- LOGIN
- MemberShip
- 2026-04-04 07:31:07
- Product
- MOHW cautions non-face-to-face Wegovy prescriptions
- by Kang Hye-Kyung Nov 13, 2024 05:55am
- The government has asked medical institutions for cooperation in making indiscriminate Wegovy prescriptions. The Ministry of Health and Welfare said on the 11th through the medical community, “Regarding Wegovy, which was recently released as a treatment for obesity, medical institutions are issuing prescriptions without sufficiently examining the patient's condition, raising concerns of its abuse and misuse.” The ministry emphasized, ”Wegovy is administered as an aid for weight management for obese patients, and side effects may occur if used regardless of the patient's medical condition.” The MOHW also asked institutions to comply with the MFDS’s approved indication, which specifies Wegovy’s use as an adjunct for managing weight in people with ▲a BMI of 30 kg/m² or greater (obesity) or ▲a BMI of at least 27 kg/m² but less than 30 kg/m² who have weight-related health problems (such as diabetes, high blood pressure, abnormal levels of fats in the blood, breathing problems during sleep called ‘obstructive sleep apnoea’ or a history of heart attack, stroke or blood vessel problems), or to reduce the risk of major adverse cardiovascular events (cardiovascular death, nonfatal myocardial infarction or stroke) in people with confirmed cardiovascular disease who are ▲either obese or overweight with a BMI of at least 27 kg/m². They also ordered that patients should be properly screened to determine if they are eligible and that they should be informed of possible side effects, including gallbladder disease, intestinal obstruction, aspiration pneumonia, pancreatitis, acute heart injury, hypoglycemia, various gastrointestinal, metabolic, neurological disorders, and suicidal thoughts. In addition, the ministry asked doctors to be careful not to mislead patients into believing that Wegoby can easily be prescribed for weight loss, and to be vigilant in preventing the misuse of such drugs during virtual visits.
- Opinion
- [Reporter's View] voluntary resignation with compensation
- by Lee, Seok-Jun Nov 13, 2024 05:54am
- As the year-end is nearing, several pharmaceutical companies are considering restructuring. Rumor has it that these companies may have plans for reducing OTC businesses and laying off staff who are age 55 or older. It remains uncertain whether it is a rumor or an actual plan, but restructuring could be possible. Over five pharmaceutical companies that were mentioned. There is a solid reason companies have chosen restructuring. It is for business efficiency. Restructuring could be an enormous benefit if laying off results in efficiency within the company. It is an approach of 'selection and focus,' losing a few if taking all is not an option. Voluntary resignation is frequently used as a method of job cuts. It is implemented to reduce the number of employees at once. It is the same as layoffs in business. However, companies utilize the voluntary resignation program instead of layoffs because layoff requirements are selective according to the Labor Standards Act and may result in conflicts and management. Korean pharmaceutical companies employ a similar method. Voluntary resignation is less frequent than that of the multinational companies, but the Korean companies have used it more often recently. At the end of last year, Korean mega-pharmaceutical companies, Ildong Pharmaceutical and GC Biopharma, conducted voluntary resignations. Likewise, Korean pharmaceutical companies are favor voluntary resignation when reducing the number of employees. Then, what is the next important step? We must consider compensation for those who are subjected to restructuring. Pharmaceutical companies' goal for restructuring is to reduce the number of employees. However, it would be better to achieve such a goal by offering a good compensation package. If restructuring is inevitable, a win-win structure must be set. Although companies may have to spend a large sum of money at once, they may have to sacrifice considering the cost-effective outcomes in a few years following restructuring. Yet, a compensation criterion has not been established for voluntary resignation. Compensation criterion vastly differs by company. Direct comparison may be difficult, but the criterion in Korean companies is different from the (consecutive years of service*2)+ N months in multinational companies. Few companies urge voluntary resignation without compensation. Companies will continue to undergo restructuring. Then, it will be necessary for Korean companies to establish a compensation criterion for voluntary resignations. Precedence will serve as a standard for those who follow. If leading restructuring companies set low compensation, latecomers will also set low standards. The situation in small and medium-sized companies will be much worse, causeing a vicious cycle. Now, an agreement between Korean companies for voluntary resignation compensation may be necessary. Better compensation will lessen the backlash from restructuring.
- Company
- Imfinzi combo drug Imjudo can be prescribed at hospitals
- by Eo, Yun-Ho Nov 13, 2024 05:54am
- Immuno-oncology drug Imfinzi's combination partner Imjudo may now be prescribed in general hospitals in Korea. According to industry sources, AstraZeneca Korea's CTLA-4 inhibitor Imjudo (tremelimumab) has passed the drug committees (DCs) of tertiary hospitals in Korea including Seoul National University Hospital and Seoul Asan Medical Center. For now, however, Imjudo is a non-reimbursed drug. AstraZeneca submitted an application for the reimbursement of the PD-L1 inhibitor Imfinzi (durvalumab) and Imjudo combination for liver cancer in June and is currently awaiting a review by the Health and Insurance Review and Assessment Service’s Cancer Disease Review Committee. Imjudo was approved by the Ministry of Food and Drug Safety in combination with Imfinzi in June last year. The first target indication for the combination is liver cancer and can be prescribed as a first-line treatment for adult patients with advanced or unresectable hepatocellular carcinoma (HCC). The specific regimen is the STRIDE (Single Tremelimumab Regular Interval Durvalumab) regimen, which consists of a single dose of Impinj 1,500 mg plus 300 mg of Imfinzi, followed by an additional dose of Impinj at regular intervals every 4 weeks. At the recent European Society for Medical Oncology (ESMO) Congress 2024, the 5-year overall survival data from the Phase III HIMALAYA trial that demonstrated the efficacy of the Imfinzi and Imjudo combination in hepatocellular carcinoma was presented. In the HIMALAYA trial, patients with inoperable HCC were treated with STRIDE (single dose of Imjudo followed by Imfinzi maintenance therapy), Imfinzi monotherapy, and sorafenib monotherapy. When comparing the results of the Imfinzi and Imjudo combination with sorafenib combination therapy in patients with unresectable HCC, patients who received the STRIDE regimen had a 5-year overall survival (OS) rate of 19.6%, compared with the 9.4% for patients who received sorafenib. The median overall survival was 16.43 months and 13.77 months, respectively, showing a 24% lower risk of death in the Imfinzi-Imjudo combination arm. “ The Imfinzi-Imjudo combination therapy has significant advantages in that it has a much lower risk of bleeding than conventional therapies and does not worsen liver function," said Hong Jae Chon, Professor of Hemato-Oncology at CHA Bundang Medical Center. “In particular, the combination shows potential for longer survival than existing therapies."
- Company
- Treatment-refractory Dravet syndrome calls for new options
- by Whang, byung-woo Nov 13, 2024 05:54am
- Despite increased treatment options for the ultra-rare Dravet syndrome, there are still gaps in care that require attention. Even with the introduction of medical cannabis, cannabidiol, there are patients who do not respond to the drug, which is why improving access to new options should be considered. Dravet syndrome is a rare neurological disorder that begins with fever and convulsions within the first year of life, persists into adulthood, and leaves nearly all young patients moderately to severely disabled after each attack. Although it is known to be a rare disease with an estimated prevalence of 1 to 2 per 10,000 people worldwide, there is no officially investigated prevalence in Korea and was designated as an ultra-rare disease in 2022. Dravet syndrome is characterized by the onset of the first seizure, which is similar to a febrile convulsion that usually occurs with fever at 6 months. The biggest risk factor is “Sudden Unexpected Death in Epilepsy” (SUDEP). While the rate of sudden death in intractable epilepsy is 20-25%, in Dravet syndrome, up to 59% of all deaths are associated with SUDEP. The goal of treatment for Dravet syndrome is to control seizure frequency and non-seizure symptoms to reduce the patient's risk of sudden death and improve quality of life. Initial treatment includes anti-seizure medications and add-on treatments such as the anti-seizure medications stiripentol and cannabidiol are used to treat the “drug refractory” nature of Dravet syndrome. Cannabidiol is a medical cannabis preparation that was previously supplied without reimbursement through the Korea Orphan & Essential Drug Center for urgent use but then has been reimbursed since April 2021. Hoon-Chul Kang, professor of pediatric neurology at Severance Hospital, emphasized that the government's approval of medical cannabis has contributed to improving the treatment environment for Dravet syndrome. Kang said, “The government's decision was based on the desperate voices of parents and caregivers of children with Dravet syndrome, as well as objective data reported in the literature,” he explains. Limitations remain for drug-refractory Dravet syndrome...a fundamental solution is needed However, stiripentol and cannabidiol are only available through the Korea Orphan & Essential Drug Center, and the treatment process from applying for the drugs to meeting the criteria for reimbursement coverage is rather complicated. Hoon-Chul Kang, professor of pediatric neurology at Severance Hospital Hoon-Chul Kang, professor of pediatric neurology at Severance HospitalIn particular, there are still many patients with Dravet syndrome who are refractory to existing medications, leaving a blind spot in terms of seizure management. Unlike Korea, where treatment options are limited, options are increasing overseas with the emergence of new options. In the long run, experts agree that Korea also needs a fundamental treatment for seizures that reduces the quality of life for people with Dravet syndrome and their caregivers. If a new treatment option can significantly improve seizure control while also managing additional comorbidities and disabilities, it would substantially improve the treatment landscape. In addition, despite the limitations of being an ultra-rare disease, there are expectations that Dravet syndrome will benefit from the government's 'fast-track program for serious and rare diseases to reduce the burden of medical expenses’ plan. The industry predicts that the government's interest in pediatric rare and intractable diseases will continue for the time being, given the revised pediatric drug pharmacoeconomic evaluation exemption system last year and the selection of drugs for the first pilot project for the approval-evaluation-negotiation linkage system. A professor of pediatrics at a tertiary hospital said, "The government needs to make another timely decision to improve the treatment environment for Dravet syndrome, an extremely rare disease that is even more neglected than others."
- Company
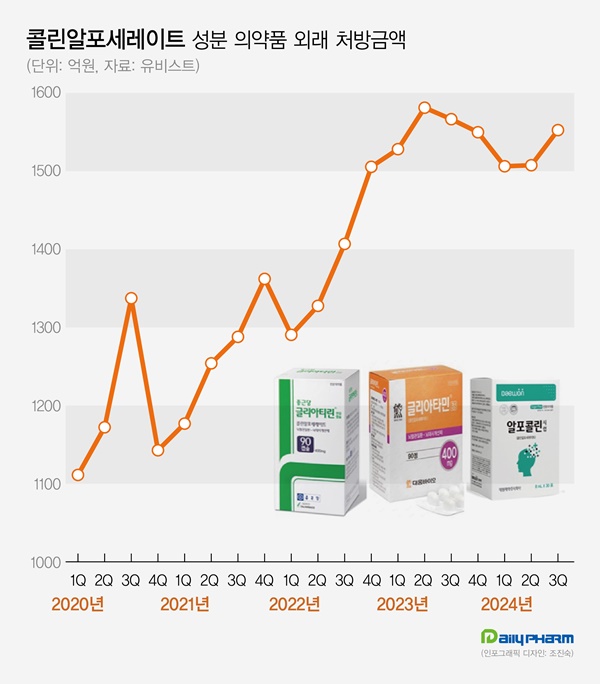
- 'Choline alfoscerate' prescription market continues to grow
- by Chon, Seung-Hyun Nov 13, 2024 05:54am
- The cognitive enhancer 'choline alfoscerate (choline products)' has expanded its presence in the prescription market. Choline products' growth slowed earlier this year but rebounded in Q3, further expanding the market size. Although a few companies withdrew from the market due to the risk of failing clinical re-evaluation, the prescription market continued sales boom. According to the pharmaceutical market research firm UBIST on November 11, the outpatient prescription market for choline products totaled KRW 155.3 billion in Q3. It decreased by 0.9% compared to Q3 of last year but increased by 3.0% compared to the previous quarter. Choline products' prescription sales recorded KRW 158.1 billion in Q2 of 2023. Then they went on a downward slide for three consecutive quarters until Q1 this year. In Q2, the sales slightly increased compared to the previous quarter. In Q3, they showed a strong rebound. Choline products' prescription size for Q3 was recorded as the third highest in history. The slowing growth of choline products in the first half of this year is likely due to steep growth in the past few years. The prescription market for choline products recorded KRW 308.8 billion in 2018. Then, it continued to renew the best record every year. Last year, the market amounted to KRW 622.6 billion, expanding more than twice in five years. Quarterly prescription sales indicate a 46.4% increase over five years from KRW 106.1 billion in Q3 of 2019. Outpatient prescription sales of pharmaceuticals containing choline alfoscerate (unit: KRW 100 million, source: UBIST). Even though choline products are under clinical re-evaluation for efficacy evaluation, clinical practices have continued to have a high demand for choline products. In June 2020, the Ministry of Food and Drug Safety (MFDS) requested companies with choline products to submit their clinical trial documents, and 57 pharmaceutical companies began clinical trials for reassessment. Previously, three indications for choline products had been approved, including ▲secondary symptoms caused by cerebrovascular deficit or degenerative organic brain syndrome ▲emotional and behavioral changes ▲senile pseudodementia. In the re-evaluation process, two out of three indications for choline products were deleted, excluding 'secondary symptoms caused by cerebrovascular deficit or degenerative organic brain syndrome.' Choline products are facing the possibility of reduced reimbursement in addition to the issue of their efficacy. In August 2022, the MOHW issued revised regulations on 'The Criteria and Scope of National Health Insurance,' indicating that patients without prior dementia diagnosis will have a copayment increased from 30% to 80%. After that, two groups of pharmaceutical companies, led by Daewoong Bio and Chong Kun Dang, filed an administrative suit to cancel the MOHW's notification. However, they all lost in the first trial in 2022. Chong Kun Dang also lost in the second trial in May. However, as the suspension of execution filed by pharmaceutical companies has been accepted, reimbursement reductions are on hold. Despite many products being withdrawn from the market after the clinical re-evaluation of choline products, the prescription market continued to grow. According to the MFDS, choline products that received Korean approval total 278 items. Among these, 134 items have been withdrawn from the market due to approval withdrawal or cancelation. Previously, the MFDS ordered the clinical re-evaluation of choline products from 134 companies. 77 companies withdrew from undergoing re-evaluation, resulting in a significant number of withdrawals from the market. More companies that commenced clinical re-evaluation of choline products are withdrawing. In two months from September, Guju Pharmaceutical, Kyongbo Pharmaceutical, PharmGen Science, YooYoung Pharmaceutical, and Medix Pharm voluntarily withdrew choline product approval. Increasing number of companies are exiting the market due to potential retrieval amounts that arise when they fail the re-evaluation for choline products. In 2020, the MOHW issued a national health insurance contract to companies with choline products, entailing 'companies failing clinical trials must return the prescription sales.' Within eight months of the negotiation order, pharmaceutical companies agreed to the term that they would return 20% of the prescription sales from the time they received IND approval to the date of deletion when the indication for the product was deleted due to failed clinical re-evaluation. The retrieval negotiations for choline products are determined by agreements between the MOHW and each pharmaceutical company, resulting in different contract details for each company. While a 20% retrieval rate from prescription sales is commonly applied, the timing of the retrieval rate varies among companies. Sources said that most companies have agreed to increase retrieval rate. For instance, companies may have agreed to set a 10% retrieval rate for this year when they fail the clinical re-evaluation of choline products, then increase to 30% after five years. As the prescription market for choline products continues to grow, companies that agreed to a gradual increase of retrieval rate would end up exponentially expanding retrieval amount due to the market growth. Pharmaceutical companies may have to face increased retrieval amounts as the market for choline products continues to grow if they fail clinical re-evaluation. For these reasons, sources said that more companies are considering exiting the market before completing the clinical re-evaluation. However, analysis suggests that the entire market for choline products continues to grow as other products replace the withdrawn products. Prescription sales by major products indicate that Daewoong Bio's Gliatamin recorded KRW 41.2 billion in Q3, a 4.4% reduction from the previous year. Chong Kun Dang's Chongkundang Gliatirin generated prescription sales of KRW 31.1 billion in Q3, up 10.9% from last year. Arlico's Choliatin recorded prescription sales of KRW 5.1 billion in Q3, a 28.7% reduction from the previous year. Daewon Pharmaceutical's Alfocholine generated prescription sales of KRW 4.9 billion in Q3, down 10.3%. Yuhan's Alfoatilin recorded KRW 3.7 billion, down 16.7% from the previous year. Dongkoo Bio's Glifos' sales increased from KRW 2.7 billion in Q3 of last year to KRW 3.7 billion in a year, up 33.9%. Mother's Pharm's Memoem recorded prescription sales at KRW 1.1 billion in Q3 of last year, then increased to KRW 3.3 billion in a year, an increase of over threefold.
- Company
- Asthma drug 'Monterizine' sales rise despite generic entries
- by Kim, Jin-Gu Nov 12, 2024 05:51am
- Hanmi's asthma treatment, 'Monterizine,' successfully expanded its prescription sales over 10% Year-over-Year (YoY), despite the release of generics. Generic drugs had been listed for reimbursement in October 2023. The analysis suggests that Monterizine's continued sales expansion is because generic prices have not been reduced and, it maintains a strong market presence with its broader scope of use. Prescription sales 14%↑despite generic releases… Monterizine Chewable Tab is the only available treatment for young children According to the pharmaceutical market research firm UBIST on November 8, Hanmi's Monterizine recorded prescription sales of KRW 4.3 billion in Q3, which is an increase of 14% over a year compared to KRW 3.7 billion in Q3 2023. Monterizine is a combination drug containing the asthma treatment, 'Montelukast,' and the third-generation antihistamine, 'Levocetrizine.' Hanmi received approval for 'Monterizine Cap' in May 2017. The following April, Hanmi changed the formulation and received approval for 'Monterizine Chewable Tab' designed to be taken by chewing. After the launch of Monterizine Chewable Tab, prescription sales quickly expanded for the Monterizine series. The combined prescription sales of Monterizine Cap and Monterizine Chewable Tab have increased by 8%, from KRW 7.9 billion in 2019 to KRW 8.6 billion in 2020. It recorded KRW 9.7 billion in 2021, surpassing KRW 10 billion when the sales amounted to KRW 12.4 billion in 2022. Last year, it recorded KRW 15.6 billion, a 21% increase from the previous year. This year, Monterizine is maintaining solid sales. In Q4 2023, it surpassed KRW 4 billion for the first time, generating KRW 4.3 billion to KRW 4.4 billion in sales each quarter. Quaterly prescription sales of the original drug Monterizine (blue) and generics (ligt blue) (unit: KRW 100 million, source: UBSIT). Interestingly, generics of Monterizine were launched in October 2023. Typically, sales of original drug slow down after the generic launches, but Monterizine is following a different trend. The analysis suggests that Monterizine Chewable Tab is showing a strong market presence with its broader scope of use. The scope of use for the original Monterizine Cap and generics to Monterizine are limited to 'adults and adolescents at the age of 15 or older.' Monterizine Chewable Tab is prescribed to 'young children at the age of 6-14.' The analysis is that Monterizine Chewable Tab is widely used in the prescription market, especially because young children require frequent prescriptions in the case of asthma drugs. Drug price has been maintained despite generic releases…the generics formulation is different from the original Additionally, one of the reasons for continued sales of Monterizine is that it remains the same price following the generics' releases. Product photo of HanmiIn September 2021, generics companies challenged four patent cases of Monterizine. In September of the following year, they successfully avoided patents one after another. After losing the first trial, Hanmi appealed to the patent court, but the company soon withdrew. Since October 2023, Monterizine generics have launched. Six products that met the requirement for the highest price by conducting bioequivalence tests became listed at KRW 886 per tablet. 14 products that did not meet the bioequivalence test because they were produced by contract manufacturing organization (CMO) became listed at KRW 753 per tablet. The drug price of Monterizine was not reduced following the generic release. The original drug price is automatically reduced by 30% when generics are released. Monterizine drug price should have been reduced from KRW 886 to KRW 620, but it was not. This is because the generics have been developed in a formulation different from the original. The Health Insurance Review and Assessment Service (HIRA) reduces the original drug price by 30% in the first year when a product with the same formulation is launched. In the following year, the HIRA further reduces the drug price of the original by 53.55%. In this case, the 'same formulation' means the active ingredient, administration route, dosage, administration method, formulation, and efficacy·effects are matched. The original Monterizine is available in capsules and chewable tablets. Generic products received approval as tablets. The government analyzed that the generics are not in the same formulation as the original Monterizine. Therefore, the drug price of the original was maintained at KRW 886.
- Company
- 'Vabysmo' for macular deg associated RVO indication expected
- by Eo, Yun-Ho Nov 12, 2024 05:51am
- Product photo of Vabysmo.The first bispecific antibody for eye diseases, 'Vabysmo,' is soon to be approved in South Korea for its indication of treating macular degeneration associated retinal vein occlusion (RVO). According to industry sources, Roche Korea's Vabysmo (faricimab) is being reviewed by the Ministry of Food and Drug Safety (MFDS) for its indication expansion. It is expected to be approved within the year. For RVO indication, it received the U.S. FDA approval in October 2023. Attention has been drawn to Vabysmo, a treatment for macular degeneration because it significantly extended the administration interval compared to Bayer's 'Eylea (aflibercept), which has been the standard therapy. In South Korea, Vabysmo's prescription became available after it was approved for reimbursement listing for neovascular age-related macular degeneration (nAMD) and diabetes-related macular edema (DME) in October last year. Existing macular degeneration drugs used in South Korea are vascular endothelial growth factor-A (VEGF-A) drugs such as Novartis' 'Lucentis (ranibizumab),' 'Beovu (brolucizumab),' and Eylea. Unlike existing VEGF drugs, like Lucentis and Eylea, Vabysmo can also block the angiopoietin-2 (Ang-2) pathway, thus inhibiting new blood vessel formation. The analysis suggests that blocking two independent pathways can more effectively stabilize blood vessels and reduce inflammation, abnormal vessel growth, and fluid leakage than the VEGF-A pathway alone. RVO is Vabysmo's third indication. Its efficacy has been confirmed through the Phase 3 BALATON and COMOINO studies. In these two clinical trials, Vabysmo achieved non-inferiority in the patient's vision improvement compared to Eylea. When treated with Eylea, the patients had continual vision improvements from the early stage. The safety profile of the trials was similar to previous study reports. Meanwhile, Vabysmo has seen an increase in sales this year. Its sales for Q1 amounted to 847 million francs, up 88.6% from the previous year. It recorded sales of 947 million francs (about 1.51 trillion won) in Q2, up 86.4% from last year. In Q2, Vabysmo surpassed the sales of the market leader Eylea (about 1.26 trillion won) for the first time.
- CG Invites' personalized cancer vaccine shows promise
- by 강은희 Nov 12, 2024 05:51am
- CG Invites (Co-CEOs Chung In-chul and Oh Su-yeon), a frontrunner in digital genomics-driven drug development, announced groundbreaking preclinical results for their personalized cancer immunotherapy vaccine on the 11th. Cancer vaccines epitomize personalized medicine, harnessing tumor-specific antigens to activate the patient's immune system for targeted malignant cell elimination. With their exceptional safety profile and minimal adverse effects, these immunotherapeutics are positioned as leading candidates for next-generation cancer treatment. The field has garnered global attention following Moderna's encouraging clinical outcomes for their melanoma vaccine, presented at the American Society of Clinical Oncology (ASCO 2024). CG Invites' Personalized Cancer Vaccine: Activating the Body's Natural Defense System The therapeutic mechanism of cancer vaccines parallels the body's intrinsic immune response pathways. Analogous to how influenza vaccines prime immune responses through viral antigen presentation, cancer vaccines stimulate anti-tumor immunity by exposing immune cells to tumor-specific neoantigens. However, cancer vaccines distinguish themselves through their dual mechanism of action: therapeutic intervention against existing malignancies and prophylactic protection against disease recurrence. Immune cells "programmed" to recognize tumor-specific signatures acquire targeted cytotoxic capabilities against malignant cells, generating robust antineoplastic responses while establishing durable immunological memory for recurrence prevention. The inherent heterogeneity of tumor mutations across patients necessitates individualized vaccine formulations, distinguishing these immunotherapeutics from conventional vaccines. Breakthrough anti-cancer effect demonstrated in mouse model of colon cancer In this preclinical investigation, CG Invites employed their proprietary imNEO algorithm to design cancer vaccines for colorectal carcinoma murine models. The study demonstrated significant immunogenic responses in 19 of 25 neoantigens (76% response rate). This represents a 2.8-fold enhancement over current industry benchmarks (approximately 20%) established by global leaders. CG Invites also conducted tumor growth inhibition tests by stratifying 25 tumor-specific neoantigens into 5 distinct groups. Results demonstrated complete tumor stasis in two groups and partial growth suppression in the other two groups. These findings validate the therapeutic efficacy of CG Invites' identified tumor-specific neoantigens in mediating tumor growth inhibition. Future combinatorial application of all 25 neoantigens is anticipated to yield enhanced anti-tumor efficacy. Figure. Immunogenicity and tumor growth inhibition test results using the mouse colorectal cancer model (MC38). AI-Derived Tumor Signatures: The imNEO Algorithm imNEO, developed by CG Invites, is an AI-powered algorithmic platform for precise identification of tumor-specific molecular signatures. This technology integrates 30 distinct immunological parameters, demonstrating unprecedented accuracy across multiple malignancies, including melanoma, lung cancer, colorectal adenocarcinoma, gastric cancer and pancreatic cancer. These findings transcend mere technical validation, demonstrating effective mobilization of patient immunological responses. This advancement shows promise either as a combined therapy to existing oncological treatments or as a novel therapeutic modality. “Personalized anti-cancer vaccine could be a game-changer in cancer treatment,” said experts Experts also predict that anti-cancer vaccines will revolutionize cancer treatment. Prof. Byoung-chul Cho of the Sinchon Severance Lung Cancer Center evaluated, who developed the global blockbuster Korean drug LECLAZA®, "While interpatient and intertumoral heterogeneity has presented significant therapeutic challenges, personalized cancer vaccines represent a paradigm-shifting therapeutic modality that transcends the limitations of conventional antineoplastic agents. This breakthrough offers renewed hope for both oncology patients and clinical practitioners." Prof. Bhak Jong-hwa of UNIST remarked, "In an era where clinical translation of genomic research remains exceptionally rare, CG Invites' cancer vaccine algorithm represents a landmark achievement, validating the therapeutic utility of genomic research." CEO Chung In-chul emphasized, "These results definitively demonstrate the potential of personalized cancer vaccines to pioneer novel pathways in oncological intervention." He further noted, "We are achieving significant advances in essential mRNA platform development and biomolecular delivery systems. We anticipate completing preclinical studies and initiating human clinical trials in the near term."
- Opinion
- [Reporter's View] Patience is required to foster K-Bios
- by Whang, byung-woo Nov 12, 2024 05:51am
- The performance of the domestic pharmaceutical bio industry is being evaluated with the approaching end of 2024. This year, the increased influence of domestic companies in the fields of biosimilars and CDMOs (contract development and manufacturing) and the technology transfer performance of bio ventures have been positively evaluated. However, there is also a view that the investment in venture firms has slowed down due to the overall stringent investment market and that the government's plans to foster the bio-industry are still fragmented across ministries. This is why the outlook for the domestic bio industry next year is also a mix of concerns and expectations. First of all, the biggest change is the launch of the National Bio Committee, chaired by the president. Although the Bio Health Innovation Committee under the Prime Minister was created last year, the industry has been emphasizing the need for a control tower to comprehensively manage the bio sector, so there are high expectations for the establishment of the top-level national bio policy deliberation body. While there are doubts over to what extent the National Bio Committee will be able to play its role, it is a timely creation as it will be able to address the larger discourse. In addition, the Korean version of ARPA-H, which will begin to kick off next year with the selection of companies, is also a positive bonus for the bio industry. It is also encouraging for the industry as the program is benchmarked after ARPA-H in the US and considers 'process over outcome'. In addition, the expected benefits for the bio industry from the election of Republican candidate Trump as president of the United States are also considered an opportunity for K-Bio to seize. During his candidacy, Trump considered lowering drug prices for new drugs and biosimilars to reduce the burden of medical expenses, and plans is open up the closed domestic market to overseas companies and induce competition. Korean companies will be forced to compete in the global market, but another way of looking at it is that the door will become open to all. Despite this good news, the industry is still arguing that it needs 'time' to foster the industry. While there are domestic pharma and biotech companies that are performing well overseas, it is strictly limited to a few companies. Another limitation is that the government's efforts, such as the National Bio Committee and the Korean ARPA-H project, are still in their early stages. In fact, there is no guarantee that things will go as expected, just as the government's ambitious K-Bio-Vaccine Fund investment project is struggling to raise funds due to low expectations. Government interest is like a double-edged sword. They want to see quick results for their support. For this reason, the industry is emphasizing the creation of an ecosystem rather than a near-sighted view to foster the industry. This is also a challenge for governments, who need to show results, as building ecosystems is vague and hard to visualize. However, relying on a few companies to produce tangible results may not be enough to foster the growth of the entire bio industry. Just as a deeply rooted tree on a solid foundation is not easily shaken, deep consideration of the ecosystem is necessary to establish the bio industry as Korea’s core national strategic business.
- Company
- Generic companies seek early entry into KRW 100 bil Tagrisso
- by Kim, Jin-Gu Nov 12, 2024 05:51am
- Patent challenges to Tagrisso (osimertinib), a non-small cell lung cancer treatment that posts annual sales of KRW 110 billion, are expanding. The companies that have filed patent challenges seek to first evade the product patent, which expires in 2035, and then launch generics early after November 2033, when the substance patent expires. According to the pharmaceutical industry on the 11th, Kwangdong Pharmaceutical recently filed a passive trial on the scope of rights against AstraZeneca for the Tagrisso formulation patent (10-2336378). The patent expires in January 2035. In addition to the formulation patent, Tagrisso has two other patents listed on the MFDS Green List. They are the product patents (10-1410902-10-1422619) that expire in November and December 2033, respectively. Kwangdong Pharmaceutical plans to launch Tagrisso generics early upon expiration of the substance patent in 2033, having first avoided the product patent, which expires in 2035. It is also possible that the company could further accelerate the early launch by taking advantage of the extended life of the substance patent. Prior to Kwangdong Pharmaceutical, Chong Kun Dang filed a patent evasion trial on Tagrisso’s product patent on the 25th of last month. With Kwangdong Pharmaceutical joining the patent challenge within 14 business days of Chong Kun Dang’s filing, the two companies have secured the “file of initial claims” requirement, which is one of the requirements for obtaining first generic exclusivity rights. Separately, Chong Kun Dang is developing a new drug for the treatment of non-small cell lung cancer. The candidate, CKD-702, has a dual antibody mechanism of action that simultaneously targets cMET and EGFR, and is currently in a global Phase 1 clinical trial. Chong Kun Dang’s strategy is to target the NSCLC treatment market by developing both new drugs and generics. According to the drug market research institution IQVIA, Tagrisso's sales in the Korean market were KRW 111 billion last year. This is up 4% from KRW 106.5 billion it had posted in 2022. The sales are expected to have increased significantly since the drug's reimbursement was expanded to cover “first-line treatment for locally advanced or metastatic NSCLC with certain gene mutations” this year. Leclaza (lazertinib), whose reimbursement was also expanded to first-line treatment along with Tagrisso, generated KRW 22.6 billion in sales last year.