- LOGIN
- MemberShip
- 2026-04-04 07:31:05
- Company
- Oral psoriasis drug 'Sotyktu' lands at Big 5 hospitals
- by Son, Hyung Min Nov 08, 2024 05:46am
- Product photo of Oral psoriasis drug 'Sotyktu' is now available for prescription at tertiary general hospitals. According to industry sources, BMS Korea’s TYK2 inhibitor, Sotyktu (deucravacitinib), has passed the drug committees of the 'Big 5' tertiary hospitals, including Samsung Medical Center, Seoul University Hospital, Seoul St. Mary's Hospital, Seoul Asan Medical Center, and Sinchon Severance Hospital. Prescription code is also available in major hospitals nationwide, including Gangnam Severance Hospital, Konyang University Hospital, Soonchunghyang University Bucheon Hospital, Seoul National University Bundang Hospital, Soonchunghyang University Hospital Seoul, Ewha Womans University Medical Center, Chosun University Hospital, and Soonchunghyang University Cheonan Hospital. Since being added to the reimbursement list in April, this drug is increasingly available in hospitals. Sotyktu is the first TYK2 inhibitor approved for adults with moderate-to-severe plaque psoriasis. It was approved in August 2023 in South Korea to treat adult patients with moderate-to-severe plaque psoriasis who are candidates for phototherapy or systemic therapy. The insurance coverage was made available eight months after receiving approval. It is covered by insurance reimbursement for the treatment of patients over 18 with chronic severe plaque psoriasis who have symptoms lasting over six months. Coverage requirements include ▲Plaque psoriasis with over 10% of the total skin areas ▲Patients who have Psoriasis Area and Severity Index (PASI) score of over 10 ▲Patients who have undergone methotrexate or cyclosporine for over 3 months but cannot continue the treatment due to no response or side effects ▲Patients who have undergone light therapy or UPV photo therapy for over 3 months but cannot continue the treatment due to no response or side effects. Clinical efficacy of Sotyktu was demonstrated in Phase 3 POETYK PSO-1 and POETYK PSO-2 clinical studies, which compared the drug with placebo or Otezla in 1,684 adult patients 18 years or above with plaque psoriasis. POETYK PSO-1 studies have shown that the Sotyktu treatment group had a PASI 75 response rate of 58.4% at week 16, which was significantly higher than the apremilast group's 35.1% and the placebo group's 12.7%. Furthermore, 53.6% of the Sotyktu treatment group achieved sPGA score of 0 or 1, higher than 32.1% in the apremilast group and 7.2% in the placebo group. In POETYK PSO-2 study, the Sotyktu treatment group had a PASI 75 response rate of 53.0% at week 16, which was significantly higher than the apremilast group's 39.8% and the placebo group's 9.4%. Additionally, 49.5% of the Sotyktu treatment group achieved sPGA score of 0 or 1, higher than the apremilast group's 33.9% and the placebo group's 8.6%. Sotyktu’s high response was maintained for up to 52 weeks. "Previously, patients who did not respond to or have had side effects when treated with conventional treatments, such as systemic therapy or light therapy, had biological agents as their only option. Sotyktu, which offers the convenience of once-daily oral administration, is expected to meet the unmet needs of psoriasis patients," Choe Yong-beom, President of the Korean Society for Psoriasis, said.
- Policy
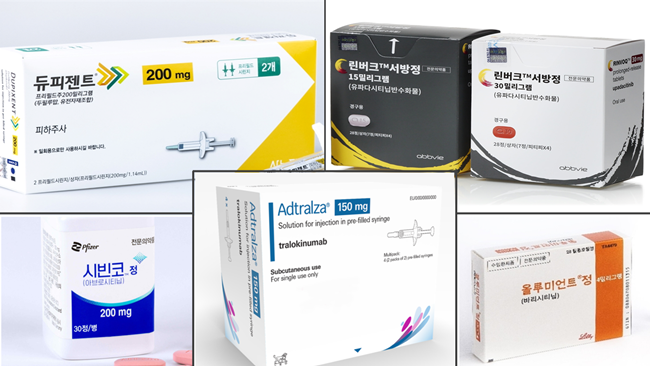
- Drug switching for atopic dermatitis measure submitted
- by Lee, Tak-Sun Nov 07, 2024 05:47am
- Pharmaceutical companies have submitted their measures for voluntary drug price reduction as part of financial allotment following expanded reimbursement. Discussions about allowing drug switching between biological agents and JAK inhibitors used to treat severe atopic dermatitis are speeding up. The Health Insurance Review and Assessment Service (HIRA) has already prepared the reimbursement criteria, and pharmaceutical companies have submitted their measures for voluntary drug price reduction as part of financial allotment following expanded reimbursement. Attention is drawn to whether it will pass the Drug Reimbursement Evaluation Committee (DREC) review, the final stage for reviewing reimbursement appropriateness. According to industry sources on November 6th, pharmaceutical companies Pfizer for Cibinqo and Lily for Olumiant submitted their financial impact analysis to the HIRA. The drug pricing reduction system for expanded scope of use requires the submission of financial impact analysis reports. The government excludes drug price reduction when the estimated additional claim amount following the expanded scope of use is below KRW 1.5 billion. The drug price reduction is applied for products over KRW 1.5 billion and below KRW 10 billion. In this case, pharmaceutical companies are required to submit a financial impact analysis, including measures to voluntarily lower prices, to the HIRA. In mid-October, the HIRA required each pharmaceutical company to submit financial impact analysis. Earlier this month, pharmaceutical companies have started to submit their reports. The issue of allowing drug switching for the treatment of severe atopic dermatitis was dealt with in the parliamentary audit held last month, so the health and welfare authority appears to be rushing the measure. The HIRA has been discussing with experts since September about whether to allow drug switching between biological agents and JAK inhibitors. The reimbursement criteria have been prepared, reflecting the latest evidence and clinical application. The remaining step is to minimize the financial impact following the expanded scope of use. Pharmaceutical companies' percentage of voluntary drug price reduction is the key. As pharmaceutical companies have submitted their measures for voluntary price reduction, the the Drug Reimbursement Evaluation Committee (DREC) review of the HIRA will make a final decision. The 11th DREC review is scheduled for July 2024. Attention is drawn to whether drug switching for severe atopic dermatitis will be considered for the agenda. When it passes the DREC review, the final reimbursement expansion will be decided through negotiation with the National Health Insurance Service (NHIS). Meanwhile, treatments for severe atopic dermatitis have been listed for reimbursement, starting with the biological agent, 'Dupixent Inj,' approved in January 2020. It was followed by the JAK inhibitors, Olumniant Tab, Linvoq ER Tab, and Cibinqo Tab. In May, another biological agent, Adtralza PFS, was listed for reimbursement. As a result, patients have more drug options. However, reimbursement and special cases do not cover drug switching between biological agents and JAK inhibitors, so it remains an issue. When clinical practices initially use high-cost drugs, and then the effects seen are not apparent, they may continue administering first-line treatments. Therefore, the medical community has been demanding the allowance of drug switching.
- Company
- Reimb discussions discontinued for NMOSD drug Uplizna
- by Eo, Yun-Ho Nov 07, 2024 05:47am
- Uplizna, a new drug for neuromyelitis optica spectrum disorder (NMOSD) that is administered twice a year, has failed to pass the final gateway to reimbursement in Korea. According to Dailypharm coverage, Mitsubishi Tanabe Pharma Korea’s pricing negotiations with the National Health Insurance Service (NHIS) for Uplizna (inebilizumab), a treatment used to treat adult patients with for neuromyelitis optica spectrum disorder (NMOSD) who are positive for anti-Aquaporin-4 (AQP4) antibodies, has been discontinued recently. The company had accepted the “below the evaluated amount” condition set by the Health Insurance Review and Assessment Service's Drug Reimbursement Evaluation Committee for the reimbursement of Uplizna (inebilizumab) in August but was unable to reach an agreement. NMOSD occurs when AQP4 autoantibodies, a disease-specific biomarker produced by B cells, bind to AQP4, a target antigen present on glial cells in the central nervous system, and activate the immune responses, causing nerve damage. Uplizna is an anti-CD19 human monoclonal antibody that selectively binds to CD19, a B cell-specific surface antigen, depleting B cells that produce AQP4 antibodies, thereby preventing disease relapse. The safety and efficacy of Uplizna were demonstrated in the N-MOmentum study, which evaluated the use of Uplizna monotherapy in 230 patients without the use of concomitant immunosuppressive agents. Study results showed that 89% of patients treated with Uplizna did not experience a relapse during 197 days of follow-up, resulting in a 77.3% reduction in the risk of relapse compared to placebo. Safety evaluations Uplizna also showed comparable rates of adverse events to the placebo group. Also, in an extension study, Uplizna continued to reduce the risk of relapse for at least 4 years, with an 87.7% relapse-free rate. In terms of long-term safety profile, Uplizna was generally well tolerated, with no increase in infection rates due to B-cell depletion. NMOSD is a serious autoimmune disease in which most patients experience persistent relapses and incomplete recovery, resulting in accumulated nerve damage that can cause vision loss, gait disturbances, and even death from respiratory failure.
- Product
- KTIC, ’21s Wegovy prescriptions, not a telemedicine issue'
- by Kang, Hye-Kyung Nov 07, 2024 05:47am
- the platform industry has spoken out to state its opinion regarding the government’s move to restrict non-face-to-face prescriptions in response to the criticism that 'obesity drugs are being easily prescribed – just in 21 seconds - through non-face-to-face medical treatment platforms.’ While the platform industry sympathizes with social concerns, the essence of the problem, such as the misuse of Wegovy, does not lie in the mode of consultation, whether it is face-to-face or non-face-to-face. On the 6th, the Korean Telemedicine Industry Council (represented by Jae-won Sun, Seul Lee, KTIC) announced its ‘Position regarding the concerns over misuse of obesity drugs through non-face-to-face medical treatment platforms,’ stating, “Since the recent launch of new obesity drugs, some influencers and patients have been found to have used non-face-to-face medical treatment in the process of prescribing the drugs, raising concerns. But the root of the problem of misuse of obesity drugs does not lie in the mode of consultation.” KTIC claims that the misuse of anti-obesity medications can occur regardless of the mode of consultation and that the problem stems from the prescribing and medication guidance process. “Therefore, various social efforts, such as strengthening DUR registration to improve transparency and the doctors’ and pharmacists' compliance with laws and regulations, are essential to essentially solve the problem of off-label drug abuse, including obesity drugs. The platform does not directly intervene in prescribing and dispensing, but will contribute to the doctors’ and pharmacists’ law abidance and prevention of drug abuse through technical support.” The association also emphasized that telemedicine overseas focuses on “preparing safety measures” rather than “putting restrictions.” Rather than looking at telemedicine as the cause of the problem, KTIC emphasized the need to put in place specific controls to ensure the safety of prescribing and medication use. The UK has implemented transparency improving guidelines for pharmacies and medical institutions to manage certain medicines, and the US is known to be improving its management system to ensure the safety of generic obesity drugs. The KTIC pledged to prepare practical and concrete management measures to improve the accessibility and safety of non-face-to-face medical treatment. “It is a clear fact that the high convenience of non-face-to-face medical treatment, which enables access to medical services without time and space constraints, enhances people's rights and interest in healthcare, but apart from this good function, we deeply sympathize with the social concerns that have been raised with the recent launch of new obesity drugs,” they said. As a member of the non-face-to-face healthcare delivery system, we will strive to enhance people's medical access and safety while addressing various social concerns that have been raised with the launch of new obesity drugs.” The council will work closely with the relevant authorities to provide input to help formulate measures to improve access to healthy healthcare, while actively guiding participating medical institutions and pharmacies to comply with relevant laws and MFDS indications in the prescribing and dispensing process. “The council will continue to pay attention to various concerns and opinions on-site to protect the public's health and make efforts to establish telemedicine as a trusted medical service.”
- Policy
- Novartis cuts price of Cosentyx after PVA negotiations
- by Lee, Tak-Sun Nov 07, 2024 05:46am
- Cosentyx UnoReady Pen The price of Novartis’s Cosentyx(secukinumab) is set to be reduced due to its increased use. This is because the National Health Insurance Service recently reached an agreement during Price-Volume Agreement negotiations with the company for Cosentyx. According to industry sources on the 6th, the insurance price ceiling of Cosentyx was adjusted for the second time under the PVA system after 2021. The affected products are the Cosentyx SensoReady Pen and Cosentyx UnoReady Pen. Cosentyx was listed for reimbursement in Korea in August 2017. Currently reimbursed indications include ▲chronic severe plaque psoriasis, ▲active and progressive psoriatic arthritis, and ▲severe active ankylosing spondylitis. According to the market research institution IQVIA, Cosentyx SensorReady Pen has seen an increase in sales every year since its reimbursement. In 2019, sales reached KRW 12.3 billion, then KRW 19.9 billion in 2020, KRW 25.1 billion in 2021, KRW 30.3 billion in 2022, and KRW 31.7 billion in 2023. In addition, Cosentyx UnoReady Pen, a high-dose product that was launched last year, generated sales of KRW 4.5 billion. Due to such high demand, the insurance price ceiling of Cosentyx had been adjusted in 2021 under the PVA system. At the time, the price of Cosentyx Sensoready Pen dropped by 7.3% from KRW 682,938 to KRW 630,084. At the time, 'Type A’ of the PVA negotiations was applied. Type A negotiations are applied to listed new drugs whose claims in the same product category have increased by more than 30% from the expected claims amount. In 2020, the drug had already well exceeded the expected claims amount. Three years have passed, and this year, ‘Type B’ PVA negotiations were applied. Type B negotiations are applied when the claims for the same product group, whose insurance price ceiling had been adjusted by Type A negotiations, or four years passed after the date of initial listing or the date the price ceiling was adjusted through negotiations, have increased by 60% or more than the previous year's claims, or by 10% or more but the increase is more than KRW 5 billion. Since Cosentyx had its price ceiling adjusted through Type A negotiations, it is possible that the additional negotiation for price adjustments was ordered because its claims increased by more than 60% or more than KRW 5 billion over the previous year's claims. Given that Cosentyx Sensoready’s sales grew 5% YoY in 2023 by KRW 1.4 billion (IQVIA), the introduction of Cosentyx UnoReady Pen, which falls in the same product group, may have triggered the PVA negotiation. Combined, the increase in sales amounted to more than KRW 5 billion compared to the previous year. Upon the agreement, the final price of Cosentyx is expected to be announced after deliberation by the Ministry of Health and Welfare’s Health Insurance Policy Review Committee.
- Policy
- 'Eutropin·Biktarvy' price drop amid sales hike
- by Lee, Tak-Sun Nov 06, 2024 05:52am
- Product photos of Eutropin and Biktarvy. Due to the increased volume of usage, drug pricing for 'Eutropin Inj (somatropin, LG Chem)' and 'Biktarvy (Bictegravir/Emtricitabine/Tenofovir Alafenamide, Gilead Science),' which recently showed a sales hike, is expected to be reduced. Sources said on November 5th that the Health Insurance Review and Assessment Service (HIRA) recently agreed on drug pricing adjustment of these products through the 'Type-Na' negotiation. As a result, the final adjusted prices will be listed on the reimbursement list following a review from the Health Insurance Policy Review Committee. Eutropin has been a top-selling growth hormone drug in the Korean market. 'Eutropin S Pen,' introduced in April 2022, is skyrocketing in sales. Eutropin S Pen's expiration date has been extended from 18 months to 24 months, and, based on UBIST, it generated sales of KRW 81.5 billion last year. Eutropin recorded KRW 21.3 billion in sales in the same period. The combined sales growth rate for the two products is 24% compared to last year. Biktarvy also successfully achieved two-digit sales figures. Based on IQVIA last year, it generated sales of KRW 54.5 billion, up 11% from the previous year (KRW 49.1 billion). Biktarvy received the MFDS approval in January 2019, and it has been in sales following reimbursement listing in July. It is a single-capsule combination drug for the treatment of HIV containing three active ingredients: Bictegravir, Emtricitabine, and Tenofovir alafenamide. With Yuhan and Gilead Sciences in a joint-sales agreement, Biktarvy is a top-selling drug in the HIV market. "Eutropin and Biktarvy show rapid growth in the market," an expert in the pharmaceutical industry said. "With the increased usage volume, reducing its ceiling price according to the Price-Volume Agreement (PVA) is inevitable." In 2022, the ceiling prices of Eutropin and Biktarvy were lowered due to the PVA agreement. Eutropin was reduced by 2.9%, and Biktarvy was reduced by 3.0%. Meanwhile, the negotiation of the PVA 'Type-Na' is conducted when the ceiling price is adjusted by the type-Ga price or when the bill amount of the same product has increased by more than 60% or 10% from the previous year's bill, and the increase is more than KRW 5 billion.
- Company
- 20-valent pneumococcal conjugate vaccine set to launch
- by Moon, sung-ho Nov 06, 2024 05:52am
- Attention has been drawn to another competitor set to launch in the market for the 'pneumococcal vaccine.' This year, the pneumococcal vaccine market has been fiercely competitive with newly released products. As the 20-valent vaccine is likely to come out right after the 15-valent conjugate vaccine has entered the market after 13 years, competition between global pharmaceutical companies to take the market share is becoming intense. As clinical practices draw attention to the next generation of vaccines, they expect that 'price' will be a determining factor in the non-reimbursed market. According to pharmaceutical industry sources, the Ministry of Food and Drug Safety (MFDS) granted approval of Pfizer Korea's 'Prevenar 20 Prefilled Syringe (hereafter, Prevenar 20).' Prevenar 20 is Pfizer Korea's next-generation product that follows 'Prevenar 13,' which currently dominates the Korean market for pneumococcal vaccines. In addition to serotype components in the 13-valent vaccine, Prevenar 20 contains seven additional serotypes, including 8, 10A, 11A, 12F, 15B, 22F, and 33F. The MFDS has approved the drug's indication to prevent invasive pneumococcal disease, pneumonia, and acute otitis media caused by pneumococcus in infants, children, and adolescents aged six weeks and below 18 years old. It is also used to prevent invasive pneumococcal disease and pneumonia caused by pneumococcus in adults 18 years of age and above. As Prevenar 20 has been officially approved, Pfizer Korea will prepare to launch the product. Pfizer Korea will likely aim to launch the product in the first half of 2025. Analysis suggests that an official launch is possible when Prevenar 20 is included in the vaccine guidelines of major medical sciences organizations and the National Immunization Program (NIP). The process is similar to that of MSD's 15-valent vaccine, 'Vaxneuvance,' which has been officially launched into the clinical practices. MSD Korea received the official approval of Vaxneuvance from the MFDS on October 31, 2023. Considering Pfizer Korea's Prevenar 20 was approved on October 31, 2024, Vaxneuvance was approved precisely a year before. After the approval, Vaxneuvance was included in the Korean Society of Infectious Diseases (KSID)'s '2024 Updated Guidelines for Adult Immunization' that "KSID recommends use of the 15-valent pneumococcal conjugate vaccine (PCV15, MSD's Vaxneuvance) over the 13-valent pneumococcal conjugate vaccine (PCV13, Pfizer's Prevenar 13) for individuals subjected to adult pneumococcal conjugate vaccine (PCV) immunization." MSD launched the vaccine in April this year soon after its inclusion in the NIP, and Pfizer Korea is likely to follow a similar launching schedule. "We have been trying to receive Prevenar 20's approval. The specific schedule for launch has not been decided," a representative from Pfizer Korea said. "We will prepare for the launch next year." "We have yet to confirm our Korean partner for Prevenar 20," a representative from Pfizer Korea said. "We are in a partnership with Chong Kung Dang for Prevenar 13 for adults, and we have an agreement with 'Koreavaccine' for Prevenar 13 for young children." The industry draws attention to the competition in the pneumococcal vaccine market for the clinical practice. The question is who will dominate the market, given the competition between two global pharmaceutical companies. Following the withdrawal of the 10-valent PCV vaccine (Synflorix, GSK) from the Korean market, the 13-valent vaccine Prevenar 13 and the 15-valent vaccine Vaxneuvance are currently competing in the clinical practice. In particular, MSD Korea chose Boryung Biopharma as its Korean partner. Upon launching Vaxneuvance, MSD Korea actively engaged in sales‧marketing by hiring Paik Jong Won, a businessman, TV celebrity, and CEO of TheBorn Korea, as its commercial model. According to a pharmaceutical market research firm UBIST, the number of NIP and non-reimbursed immunizations for adults in nationwide hospital‧private clinics is rising after Vaxneuvance launched in April. In detail, based on UBIST, the cumulative number of PCV vaccine immunizations from April to June this year is 994 counts. Among these, 809 immunizations were with Prevenar 13. 185 Vaxneuvance immunization was made. After MSD Korea launched Vaxneuvance in April this year, they chose Paik Jong Won, TheBorn Korea CEO, as their commercial model, putting efforts into raising awareness. To the right is the product photo of Prevenar 20, Pfizer Korea's pneumococcal vaccine, which is expected to launch in the first half of 2025. The number of immunizations with Prevenar 20 is below that of market dominating Prevenar 13, but the number is rising. Based on the assumption that Vaxneuvance has been well established in clinical practice as a new vaccine, the number of immunizations were likely increased. In fact, a director of the pediatrics and adolescent clinic said, "When we used Prevenar 13 for immunizations, we had several inquiries if it's possible to switch to Vaxneuvance. Since medication switching is possible during the immunization course, we answer patient inquiries." And added, "If patients have not completed the immunization schedule, medication switching is possible. We had several cases of switching after inquiries." Considering changes brought by the Vaxneuvance launch this year, Prevenar 20 will likely bring changes to clinical practices such as the department of internal medicine and the department of pediatrics and adolescents. Sang Hyuk Ma, a Director of Pediatrics and Adolescents at Changwon Fatima Hospital, said, "Following Prevenar 13, Vaxneuvance and the 15-valent vaccine have been introduced to South Korea. We must consider clinically if a vaccine with more serotypes is appropriate for South Korea. Pneumococcal serotypes tend not to mix, and serotypes common in South Korea must be accounted for vaccination." Ma added, "In other words, the vaccines were developed towards what's needed in the United States. We must look into common serotypes in South Korea and discuss whether the latest vaccines are necessary for immunizations in Korean citizens, including infants and children." Gwak Kyung-Keun, the President of Seoul Physicians' Association, said, "In my opinion, there is no difference between the 15-valent and the 20-valent vaccines. There is no basis for which one is more superior." Gwak anticipated, "In the end, it will be the sales‧marketing competition between companies. One would argue that the 20-valent vaccine has more serotypes but will be a marketing fight without a comparison basis."
- Company
- Roche seeks digital solutions for its healthcare ecosystem
- by Whang, byung-woo Nov 06, 2024 05:52am
- With the importance of personalized healthcare being emphasized more than ever with the emergence of innovative new drugs, Roche Diagnostics Korea is aiming to lead the market by focusing on digital solutions. The company plans to lead the healthcare sector through digital transformation in line with the paradigm shift in treatment, from early diagnosis and precision medicine to predictive medicine. (From the left) Tae-Hyun Um (Director of Insurance Policy Affairs, KSLM), Yeo-Min Yun, Director of Scientific Affairs, KSLM), Sail Chun (CEO&Chaiarman, KSLM), Kit Tang (General Manager of Roche Diagnostics Korea), Muhwan Yun (head of Digital Insights, Roche Diagnostics Korea), Sungho Cho (Head of Core Lab & POC, Roche Diagnostics Korea) On the 5th, Roche Diagnostics Korea held a press conference on the theme of “Future Healthcare and Innovation Presented by Diagnostic Tests” and emphasized the importance and opportunities of digital transformation in the healthcare sector. With the launch of the Digital Insights division, the company has been focusing on providing data-driven insights and marking a new turning point in personalized medicine. “Digital transformation in healthcare is the new normal, and we have already entered the era of artificial intelligence (AI) transformation beyond digital transformation,” said Muhwan Yun, head of Digital Insights at Roche Diagnostics Korea. ”Roche Diagnostics has set its own ethical standards for applying AI to healthcare, and is aiming to expand and innovate its digital portfolio, including continuous R&D investment and collaboration.” One representative project is Smart Lab, a digital transformation of diagnostic laboratories. The goal is to make existing laboratory operations more efficient, flexible, and data secure, to ultimately increase insight from analyzing data. For this, Roche Diagnostics has been emphasizing 'digital solutions' and building an enabling ecosystem. It focuses on incorporating significant data of a certain scale, advanced analytics, and digital technologies to support medical decisions and improve the treatment experience. The company’s representative technology, NAVIFY, is a cloud-based data platform that analyzes vast amounts of healthcare data in a standardized format to enable precision medicine. “In practice, we have seen significant improvements in time, manpower, and cost metrics for testing and analysis after implementing Roche Diagnostics Digital Insight Solution’s NAVIFY portfolio,” said Yun, ”and the satisfaction rate among healthcare providers who have used them is over 90%.” “With South Korea set to enter a super-aged society next year, the role of diagnostic test data for efficient and effective treatment is expected to increase further,” said Kit Tang, General Manager of Roche Diagnostics Korea. ”Roche Diagnostics Korea will continue to strive to contribute to an efficient healthcare system and improved patient outcomes with innovative diagnostic solutions covering a wide range of disease areas.” The hurdle to building a digital ecosystem for diagnostic tests is in the “maturity” of the digital transformation In the long run, digital transformation in diagnostics is essential, but there are hurdles. Not only are institutional improvements required for the digital transformation of the existing system, but the nature of healthcare is such that it is important to integrate the different perspectives of each stakeholder, including hospitals, manufacturers, patients, and academic societies. Yeo-Min Yun (Director of Scientific Affairs, KSLM)In response to this, Yeo-Min Yun, Director of Scientific Affairs at KSLM of the Korean Society for Laboratory Medicine (Konkuk University Hospital), emphasized that the emergence of technology should be supported by measures that can encourage their use in practice, such as by assigning service fees. “If you look at imaging, the technology that analyzes the images through AI technology is being applied and utilized to create values that have never existed before,” said Director Yun. ”Referring to this precedent, I think that efforts should be made to apply a service fee for such diagnostic tests and recognize their value in clinical practice.” Ultimately, Roche Diagnostics' challenge is to collect and utilize fragmented data. To this end, the company expects the newly launched division to serve as a base to build the ecosystem. “In Korea, data compatibility is relatively poor, so we are striving to collect and utilize data well,” said Director Yun, adding, ”As the implementation of digital ecosystems is not fully mature yet, it will be important for each component, including the company, to be integrated.”
- Company
- CKD’s CKD-508 receives approval to initiate P1T in the U.S.
- by Son, Hyung Min Nov 06, 2024 05:52am
- Chong Kun Dang announced on the 4th that it has received approval from the U.S. Food and Drug Administration (FDA) for the Phase I clinical trial of CKD-508, its new drug candidate for dyslipidemia. In the trial, Chong Kun Dang will confirm the safety and lipid-improving effects of CKD-508 and explore the optimal dose for a Phase II trial. CKD-508 is a treatment for dyslipidemia that works by inhibiting the activity of cholesteryl ester transfer protein (CETP), which facilitates the transport of cholesteryl esters (CE) and triglycerides (TG) between lipoproteins in the blood, thereby lowering low-density cholesterol (LDL-C) levels and increasing high-density cholesterol (HDL-C) levels. In a non-clinical efficacy trial conducted by Hyosung Research Center, Chong Kun Dang confirmed the LDL-C-lowering and HDL-C-raising effects of CKD-508 and demonstrated a significant reduction in apolipoprotein (Apo-B), a key marker of dyslipidemia. “CKD-508 is an innovative drug that is expected to be effective at low doses by solving the problems of previous CETP inhibitors that were discontinued due to drug accumulation and blood pressure increase based on its strong binding to CETP,” said Chong Kun Dang. ”If successful, it is expected to become a new treatment option for patients with statin-resistant dyslipidemia that cannot be controlled with statins.”
- Policy
- How will the new drug review process change with fee hike?
- by Lee, Hye-Kyung Nov 06, 2024 05:52am
- The Ministry of Food and Drug Safety (MFDS) announced plans to raise the fee for new drug approvals to KRW 410 million from January 1 next year. The agency has prepared the approval and review process to implement the plan and has begun collecting industry opinion. According to MFDS’s press corp coverage, the MFDS recently delivered a revised version of the “Operating Procedures for Approval and Examination of New Drug Products (Guideline for public officials)” to the pharmaceutical and biotech industries and relevant associations. The guidelines contain specific measures to implement the “Innovative Measures for New Drug Approval” that was announced by the MFDS on Sept. 9, including the operation of a dedicated review team for each product and prioritizing GMP-GCP on-site inspections. “Upon the announcement of the rise in the approval fee of new drugs under the Innovative Plan for New Drug Approval, the pharmaceutical industry raised the question of whether the actual approval period will be shortened,” said an MFDS official. ”By recruiting reviewers, GMP inspections will be prioritized to be completed within 90 days from the date of the application receipt, and pre-registration of supplementary materials and explanatory meetings will reduce unnecessary delays in the approval process, dramatically shortening the approval period for new drugs from 420 days to 295 days.” The enactment of the Guideline on Operating Procedures for New Drug Approval and Review was promoted in line with the redetermination of new drug approval fees, which will take effect on January 1, 2025, and includes various changes to differentiate it from the existing approval process, such as the formation of a dedicated team, the establishment of explanatory meetings, and the introduction of a procedure for pre-registration of supplementary materials. From next year, when a new drug approval application is filed, MFDS will conduct an independent preliminary review within 7 days. During the preliminary review period, 80% of the fee will be refundable, and the MFDS will check the submission requirements. If there are any missing materials during this process, the applicant will have 14 days to submit them before an “initiation meeting” is held. Given the overall review timeline, the preliminary review process is crucial as data not submitted by the initiation meeting will be difficult to submit later and the application will be categorized as an application that requires primary supplementary data. A key part of this process is the establishment of a dedicated team for each item before the initiation meeting. The head of the approval division will lead the team, and a representative from the approval division will take charge as each item’s manager, and the team will be composed of representatives from ▲safety, efficacy, ▲quality control, ▲GMP (good manufacturing practice), and ▲GCP (good clinical practice). In addition, an initiation meeting is held within 14 days of receipt to coordinate the entire approval and review process and to provide the applicant with the necessary materials and procedures. The initiation meeting may be held face-to-face, via video, or in parallel, which is expected to increase the efficiency of the new drug review process. According to the proposed Operating Procedures for Approval and Examination of New Drug Products, GMP inspections will be conducted within 90 days from the date of application receipt, and GCP inspections will be conducted within 60 days after the first supplement. By accelerating the inspection schedule compared to the existing procedure, the verification required for the manufacturing and clinical management procedure will be completed at an earlier stage. This is intended to speed up the approval process and ensure adequate quality control. A number of improvements have also been made to the supplement requests. Previously, there were only first and second supplementary notifications, but the reform introduces a first supplementary request briefing session and a second supplementary request briefing session to ensure that the applicant fully understands the supplementary request. In particular, the explanatory meetings provide detailed information on the scope and requirements of the required materials and provide time to answer the applicant’s questions. In particular, for the first round of supplemental data submissions, a pre-registration process and a request for an information session can be made by the applicant. Applicants can pre-register their supplementary data and request the KFDA to hold an explanatory meeting. This allows the adequacy of the supplementary data to be reviewed in advance, reducing additional supplementary work and increasing the efficiency of the approval process. In addition, a final meeting will be held 5 days before the final approval target date to discuss the results of the approval review. “In addition to the effect of shortening the approval period, the revised procedure will improve the transparency of the approval and review process and help pharmaceutical and biotech companies develop new drugs efficiently while protecting the public's health,” said an MFDS official. ”We will collect opinions from relevant associations and industries to prepare the final draft and continue to optimize and implement the approval review service for the development of the domestic pharmaceutical industry.”