- LOGIN
- MemberShip
- 2026-04-07 22:25:29
- Policy
- Atopic dermatitis drug Adtralza will be reimb in May
- by Lee, Jeong-Hwan Apr 22, 2024 05:45am
- Leo Pharma Korea’s atopic dermatitis treatment, Adtralza (tralokinumab), will be reimbursed from May 1 in Korea. The reimbursement standards for osteoporosis drugs will also be improved, and patients who show some improvement in bone mineral density will be able to continue receiving reimbursement. As such, JW Pharmaceuticals' iron injection Ferinject will also be reimbursed. On the 19th, the Ministry of Health and Welfare issued an administrative notice of the 'Partial Amendment to the Details on the Standards and Methods for Applying Medical Benefits (Pharmaceuticals)' that contained the contents above. It will collect opinions until the 23rd for its implementation on the 1st of next month. The new drugs covered by the reimbursement through the administrative notice are Adtralza, a drug for atopic dermatitis, and Ferinject, an iron injection. Adtralza is a humanized monoclonal antibody that binds to interleukin-13. It controls atopic dermatitis by inhibiting downstream interleukin receptor signaling that triggers the inflammatory response. Adtralza is reimbursable for the treatment of chronic severe atopic dermatitis in adults (18 years and older) and adolescent patients (12 years to 17 years of age) whose condition has remained symptomatic for at least 3 years. Patients must have received at least 4 weeks of topical therapy (at least moderate-potency corticosteroids or calcineurin inhibitors) as a first-line treatment but were unable to adequately control their condition, followed by at least 3 months of systemic immunosuppressive agents (cyclosporine or methotrexate) that did not result in a 50% or greater reduction in Eczema Area and Severity Index (EASI) or were unable to continue their use due to side effects. Patients with an EASI of 23 or greater prior to Adtralza use are recognized as eligible for reimbursement. However, the patient should be able to confirm a history of topical and systemic immunosuppressive therapy within 6 months before initiating treatment with Adtralza for reimbursement. After 14 weeks of Adtralza treatment, if a patient’s EASI is reduced by 75% or greater at the 16th-week assessment, the patient may continue to use Adtralza with reimbursement for 6 additional months. Patients will continue to be evaluated every 6 months thereafter and may maintain their reimbursed status if they maintain their initial results. Inpatient prescriptions are required in principle, and a maximum of 4 weeks’ supply is allowed to be prescribed per single prescription upon discharge for outpatient use. However, a maximum of 8 to 12 weeks' supply is allowed for patients with stable disease activity and no adverse events after 24 weeks from the initial dose, and switching between Adtralza and the JAK inhibitor Dupixent is not allowed. The reimbursement standards for osteoporosis treatments have been extended to allow borderline patients who have reached their T-score treatment goal to continue to be eligible for reimbursement. As a result of clinicians and academic society input, patients who have been recognized to be eligible for reimbursement for a T-score of -2.5 or less (T-score≤-2.5) when measuring bone mineral density using Dual-Energy X-ray Absorptiometry (DXA) of the central bone, will be eligible for an additional year of reimbursement if their T-score improves during treatment to a T-score greater than -2.5 but less than or equal to -2.0. If the T-score remains greater than -2.5 but less than or equal to -2.0 thereafter, reimbursement will be allowed for another year. In the additionally approved period, patients will be allowed to switch between raloxifene, bazedoxifene, bisphosphonate, and denosumab. The patients may also switch to zoledronic acid injections. Lastly, the MOHW added the iron injection Ferinject to the reimbursement standards. However, it excluded Ferinject’s use in patients with chronic kidney disease on dialysis from the reimbursement standards.
- Policy
- KMPG ‘We ask for the prompt reimbursement of Xpovio’
- by Kim, Jung-Ju Apr 22, 2024 05:45am
- Patients’ demand for the reimbursement listing of ‘Xpovio 20mg (selinexor),’ a new drug for multiple myeloma that was developed by the Chinese pharmaceutical company Antengene, is growing. The patients are claiming that they are becoming ‘medical poor’ due to the delay in the reimbursement of the only option available in the area. Xpovio’s reimbursement agenda has been stuck at the reimbursement stage for more than 3 years since it was approved in Korea and is being delayed without a viable alternative. The Korea Multiple Myeloma Patient Group issued a statement on the 19th and strongly urged the authorities to consider the patients’ situations. The KMPG chairman said, "We strongly urge the Drug Reimbursement Evaluation Committee, which will meet in May, to expedite the reimbursement of Xpovio, the only light of hope remaining for the fading flickers of life for our multiple myeloma patients.” Multiple myeloma is a blood cancer caused by the abnormal differentiation and proliferation of plasma cells, a type of white blood cells responsible for the immune system, in the bone marrow. Xpovio Tab 20mg, which was developed to treat the disease, was approved by the MFDS and introduced to Korea on July 29, 2021. On June 14, 2023, the drug’s reimbursement application passed HIRA’s Cancer Disease Review Committee but suffered a setback right before its reimbursements when it was deemed non-reimbursable by DREC in November of the same year. Min-Hwan Baek, Chairman of KMPG, said, “Xpovio is currently being reimbursed in not only the A8 countries but A9 countries, being granted reimbursement in the United States, Germany, Canada, the United Kingdom, and Australia. But in Korea, patients are dying due to the drug’s non-reimbursement. I lay awake night after night as chairman of KMPG, worrying about the patients who are dying needless deaths due to lack of other options than the non-reimbursed, unaffordable Xpovio.” "It has come to the point where many patients who turned medical poor due to Xpovio’s non-coverage are giving up on treatment," said KMPG, urging for the prompt reimbursement of Xpovio.
- Company
- AstraZeneca focuses on improving competitiveness of NMOSD
- by Eo, Yun-Ho Apr 22, 2024 05:45am
- (From upper left) Soliris, Ultomiris. AstraZeneca focuses on improving the competitiveness of neuromyelitis optica spectrum disorder (NMOSD) treatments. The company aims to target the market as its new blockbuster drugs, 'Soliris (eculizumab),' which is used to treat paroxysmal nocturnal hemoglobinuria (PNH), and 'Ultomiris (ravulizumab),' which is a treatment for neuromyelitis optica spectrum disorder (NMOSD), expand indications. In 2021, Soliris received approval for an expanded indication for NMOSD in South Korea. After a prolonged discussion about its listing for insurance reimbursement, Soliris can now be reimbursed for insurance under expanded criteria starting this month (April). The specifics of reimbursement criteria include adult NMOSD patients aged 18 and above who test positive for anti-aquaporin-4 (AQP-4) antibodies, with at least two symptom relapses within the past year or at least three relapses within the past two years (including at least one relapse in the past year). Soliris can be prescribed when 'Mabther (Rituximab)' or ' Enspryng (satralizumab)' treatment cannot be continued due to relapses or side effects following administration. In terms of Ultomiris, after a failed attempt last September, it secured U.S. FDA approval for an additional indication last month. Ultomiris has a higher adherence rate than Soliris. While Soliris requires injections every two weeks, Ultomiris is administered every eight weeks, resulting in longer intervals between doses. The expansion of Ultomiris indications is based on the global Phase 3 CHAMPION-NMOSD study results. During the 73-week treatment period (median value), none of the patients who received Ultomiris relapsed, resulting in a reported 98.6% reduction in relapse risk compared to the control group. On average, NMOSD is a chronic autoimmune disorder that affects the central nervous system. It is estimated to impact approximately 200,000 people worldwide and is more commonly found in women aged 30 to 40. Initially recognized as a variant of multiple sclerosis, a chronic inflammatory demyelinating disease of the central nervous system, NMOSD was distinguished from multiple sclerosis upon the discovery of disease-specific antibodies (aquaporin-4 [AQP4]-IgG) in opticospinal myelitis patients' serum. According to the Korean Society of Neuroimmunology, as of 2017, about 3.56 individuals per 100,000 are estimated to be affected by NMOSD. It is estimated that there are over 1,000 NMOSD patients in South Korea. The average onset age of NMOSD in South Korea is reported to be 43 years old, with a prevalence that is 4.7 times higher in females than in males.
- Policy
- Will the 37th new drug in Korea receive approval?
- by Lee, Hye-Kyung Apr 22, 2024 05:45am
- Jeil Pharmaceutical. The approval for the 37th new drug in Korea is approaching. According to industry sources on the 22th, the Ministry of Food and Drug Safety (MFDS) completed the safety and efficacy assessment of the three zastaprazan-containing products from Onconic Therapeutic, Jeil Healthscience, and Jeil Pharmaceutical. The completion of the safety and efficacy assessments by MFDS indicates that approval is nearing. Jeil Pharmaceutical simultaneously applied for new drug approvals from three affiliated companies, including Jeil Healthscience, which manufactures over-the-counter drugs, and its subsidiary, Onconic Therapeutic, which specializes in new drug R&D. These three products contain the active ingredient of zastaprazan. Upon completion of the safety and efficacy assessment by MFDS, approval for these three products is anticipated. In June last year, Onconic Therapeutic submitted the New Drug Application (NDA) to the MFDS for domestic launch. The NDA was based on the Phase 3 clinical trial, which evaluated 298 patients with gastroesophageal reflux disease (GERD) in 28 medical facilities in South Korea. Notably, there has not been a single case of MFDS-designated new drug in South Korea following the 36th new drug in Korea, 'Envlo,' in November 2022. Therefore, approval of zastaprazan would lead to approval of new drugs in South Korea. Zastaprazan is a new drug that belongs to the class of novel potassium-competitive acid blocker (P-CAB). P-CAB is expected to replace the proton pump inhibitors (PPI) in the anti-peptic ulcer drug market, including the treatment for gastroesophageal reflux disease (GERD). P-CAB class products in Korea include HK inno.N’s 'K-CAB,' the 30th new drug in Korea, and Daewoong Pharmaceutical’s 'Fexuclue,' the 35th new drug in Korea. According to medical market research firm UBIST, K-CAB recorded prescription sales of KRW 30.4 billion in 2019. Sales grew rapidly to generate KRW 77.1 billion in 2020, KRW 110.7 billion in 2021, and KRW 132.1 billion in 2022. Since its launch in July 2022, Fexuclue has recorded cumulative prescription sales of KRW 61 billion up to November 2023, changing the market paradigm for gastroesophageal reflux disease.
- Company
- Will Padcev be deemed cost-effective and receive DREC review
- by Eo, Yun-Ho Apr 22, 2024 05:45am
- Whether the reimbursement discussions for Padcev, a new antibody-drug conjugate for bladder cancer, will make progress is gaining attention. According to industry sources, Astellas Pharma Korea’s Padcev (enfortumab vedotin), which passed the Health Insurance Review and Assessment Service's Cancer Disease Review Committee in February, has completed pharmacoeconomic evaluations and is being reviewed by the subcommittee. Whether the agenda will pass the subcommittee review and be presented to the Drug Reimbursement Evaluation Committee remains to be seen. The drug is recommended as Category 1 in the National Comprehensive Cancer Network (NCCN) guidelines. It is a new treatment option for urothelial cancer patients whose cancer has progressed or recurred even after receiving treatment with immunotherapy drugs and platinum-based chemotherapy, for whom no standard of care has existed as of yet. The drug was approved in March in Korea for the treatment of adult patients with locally advanced or metastatic urothelial cancer who have received prior treatment with PD-1 or PD-L1 inhibitors and platinum-based chemotherapy. The key to Padcev’s successful reimbursement is in receiving a beneficial ICER threshold from the government in recognition of its innovativeness, like Enhertu (trastuzumab deruxtecan), which was listed in April. Padcev is also known to meet the government's preferential treatment standards for innovative new drugs. The standards are ▲ there is no substitute or therapeutically equivalent product or treatment ▲ demonstrated clinically meaningful improvement, such as a significant extension in survival ▲ the new drug has been approved by the Ministry of Food and Drug Safety under Article 35(4)(2) of the Pharmaceutical Affairs Act (designation of priority review) and were approved through the fast-track (GIFT) or received a breakthrough therapy designation (BTD) by the US FDA or a priority review (PRIME) by the European Union’s EMA. Padcev demonstrated its efficacy through the EV-301 study, an open-label, Phase III trial that was conducted on 608 patients with locally advanced or metastatic urothelial cancer who have previously been treated with platinum-based chemotherapy and PD-1 or PD-L1 inhibitors. Study results showed that Padcev reduced the risk of death by 30% compared to chemotherapy. The median overall survival (OS) of the Padcev group was 12.9 months, demonstrating a significant improvement in survival compared to chemotherapy's 9.0 months. In addition, Padcev significantly improved progression-free survival (PFS) with a 39% reduction in disease progression or death risk, with the median progression-free survival (PFS) for Padcev being 5.6 months and 3.7 months for the control group. Mi-so Kim, Professor of Oncology at Seoul National University Hospital, explained, “Urothelial cancer progresses quickly and requires continuous treatment, but patients were left to use chemotherapy for later line therapies after using immunotherapy in the second-line due to the lack of a standard later line treatment option. Padcev can be used in patients whose cancer had progressed or relapsed after chemotherapy (first-line therapy) and immunotherapy (second-line therapy or first-line maintenance therapy), and will open a new paradigm in the treatment of patients with locally advanced or metastatic urothelial cancer.”
- Policy
- Reimb expansions of Keytruda have failed the 4th CDRC
- by Lee, Tak-Sun Apr 19, 2024 06:24am
- MSD Korea’s cancer immunotherapy drug Keytruda’s reimbursement expansion item did not cross the Cancer Disease Review Committee (CDRC) threshold for the fourth time following its expansion in June last year. The analysis is that this attempt is particularly significant as all 15 indications were reviewed by the CDRC, and MSD’s financial contribution proposal was discussed for the first time. During the 3rd CDRC in 2024, held on April 17th, the reimbursement expansion of MSD Korea’s cancer immunotherapy drug 'Keytruda (pembrolizumab)' was reviewed but failed in securing reimbursement expansion. The CDRC announced during the meeting, it decided to postpone setting reimbursement criteria for 15 indications, including endometrial cancer. It was decided that the setting of reimbursement criteria would be reconsidered upon the additional submission of financial contribution proposal. All 15 indications included in the reimbursement expansion application were reviewed. In June of last year, MSD Korea submitted Keytruda’s reimbursement expansion application to the Health Insurance Review and Assessment Service (HIRA) for 13 indications with high unmet needs in medical fields in Korea. MSD's application includes 13 indications: ▲ Early-stage triple-negative breast cancer ▲ Metastatic or recurrent triple-negative breast cancer ▲ Metastatic or recurrent head and neck cancer ▲ Advanced or metastatic esophageal cancer ▲ Adjuvant therapy after renal cell carcinoma surgery ▲ Non-invasive bladder cancer ▲ Persistent, recurrent, or metastatic cervical cancer ▲ Advanced endometrial cancer ▲ Metastatic endometrial cancer with MSI-H or dMMR ▲ Metastatic rectal cancer with MSI-H or dMMR that cannot be removed with surgery ▲ Metastatic small intestine cancer with MSI-H or dMMR ▲ Metastatic ovarian cancer with MSI-H or dMMR ▲ Metastatic pancreatic cancer with MSI-H or dMMR. The company recently added two additional indications. It was reported that MSD applied for the reimbursement expansion of two indications last December: gastric cancer with MSI-H and bile duct cancer with MSI-H. Three indications were assessed at the CDRC meeting in October last year, where Keytruda's reimbursement expansion was first discussed. Then, 4 indications were assessed in November of the same year, and 6 indications were assessed in January of this year. The CDRC said at each review, “We will prioritize reviewing the medical validity and clinical necessity of multiple indications for reimbursement expansion. Upon receiving the pharmaceutical companies' financial contribution proposal towards proven indications, we have decided to analyze its impacts to establish reimbursement criteria.” Furthermore, during this CDRC review, 15 indications, including the previously discussed 13 and the two additional indications applied for in December, were all brought to the review table. This indicates that the CDRC prioritized reviewing the medical validity and clinical necessity of multiple indications for reimbursement expansion. In other words, the initial assessment of indication has been completed. The committee will discuss establishing final reimbursement criteria for the next step after reviewing the pharmaceutical companies' financial contribution proposal. It was reported that the financial contribution proposal submitted by MSD Korea was added as an additional agenda item just two days before the CDRC review. This suggests that pharmaceutical companies deliberated on their financial contribution proposals until the last minute. However, it has been reported that members of the CDRC were not satisfied with the financial contribution proposal submitted by MSD. Keytruda's reimbursement expansion in the future depends on how much MSD is willing to compromise in their financial proposal. “Keytruda is reimbursed for 7 indications, and its domestic sales reach KRW 400 billion,” an industry expert explained. He added, “If the pharmaceutical company does not submit a new financial contribution proposal, it may not be possible to reimburse all 15 indications at once.”
- Company
- Global companies are affected by doctors’ resignation
- by Moon, sung-ho Apr 19, 2024 06:24am
- Two months have passed since the government announced its plan to raise the medical school quota by '2,000' students. Pharmaceutical companies are facing more marketing challenges amin the ongoing dispute between the medical community and the government. In particular, global pharmacies that launched new drugs after overcoming reimbursement hurdles face challenges as new patients decline. In addition to local pharmaceutical companies, global companies are also impacted by doctors’ strikes. According to industry sources on April 6th, the Ministry of Health and Welfare (MOHW) approved the new listing and reimbursement expansion of 18 drugs until April. Due to the new listing and reimbursement expansion, approximately KRW 434.2 billion from the annual health insurance budget will be allocated toward these items this year. Last year, approximately KRW 381.5 was spent in 32 reimbursed drugs (24 newly listed and 8 reimbursement expanded drugs), whereas this year’s expenditure has already surpassed last year’s even before the end of the first half of the year. Breaking down the pricing by drugs, there are items that have gathered attention in the clinical field in South Korea. For example, the list includes Tagrisso (osimertinib) and Leclaza (lazertinib, Yuhan), which competed for reimbursement expansion for the first-line treatment of non-small cell lung cancer (NSCLC). According to estimates from the MOHW, Tagrisso and Leclaza will receive KRW 204.6 billion and KRW 137.7 billion, respectively, in finances this year. Both drugs were approved for reimbursement expansion in January. Out of the total finance, the expected increase due to reimbursement expansion would be KRW 92 billion and KRW 88.1 billion for these two drugs, and 2634 patients are expected to benefit from coverage of the first-line treatment. For these two drugs, an increase in financial spending by KRW 180 billion is expected. Among the drugs newly listed up to April, the antibody-drug conjugate (ADC) 'Enhertu (trastuzumab deruxtecan, Daiichi Sankyo Korea, AstraZeneca)' accounted for the largest part of the financial spending. The MOHW has calculated the expected financial spending for Enhertu, which is reimbursed for breast cancer and gastric cancer, to be KRW 134.7 billion. Among the reimbursement expanded and newly listed drugs, this is the single item with the highest financial spending. In addition to anticancer drugs, drugs for chronic diseases, such as the 'chronic kidney disease' drug Kerendia (finerenone, Bayer), are among the drugs gaining attention. As it can be prescribed in endocrinology and nephrology departments within university hospitals, it is considered a notable new drug from a global pharmaceutical company. This is supported by the government’s estimate that 29,350 patients can benefit from the reimbursement listing. Since the announcement on February 6th of an increase in medical school quotas, there have been mass resignations extending beyond resident physicians to professors at nationwide university hospitals, resulting in continued disruptions in medical services. The pharmaceutical industry, which must actively engage in sales and marketing following successful new drug approvals or reimbursement expansion, is in a challenging environment. As a result, they remain cautiousness about holding medical symposiums after new drug launches. So far, challenges have primarily been experienced by suppliers of surgical materials, pharmaceuticals, medical devices, wholesalers, and domestic pharmaceutical companies. However, this situation could also impact global big pharma with new drugs. AstraZeneca and Yuhan have been proactively preparing through prior processes, such as clearing the Drug Committees (DC) at hospitals for Tagrisso and Leclaza in line with the reimbursement expansion in January. This effort was aimed at gaining an advantage in the non-small cell lung cancer market through medical institution coding work. As a result, by the end of March, AstraZeneca cleared the DC review of Tagrisso in 94 hospitals, and Yuhan cleared the DC review of Leclaza in 103 hospitals. However, university hospitals face difficulties treating new patients with prolonged medical service disruptions. As a result, the expansion of the field for these two treatments, Tagrisso and Leclaza, is unfolding differently than anticipated. Enhertu, which became reimbursed in April, may have faced similar circumstances in a clinical setting specializing in oncology. Daiichi Sankyo and AstraZeneca, in joint sales and marketing, have obtained approval from the DC in 60 hospitals for Enhertu. “Due to structural challenges, our focus is currently on existing patients, but we must see new patients,” a professor of Hematology-Oncology at Hospital A commented. He added, “Despite efforts to prioritize patient care as much as possible, there has been a decline in the number of new patient treatments, which may result in reduced drug usage.” “Because leukemia patients are still being accepted through the emergency room, new patient intake has not stopped. The problem is that it is now becoming a chronic situation,” another professor of Hematology at a different university hospital stated. He added, “Even if the range of treatments expands, it will be challenging for pharmaceutical companies due to the structural constraints in the clinical setting, making it difficult to utilize them effectively.” Global big pharma headquarters are now increasingly concerned about the consequences of the increase in medical school quotas for the clinical field in South Korea. At the same time, there is a sense of urgency to evaluate the extent of revenue decline in the first quarter of this year due to the controversy surrounding the expansion of medical school quotas. "The headquarters closely monitors the long-term treatment disruption issue in the domestic clinical field. This issue is a major topic in every video conference," an anonymous executive from the Korean subsidiary of a global pharmaceutical company stated. He added, "As we are in a situation where the introduction of new drugs or their reimbursement is at stake, we are proactively evaluating and communicating the potential consequences of this situation."
- Policy
- Impurity detected in single-agent entacapones
- by Kang, Shin-Kook Apr 19, 2024 06:24am
- t has been confirmed that impurities have been detected in entacapone, a single-agent drug used to treat symptoms of Parkinson's disease. According to medical and pharmaceutical organizations on the 18th, the Ministry of Food and Drug Safety began reviewing safety measures, including setting necessary temporary permissible limits based on the submitted test and inspection results, following a company report that nitrosamine impurities (NDEA, N-nitrosodiethylamine) were detected in its entacapone-containing drugs. In other words, if there is a medical need for the drug or there is a concern about supply interruptions (shortage), the government plans to apply a temporary permissible limits for the drugs’ distributions for a certain period of time. For this, the MFDS is collecting opinions from doctors to prepare for the possibility of unstable supply and demand of entacapone-containing drugs if recall measures or suspension of production and import measures are applied due to necessity until impurities are reduced. This is a preliminary investigation into the medical necessity of entacapone, the impact on patient care in the event of a supply interruption (shortage) of entacapone single-agent drugs, the current status of substitute drugs, and the establishment of temporary permissible limits. The entacapone single tablets currently approved in Korea include Comtan Tab 200 mg, Myungin Entacapone Tab 200 mg, and Entapone Tab 200 mg.
- Policy
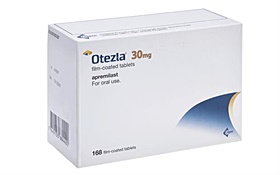
- Four Otezla generics were authorized simultaneously in Korea
- by Lee, Hye-Kyung Apr 19, 2024 06:24am
- Amgen Four generic versions of Amgen's oral psoriasis drug Otezla (Apremilast) have been approved simultaneously in Korea. With the resolution of Otezla’s 'use patent' issue, which had served as an obstacle to the launch of generics, domestic pharmaceutical companies that have previously succeeded in avoiding the drug’s composition patent seem to have applied for approval of their generic versions at once. On the 17th, the Ministry of Food and Drug Safety approved four apremilast-based formulations, including Dong-A ST's ‘Otelia Tab,’ Daewoong Pharmaceutical's ‘Apsola Tab,’ Dongkoo Biopharm’s ‘Otemila Tab,’ and Chong Kun Dang’s ‘Otebell Tab.’ Otezla, the original apremilast formulation of these generics, was the first oral psoriasis treatment to be approved by the MFDS in November 2017, but the company voluntarily withdrew the drug’s license in June 2022 after failing to cross the reimbursement threshold. In the process, domestic pharmaceutical companies began filing patent suits to launch their generic versions of Otezla. Otezla has 3 registered patents, including a use patent (10-0997001) and two formulation patents (10-2035362 and 10-2232154). In the case of the 2 formulation patents, 7 companies, including Daewoong Pharmaceutical, Dong-A ST, DongKoo Bio&Pharma, Huons, Chong Kun Dang, Mothers Pharm, and Cosmax, filed to confirm the passive scope of rights in May 2021 and November 2022 and succeeded in avoiding the patents. However, the companies still had to overcome the use of patents to launch their generic versions. Otezla’s use patent was set to expire on March 18, 2028. The companies must invalidate the use patent as well to launch their generic versions, and on August 24 last year, the court decided to ‘partially reject and dismiss’ the use patent’s invalidation trial. Despite withdrawing from the Korean market, Amgen appeared to be vigorously defending its patent rights, leading to criticism that it was blocking the entry of generics that were preparing to launch in Korea. And with no further news of the domestic pharmaceutical companies filing appeals to evade the use of patents since August last year, the news of the 4 generic Otezla approvals drew attention to their background. A representative from a relevant Korean pharmaceutical company said, "We have been continuously making an agreement with Amgen regarding the use of the patent. Amgen withdrew its license for Otezla in 2022 and had no intention of launching it in the future, so there was no reason to prevent generics from entering the market." "Amgen said that it didn't want to continue litigating a product that they had no intention of launching in the country. They agreed that the patent would not be a barrier to generic entry in the future, which is why we went through the process." Currently, the legal status of Otezla's use patent, (+)-2-[1-(3-ethoxy-4-methoxyphenyl)-2-methylsulfonylethyl]-4-acetylaminoisoindoline-1,3-dione, and methods of synthesis and compositions thereof" is indicated as “revoked," according to Korea’s Intellectual Property Trial and Appeal Board bulletin. An industry official said, "The Otezla generics have not been jointly developed by the companies, but seem to have been approved at the same time upon the resolution of the patent issue. We plan to apply for reimbursement immediately upon approval and start drug price negotiations."
- Company
- Adcetris’s IPS requirement for reimb may be removed
- by Eo, Yun-Ho Apr 19, 2024 06:23am
- A green light has been given for the removal of the IPS score requirement for Adcetris, which had been a long-standing wish of the patients. According to industry sources, Takeda Pharmaceutical Korea’s reimbursement expansion application to remove the International Prognostic Score (IPS) score criteria for prescribing its Adcetris (brentuximab vedotin) in combination with chemotherapy (AVD, adriamycin+bleomycin+dacarbazine) in Hodgkin's lymphoma passed the Health Insurance Review and Assessment Service's Cancer Disease Review Committee review on the 17th. Adcetris )’ was granted reimbursement as a first-line treatment for Hodgkin’s lymphoma in 2021, but a restriction of "IPS score of 4" was applied, meaning that the drug was only reimbursable for patients with severe disease. As a result, patients with an IPS score of 2-3 have been prescribed an existing chemotherapy combination, ABVD (adriamycin+bleomycin+vinblastine+dacarbazine), instead of Adcetris. Even patients with stage IV disease were unable to benefit from Adcetris and its better efficacy if they had a low IPS score. However, when analyzing the results of the 1,344 patients in the ECHELON-1 trial, Adcetris demonstrated efficacy in high-risk patients who were Stage IV, male, younger, and had an IPS score of 4-7. The National Comprehensive Cancer Network (NCCN) guidelines recommend Adcetris+AVD for the treatment of patients with stage III and IV disease, and the European guidelines allow Stage IV patients to be treated with Adcetris regardless of the IPS score. Therefore, the medical community has been pushing for the removal of the IPS score requirement from Adcetris’s reimbursement criteria, and expectations are rising with its passage of the CDDC review this time. However, it remains to be seen if Adcetris can clear the remaining hurdles, including the Drug Reimbursement Evaluation Committee and drug price negotiations, and remove the IPS score requirement from its reimbursement criteria. In the Phase III ECHELON-1 trial, Adcetris demonstrated superior clinical efficacy compared to ABVD in previously untreated patients with stage III or IV classical Hodgkin lymphoma. Its three-year progression-free survival (PFS) analysis showed that the Adcetris combination therapy (PFS rate 83.1%) reduced the risk of disease progression by 30%, compared with ABVD (PFS rate 76%).