- LOGIN
- MemberShip
- 2026-04-07 18:50:36
- Policy
- Celltrion Pharm completes localizing 3 Edarbi products
- by Lee, Hye-Kyung May 09, 2024 05:52am
- Celltrion Pharm has completed the localization of Edarbi Tab (azilsartan medoxomil potassium), a treatment for essential hypertension. On the 8th, the Ministry of Food and Drug Safety (MFDS) simultaneously withdrew and approved the authorization of Celltrion Pharm's Edarbi Tab 20mg. In December 2020, Celltrion Pharm acquired the entire rights, including sales and patents, of Takeda Pharmaceuticals' 12 ETC and 6 OTC drugs in 9 countries in the Asia-Pacific region for USD 278.3 million (approximately KRW 307.4 billion). The Edarbi license that was withdrawn this time is the item Takeda Pharmaceuticals received approval for in May 2017 and then sold to Celltrion Pharm. Celltrion Pharm had previously received approval for its Edarbi 40 mg and 80 mg in December 2023 and then withdrew the licenses for the Edarbi Tab 40 and 80 mg that the company acquired from Takeda Pharmaceuticals in March 2024. With the simultaneous approval and withdrawal of Edarbi 20mg, the company completed preparations to localize the production of the hypertension treatment. Celltrion Pharm acquired all rights to Edarbi and has been preparing to self-produce and sell the drug to ensure stable product distribution. Edarbi is an angiotensin II receptor blocking (ARB) class drug used for the management and treatment of hypertension. The drug lowers blood pressure by blocking angiotensin II receptors and suppressing angiotensin II, a hormone that increases blood pressure. Adults may take 40 mg of Edarbi once daily, with or without food, and may increase the dose to a maximum of 80 mg per day if the patient’s blood pressure is not adequately controlled. Edarbi 20 mg is indicated as an initial dose for the very elderly (75 years or above) who are at risk for hypotension, patients with mild-to-moderate hepatic impairment, and patients with possible intravascular volume depletion. Following the launch of its alogliptin+metformin combination therapy for diabetes the past year, Celltrion Pharm has received approval for its Edarbi as ‘Celltrion Azilsartan Medoxomil Tablets’ for export and as ‘Edarbi Tab’ for domestic use.
- Policy
- Name of transferred and aquired drugs can be changed in KOR
- by Lee, Hye-Kyung May 09, 2024 05:52am
- Korea’s regulations now stipulate that brand names of a drug transferred to a different company can be changed. According to industry sources on the 7th, the Ministry of Food and Drug Safety recently announced the revision of the 'Casebook for Drug Product Naming Practices (Citizen's Guide)' and announced the allowance of brand name changes. The MFDS changed the regulation that only allowed the company name of a product to be changed upon transfer of a drug that was specified in 2016 to allow the product name to be changed as well. However, in response to inquiries from the pharmaceutical industry regarding the transferred drug’s product name, the MFDS has been responding that "the product name is determined after comprehensive consideration on its domestic use (including similar product names) at the time of completion of the marketing authorization (report).” The pharmaceutical industry had also inquired whether a pharmaceutical company that voluntarily withdraws its marketing authorization for the product and acquires a product with the same ingredient from a third party can apply for the grant to change the product name of the new item to the name previously held by the company. In general, in order to change the product name upon receiving approval for the change in manufactured and imported items (report) due to transfer or assignment, Article 10 (9) of ‘Regulation on Pharmaceuticals Approval, Notification, and Review’ allows for change of a product’s name if it meets the criteria of Article 10 (8) of the same regulation. If a company wishes to use the same product name as the withdrawn item, the company may change the product name to the other name if it is clear that the withdrawn item was not distributed in the market and lacks production and import performance within the period of use granted for the withdrawn item before the transfer. However, the authorities added that the situation at the time of marketing authorization would need to be considered. However, during the revision of the citizen’s guide, the MFDS has given a clear answer that "it is possible to use” the previous product’s name in the event of a product transfer (merger or acquisition, etc.). The MFDS may grant permission to a company to change the name of a product in accordance with Article 11, paragraph 2 of the 'Rules on the Safety of Medicines, etc.' if the product name the company seeks to use is in accordance with Article 11, paragraph 2 of the 'Rules on the Safety of Medicines, etc.’ Meanwhile, the MFDS also reviews the conformity of the product name change as part of the drug's labelling during marketing authorization reviews.
- Policy
- AZ renews orphan drug Strensiq’s risk-sharing agreement
- by Lee, Tak-Sun May 09, 2024 05:52am
- The risk-sharing agreement (RSA) for Strensiq Inj (asfotase alfa, AZ), a treatment for hypophosphatasia, has been successfully renewed in Korea. The drug was last covered under the RSA scheme in June 2020, when it was listed for reimbursement in Korea under the Expenditure Cap Type and Refund Type RSA. According to the National Health Insurance Service on the 8th, AstraZeneca and NHIS recently completed negotiations to renew the risk-sharing arrangement for 5 Strensiq Inj items. Both sides reportedly settled on appropriate terms for the agreement. The 4-year RSA term for Strensiq Inj was set to expire on May 31, but with the renewal, the drug will continue to be reimbursed for 5 years thereafter. The drug’s prior authorization requirement was also removed recently. Previously, if a patient with perinatal/infantile- and juvenile-onset hypophosphatasia wished to use Strensiq as a long-term enzyme replacement therapy to treat bone symptoms, he or she had to be reviewed on an individual basis by the Heath Insurance Review and Assessment Service’s review panel. After 4 years of prior authorizations, Strensiq’s reimbursement status has been converted to post-approvals due to its high prior authorization rate and lack of fluctuation in the number of patients. When it was first introduced, Strensiq was distributed domestically by Handok but has been supplied by AstraZeneca Korea since last year. The exclusion of the prior authorization requirement and RSA renewal are expected to improve patient access to Strensiq in Korea.
- Company
- Novel targeted therapy for gastric cancer 'Vyloy' may launch
- by Eo, Yun-Ho May 09, 2024 05:52am
- 'Vyloy,' a targeted therapy for gastric cancer with a novel mechanism of action, is expected to be launched in South Korea. According to industry sources, Astellas Korea has submitted an application for approval of Vyloy (zolbetuximab), a treatment for claudin 18.2 (CLDN18.2) positive, HER2-negative, unresectable, advanced, or recurrent gastric cancer. In March, it acquired the final approval in Japan. Vyloy is a monoclonal antibody targeting CLDN18.2, which is expressed in the stomach. Its mechanism of action involves binding to CLDN18.2, a protein expressed on the cancer cell membrane of gastric epithelial cells. The efficacy of Vyloy was proven through the Phase 3 SPOTLIGHT and GLOW clinical trials. The SPOTLIGHT trial, which enrolled 557 adult patients with metastatic gastric or gastroesophageal junction (GEJ) adenocarcinoma, compared the efficacy and safety of zolbetuximab plus mFOLFOX6 combination therapy (fluorouracil, leucovorin, and oxaliplatin) to placebo plus mFOLFOX6. In the clinical trial, zolbetuximab combination therapy recorded progression-free survival (PFS), the primary endpoint, of 10.6 months, 8.7 months longer than the comparator. Additional clinical results showed that zolbetuximab combination therapy had an overall survival (OS) of 18.2 months, which was improved by 2.7 months compared to the comparator. In the GLOW study, zolbetuximab combination therapy improved PFS and OS. The study compared zolbetuximab plus CAPOX therapy (capecitabine and oxaliplatin) to placebo plus CAPOX therapy. The clinical results demonstrated that zolbetuximab combination therapy had a PFS of 8.2 months, 1.4 months longer than the comparator. The OS of zolbetuximab combination therapy was 14.4 months, compared to 12.2 months for the comparator. The common side effects related to safety were nausea and vomiting. Meanwhile, Vyloy is in progress for the discussion with the U.S. FDA, Europe, and China approvals. In the United States, its approval was rejected once over manufacturing issues.
- Company
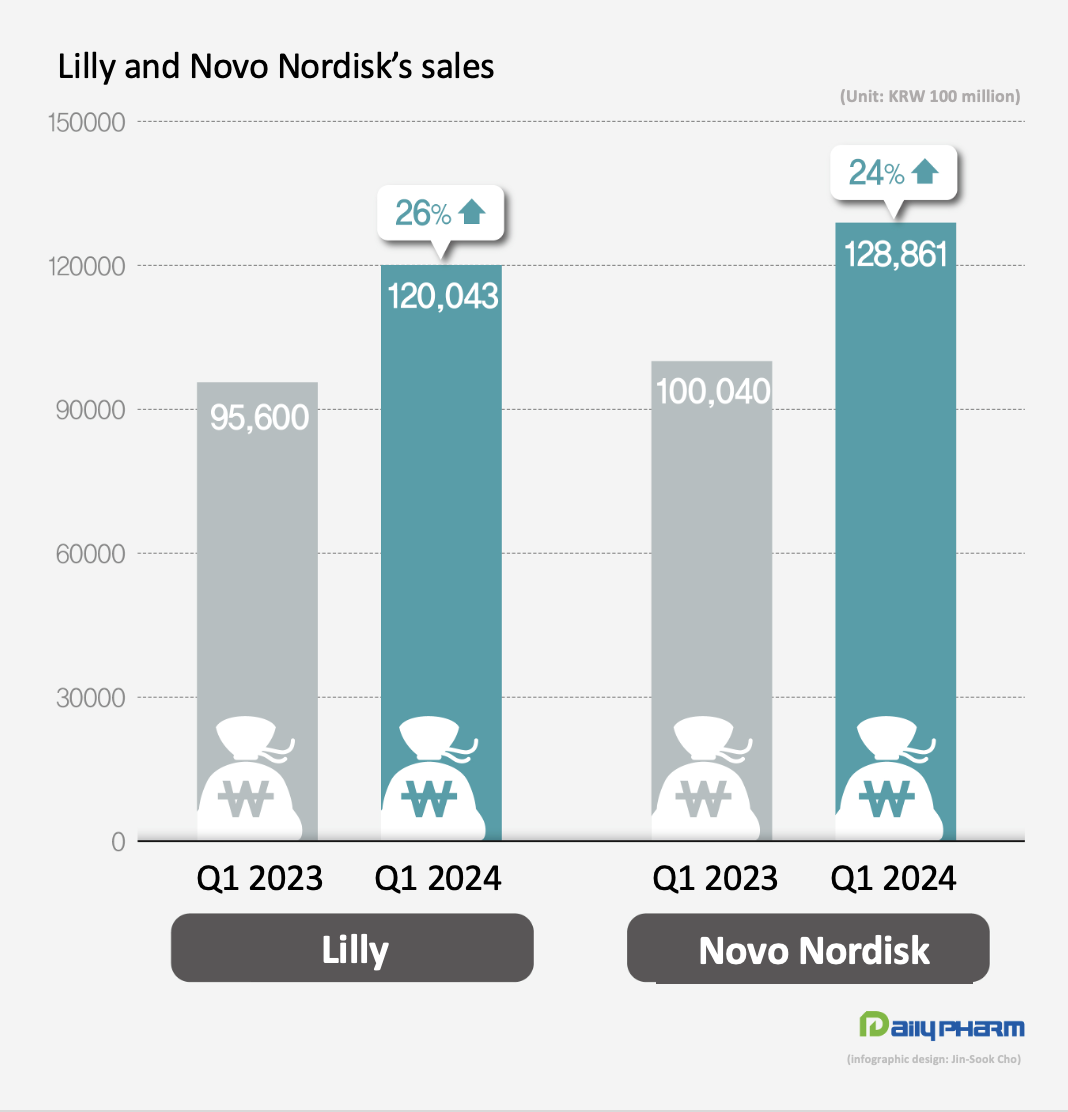
- Novo Nordisk and Lilly post strong performance
- by Son, Hyung-Min May 08, 2024 05:48am
- The performance of Novo Nordisk and Eli Lilly, which released blockbuster diabetes and obesity drugs, have improved greatly in Q1 this year. Novo Nordisk's GLP-1 class diabetes drugs together generated KRW 10 trillion in quarterly sales. Lilly's obesity drug Zepbound posted sales of KRW 700 billion in Q1 this year, reaching nearly 1 trillion won in only half a year since its launch. According to KoreaBio data on the 3rd, Novo Nordisk's first-quarter sales rose 24% YoY to KRW 12.886 trillion (DKK 65.349 billion). The exchange rate was based on the basic rate of exchange as of May 2. Novo Nordisk's diabetes medicines generated sales of KRW 9.8456 trillion, up 24% YoY. In particular, sales of GLP-1 diabetes medicines surged 32% YoY to KRW 6.8981 trillion. Novo Nordisk's GLP-1 class diabetes drugs include Ozempic, Rybelsus, and Victoza, which are semaglutide and liraglutide drugs. Sales of obesity treatments totaled at KRW 2.1759 trillion, up 42% from Q1 2022. Specifically, Wegovy’s sales rose 107% to KRW 1.849 trillion. As of February 2024, the global market share of Novo Nordisk's diabetes treatments was 34%, including 35% in North America, 30% in Europe and 32% in China. Its global share of obesity medicines amounts to 85%. Lilly sees strong sales of diabetes and obesity drugs Lilly also reported first-quarter sales of KRW 12.43 trillion (USD 8.77 billion), up 26% YoY, driven by new diabetes and obesity drugs. The increase was driven by the company’s new products. Sales of its new products launched in 2022 and thereafter jumped 495%, bringing in KRW 3.27 trillion in Q1, up from KRW 550 billion in Q1 last year. Mounjaro’s revenue increased 218% from KRW 780 billion in Q1 last year to KRW 2.48 trillion in Q1 this year, recording a revenue of KRW 2.08 trillion in the U.S. alone. Mounjaro is a type 2 diabetes treatment developed by Eli Lilly that has a dual effect on glucagon-like peptide-1 (GLP-1) and glucose-dependent insulinotropic polypeptide (GIP). By acting on GLP-1 and GIP, Mounjaro provides further blood glucose control. Mounjaro has also been shown to have dramatic weight loss benefits. Last year, Lilly also launched Zepbound, an obesity drug that contains the same substance as Mounjaro, after confirming its weight-loss benefits in clinical trials. The drug has become a global blockbuster only half a year after its launch, posting sales of KRW 71 billion in Q1. Demand for GLP-1 class drugs surge...company will address supply issue Novo Nordisk and Lilly are working to ensure smooth production of their GLP-1s, which are experiencing supply shortages. Currently, GLP-1 medicines are experiencing global stock-outs due to a surge in demand. Novo Nordisk plans to accelerate production by acquiring Catalent’s plant from Novo Holdings. Since Novo Holdings acquired Catalent last year, it has handed over 3 plants to its subsidiary, Novo Nordisk. Lilly has recently signed contract development and manufacturing organizations (CDMOs) with National Resilience in the U.S. and BSP Pharmaceuticals in Italy to increase its production capacity. To further increase its production facility in the mid-to long-term, Lilly recently acquired Nexus Pharmacutical's manufacturing facility in the U.S. and broke ground on a parenteral (injectable) manufacturing plant in Germany, both of which are expected to be operational by the end of 2025 and 2027, respectively.
- Policy
- MSD’s Cancidas will retain premium pricing for up to 5 yrs
- by Lee, Tak-Sun May 08, 2024 05:48am
- MSD's antifungal injection Cancidas’s premium pricing will be maintained for 5 years after generic entry. Originally, the price should have been adjusted to 53.55% of the highest price, but for various reasons, its price has remained at the level of 70% of the highest price. According to the industry sources on the 6th, Cancidas’s drug pricing premium was supposed to end on the 1st of this month, but the pricing premium was extended for another year according to the opinion of the Drug Reimbursement Evaluation Committee. As a result, the price of Cancidas 50mg will remain at KRW 215,977 and Cancidas 70mg at KRW 275,989. Sam Chung Dang Pharm’s Casfun Inj, the only generic version of Cancidas, also maintained its markup, and its price ceiling will remain at KRW 188,198 for Casfun Inj 50mg and at KRW 238,253 for Casfun Inj 70mg for another year. In principle, after the entry of generics, the price of the original drug drops to 70% of the highest price through ex officio adjustment, and after 1 year, it becomes the same price as its generic, set at 53.55% of the highest price. However, the price of Cancidas remains at the 70% level for 5 years. In the meantime, the principle of equal pricing for generic drugs has been broken. In June 2020, the price of Cancidas was adjusted to its current price following generic entry. The adjustment was temporary and set to last until May 2021. However, a year later, the number of generic companies remained below three, so the markup was maintained for 2 more years. Then, in April of last year, the 3-year premium pricing period passed, but the company wanted to extend the period for reasons such as ensuring a stable supply of the product, and DREC accepted the request and decided to maintain the premium pricing for one more year. This year, despite the expiration of the premium pricing period (3+1year), the premium pricing period was extended another year until May 2025 on the basis that if a drug manufacturer, contract manufacturer, distributor, or importer wants to extend the premium pricing period for reasons such as ensuring a stable supply of products, the period can be extended following DREC opinion. In January 2021, the Ministry of Health and Welfare reorganized the premium pricing system to allow up to 5 years of premium pricing through 1+2+2, including 1 year of markup upon initial registration, up to 2 years of premium pricing maintenance for drugs with 3 or fewer manufacturers, and 2 years of additional premium pricing granted after DREC deliberation when the pharmaceutical company wishes to extend the premium pricing. Cancidas’s premium pricing is being maintained for up to 5 years through the new system. Cancidas’s sales in Korea had been KRW 8 billion in 2022 (IQVIA). The drug is used for ▲ Empirical therapy for presumed fungal infections in febrile, neutropenic patients; ▲treatment of Candidemia and Other Candida Infections; ▲ treatment of esophageal candidiasis; and ▲ treatment of invasive aspergillosis. Sam Chung Dang Pharm was the only Korean pharmaceutical company to launch a generic version of Cancidas in May 2020. Since then, Sam Chung Dang Pharm ‘Casfun Inj ‘ has been the only generic version that entered the market.
- Company
- Prescriptions for cephalosporin antibiotics double in 3yrs
- by Chon, Seung-Hyun May 08, 2024 05:48am
- The popularity of antibiotics in the outpatient prescription market continues. The prescription market for cephalosporin-class antibiotics has nearly doubled in the past 3 years. This ‘heyday’ in the cephalosporin prescription market is continuing, fueled by the steady increase in flu and cold cases since the end of the pandemic. According to the market research institution UBIST, the outpatient prescription market for cephalosporin antibiotics was KRW 80.4 billion in Q1 last year, up 11.3% year-on-year. Compared to the KRW 64.1 billion it posted in Q1 2022, the market has expanded 24.4% in 2 years. Cephalosporin antibiotics, which are also called ‘Cepha antibiotics,’ are antibiotics used to manage a wide range of conditions including pneumonia, sore throat, tonsillitis, and bronchitis. The prescription market for cephalosporin antibiotics has undergone significant changes during the pandemic. The prescription market for cephalosporin antibiotics shrank by 29.3% from KRW 60.2 billion in Q1 2020 to KRW 42.6 billion in Q1 2021. In the early days of the COVID-19 pandemic, the prescription market for cephalosporin antibiotics shrank significantly, with the number of patients with infectious diseases such as flu and colds plummeting due to increased personal hygiene measures such as hand washing and mask-wearing. However, the demand for cephalosporin antibiotics surged with the explosive rise in the number of COVID-19 cases in late 2021. In Q1 2022, the prescription value of cephalosporin antibiotics began to rebound, reaching KRW 64.1 billion, up 50.6% YoY. Even after the end of the pandemic last year, the demand for cephalosporin antibiotics increased due to a steady number of confirmed daily COVID-19 cases and the rising number of flu and cold cases that arose after the lifted mask mandate. In Q1, the prescription market for cephalosporin antibiotics soared 88.8% in 3 years compared to Q1 2021. Cefaclor, the leading cephalosporin antibiotic, has recently grown significantly after the prescription market contracted in the early days of the COVID-19 outbreak. The outpatient prescription market for Cefaclor shrank by 23.2% from KRW 31.8 billion in Q1 2020 to KRW 24.4 billion in Q1 2021. However, from the end of 2021, the prescription market expanded at a rapid pace. In Q1, Cefaclor’s prescription size was KRW 40.6 billion, which is a 66.4% rise in 3 years from Q1 2021. Daewoong Bio was the dominant player in the Cefaclor market. Prescriptions of Daewoong Bio's Ceclor in Q1 was KRW 3.4 billion, up 20.1% YoY. Prescriptions of Ceclor have nearly doubled in 3 years from 1.8 billion won in Q1 2021. Prescription sales of Withus Pharamceutical’s Withus Cefaclor have increased sixfold from KRW 500 million in Q1 last year to KRW 2.8 billion in a single year. Other companies in the cephalosporin market, such as HLB Pharma, Mothers Pharmaceutical, Celltrion Pharm, Samsung Pharm, and Theragen Etex, generated over KRW 1 billion in prescriptions in Q1. Cephalosporin antibiotics have recently emerged as a new cash cow for pharmaceutical companies due to such a significant increase in demand. Many pharmaceutical companies had been considering withdrawing from the cephalosporin antibiotics business due to the declining market rising raw material costs and low drug prices. However, with the recent increase in demand, pharmaceutical companies are rather facing an urgent need to secure more supply. Daewoong Bios, which has been leading the market for cephalosporin antibiotics, announced last year that it would invest KRW 50 billion to build a new plant dedicated to the production of cephalosporin antibiotics. The current cephalosporin antibiotics plant in Seongnam, South Korea, will cease operation after this year due to outdated facilities. Once the new plant is completed, the company plans to further expand its antibiotic business. Separation of factories has become mandatory for the production of cephalosporin antibiotics since 2011. Since separate manufacturing facilities are required for its production, not many companies own dedicated facilities. One pharmaceutical company official said, "Cephalosporin antibiotics are a representative drug supplied through active consignment agreement. With the recent surge in demand, we have been struggling to secure sufficient supplies from our consignees.”
- Policy
- Will oral anemia drugs for dialysis patients be reimbursed?
- by Lee, Tak-Sun May 07, 2024 05:50am
- AstraZeneca recently withdrew Evrenzo, an oral treatment for anemia in chronic kidney disease, from the Korean market Industry interest in rising on whether oral treatments used to treat anemia in dialysis patients will be covered by health insurance in Korea. The drugs have long been approved in Korea since 2020, but one of the products recently withdrew its license. The other two have yet to be reviewed for reimbursement, raising interest. According to industry sources on the 3rd, AstraZeneca dropped the license for its Evrenzo Tab (Roxadustat, AstraZeneca), which is used to treat symptomatic anemia in patients with chronic kidney disease, on April 30. The market withdrawal comes more than 3 years after its approval in July 2021. The drug was praised for improving the convenience of anemia treatment for patients with kidney disease who previously had to depend on injections by switching to oral medication. Historically, patients with chronic kidney disease have been given injections to stimulate red blood cell production called erythropoietin (EPO) or erythropoiesis-stimulating agents (ESA) to treat anemia, a common complication of chronic kidney disease. However, Evrenzo was developed as an oral formulation that works by activating hypoxia-inducible factor (HIF), a gene involved in erythropoiesis that regulates gene expression and raised expectations on improving the convenience of treatment for the patients. After Evrenzo, JW Pharmaceuticals received approval for Enaroy in November 2022, and Mitsubishi Tanabe Pharma Korea received approval for Vadanem in March last year. Both are reportedly working to list their respective drugs for reimbursement. Under these circumstances, Evrenzo's market withdrawal has raised concerns that the authorities may have a negative stance toward the reimbursement of oral anemia drugs for chronic kidney disease. The reimbursement of these drugs also rose as an issue at the National Assembly audit held in October last year. At the time, Democratic Party lawmaker Sun-woo Kang questioned the denial of reimbursement, to which the HIRA responded that the drugs had not been reviewed for reimbursement in key reference countries and lacked clinical evidence. In a written QA, the HIRA said, "Although the drugs are oral formulations unlike existing ESA preparations that are injectables, additional advantages such as the convenience of dosing in hemodialysis patients, which is the target patient group for approval, are unclear. Although it may be potentially useful in some patient groups such as cancer patients for whom ESA administration increases the risk of tumor progression and recurrence and patients with adverse reactions or poor response to high-dose ESA therapy, it lacks direct clinical evidence of effectiveness and safety in these patient groups.” HIRA added that it would review any additional evidence of their therapeutic benefit in future reapplications or supplemental data submitted, as well as the results of overseas assessments, but was not deliberated by the Drug Reimbursement Evaluation Committee on reimbursement adequacy thereafter. However, as Vadanem received US FDA approval in March and reapplied for reimbursement, it remains of interest whether HIRA will start a reimbursement review for the drug soon.
- Company
- Another petition for reimb listing of TNBC drug 'Trodelvy'
- by Eo, Yun-Ho May 07, 2024 05:50am
- Gilead Science Korea’s Trodelvy (Sacituzumab govitecan). There is now yet another petition requesting reimbursement listing of the new breast cancer drug 'Trodelvy'. There was already a previous posting with 50,000 votes in January of last year. It appears that another petition was posted because it has been three months without any updates. A petitioner, who introduced themselves on the national petition platform on May 2nd as a stage 4 triple-negative breast cancer (TNBC) patient, pleaded for support in need of Trodelvy reimbursement, stating, "The tumor is growing too fast. I earnestly request the community to participate to extend the treatment option to me as a stage 4 patient." There has not been any updates regarding Gilead Science Korea’s Trodelvy (Sacituzumab govitecan), a treatment of triple-negative breast cancer (TNBC), being considered for the Drug Reimbursement Evaluation Committee (DREC) after the clearance of the Cancer Disease Review Committee of the Health Insurance Review and Assessment Service (HIRA) in November of last year. Trodelvy is the first Antibody-Drug Conjugate (ADC) composed of a monoclonal antibody that binds to the cell surface antigen Trop-2 and a DNA TOP1 inhibitor payload called 'SN-38,' which destroys cancer cells. It received approval as a treatment for adult patients with unresectable locally advanced or metastatic triple-negative breast cancer (mTNBC) who have received two or more prior systemic therapies, including at least one for metastatic disease, from the Ministry of Food and Drug Safety (MFDS) in May last year. Although Trodelvy is the only drug to have demonstrated improvements in overall survival period among those used for second-line treatment or more of metastatic TNBC, it is stalled at getting the reimbursement listing due to cost-effectiveness evaluation. One factor that can anticipate reimbursement coverage is recognition for innovativeness, as seen in the case of Enhertu (trastuzumab deruxtecan), which was listed in April and received a benefit from the government based on its ICER value. Whether Trodelvy will be considered for the upcoming DREC review is to be watched. Meanwhile, Trodelvy demonstrated its clinical effectiveness through the Phase 3 ASCENT clinical study. It has shown a 49% reduction in the risk of death and a 57% improvement in progression-free survival (PFS) in adult patients with unresectable locally advanced or metastatic triple-negative breast cancer (mTNBC) who have received two or more prior systemic therapies, including at least one for metastatic disease, compared to patients who received single-agent chemotherapy (TPC, Treatment of Physician’s Choice). These effects were observed regardless of the presence of brain metastasis.
- Company
- Hyperparathyroidism drug Orkedia may be reimb in Q3
- by Eo, Yun-Ho May 07, 2024 05:50am
- ‘Orkedia,’ a drug for secondary hyperparathyroidism, has entered its final gateway to insurance reimbursement in Korea. According to industry sources, Kyowa Kirin Korea recently entered into pricing negotiations with the National Health Insurance Service for Orkedia (evocalcet), a new drug for secondary hyperparathyroidism that can develop in patients with chronic kidney disease on dialysis. If the company and the authorities reach an agreement within the 60-day negotiation deadline, the drug could be launched with reimbursement in Q3 this year. The drug passed the Health Insurance Review and Assessment Service's Drug Reimbursement Evaluation Committee last month after receiving domestic approval in November last year. Orkedia is a calcimimetic agent and acts on calcium receptors on the surface of parathyroid cells to inhibit excessive parathyroid hormone (PTH) secretion, thereby reducing serum parathyroid hormone levels Secondary hyperparathyroidism is a condition in which excessive secretion of parathyroid hormone persists due to hypocalcemia caused by decreased kidney function, leading to complications such as bone disease. Orkedia is expected to help expand treatment options for patients who are taking existing medications for secondary hyperparathyroidism. Meanwhile, Kyowa Kirin acquired the rights to cooperative research, develop, market, and manufacture Orkedia in Japan and some parts of Asia with Mitsubishi Tanabe in March 2008 and has since been developing the drug.