- LOGIN
- MemberShip
- 2026-04-03 10:38:09
- InterView
- ‘SGLT2i+TZD promising for diabetics with fatty liver risk’
- by Kim, Jin-Gu May 26, 2025 05:55am
- A large-scale epidemiological study conducted on a Korean population has found that metabolic dysfunction-associated steatotic liver disease (MASLD), which commonly accompanies diabetes, increases the risk of cardiovascular disease and mortality. MASLD is not merely a liver disease but a cause that exacerbates overall metabolic disorders, making it a focus of recent attention. As a result, proactive treatment strategies to address it early are gaining prominence. Professor Cheol-Young Park of the Endocrinology Department at Gangnam Samsung Hospital said, “Combining SGLT-2 inhibitors and TZD-class diabetes medications early on could be a promising treatment option for diabetes patients with MASLD. This combination therapy not only improves blood sugar control but also regulates lipid metabolism and insulin resistance, while also mitigating the side effects of each medication.” A follow-up study on 500,000 diabetes patients revealed that those with MASLD had a 1.2-fold increased risk of death. Professor Cheol-Young Park, along with Professor Kyung Soo Kim from Bundang Cha Hospital, Professor Sangmo Hong of Hanyang University Guri Hospital, and Professor Kyung-do Han of Soongsil University, conducted a large-scale epidemiological study to examine the correlation between MASLD, cardiovascular disease, and mortality in patients with Type 2 diabetes. MASLD is a condition previously known as non-alcoholic fatty liver disease (NAFLD). It refers to the accumulation of fat in the liver in the presence of metabolic abnormalities (such as diabetes, hyperlipidemia, and obesity), regardless of alcohol consumption. It is not merely a liver disease but is considered a manifestation of systemic metabolic issues and is associated with cardiovascular disease, kidney disease, and certain cancers. Professor Cheol-Young Park’s team conducted a follow-up study on 500,000 patients with type 2 diabetes among 7.7 million participants in the National Health Insurance Service's health screening program in 2009. The results showed that patients with diabetes and MASLD had a 1.37 times higher risk of developing major cardiovascular diseases such as myocardial infarction and ischemic stroke. The risk of death from all causes also increased significantly by 1.21 times. This trend was consistently observed even in those with mild NAFLD (grade 1). In patients with NAFLD but without diabetes, the absolute risk of developing cardiovascular disease within 5 years was 1.23% to 1.42%. In contrast, patients with NAFLD and diabetes had a significantly higher absolute risk of developing cardiovascular disease within five years, ranging from 3.34% to 4.66%. The risk of death also showed a similar trend. Professor Cheol-Young Park explained, “This study shows that MASLD does not simply affect liver health, but also has a significant impact on overall cardiovascular outcomes. In particular, the results showing that patients with diabetes and NAFLD have a higher risk of cardiovascular disease and death highlight the importance of treating both conditions together.” “SGLT2i+TZD combination, a promising option for treating MASLD in patients with diabetes” Regarding these complex metabolic disorders, Professor Park proposed the SGLT-2 inhibitors and TZD drug combination as a promising treatment strategy. The two drugs have complementary effects in alleviating insulin resistance and reducing fat accumulation in the liver through different mechanisms. TZD class drugs promote the differentiation of subcutaneous adipocytes, redistribute fat from visceral depots, suppress hepatic glucose production, and enhance glucose uptake in muscle and adipose tissue, thereby improving insulin sensitivity. SGLT-2 inhibitors induce energy loss by excreting glucose through the kidneys, thereby indirectly promoting fat oxidation and reducing liver fat. Professor Park explained, “TZDs redistribute fat outside the liver to reduce fat accumulation within the liver, while SGLT-2 inhibitors reduce blood glucose levels and encourage the use of fat as an energy source. When used in combination, they regulate the metabolic environment through different pathways, producing a synergistic effect in improving MASLD.” Furthermore, Professor Park emphasized that the combination of the two drugs has the potential to offset each drug’s side effects. TZD-class drugs are associated with side effects such as weight gain and edema. In this context, the weight-reducing and diuretic effects of SGLT-2 inhibitors can partially offset these side effects. Additionally, by fundamentally improving insulin resistance, they reduce the burden on insulin-secreting cells (β-cells), thereby alleviating hyperinsulinemia and potentially contributing to reduced insulin dependence in the long term. Professor Park added, “Combination therapy is an integrated strategy that can regulate not only blood sugar levels but also lipid metabolism, insulin resistance, weight, and lipid status. It is necessary to consider this treatment approach early on for patients with both diabetes and MASLD. “MASLD, a key risk factor for cardiovascular disease… Early use of combination therapy required to improve prognosis” Professor Park explained that considering the high prevalence of MASLD in Korea, early combination therapy is important for patients with both diabetes and MASLD. He emphasized that since MASLD is not merely an indicator of liver health but also increases the risk of metabolic disorders and cardiovascular disease, an integrated management approach is required from early on. MASLD is deeply associated with various metabolic abnormalities such as insulin resistance, dyslipidemia, visceral obesity, and hypertension. These metabolic abnormalities interact synergistically to negatively impact the cardiovascular system. For example, insulin resistance accelerates fat accumulation in the liver while also causing dyslipidemia through increased triglycerides, reduced HDL cholesterol, and changes in LDL cholesterol particle size. Visceral obesity increases systemic inflammation by secreting inflammatory cytokines from fat cells, while hypertension increases atherosclerosis and cardiac burden, making it a direct risk factor for cardiovascular disease. Professor Park stressed, “The factors do not exist independently. When one worsens, it has a cascading effect on the others. MASLD originates at the core of this metabolic imbalance and should be recognized not just as a simple liver disease but as the starting point of a systemic disease.” Professor Park noted, “For patients with MASLD accompanying diabetes, strategies that not only control blood sugar but also reduce liver fat accumulation and improve the overall metabolic environment should be used. The combination of SGLT-2 inhibitors and TZDs could be an effective approach that can simultaneously target these complex goals.”
- Company
- Pfizer’s Lorviqua is granted reimbursement in Korea
- by Whang, byung-woo May 26, 2025 05:54am
- Lorviqua (lorlatinib), a treatment for ALK-mutated non-small cell lung cancer (NSCLC), has been approved for reimbursement as a first-line treatment, heralding a tectonic shift in the field. Experts saw this as a positive development in addressing unmet patient needs and improving access to treatment. In this sense, the presence of third-generation treatments for ALK-positive metastatic non-small cell lung cancer is expected to grow further. On the 21st, Pfizer Korea held a press conference to commemorate the expanded reimbursement of Lorviqua (lorlatinib), a first-line treatment for ALK-positive metastatic non-small cell lung cancer (NSCLC), highlighting the significance of the milestone. Ji-Youn Han, Professor of Hematology and Oncology at the National Cancer Center Lorviqua is a third-generation ALK tyrosine kinase inhibitor (TKI) designed to be effective against ALK mutations and to effectively penetrate the blood-brain barrier (BBB). In May 2022, it was granted reimbursement as a first-line treatment, 3 years after the indication was expanded to include ALK-positive metastatic non-small cell lung cancer as a first-line treatment. Patients with ALK-positive non-small cell lung cancer account for over 80% of all lung cancer cases and are characterized by relatively young age and a history of minimal or no smoking. Lorviqua’s efficacy as a first-line treatment was confirmed in the global Phase III CROWN trial. According to the 5-year follow-up results of the global Phase III CROWN clinical trial, Lorviqua demonstrated an 81% reduction in the risk of disease progression or death compared to the crizotinib group in patients with no prior treatment experience. Also, the median progression-free survival (PFS) for Lorviqua was not reached at 60.2 months of follow-up, regardless of brain metastasis status, while the median PFS for crizotinib was 9.1 months at 55.1 months of follow-up. According to a 5-year analysis of the CROWN trial, this is the longest progression-free survival rate achieved among ALK-positive non-small cell lung cancer treatments to date. Ji-Youn Han, Professor of Hematology and Oncology at the National Cancer Center who presented at the event, said, “Among non-small cell lung cancer, which accounts for the majority of lung cancer cases, treatment for ALK-positive non-small cell lung cancer, a major genetic mutation, has progressed from the first-generation crizotinib to the third-generation Lorviqua. According to the five-year follow-up analysis of the CROWN study, this is the treatment with the longest median progression-free survival (mPFS) among ALK-positive NSCLC treatments to date.” Han added, “There are studies showing that 25-30% of patients with ALK-positive metastatic non-small cell lung cancer do not receive later-line treatment and that the main cause is rapid clinical deterioration due to tumor progression. The reimbursement of Lorviqua as a first-line treatment is significant in that it addresses unmet needs and greatly improves access to treatment.” Furthermore, the efficacy and safety profile of Lorviqua was consistently demonstrated in studies involving patients in Asia, including Korea. In a study of Asian patients, after 5 years of follow-up, the median PFS for Lorviqua was not reached, while the median PFS for crizotinib was 9.2 months. Furthermore, Lorviqua demonstrated a 99% reduction in the risk of disease progression and death compared to crizotinib at the 5-year mark. However, some have expressed concerns about the risk of central nervous system side effects associated with Lorviqua. One percent of patients who experienced cognitive impairment as an adverse reaction after receiving Lorviqua discontinued treatment. Nevertheless, given that Lorviqua demonstrates superior efficacy compared to existing treatments, Han believes it can become the preemptive choice as a first-line treatment. He stated, “Based on the data, Lorviqua shows positive results in patients with over 5 years of follow-up, so there seems to be no room for second-generation treatments. While there may be cases where the drug is replaced due to CNS side effects, using Lorviqua, which maintains PFS for over 5 years, as a backup and using second-generation treatment first is something patients would not agree to.” In fact, Lorviqua was included in the 2025 National Comprehensive Cancer Network (NCCN) guidelines (recommendation level: Category 1), the 2024 American Society of Clinical Oncology (ASCO, recommendation level: Strong), and the 2023 European Society for Medical Oncology (ESMO, recommendation level: Tier I-A) guidelines as one of the first-line treatments for ALK-positive metastatic non-small cell lung cancer.
- Company
- Accomplishments of the approval-drug price pilot project
- by Eo, Yun-Ho May 23, 2025 05:52am
- Winrevair·Fintepla·Limcato expected to be commercialized With the drugs that were reviewed for approval and drug prices simultaneously about to be commercialized, the industry gathers attention. The Ministry of Health and Welfare (MOHW) has been running the 'Pilot Project for Integration of Product Approvals, Reimbursement Coverage Reviews, and Drug Price Negotiations' since 2023 to improve treatment access for life-threatening severe·rare diseases. The project conducts approval, reimbursement evaluation, and drug price negotiations simultaneously, aiming to shorten the time required for new drugs to be included in the National Health Insurance list. The first pilot project is at the final stage, showing accomplishments. Three drugs were selected in the second pilot project, which recently completed the selection of items. Typically, it takes approximately 330 days for a new drug to be introduced into the market. This pilot program aims to significantly reduce the time for ▲Product Approval (Ministry of Food and Drug Safety, MFDS) for 120 days ▲DrugReimbursement Evaluation (Health Insurance Review & Assessment Service, HIRA) for 150 days ▲ Drug Price Negotiation (National Health Insurance Service, NHIS) for 60 days. The two drugs selected for the first pilot program (2023) are either already on the reimbursement list or nearing their evaluation process's completion. Following this, ten drugs were submitted for the second pilot program, launched in 2024, and three new drugs were selected. These three are 'Winrevair (sotatercept),' a pulmonary hypertension treatment from MSD Korea; 'Fintepla (fenfluramine),' a Dravet syndrome treatment from UCB Pharma Korea; and 'Limcato,' a large B-cell lymphoma treatment from the Korea-based company Curocell. All three drugs are currently expected to be commercialized in Korea this year. Among them, Winrevair is receiving particular attention. This drug is a first-in-class innovative new drug with a novel mechanism, marking its appearance 20 years after 'Sildenafil', which targeted the NO-sGC-cGMP pathway, in 2005. As of 2023, the number of pulmonary hypertension patients in Korea is approximately 3,600. The average age of these patients is in their 40s, a demographic that plays a crucial role in society and family. Although the 5-year survival rate has significantly improved compared to the past, 3 out of 10 Korean pulmonary hypertension patients still die within 5 years. Furthermore, most patients experience significant difficulties performing daily activities such as housework, childcare, and light outings. Pulmonary hypertension is a rare, intractable, and progressive disease, where delaying the worsening of the condition directly impacts patients' quality of life and survival. To date, no cure through drug treatment has been discovered, and the mechanism of existing drugs primarily aims to alleviate symptoms by relaxing thickened pulmonary arteries. It remains to be seen how quickly drugs undergoing the concurrent approval-evaluation process, including Winrevair, will be listed. The selection criteria for the second pilot program considered drugs that met all of the following conditions: ▲ drugs for which approval and reimbursement decisions can be applied by June 2025 ▲ drugs with sufficient efficacy intended for treating life-threatening diseases with a life expectancy of less than one year, or for rare diseases ▲drugs for which no existing treatment is available or that show clinically significant improvement over current treatments ▲drugs that have been designated for or are eligible to apply for, MFDS's Global Innovative Products on Fast Track (GIFT) program. The MOHW announced December 2024 that it had selected three drugs for the second pilot program. This selection considered factors such as disease severity, availability of alternative drugs, urgency, treatment efficacy, and expert opinions from the submitted applications.
- Company
- CKD-Novartis new drug advances to the next phase trial
- by Chon, Seung-Hyun May 23, 2025 05:52am
- Chong Kun Dang (CKD)'s new drug candidate out-licensed to Novartis is entering the next clinical stage. CKD has secured its first milestone payment of KRW 7 billion, 1 year and 6 months after the technology export contract. CKD announced on May 22 that it expects to receive a milestone payment of USD 5 million (KRW 7 billion) from Novartis, upon the achievement of a stage-based milestone for CKD-510. As Novartis submitted its Investigational New Drug (IND) application for a Phase 2 clinical trial of CKD-510 to the U.S. Food and Drug Administration (FDA), the condition for the milestone payment, as per the contract, was met. CKD is set to receive an additional milestone payment for the first time since the CKD-510 technology export. CKD-510 is a new drug candidate that CKD out-licensed to Novartis in November 2023. It was a blockbuster technology export agreement with a non-refundable upfront payment of USD 80 million. The maximum milestone payment, contingent on development and approval stages, reaches up to USD 1.225 billion. CKD-510 is a new drug candidate researched and developed by CKD. It is an HDAC6 inhibitor developed using a highly selective non-hydroxamic acid (NHA) platform technology. In preclinical studies, the efficacy of the candidate was confirmed in various HDAC6-related diseases, including cardiovascular diseases. Its safety and tolerability were demonstrated in Phase 1 clinical trials conducted in Europe and the United States. CKD has completed the European Phase 1 trial of CKD-510 for Charcot-Marie-Tooth (CMT) disease. CMT is a hereditary peripheral neuropathy where motor and sensory nerves are damaged due to gene mutations, making normal walking and daily life difficult. It is a rare disease, but there is currently no definitive treatment. CKD has strategized to derive optimal drugs for various diseases based on the fundamental structure of HDAC6 inhibitors. CKD is developing new drugs applying its HDAC6 platform technology to CMT disease, Huntington's disease, Alzheimer's disease, hematologic cancers, and autoimmune diseases. Novartis enters a Phase 2 clinical trial for the first time since in-licensing CKD-510. However, Novartis has not disclosed the target indication for CKD-510.
- Policy
- Myung In’s Nuvigil first generic listed for reimb in Korea
- by Lee, Tak-Sun May 23, 2025 05:52am
- Myung In Pharm’s first generic version of the narcolepsy drug Nuvigil (amodapine) is expected to be sold in earnest following its reimbursement listing in June. Currently, narcolepsy treatments are primarily composed of two ingredients: modafinil and amodafinil. With Mitsubishi Tanabe Pharma's ' 'Wakix (pitolisant)' recently withdrawing from the Korean market, fierce competition among existing treatments is anticipated. Currently, modafinil and armodafinil are primarily used for the treatment of narcolepsy. With Mitsubishi Tanabe Pharma Korea's recent withdrawal of 'Wakix (pitolisant)' from the Korean market, the competition among existing treatments is expected to intensify further. Amidst this situation, Myung In Pharm lowered the price of its generic product at a price below the calculated price and announced plans for aggressive market entry. According to industry sources on the 22nd, two dosages (150 mg and 250 mg) of its Amonil Tab, the first generic version of Nuvigil, will be listed for reimbursement on June 1. This drug is the first generic version of armodafinil, the active enantiomer of Provigil (modafinil). It works by activating dopamine in the brain to promote wakefulness and is characterized by its improved duration of action compared to the original drug. The original amodafinil drug is Teva-Handok’s Nuvigil. Nuvigil was approved for reimbursement in Korea in June 2018 and is competing with existing modafinil preparations. Last year, the amount of outpatient prescriptions for the drug amounted to KRW 1.1 billion, according to UBIST. Narcolepsy is a neurological disorder characterized by excessive daytime sleepiness, hallucinations when falling asleep or waking up, sleep paralysis, and sleep attacks. It is designated as a rare and intractable disease in Korea due to the low number of patients. The prevalence rate in Korea is estimated to be 0.002–0.18%, and it primarily occurs during adolescence or early adulthood before the age of 30. Due to the small number of patients, there are few medications available. For modafinil, there are two products: JW Pharmaceutical's Provigil Tab 200mg and Hanmi Pharmaceutical's Modanil Tab 200mg. For amodafinil, there are only two brands: Nuvigil Tab and the newly approved Amonil Tab. Even Wakix, a new drug that was approved in 2021 and listed for reimbursement that same year, withdrew from the Korean market last year due to the small number of patients and low drug prices. From the perspective of pharmaceutical companies, the domestic market for narcolepsy treatments is not so attractive. Nevertheless, Myung In Pharm is showing enthusiasm, lowering its drug’s price during reimbursement listing. Amonil 150mg is priced at KRW 1,206 per tablet, which is about 59% of the price of the original Nubizil 150mg at KRW 2,027. The price of Amonil 250mg is also significantly cheaper at KRW 1,766, compared to Nubizil 250mg at KRW 2,968. Amonil Tab is used for excessive daytime sleepiness associated with adult narcolepsy, with a recommended dosage of 150 mg once daily in the morning. The maximum daily dose is 250 mg. Therefore, attention is now focused on whether Myung In Pharm, which has been making notable strides in the central nervous system treatment area, will also establish a strong presence in the narcolepsy treatment market with its first generic version of Nuvigil.
- Company
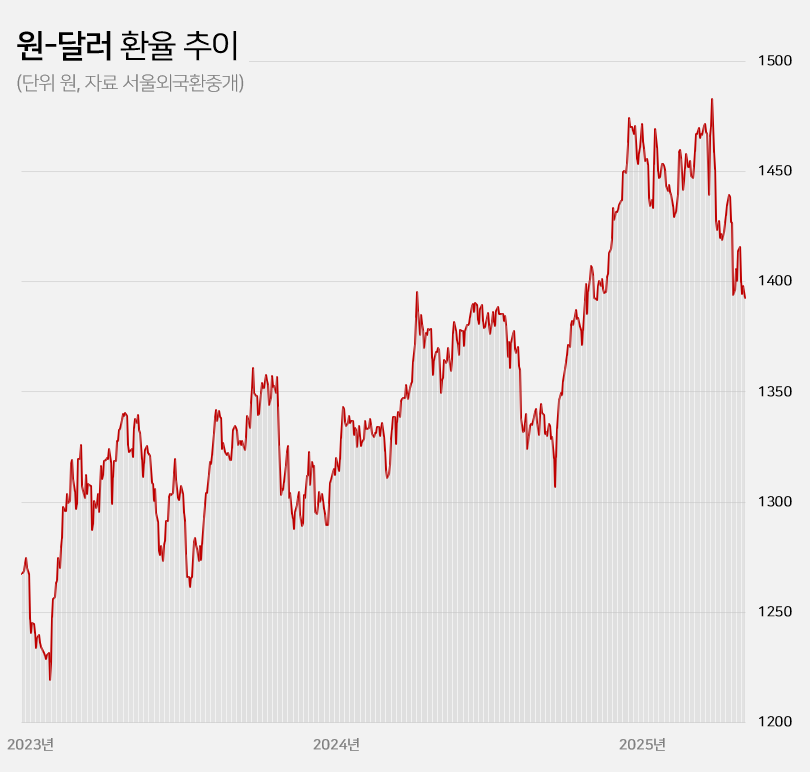
- Won-dollar rate lowest in 6mths... industry mixed
- by Kim, Jin-Gu May 23, 2025 05:51am
- With the won-dollar exchange rate falling below KRW 1,400, pharmaceutical and biotech companies are experiencing a mix of anticipation and concern. If the prolonged high exchange rate returns to previous levels, API imports and overseas clinical trial costs are expected to decrease, leading to an improvement in the cost structure. On the other hand, some predict that the asset value of pharmaceutical and biotech companies with a high proportion of drug exports will decline. Won-dollar exchange rate falls below KRW 1,400... Will the burden of API purchases and overseas clinical trials decrease? According to industry sources on the 22nd, the won-dollar exchange rate closed at KRW 1,392.60 on the 21st, down KRW 5.40 from the previous trading day. This is a 6.1% (KRW 90.30) decrease from the KRW 1,482.90 on the 10th of last month when concerns over the U.S.’s mutual tariffs were at their peak. The won-dollar exchange rate has remained high for a long time since entering the KRW 1,300 range in March 2023. It rose further to over KRW 1,350 from the middle of last year and broke through KRW 1,400 after the martial law crisis at the end of last year. With mutual tariffs by the US adding to the concern, the exchange rate soared to exceed KRW 1,480 at the beginning of last month. However, the won-dollar exchange rate has been falling this month amid expectations that trade tensions between the US and China will ease. In particular, the exchange rate has remained in the KRW 1,300 range for 4 consecutive trading days since the 16th, raising expectations that the prolonged high exchange rate trend will return to normal levels. KRW to USD exchange rate The pharmaceutical industry is hopeful that the decline in the won-dollar exchange rate will continue, leading to an improvement in the cost structure. Over the past 2 years, the rise in the won-dollar exchange rate has had a negative impact on the cost structure of domestic pharmaceutical and biotechnology companies. Due to their high dependence on imported APIs, the exchange rate increase directly led to higher manufacturing costs. Additionally, since these companies purchase APIs from China and India, with a high import dependency, they were significantly affected by the rise in the won-dollar exchange rate. As of 2023, the self-sufficiency rate for domestically produced APIs for pharmaceuticals stands at 25.4%. Among these, imports of APIs from China account for 30.5% of the total, while those from India make up 15.2%. Together, these two countries account for nearly 50% of the total API imports. Although manufacturing costs rose due to the increase in the won-dollar exchange rate, unlike other consumer goods, the price of finished drugs cannot be arbitrarily set by companies. As a result, the deterioration of the cost structure in the pharmaceutical industry has become more pronounced over the past 2 years. Additionally, the burden of clinical trial costs conducted overseas has steadily increased during the prolonged high exchange rate environment. Most clinical trials targeting the US and European markets are conducted locally. When the won-dollar exchange rate rises, global clinical trial costs also increase accordingly. In this situation, there are expectations that a decline in the won-dollar exchange rate will reduce the cost of importing APIs and the burden of global clinical trials, ultimately improving cost burdens. In the long term, improvements in the cost structure are expected to contribute positively to performance recovery. Asset value decline is inevitable for companies with high export ratios... Samsung Biologics to lose KRW 91.6 billion if exchange rate falls by 10% However, companies with high export ratios, such as Samsung Biologics and Celltrion, are expected to see their asset values decline due to the decline in the won-dollar exchange rate. These companies earn a large amount of assets in dollars overseas, so when the exchange rate rises, their asset values increase. Conversely, when the exchange rate falls, their asset values decline. Samsung Biologics had an overseas sales ratio of 96.5% in the first quarter. Of the KRW 1.2983 trillion in sales rendered in the first quarter, KRW 1.2528 trillion came from overseas. As the proportion of overseas sales is high, its performance is expected to be greatly affected by fluctuations in the exchange rate. Samsung Biologics explained in its quarterly report that a 10% increase or decrease in the won-dollar exchange rate would result in a KRW 91.6 billion increase or decrease in pre-tax income. Considering how the current won-dollar exchange rate has fallen by 1.5% compared to the average exchange rate (KRW 1,452.66) at the time of the quarterly report, the recent decline in the exchange rate is estimated to have reduced asset value by approximately KRW 14 billion. Samsung Biologics has seen an increase in cash and cash equivalents over the past 2 years due to the impact of high exchange rates. The figures were KRW 12.2 billion in 2023 and KRW 41.6 billion last year. This was due to the average won-dollar exchange rate rising from KRW 1,291.95 in 2022 to KRW 1,307.90 in 2023 and KRW 1,363.09 in 2024. The same goes for Celltrion and SK Biopharm. Given their high proportion of overseas sales, they are relatively more affected by fluctuations in the exchange rate. Celltrion saw its cash and cash equivalents increase by KRW 52.4 billion last year due to exchange rate changes. The company explains that, assuming all other variables remain constant, an 8% change in the won-dollar exchange rate would result in a KRW 33.8 billion increase or decrease in pre-tax profit. For SK Biopharm, the change in cash and cash equivalents due to exchange rate fluctuations last year amounted to KRW 3.6 billion. Based on the exchange rate at the end of last year, a 10% change in the exchange rate would result in an increase or decrease of KRW 14.1 billion in the company's pre-tax profit. It is expected that the fluctuation in pre-tax profit due to exchange rate changes will widen further this year if sales of Xcopri increase in the United States.
- Opinion
- [Reporter's View] 'Samsung Epis' as independent firm
- by Whang, byung-woo May 23, 2025 05:51am
- Samsung Biologics officially announced on May 22 that its shareholders determined to conduct a spinoff of Samsung Bioepis and establish 'Samsung Epis Holdings.' The business independence of Samsung Bioepis, which expanded its focus on biosimilars, was conducted earlier than anticipated. It has been two years since Samsung Biologics acquired Biogen's entire share in Epis. The current spinoff is meaningful in a way more than simply reorganization of the firm. This indicates Samsung Bioepis' performance over 10 years, future market changes, and efforts to achieve the technological leap. Samsung Bioepis was established as a joint venture between Samsung Biologics and the biotech firm Biogen in 2012. In 2022, Samsung Biologics acquired 49.9% of the U.S.-based Biogen's share in Samsung Bioepis for approximately USD 2.3 billion, acquiring Epis as a 100% subsidiary. After that, Samsung Bioepis established a partnership-based collaborative structure that is flexible and strategic rather than simply a shareholder relationship. Its performance in the past two years can be seen as a period of testing the firm's 'independent survival.' Samsung Bioepis will become a completely independent entity after the spinoff. At the same time, Samsung Epis Holdings will strengthen its role in Samsung Group's biotech business. Samsung Bioepis' performance in the global market is notable. According to IQVIA, Samsung Bioepis ranked No.2 in the market share of Humira biosimilars as of 2023. However, the company's competitors are fiercely rising. For instance, Celltrion has established a competitive ground as it obtains approval and distributes new products in addition to existing biosimilars. Samsung Bioepis' new project is 'new drug discovery,' preparing beyond biosimilars. Whereas the past performance focused on proving equivalence against original products and expanding market presence, the company will likely focus on securing a proprietary new drug pipeline for the next 10 years. Samsung Bioepis has already started investing in R&D of proprietary new drug discovery. It has begun searching for next-generation technologies, such as establishing an AI-based new drug discovery platform and antibody-drug conjugate (ADC). However, the company's new drug pipeline for clinical trials has not been officially announced. The company will likely seek an R&D model that could balance profitability and risk rather than delving into new drug discovery, requiring time and funds but carrying low success. This approach has both pros and cons. This means there is an opportunity to choose and focus on an area with a high probability of success rather than taking risks and being reckless. Celltrion employs a similar strategy. Samsung Epis Holdings' establishment can be seen as Samsung Group entering a new stage of 'comprehensive biotech strategy' beyond biosimilars. This is likely to be followed by efforts to expand into fundraising, co-development with global big pharma, and the rare disease and vaccine sectors. There's still a long way to go. Their experience in new drug discovery remains in the early stages, and there are many challenges to overcome, such as securing high-value pipelines, navigating the regulatory environment, and securing talent. The company's journey was like 'starting from scratch' and turning question marks from external and internal sources into exclamation points. Conversely, it was also an expression of pride in leading the company to its current level with a relatively minor organization compared to large entities like Samsung Electronics and Samsung C&T. The launch of Samsung Bioepis Holdings signifies confidence in past achievements and represents a bold first step toward new challenges and responses. We anticipate Samsung Bioepis' continued strong presence in future competition, leveraging their 'original vision' and proven competitiveness in the biosimilar industry.
- Company
- Will Tibsovo be reimbursed for cholangiocarcinoma this time?
- by Eo, Yun-Ho May 22, 2025 06:10am
- Attention is focused on whether the targeted anticancer drug “Tibsovo” for cholangiocarcinoma and acute myeloid leukemia will be successful in its attempt to be reimbursed by health insurance in Korea. Servier Korea’s IDH1 (isocitrate dehydrogenase 1) genetic mutation targeting therapy recently passed the Health Insurance Review and Assessment Service's Cancer Disease Review Committee. The drug is indicated for use in IDH1-mutation-positive patients in combination with azacitidine for newly-diagnosed acute myeloid leukemia (AML) with a susceptible isocitrate dehydrogenase-1 (IDH1) mutation in adults aged ≥75 years, or who have comorbidities that preclude use of intensive induction chemotherapy, or as monotherapy for locally advanced or metastatic cholangiocarcinoma in previously treated adults with IDH1 mutation. The dual AML indication passed the Health Insurance Review and Assessment Service's Cancer Disease Review Committee’s review in October last year, but the cholangiocarcinoma indication had not been approved until now. It remains to be seen whether Tibsovo will be submitted to the Drug Reimbursement Evaluation Committee and complete the reimbursement process in the field of cholangiocarcinoma, where treatment options are limited. Cholangiocarcinoma is a highly aggressive cancer with a poor prognosis, with a 5-year relative survival rate of just 28.9%. In particular, 65% of patients with intrahepatic cholangiocarcinoma are diagnosed at an advanced stage where surgery is not feasible. Tibsovo is the only targeted therapy recommended by the National Comprehensive Cancer Network (NCCN) in the highest category (Category 1) as a second-line treatment for bile duct cancer. According to the Phase III ClarlDHy trial, Tibsovo reduced the risk of disease progression by 63% compared to placebo, with a median progression-free survival (PFS) of 2.7 months (1.4 months in the placebo group). Additionally, the median overall survival (mOS) was 10.3 months in the Tibsovo group, more than twice as long as the 5.1 months in the placebo group. Do-Youn Oh, Professor of Hematology-Oncology at Seoul National University Hospital, stated, “The development of drugs for cholangiocarcinoma has accelerated significantly in recent years. As new drugs are being developed, many companies are also actively working on the development of treatments for cholangiocarcinoma. Patients with cholangiocarcinoma must follow the doctors’ guidance and receive appropriate treatment rather than feeling discouraged, so they can participate in clinical trials and access new treatment opportunities.” Meanwhile, in the Phase III AGILE study that enrolled patients with acute myeloid leukemia, Tibsovo demonstrated improved event-free survival (EFS) when used in combination with azacitidine, and also significantly improved overall survival (OS). The median OS in the Tibsovo-treated group was 24.0 months (7.9 months in the placebo group), and long-term follow-up results showed that the median OS with Tibsovo combination therapy was 29.3 months, over 3.7 times longer than with placebo combination therapy. Kyu-Pyo Kim, Professor of Oncology at Asan Medical Center, said, “Tibsovo is indicated for IDH1 mutation-positive cholangiocarcinoma and acute myeloid leukemia, and has demonstrated excellent efficacy and safety in clinical studies. Considering that both studies allowed crossovers, the significant improvement in survival is a very meaningful result.” He added, “Given the limited treatment options for cholangiocarcinoma and acute myeloid leukemia, the emergence of a new targeted therapy with promising efficacy is expected to contribute to improving treatment outcomes and quality of life for patients with these diseases in Korea.”
- Company
- SK Bioscience wins the '8-year Prevenar patent dispute'
- by Kim, Jin-Gu May 22, 2025 06:09am
- In a patent infringement trial against Pfizer involving SK Bioscience's export of 'SKYPneumo' raw solution, SK Bioscience has won after eight years. The latest ruling is expected to enable SK Bioscience's global export of its pneumococcal conjugate vaccine 'SKYPneumo' raw solution. Ruling that 'Exporting SK Bioscience's SKYPneumo raw solution does not infringe on the patent'…an opportunity has been presented to export According to SK Bioscience, on May 21, the Supreme Court of Korea ruled in favor of SK Bioscience in a legal dispute filed by Pfizer for 'Cessation of infringement of Pfizer's Prevenar 13 (PCV13) pneumococcal conjugate vaccine patent.' The Supreme Court recently upheld the second trial ruling, which ruled in favor of SK Bioscience. SK Bioscience and Pfizer long fought over whether the overseas supply of SKYPneumo's raw solution infringes on Pfizer's 'Prevenar 13.' This eight-year legal battle began in 2017 when Pfizer filed a patent infringement lawsuit. The Supreme Court ruled that "Producing and supplying PCV13 finished products for research purposes does not infringe on the patent," adding, "Individual conjugates that make up PCV13 do not fall within the scope of the patent claim." It is analyzed that the Supreme Court's final ruling favoring SK Bioscience is expected to open the door for SK Bioscience to export SKYPneumo raw solution overseas. SK Bioscience says, "While supplying PCV13 finished products is not permitted, the latest ruling presents an opportunity for supplying individual conjugates overseas," and added, "We plan to initiate a new project to supply raw solution of individual conguates to regions with high vaccine demand, such as Southeast Asia and Central and South America. We also intend to pursue technology transfer through local partnerships." SK Bioscience vs. Pfizer: Patent Invalidation·Infringement Lawsuits…3 out of 4 cases transferred to the Supreme Court In addition to this case, the dispute between SK Bioscience and Pfizer surrounding the Prevenar 13 patent has been ongoing for a long time, including 'patent invalidation' lawsuits filed by SK Bioscience and 'cessation of patent infringement' lawsuits counter-filed by Pfizer. First, SK Bioscience challenged two related patents after developing SKYPneumo, a follow-up product to Prevenar 13. SK Bioscience challenged two of Pfizer's Prevenar 13-related patents: ▲Expiring in March 2026, 'a novel formulation that stabilizes immunogenic compositions and prevents precipitation' patent ▲Expiring in April 2027, 'a multivalent pneumococcal polysaccharide-protein conjugate composition' patent. SK Bioscience filed invalidation trials for both patents. Among these, the patent expiring in 2026 was successfully invalidated. The Intellectual Property Trial and Appeal Board (IPTAB) ruled in favor of SK Bioscience in January 2020, and the decision was finalized without an appeal from Pfizer. On the other hand, the patent expiring in 2027 ultimately failed to be invalidated. SK Bioscience argued for the invalidation of this patent, but received a dismissal from IPTAB in June 2015. SK Bioscience also lost in the second trial, which proceeded with SK Bioscience's appeal, in November 2017. SK Bioscience decided to appeal to the Supreme Court. However, they again received a losing ruling from the Supreme Court in 2018. In conclusion, the patent expiring in March 2026 was successfully invalidated, while the patent expiring in April 2027 was not. Consequently, domestic sales of SKYPneumo are prohibited until April 2027. Pfizer counter-filed a patent infringement prohibition lawsuit against SK Bioscience during this process. In 2017, Wyeth LLC, the patent holder of Prevenar 13, and Pfizer Korea, the domestic distributor, filed a lawsuit claiming that SKYPneumo infringed on their patent rights. This lawsuit also went to the third trial. Ultimately, the Supreme Court issued a recommendation for settlement in 2019, essentially siding with Pfizer. The Supreme Court ruled that Pfizer's patent was valid and prohibited the production and launch of SK Bioscience's SKYPneumo until 2027. SK Bioscience voluntarily withdrew the product approval for SKYPneumo. However, they re-obtained approval for SKYPneumo in June 2021. The conflict between the two companies continued thereafter. Facing difficulties with the domestic launch, SK Bioscience signed a license agreement to transfer related technology to a Russian pharmaceutical company to develop vaccines in that region. SK Bioscience supplied raw solution for its self-developed pneumococcal vaccine and finished pharmaceutical products for research. Pfizer and Wyeth LLC also interfered with this, filing a cession of patent infringement lawsuit in 2020. SK Bioscience countered, arguing that supplying raw solution for research and testing purposes, not finished products, to overseas markets was outside the scope of patent infringement. In August 2023, Pfizer won in the first trial. SK Bioscience appealed, and ultimately, the Patent Court overturned the first trial ruling and sided with SK Bioscience. This time, Pfizer appealed to the Supreme Court after losing in the second trial. Finally, the Supreme Court upheld the original ruling in the appeal. The two companies engaged in a long-term battle, going to the Supreme Court for three out of four cases, including patent invalidation lawsuits and patent infringement lawsuits. With this, most disputes between the two companies concerning the Prevenar 13 patent have been concluded. However, a trade commission dispute remains. The Korea Trade Commission under the Ministry of Trade, Industry and Energy is investigating SK Bioscience's unfair trade practices. This is because Pfizer and Wyeth LLC have filed a complaint requesting a cessation on exporting SK Bioscience's pneumococcal raw solution. In the pharmaceutical industry, there is an outlook that the trade commission dispute surrounding the export of pneumococcal raw solution will also be concluded soon due to this Supreme Court ruling.
- Policy
- Govt-ind sees consensus on 'AI-based new drug governance'
- by Lee, Jeong-Hwan May 22, 2025 06:09am
- The current administration has promised support and promotion for the establishment of a national governance system for AI-based new drugs, which the domestic pharmaceutical industry has called for the attention of the presidential election candidates. The National Bio Committee, which reports directly to the president, has selected AI-based new drug development as one of the 10 key tasks that the government will focus on for research and development (R&D) in the future. The vision is to discover optimal new drug candidates based on AI and big data, and to fulfill national responsibilities by supporting the entire cycle, including design and validation. On the 20th, the pharmaceutical and biotechnology industry expressed their broad consensus on the needs of the pharmaceutical industry in response to the announcement of the vision of the National Bio Committee. The domestic pharmaceutical industry has repeatedly suggested that the infrastructure for new drug development utilizing AI and big data should be established at the national level. In fact, the Korea Pharmaceutical and Bio-Pharma Manufacturers Association has proposed a policy to establish AI-based drug development governance for each candidate ahead of the June 3 presidential election. The demand is that the government should aggressively provide budget and policy support to utilize AI and big data to revitalize new drug development, which requires astronomical investment and time, and has a significantly low success rate. In particular, global big-tech companies such as Google AlphaFold 3-AlphaProteo, Nvidia BioNeMo, and Microsoft are developing AI super gap technology to be utilized in the whole stage of drug development based on large-scale investments and resources from global pharmaceutical companies. The KPBMA believes that Korea must not fall behind in the AI drug development competition with advanced countries to become a new drug powerhouse. Therefore, it is suggested that the government's full support is needed to create a national data-based open innovation ecosystem, build a big data platform for new drug development, and develop an AI-based intelligent autonomous laboratory optimization model. The next administration should collect national bio R&D public data for each field of drug development and create a new drug development big data platform for pharmaceutical companies’ access. Also, KPBMA’s policy proposal includes securing skilled professionals through an academia-industry collaborative training program for AI-driven drug discovery, as well as carrying out a joint platform project for shared use of AI-bio computing resources. As the National Bio Committee directly agreed with the pharmaceutical industry's policy proposal, there are expectations that the green light has been given for national-level budget and policy support for AI-based drug development even after the presidential election. An industry official said, “All previous administrations have set the goal of fostering the pharmaceutical industry as a future growth driver for the country, including developing new domestic drugs and discovering indigenous blockbusters. The government that will be established after the presidential election should realize that establishing governance for AI drug development is a prerequisite for creating domestic blockbusters, and should provide policy support and budgetary investment at the entire government level.” “The National Bio Committee's selection of developing infrastructure for AI-based drug development as one of the top 10 key R&D tasks has become a cog in the domestic pharmaceutical industry's policy proposals. We expect the new government to recognize that it is difficult for the private sector to create AI and big data drug development platforms, which are costly, and to take active policy and budgetary measures.”