33.9%), and 88% believed that reimbursement and access to new drugs in South Korea need to be improved to the level of the top 10 OECD countries. Medical professionals identified “enhancing access to innovative new drugs” as the top priority among the four key strategies of the government's Second Comprehensive National Health Insurance Plan (2024-2028). As the government pushes policies to reduce drug costs in response to an aging society, 67% of medical professionals expressed the view that “the budget savings should be reinvested into the health insurance fund.” As the survey respondents were clinical experts, they also requested that the opinions of those working on-site be more actively taken into account in the reimbursement decision-making process. Eighty-eight percent of medical professionals responded that “the opinions of medical professionals should be better reflected in the process of registering drugs for health insurance coverage,” and 80% said that “medical professionals should also be involved in the process of selecting patient population eligible for health insurance coverage.” A KRPIA official stated, “Medical professionals who care for patients on the front lines are deeply concerned about the difficulties patients face in receiving treatment due to delays in the introduction of innovative new drugs. They hope that new drugs will be listed for health insurance reimbursement more swiftly and with a broader scope. We anticipate that the results of this survey will contribute to the government’s fostering of a patient-centered treatment environment and policy design.”
- Company
- Arexvy opening the era of RSV vaccine
- by Whang, byung-woo May 15, 2025 06:22am
- As GSK launches the respiratory syncytial virus (RSV) vaccine Arexvy in South Korea, it will challenge the market on a full-scale. Arexvy is already expanding its market dominance in the global market with its strength as the first RSV vaccine. The company will likely focus on expanding vaccine awareness as it opens the RSV vaccine market for the first time. Dr. Ji-Yong Moon, Professor of Konkuk University On May 14, GSK Korea convened a press conference celebrating the launch of Arexvy, the world's first RSV vaccine. The company showcased a preventative strategy for seniors against RSV and Arexvy's clinical significance. Arexvy received approval from the Ministry of Food and Drug Safety (MFDS) at the end of December 2024 for the 'Prevention of lower respiratory tract disease (LRTD) caused by RSV in adults over 60 years of age and older.' Approval of Arexvy was based on results from two Phase 3 studies, 'RSV OA=ADJ-006' and 'RSV OA=ADJ-004,' involving adults 60 years of age and older. The study results showed that during the first RSV season, Arexvy significantly lowered the RSV-LRTD risk by 82.6% and severe RSV-LRTD risk by 94.1% in participants 60 years of age and older compared to placebo. Furthermore, the efficacy of the vaccine regarding RSV-A-associated LRTD increases and RSV-B-associated LRTD increases were 84.6% and 80.9%, respectively. Dr. Ji-Yong Moon, Professor of Konkuk University's Department of Respiratory-Allergy and Clinical Immunology, explained, "RSV infection causes complications, such as pneumonia, in adults aged 60 years or older and it may require hospitalization or lead to death in severe cases," and added, "Based on a retrospective study, 56.8% of the hospitalized adults over age of 65 had pneumonia and 10.6% of those died." Dr. Moon added, "Despite the high disease burden, awareness of RSV infection is poor, and differential testing is not well implemented, so the disease burden of RSV infection has been underestimated." He said, "RSV infection is as contagious as influenza, but there is no specific treatment other than supportive care, so prevention is most important." (from left) Professor Jacob Lee, Professor of the Division of Infectious Disease at Hallym University Kangnam Sacred Heart Hospital, and, Dr. Ji-Yong Moon, Professor of Konkuk University Experts consider that the launch of the first RSV vaccine, Arexvy, is expected to be significant from a preventive standpoint. Professor Jacob Lee, Professor of the Division of Infectious Disease at Hallym University Kangnam Sacred Heart Hospital, reported that RSV vaccination is already recommended in the United States. Dr. Lee said, "Arexvy showed a preliminary efficacy of 94.6% against RSV-LRTD in adults with one or more comorbidities," and emphasized, "Considering that 84% of domestic adults aged 65 and over have one or more chronic diseases, these data are noteworthy." Dr. Lee stated, "Arexvy was approved in the U.S. in 2023, real-world data on its use have accumulated, and excellent preventive efficacy has been confirmed in actual clinical settings." He said, "The U.S. Advisory Committee on Immunization Practices (ACIP) recommends RSV vaccination for high-risk individuals aged 60–74 and all adults aged 75 and older." Cost and awareness remain challenges… "NIP is necessary from a long-term perspective" Regardless of the preventive value of RSV through Arexvy, it remains uncertain how much influence it will exert in the market. Currently, Arexvy remains non-reimbursed in South Korea. Without a recommendation like that of the U.S. ACIP, there is a need to improve awareness. In particular, the fact that the recommended vaccination age in the U.S. is higher at 75 years old, compared to the domestic approval age of 60 and over, also raises questions. Hyunji Kwon, Business Unit Head at GSKRegarding this, Dr. Lee explained, "In South Korea, the Korean Society of Infectious Diseases is expected to announce a recommendation within this year, but it is unlikely to differ significantly from the U.S.," and added, "To enter the National Immunization Program (NIP), sufficient disease burden research needs to be conducted." Dr. Lee continued, "There is a cost burden and domestic data are lacking, but as the population structure changes, the number of vaccination targets will also increase. Through research data and cost-effectiveness, we can hope for RSV prevention through the NIP in the long term." GSK Korea has stated that it will work to improve awareness through TV advertisements and other means alongside the launch of Arexvy. Hyunji Kwon, Business Unit Head at GSK, added, "RSV is a disease with a great unmet medical need yet remains unfamiliar, so we will work to improve awareness among medical staff and high-risk patients," and added, "In a super-aged society in which, for the first time, the population in their 60s exceeds that in their 40s, we will collaborate with the medical community and the government to increase patient access."
- Company
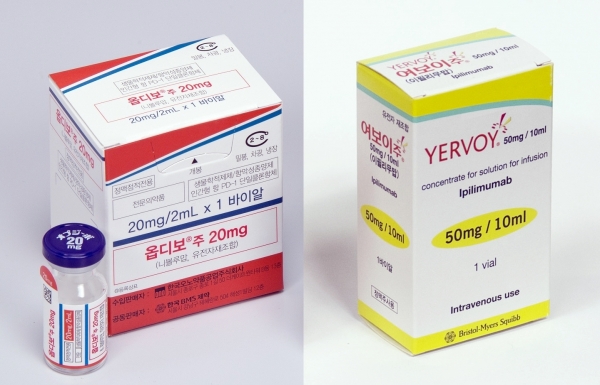
- Will a new trend emerge for liver cancer treatment?
- by Moon, sung-ho May 15, 2025 06:22am
- With new anticancer drugs entering the liver cancer treatment market, where combination therapy has been gaining prominence, attention is focused on whether a paradigm shift will occur. This is because a new competitive landscape is forming with the arrival of newly approved drugs and newly reimbursed drugs. # According to industry sources on the 7th, the US Food and Drug Administration (FDA) recently approved the combination therapy of Bristol Myers Squibb (BMS) and Ono Pharmaceutical's Opdivo (nivolumab)+Yervoy (ipilimumab) combination as a first-line treatment for adult patients with unresectable or metastatic hepatocellular carcinoma (HCC). The approval was based on the results of a randomized, open-label, global Phase III clinical trial (CheckMate-9DW) comparing the combination therapy of Opdivo and Yervoy (335 patients) with either lenvatinib or sorafenib monotherapy (333 patients). The trial was conducted on patients with unresectable or metastatic HCC who had not previously received systemic therapy. The CheckMate-9DW results showed that the median overall survival (mOS) in the Opdivo+Yervoy combination therapy group was 23.7 months (95% CI: 18.8-29.4), compared to 20. 6 months (95% CI: 17.5-22.5) in the control group (n=333), reducing the risk of death by 21% (HR=0.79; P=0.0180). In addition, the 3-year survival rate was 38% in the Opdivo+Yervoy combination therapy group, higher than the 24% in the control group. The objective response rate (ORR) was also significantly higher in the combination therapy group at 36.1% (95% CI: 31–41.5) compared to 13.2% (95% CI: 9.8–17.3; P
- Company
- 'Adempas' for pulmonary hypertension closer to obtain reimb
- by Eo, Yun-Ho May 14, 2025 06:11am
- Product photo of Adempas A new reimbursable treatment option for pulmonary hypertension is anticipated to be introduced. According to industry sources, Bayer Korea has finally reached an agreement with the National Health Insurance Service (NHIS) for its 'Adempas (riociguat).' Accordingly, after 10 years since it was approved in South Korea, Adempas will likely be included in the reimbursement list. Adempas' negotiation was a dosage negotiation, rather than a ceiling price negotiation. Bayer accepted the price 100% below the weighted average price (WAP) of a substitute drug and passed the Drug Reimbursement Evaluation Committee (DREC) of the Health Insurance Review and Assessment Service (HIRA) in February. Adempas was exempted from the drug pricing negotiations. This drug obtained approval in South Korea as an orphan drug in June 2014. Five products with different doses are available, and it has the efficacy and the effect in ▲patients with persistent/recurrent chronic-thromboembolic pulmonary hypertension (CTEPH, WHO Group 4) after surgical treatment or inoperable CTEPH, to improve exercise capacity ▲adult patients with arterial pulmonary hypertension (PAH, WHO Group 1) who have WHO functional class 2-3, to improve exercise capacity. Adempas has been known as the first novel drug to treat CTEPH. CTEPH occurs in patients with chronic pulmonary embolism who progress to chronic obstructive pulmonary disease (COPD) and develop fibrotic stenosis and occlusion, leading to pathological vascular remodeling and increased resistance in the pulmonary arteries. CTEPH is a chronic disease that causes progressive shortness of breath and right heart failure. Symptoms include dyspnea, fatigue, chest pain, dizziness, peripheral edema, cough, and hemoptysis, significantly impacting quality of life. Ultimately, it can progress to heart, kidney, and liver failure, potentially leading to death. Meanwhile, Adempas is a stimulator of soluble guanylate cyclase (sGC), an enzyme found in cardiovascular organs. The efficacy of the drug in patients with chronic thromboembolic pulmonary hypertension (CTEPH) was confirmed in Phase 2 and Phase 3 clinical trials. The clinical trial results showed that Adempas improved the study's primary endpoint physical activity and demonstrated superior tolerability. No unusual adverse reactions were reported. In the CHEST-1 study, results from comparing the 6 Minute Walking Test (6MWT) at 16 weeks from baseline showed that the patient group treated with riociguat had statistically significant improvement compared to the placebo group. In the PATENT-1 study, comparison of changes in 6MWT values at 12 weeks to the placebo group demonstrated statistically significant improvement, meeting the primary endpoint.
- Company
- U.S. executive order on drug price cuts raises hope
- by Kim, Jin-Gu May 14, 2025 06:10am
- U.S. President Donald Trump has signed an executive order to significantly reduce drug prices in the United States. This measure, which focuses on significantly reducing U.S. drug prices in line with overseas prices, is expected to have a positive effect on domestic biosimilars. However, some predict that the expected effects of this executive order will not be sufficient and that there will be no significant impact. Trump signs executive order to lower drug prices in the US... “Up to 90% reduction” According to local media reports on the 13th, President Trump signed an executive order on the 12th (local time) to lower the prices of prescription drugs in the US to the same level as other countries. The order essentially applies the most-favored-nation policy to drug prices in the US. Under the executive order, the US Secretary of Health and Human Services must promote a program that allows US patients to purchase drugs directly from pharmaceutical companies at a “most-favored-nation” price. In addition, the Secretary of Health and Human Services must impose the most-favored-nation pricing on companies within the US pharmaceutical industry within 30 days. This measure is interpreted as a move made to present a kind of price cap to pharmaceutical companies compared to drug prices overseas, and pressure them to lower their prices accordingly. Ultimately, this is expected to improve the intermediate distribution structure of Pharmacy Benefit Managers(PBMs) and further reduce the prices of expensive drugs. President Trump said at a press conference, “What we are trying to do is level the playing field for drug prices,” adding, “The American people will be able to purchase drugs at the lowest prices in the world.” He did not specify which drugs would be subject to price reductions. However, the administration has hinted that the price reduction could be as high as 90%. President Trump said at the press conference, “Drug prices in the United States could be reduced by 59%, 80%, or even 90%.” A day earlier, he had posted on his Truth Social account, “Prescription Drug and Pharmaceutical prices will be REDUCED, almost immediately, by 30% to 80%.” “Original drug price cuts expected to expand market for Korean biosimilars” The domestic pharmaceutical and biotech industry has responded with more optimism than concern to these measures. There are expectations that the price cuts targeting original drugs will have a positive impact on biosimilars. Celltrion said it expects the measure to create “a better business environment.” With the simplification of the intermediate distribution structure, it is expected that “this will provide positive opportunities for Celltrion's U.S. business activities,” and that “the dominance of pharmaceutical companies making high profit selling original products is expected to weaken, which could present market expansion opportunities for biosimilar companies.” Furthermore, it is anticipated that “biosimilar manufacturers will be able to negotiate drug prices directly with the government rather than through intermediaries such as PBMs, which could benefit both the government and manufacturers.” Additionally, it predicted that the prescription of biosimilars will increase as the prices of high-priced drugs are reduced. Currently in the US, through insurance companies and PBM systems, high-priced original drugs are prioritized for inclusion in formularies, followed by limited competition among biosimilar products, resulting in the addition of 2–3 products. During this process, rebates are paid to intermediary distributors, so the burden associated with these rebates will be significantly reduced with the new system. Celltrion stated, “Previously, biosimilar prices were set at the same high level as originals, making it impossible to provide substantial benefits to patients. However, if the intermediate distribution structure is improved through this executive order, the actual prescription prices of biosimilars will decrease, ultimately expanding biosimilar prescriptions to European levels.” Furthermore, it is also expected to provide an opportunity to launch new products in the US market. Celltrion explained, “If parallel imports are activated to supply medicines at most-favored-nation prices in accordance with this executive order, Celltrion will secure the opportunity to launch additional products that have not yet been introduced in the US market.” Another biosimilar company, Samsung Bioepis, is said to have shown a similar response. A positive outlook on the newly announced measure is dominant both inside and outside the company. The background is the expectation that the preferential policy for biosimilars will be strengthened to reduce medical costs. However, a Samsung Bioepis official said, “We are closely monitoring this policy,” without further elaboration. “It is premature to make specific judgements… Its impact on the Korean pharmaceutical industry will be limited The key issue is the strong opposition from the U.S. pharmaceutical and biotech industry. The U.S. pharmaceutical and biotech industry has consistently opposed the U.S. government's repeated attempts to lower drug prices, either by blocking them altogether or minimizing their impact. As a result, the actual evaluated effect of the drug price reductions was then minimal. In fact, President Trump signed an executive order for drug price cuts during his first term in 2018, but it was ultimately scrapped due to opposition from the pharmaceutical industry. At the time, an attempt was made to lower drug prices through an international reference pricing system, but a federal court raised procedural issues and put the brakes on the plan. President Joe Biden also conducted drug price negotiations with major pharmaceutical companies under the Inflation Reduction Act (IRA). However, the actual drug price reductions were limited to 10 drugs, including Eliquis, Xarelto, Januvia, Forxiga, Entresto, Enbrel, Imbruvica, Stelara, and Fiasp. Even these are limited to those who have Medicare - people aged 65 and older - in the United States. The drug price reduction measures will take effect in 2026, and the annual savings in medical costs are estimated at USD 6 billion (approximately KRW 8 trillion). This is considered insignificant compared to the total drug costs in the United States, which amounted to USD 805.9 billion (approximately KRW 1,142 trillion) last year. In this situation, the Trump administration's push for more stringent drug price reduction policies in the second term is expected to face strong opposition from the U.S. pharmaceutical industry. Additionally, criticism has emerged locally that the executive order lacks specific policies. The New York Times (NYT) criticized the executive order, stating that it “did not include specific policies such as pushing for legislation to reduce drug prices or revising drug payment regulations under government health programs.” In the same line, there are also projections that its impact on the domestic pharmaceutical and biotech industry will be limited. A representative from the pharmaceutical industry stated, “So far, only the direction to pursue drug price cuts has been outlined; there are no specific methods for how the cuts will be implemented, nor have the target products been determined.” The official added, “Its impact on domestic pharmaceutical and biotech companies is currently unclear, and even if there is an impact, it is expected to be minimal.” Another industry insider noted, “The recent administrative order requiring the manufacture of pharmaceuticals within the United States, as well as the upcoming pharmaceutical tariff policy expected next week, are likely to have a greater impact on the domestic pharmaceutical and biotech industry than the recent drug price reduction administrative order.”
- Product
- Patient organization proposes 6 policies as election pledges
- by Kang, Hye-Kyung May 14, 2025 06:10am
- A patient group has proposed six policies, including a “rapid reimbursement for new drugs and post-marketing adjustment system.” The Korea Alliance of Patients Organization submitted a 'Statement regarding Six Major Patient Policies' to political parties on the first day of the official campaign for the 21st presidential election on the 12th, urging them to reflect the proposals in their election pledges. The six proposed policies are: ▲Enactment of a Framework Act for patients to promote their treatment journey and protect their rights ▲Establishment of a Patient Policy Division within the Ministry of Health and Welfare ▲Establishment of an integrated support platform for patients fighting illness ▲Introduction of a system for rapid reimbursement and post-marketing adjustments for new drugs directly related to life ▲Institutionalization of a nursing care system ▲Promotion of a national responsibility system for essential organ transplant costs. KAPO stated, “The medical crisis that has lasted over a year and three months, triggered by the government's policy to increase medical school quotas. The resulting conflict with the medical community has caused immense suffering and harm to us patients. The conclusion of this medical-political conflict has left only a visual lesson: the government cannot defeat doctors by using patients' lives as a tool. Now, we patients and patient groups demand a government that will protect patients' lives and rights no matter what medical crisis arises.” They called for a government that will not hesitate to push forward policies and legislation for patients' treatment and rights even in the face of opposition from the medical community, and for a presidential candidate who will create a “patient-centered healthcare environment” where patients are no longer objects or targets but as “subjects” that actively participate for their treatment and rights. They stated, “We have conveyed patients' voices to presidential candidates as 6 major patient policies and hope they will be adopted as campaign pledges.” KAPO was established on February 4, 2010, under the slogan “Listen to Patients,” and currently includes the Korean Leukemia Patients Organization, the Korean GIST Patients Association, the Korea Kidney Cancer Association, All. Can Korea, the Korea Congential Heart Disease Patient Group, the Korea Psoriasis Association, the Korean Society of Type 1 Diabetes, the Korea Neuroendocrine Tumor Society, the Korean PROS Patient Organization, and the Korean Parkinson's Hope Association, among others, with over 92,000 patient participants.
- Company
- How has Leqembi changed the clinical site in 6 months?
- by Moon, sung-ho May 14, 2025 06:09am
- The overall treatment system on-site has been changing with the introduction of Leqembi, a new dementia drug that was released 6 months ago in Korea. In response, experts are evaluating the therapeutic effects and side effects of Leqembi (lecanemab, Eisai Korea) while demanding improvements to the health insurance reimbursement system in various areas, such as testing costs. At the same time, they are expressing anticipation for other new dementia drugs from global pharmaceutical companies that are likely to be introduced in Korea. The introduction of just one new global drug has changed the atmosphere on-site and in academic activities. #According to the medical community on the 12th, Eisai Korea’s Leqembi (Lecanemab), which was launched in Korea at the end of last year, has passed the Drug Committees (DCs) of major university hospitals and is rapidly expanding its prescriptions in Korea. Leqembi is a new treatment that removes amyloid beta (Aβ), one of the main causative substances of Alzheimer's disease. It specifically binds to soluble amyloid beta protofibrils and insoluble amyloid beta fibrils—the most toxic forms of amyloid beta—to reduce amyloid beta plaques in the brain. In particular, it is the first antibody therapy to receive full approval from the FDA in July 2023 for its efficacy and safety in delaying the progression of Alzheimer's disease and cognitive decline by removing the causative agent. In Korea, it was approved by the MFDS in May last year and has been prescribed in general hospitals since the end of November. However, there are limitations in that it can only be used in medical institutions that meet certain standards, as it requires collaboration with radiologists, neurologists, or other specialists to assess amyloid-related imaging abnormalities (ARIA) such as cerebral vascular lesions and cerebral hemorrhage, as well as facilities capable of administering Leqembi intravenously every 2 weeks and personnel to monitor adverse drug reactions. Nevertheless, Lecanemab is being rapidly adopted and utilized on-site not only at university hospitals but also at regional hub general hospitals, due to the disease's characteristic lack of a fundamental cure. According to an internal evaluation by the Korean Dementia Association, approximately 700 cases have been treated using the medication 6 months after its introduction in Korea. Seong Hye Choi, president of the Korean Dementia Association (Department of Neurology, Inha University Hospital), stated, “A patient weighing 50kg bears approximately KRW 1 million for a single administration of Leqembi. Considering that it is administered every 2 weeks, this amounts to KRW 2 million per month, and KRW 24 million per year for the patient. For a 40kg patient, the cost per administration is approximately KRW 800,000. Some patients are covered with indemnity insurance, and the treatment being well received onsite, even better than expectations.” Additionally, in clinical settings, there is a projection that the incidence of adverse reactions related to brain edema, particularly ARIA, observed during MRI scans in the course of Leqembi’s introduction may be relatively lower than what was shown in clinical studies. Kee Hyung Park, Chair of the Strategy and Planning Committee (Department of Neurology, Gachon University Gil Medical Center), said, “Japan introduced Leqembi before South Korea, and the drug is being actively used as it is covered by reimbursement. Among approximately 8,000 cases, ARIA occurred in 537 cases, which is 6.7% of all patients.” He added, “During the initial clinical trials, the incidence rate of ARIA was reported to be between 12% and 17%.” Park added, “In Korea, there have been no cases of patients experiencing severe side effects after receiving Leqembi. While it is still too early to draw definitive conclusions, it is reasonable to expect that side effects may be less common in Asians compared to Westerners. Amidst this, doctors have suggested that reimbursement for Leqembi itself is necessary, but that reimbursement for tests such as MRI is more urgent. Currently, MRI tests are required to assess cerebral vascular lesions and ARIA in patients receiving Leqembi. The logic is that tests that bring less health insurance burden should be reimbursed first, followed by a discussion on reimbursement for the drug itself. Choi said, “According to MFDS guidelines, patients receiving Leqembi must undergo MRI scans. This means that MRI scans must be performed before the fifth, seventh, and fourteenth injections. If reimbursement is applied to this part of the treatment, it will be able to somewhat reduce the burden on patients.” Choi also pointed out that long-term support for the costs of injection rooms and dedicated nurses (coordinators) provided for severe cancer patients is also necessary for dementia. Recently, Ewha Womans University Mokdong Hospital and Ewha Womans University Seoul Hospital introduced dedicated nurses following the adoption of Leqembi, but many medical institutions find it difficult to operate such programs due to the burden of labor costs. Jee Hyang Jeong of the Department of Neurology at Ewha Womans University Seoul Hospital (Chair, Public Relations Committee, KDA) explained, “When patients and their families request Leqembi treatment, it is necessary to provide detailed explanations about the treatment process, side effects, MRI scans, and other related matters. This entire process typically takes 30 to 40 minutes. We recognize this as a critically important process and have begun operating with dedicated staff internally. This was a big and dedicated decision on the hospital’s part.” Jeong added, “Ultimately, dementia is similar to cancer. We need to hire dementia specialists to educate patients and their families. The same applies to infusion rooms. Comprehensive improvements are needed, including reimbursement for the use of infusion rooms and labor costs, similar to cancer treatment.” Following the use of Leqembi on-site, expectations are also rising for another new dementia drug, Lilly's Kisunla (donanemab). Lilly's Kisunla cleared the regulatory hurdle - FDA - last year. Kisunla demonstrated efficacy in delaying cognitive decline in patients with early-stage Alzheimer's disease in the Phase III TRAILBLAZER-ALZ2 study. In the clinical trial, it delayed cognitive decline regardless of the disease's progression or pathological stage. Particularly, compared to Leqembi, it is evaluated as having a high potential for utilization in Korea due to improved convenience in patient administration. Choi said, “Kisunla has the advantage of a relatively long administration cycle and the possibility of discontinuing treatment midway through the course. I understand that Lilly, the manufacturer of Kisunla, is in discussions with the MFDS for its introduction in Korea. With mixed results in global markets, I expect the MFDS to make a decision based on the domestic situation.” Park also noted, “Rather than focusing on why approval was granted in the U.S. but not in Europe, we should examine the characteristics of the drug and its differences from existing medications. We need to assess this and consider how patients can individually select the appropriate medication for treatment when approval is granted domestically.”