- LOGIN
- MemberShip
- 2026-04-09 03:05:54
- Company
- SK Bioscience & Hilleman Laboratories develop Ebola vaccine
- by Kim, Jin-Gu Nov 23, 2023 05:37am
- SK Bioscience announced on the 22nd that it signed a memorandum of understanding (MOU) with Hilleman Laboratories, an international non-profit research institute, for the joint development of a second-generation Zaire Ebola virus vaccine. Hilleman Laboratories was established in 2009 as an equal joint venture between MSD and Wellcome Trust, a medical research support foundation. SK Bioscience had signed an agreement with Hilleman Laboratories in October to jointly research and develop a new vaccine and platform. Through the new MOU, the parties specified the subject vaccine and the parties’ role in vaccine development. MSD’s Ervebo and Johnson & Johnson’s Zabdeno are the only Ebola virus vaccines available as of now. Hilleman Laboratories had worked with MSD to improve process efficiency and storage stability to optimize product productivity and thermostability and recently succeeded in discovering a 2nd generation Ebola vaccine candidate. Under the new MOU, SK Bioscience will work with Hilleman Laboratories to develop a 2nd generation Ervevo, which has an improved vaccine production process, production efficiency, and thermostability. When developed, SK Bioscience and Hilleman Laboratories plan to secure the price competitiveness of the vaccine and supply the vaccine to low- and middle-income countries. If commercialized, the global supply of a 2nd generation Ervevo will be manufactured on consignment by SK Bioscience at its vaccine facility, Andong L House. For this, the company signed a CDMO agreement with MSD for its 2nd generation Ebola vaccine in May this year. Dr Raman Rao, CEO of Hilleman Laboratories said, "Developing more affordable and accessible vaccines is core to our mission at Hilleman Laboratories. Through our partnership with SK Biosciences, we will commit ourselves to create positive outcomes for global public health, especially in affected regions of Central and Western Africa.” Jaeyong Ahn, CEO of SK Bioscience said, "Developing a vaccine to prevent viruses causing diseases with a high fatality rate, such as Ebola-Zaire, is essential to protect humanity. We plan to successfully complete the project and expand our cooperation with global companies and institutions and grow into a global company."
- Company
- Leukemia drug Xospata’s reimb limit may be resolved
- by Eo, Yun-Ho Nov 23, 2023 05:37am
- Astellas Korea is working to resolve the issue of the reimbursed number of cycles approved for its acute myeloid leukemia treatment ‘Xospata.’ Dailpharm’s coverage showed that the agenda on expanding reimbursement standards for Astellas Korea’s Xospata, a drug for patients with relapsed or refractory acute myeloid leukemia (AML) with a FLT3 mutation, has recently passed the Health Insurance Review and Assessment Service’s Cancer Reimbursement Evaluation Committee review. The company applied for the reimbursement extension in November, and the progress has been made 6 months after the agenda passed the Cancer Disease Deliberation Committee in May. However, since Xospata waived the pharmacoeconomic evaluation process, the drug would also have to undergo drug pricing negotiations with the National Health Insurance Service for its reimbursement extensions. Therefore, whether its reimbursement can be extended to cover more cycles will depend on when the agenda is submitted to the Drug Reimbursement Evaluation Committee for review and whether it passes the review. The drug is indicated as monotherapy for the treatment of patients with relapsed or refractory acute myeloid leukemia (AML) with a FLT3 mutation (FLT3mut+). However, only patients who are eligible for allogeneic hematopoietic stem cell transplantation can receive reimbursement, for up to 4 cycles. Other than the financial issues, there are no specific reasons to limit the number of administration cycles for Xospata. In the ADMIRAL trial, Xospata was used without limiting the treatment period, and the NCCN guidelines also issued a ‘Category 1’ recommendation for the drug without restricting its treatment period. The current best option to cure AML patients is hematopoietic stem cell transplantation, but this is accompanied by a high risk of recurrence, and transplantation is not an option for the large number of elderly AML patients that exist. Therefore, there is no suitable treatment alternative other than Xospata available for patients who cannot undergo hematopoietic stem cell transplantations, and these patients are still using the chemotherapy that was developed over 40 years ago due to ineligibility for reimbursement of Xospata. Xospata targets both types of FLT3 mutations, FLT3-ITD and FLT3-TKD, and may be self-administered at home as a single oral tablet once daily without frequent hospital visits. Also, Xostapa has demonstrated improved safety and efficacy compared with existing chemotherapy.
- Policy
- Enspryng and Jemperli will be reimbursed through RSA
- by Lee, Tak-Sun Nov 22, 2023 05:30am
- The new Neuromyelitis optica spectrum disorder (NMOSD) drug Enspryng Prefilled Syringe Inj (satralizumab, Roche) and new PD-1 inhibitor immune-oncology drug ‘Jemperli Inj (dostarlimab, GSK) will be reimbursed in Korea from December 1st this year. Also, the insurance price ceiling for Xeljanz Tab 5mg, for which reimbursement has been extended to cover ankylosing spondylitis, will be reduced. According to industry sources on the 21st, Enspryng will be reimbursed through the expenditure cap type RSA, and Jemperli through the expenditure cap type and refund type RSA from December 1st. The price ceilings set for the drugs are KRW 7,823,456/syringe for Enspryng and KRW 3,868,840 for Jemperli. Enspryng is reimbursed for the treatment of f neuromyelitis optica spectrum disorders (NMOSD) in adult patients over 18 years of age who are anti-aquaporin-4 IgG (AQP4-IgG) seropositive and satisfy all of the following conditions: ▲2 or more symptom relapses in the last 2 years (including ≥1 relapse in last year, were viable for reimbursed use of rituximab injections and had used the drug for over 3 months but experienced relapse or cannot continue rituximab due to side effects, and ▲has a 6.5 or lower Extended Disability Status Scale (EDSS) score upon administration of satralizumab. Jemperli is indicated for the treatment of adult patients with dMMR recurrent or advanced endometrial cancer. The drug is the 3rd PD-1 inhibitor immune-oncology drug introduced to the field following Opdivo (nivolumab, Ono·BMS) and Keytruda (pembrolizumab, MSD). Meanwhile, with its reimbursement standards extended to severe cover active ankylosing spondylitis Xeljanz’s insurance price ceiling will be adjusted from KRW 11,085 to KRW 10,996. In addition, the prices of Taltz Prefilled Syringe Inj and Taltz Autoinjector Inj which also received reimbursement extensions to active ankylosing spondylitis will be adjusted from KRW 802,800 to KRW 798,786. Also, Cosentyx Sensoready Pen’s insurance ceiling price will be reduced from KRW 633,084 to KRW 629,308, and Cosentyx Unoready Pen 300mg/2mL from KRW 1,202,860 to KRW 1,195,643.
- Company
- Reimb of COVID-19 drugs necessary with eased crisis level
- by Eo, Yun-Ho Nov 22, 2023 05:30am
- Whether COVID-19 treatments will be able to be listed for reimbursement before Korea's COVID-19 containment measures are lifted remains under attention. Two COVID-19 treatments, Gilead Science Korea’s ‘Veklury (remdesivir)’ and Pfizer Korea’s ‘Paxlovid (nirmatrelvir+ritonavir),’ are currently under review by the Health Insurance Review and Assessment Service after the companies filed applications for their reimbursement. The government had previously announced that it would work to reimburse COVID-19 treatments in H2 2024 as part of its COVID-19 risk level management roadmap. This means that the COVID-19 treatments that are being supplied free of charge will be converted and become purchased products like general medicine. When Phase 2 of the COVID-19 risk level management roadmap, which will further ease the disease control and prevention system, is implemented next month, COVID-19 will then be regarded as a Grade 4 infectious disease like influenza. Not dangerous because it’s an endemic?... Improving awareness remains a key issue However, the road to reimbursement for these treatments is not easy. Not only are the prices expensive, but the sharp decline in the public’s sense of crisis is also playing a significant role. COVID-19 has shifted from a pandemic into an endemic, but it is still producing variants that threaten high-risk patients such as those with underlying diseases and the elderly. The 'Excess mortality during COVID-19’ submitted by the Korea Disease Control and Prevention Agency in October to the office of Democratic Party of Korea Rep. Choun Sook Jung, a member of the National Assembly Health and Welfare Committee, that cites data from Statistics Korea, the number of excess deaths continued to be on the rise until recently due to the aftermath of COVID-19. According to the report, ‘excess deaths’ from COVID-19 had exceeded 65,000. ‘Excess deaths’ refer to additional deaths beyond the number of deaths normally expected to occur at a particular period. This figure indicates how many more deaths occurred compared to when COVID-19 had not occurred. Eun-Joo Choo, Professor of Infectious Diseases at Soonchunhyang University Bucheon Hospital, said, “COVID-19 is still a disease that requires active treatment and management, and many patients are still being hospitalized for the disease. Its treatments need to be listed with reimbursement immediately upon the closure of the free support system next year to ensure treatment access for the patients and to secure essential drugs.” Both drugs are necessary...due to different roles Although both were approved as COVID-19 treatments, Veklury and Paxlovid differ in terms of indication, and formulation, among others. Veklury has been verified to have slowed down disease progression in many COVID-19 patients, reducing the time to recovery and thereby reducing the burden on Korea’s medical system. It was approved to treat COVID-19 in adults and pediatric patients with mild-to-moderate COVID-19 who are at high risk of progressing to severe disease. It can be administered regardless of the severity of the patient’s disease and aids in quicker recovery by offering a lower risk of disease progression with earlier treatment. Also, it is a treatment whose efficacy and safety have been proven through several clinical trials and RWE and is the only COVID-19 treatment that can be used on patients with severe COVID-19. In particular, it has little drug-drug interactions (DDI), and comes in an injectable formulation that can be administered to patients who have difficulty taking oral medications. Paxlovid, which comes in an easy-to-take oral formulation, is approved to treat mild-to-moderate COVID-19 in adults who are at risk of progressing to severe COVID-19, which includes hospitalization and death. Paxlovid needs to be taken within five days of developing symptoms and is administered twice daily for 5 days. In the EPIC-HR trial that was conducted on non-hospitalized symptomatic adult subjects with COVID-19 who are at high risk of progressing to severe disease and have no prior history of vaccination, Paxlovid significantly reduced the proportion of people with COVID-19-related hospitalization or death from any cause through 28 days of follow-up by 86% compared to placebo among patients treated within 5 days of symptom onset and who did not receive COVID-19 therapeutic monoclonal antibody treatment.
- Policy
- Will PE exemption drugs be reevaluated based on RWD?
- by Lee, Tak-Sun Nov 22, 2023 05:30am
- A public hearing on measures for performance-based reimbursement management of drugs that was held on the afternoon of the 20th at the main auditorium of the Catholic University of Korea’s Institute of Biomedical Industry Will ultra-high-priced drugs or drugs exempted from receiving pharmacoeconomic evaluations be reevaluated based on actual patient administration data after being listed for reimbursement? Starting with Kymriah first last year, as drugs were applied a performance-based refund type RSA for reimbursement, an interpretation that it would now be possible to reevaluate the price of high-priced drugs or drugs that are waived PE evaluations based on real-world data has been proposed. The ‘Public hearing on measures for performance-based reimbursement management of drugs’ that was held on the 20th at the main auditorium of the Catholic University of Korea’s Institute of Biomedical Industry is likely to serve as the starting point for re-evaluation using RWD on high-priced drugs or PE exemption drugs. RWD refers to actual patient clinical data collected post-reimbursement, such as health insurance claims data, hospital medical records, surveys, and post-marketing drug survey data. Real-world evidence (RWE) is actual clinical grounds derived from RWD. The public hearing was prepared to share the results of RWD/RWE research conducted by the Korea Cancer Study Group and HIRA for the past 5 years and to collect various opinions on the use of RWD/RWE-based drug reimbursement management guidelines. On the day, Chi-Hoon Maeng, Professor of Kyung Hee University’s School of Medicine, announced the results of his study that evaluated the efficacy and safety of paclitaxel-ramucirumab combination therapy through RWD data in patients with advanced gastric cancer. In addition, Jung-hoon Ahn, Professor of Health Convergence at Ewha Womans University Graduate School, prepared a pharmacoeconomic evaluation result based on the RWE data of the paclitaxel-ramucirumab combination therapy based on Professor Maeng's interpretation of the results. The two results have significance in that the post-marketing evaluation was conducted based on RWE. Ji-Hye Byun, associate researcher at HIRA followed the two presentations with an announcement of the RWE guidelines for reimbursement management through drug performance evaluation, which contained a whole RWE utilization plan from Step 1:reimbursement plan to Step 4: reevaluations. Steps 1 and 2 contain measures identical to what has been applied to Kymriah and Zolgensma – the current performance-based refund type RSA. The plan is to reimburse drugs based on the pharmaceutical company’s RWE evaluation, and then when the medical institutions submit RWD data, the authorities evaluate the drug’s performance and receive a refund from the company for patients who fail treatment. Steps 3 and 4 evolve further from current plans. HIRA plans to work with external experts to analyze RWD data for reimbursement reevaluations. If implemented, high-cost drugs that have submitted PE data but have a highly uncertain Incremental Cost-Effective Ratio (ICER) or drugs for which PE evaluation data were not or could be submitted can be reevaluated after reimbursement with RWD. The ‘PE evaluation data waiver system,’ or ‘PE exemption system’ had been applied in 2015 to benefit rare disease drugs or anticancer drugs with no alternatives. This year, subjects were expanded to benefit drugs for pediatric patients that demonstrate an improvement in quality of life. Managing reimbursement through RWD allows for post-marketing evaluation of drugs that have unclear cost-effectiveness rather than prior evaluations, thereby offering an advantage in being able to shorten the reimbursement listing process, and strengthening access to high-priced drugs for severe and rare diseases. Also, post-marketing evaluations can help improve the financial soundness of health insurance and offer transparent management of reimbursement as decisions are made based on real-world data. On the other hand, RWD collection adds a burden to the various other parties involved, such as the burden of data collection posed on the doctors and the risk of refunding the drug cost of patients who fail treatment on the companies’ part. However, the measures presented at the public hearing are not specific guidelines and will require serious opinion-gathering processes to be implemented as an actual policy. Dong-Churl Suh, Director of the Pharmaceutical Policy Research Institute, who participated at the hearing as a panelist, pointed out, "RWD data analysis presents rise to transparency issues. The results can vary depending on the data source, so many considerations would need to be made for its implementation, including on how to secure data transparency.” However, the government was more for the use of RWD as a way to reduce uncertainties in financial management regarding high-priced drugs and PE exemption drugs. Mi-Young Yoo, Director-General of the Pharmaceutical Benefits at HIRA, said, “Since the introduction of the positive listing system in 2006, many drugs have been listed for reimbursement through various systems including the PE exemption system to enable better patient access. So this is now the time a post-marketing measure needs to be implemented. Although social consensus should be made on its need, such a management system can be a way to ensure an appropriate level of financial soundness within limited insurance.” Chang-Hyun Oh, Director of Pharmaceutical Benefits at MOHW, added, “Addressing the uncertainty that follows the listing of high-priced drugs reimbursed with PE exemption is a pending task for all. As a tool, I expect RWD to become a good way to cover the uncertainties that remain in the area.”
- Company
- Mitsubishi’s Lou Gehrig's disease drug Radicut's sale soars
- by Nho, Byung Chul Nov 21, 2023 05:48am
- The latecomer injection formulations are taking over the Lou Gehrig's disease treatment market that had been occupied by injectables in the past. According to the industry’s pharmaceutical distribution performance data, Mitsubishi Tanabe Pharma Korea's Radicut Inj (edaravone) has occupied 39% of the market share in the related treatment market 8 years after receiving marketing authorization, narrowing the gap with YooYoung Pharm's Yooritek (riluzole, 41%). Radicut, which was approved in 2015 by the Ministry of Food and Drug Safety, had posted sales of KRW 500 million, KRW 1.3 billion, KRW 1.1 billion, KRW 1.6 billion, KRW 2.2 billion, and KRW 0.9 billion in H1 of 2018, 2019, 2020, 2021, 2022, and 2023, respectively. It has outperformed Sanofi Aventis’s Rilutek since 2020. During the same period, Sanofi Aventis’s sales of Rilutek were KRW 1.2 billion, KRW 2.6 billion, KRW 0.1 billion, KRW 0.3 billion, KRW 0.7 billion, and KRW 0.4 billion, respectively. After posting record-high sales of KRW 2.6 billion in 2019, Rilutek recorded sales worth KRW 0.4 billion in H1 this year, drawing a downward-sloping performance curve. Until earlier this year, Yooyoung Pharm was the only domestic company to supply Lou Gehrig's disease treatment. Therefore the fact that it had recorded record-breaking performance of KRW 2.4 billion in 2020 and 2021 with Yooritek against global pharmaceutical companies Sanofi and Mitsubishi is one aspect to note. Clockwise from the top: Yooyoung Pharm’s Yooritek, Mitsubishi Tanabe Pharma Korea’s Radicut, and Sanofi’s Rilutek Radicut is indicated to slow down the progression of functional impairment in patients with amyotrophic lateral sclerosis (ALS). Rilutek and Yooritek are indicated to extend life or the time to mechanical ventilation for patients with amyotrophic lateral sclerosis (ALS). Lou Gehrig's disease is a rare condition that cannot be cured until now. Its annual drug cost is around KRW 3.3 million, which is relatively cheap compared to the ultra-expensive rare and anticancer drugs that cost hundreds of millions of won. Another interesting aspect is that Radicut, which contains edaravone, was initially prescribed as a treatment for cerebral infarction, and then added an indication for Lou Gehrig's disease. Radicut’s efficacy and effect in suppressing the progression of functional impairment have been confirmed through clinical trials conducted on Lou Gehrig patients in Japan. Lou Gehrig's disease is a representative rare and intractable neurological disease that selectively degenerates only motor neurons, causing irreversible paralysis of limbs and respiratory muscles. Most patients affected with Lou Gehrig's disease die within 2 to 5 years after onset without mechanical ventilation. It is estimated that there are approximately 350,000 patients worldwide and 2,000 to 3,000 in Korea. It occurs in approximately 1 to 2 people per 100,000 per year, and its incidence increases from the late 50s. Its incidence rate is 1.4 to 2.5 times higher in men than in women. Meanwhile, SK Chemicals also succeeded in listing Teglutik (200ml), a suspension formulation containing riluzole, that it imported from the Italian pharmaceutical company Italfarmaco at an insurance price of KRW 134,970 earlier this year. The ceiling insurance price is KRW 133,970 per vial for a 15-day use. Therefore, the price is the same as the price of existing tablet formulations (KRW 4,499 per day) in terms of daily dosage. As it comes in a suspension formulation, it is administered directly using oral syringes, and through percutaneous endoscopic gastrostomy (PEG) tubes for those who have difficulty taking it directly. Patients with Lou Gehrig's disease often experience dysphagia (difficulty swallowing) an early symptom due to weakened tongue and throat muscles. Although tablets are considered more convenient for intake take than injections in general, for Lou Gehrig's injections are more convenient than tablets for the reason above. This is why Mitsubishi Tanabe Pharma Korea’s injectable Radicut is rapidly expanding its market share. Therefore, patients with dysphagia had difficulty taking the table formulation of riluzole, which was administered twice a day.
- Company
- IL inhibitor reimb as 1st-line Tx for ankylosing spondylitis
- by Eo, Yun-Ho Nov 21, 2023 05:48am
- Interleukin inhibitors may soon be available as first-line treatment for ankylosing spondylitis in Korea. On the 20th, the Ministry of Health and Welfare announced an amendment notice that contains a plan to expand insurance reimbursement for the 2 IL-17A inhibitors approved in Korea - Novartis Korea's 'Cosentyx (secukinumab)' and Lilly Korea's 'Taltz (ixekizumab)’ – as treatments for ankylosing spondylitis. With the notification, the reimbursement standard extension for the two drugs, which had remained pending for over a year, is expected to be implemented within the year. Novartis and Lilly both submitted an application for reimbursement extension for their respective drugs based on the ‘ASAS-EULAR axSpA treatment recommendations 2022’ in July last year. Although the actual process for their reimbursement extension was completed earlier this year, time was wasted due to a delay in final approval on the Ministry of Health and Welfare’s part. Interleukin inhibitors are available with reimbursement as a first-line treatment for ankylosing spondylitis in over 30 countries. In Korea, both drugs are currently only approved for use with reimbursement in the second-line treatment setting, after patients are prescribed TNF-α inhibitors. The grade of recommendation and level of evidence for IL-17A inhibitors were raised to Grade A and 1a in the 2022 update of the ASAS-EULAR recommendations, to the same level as TNF-α inhibitors. The guidelines referred to how both agents are currently being used as first-line biological agents to treat ankylosing spondylitis and officially confirmed their equal therapeutic positions as treatments for ankylosing spondylitis. Moreover, the use of TNF-α inhibitors is associated with tuberculosis. So patients had to undergo prior therapy to treat latent tuberculosis before using TNF-α inhibitors. However, despite the anti-TB treatment, the occurrence of tuberculosis due to the use of TNF-α inhibitors is still reported in several studies. Therefore, another treatment option was dire for patients who are at high risk of infection such as tuberculosis, or who have comorbidities, such as heart failure, and have complications in being prescribed TNF-α inhibitors. The number of ankylosing spondylitis patients has rapidly increased by nearly 50% over the past 10 years (35,592 in 2013, 52,616 in 2022). It is a disease that mainly occurs at a young age, and domestic patients go through about 40 months of wait until they receive an accurate diagnosis. In addition, patients can receive a disability level up to Level 2 at the highest, rendering it a serious disease and eligible for exemption from military service.
- Policy
- Global bioleaders gather in Seoul for World Bio Summit
- by Lee, Jeong-Hwan Nov 21, 2023 05:48am
- Vaccine and bio leaders from around the world will gather in Seoul, Korea to discuss response strategies for the next global pandemic that may come after COVID-19. On the 19th, the Ministry of Health and Welfare and the World Health Organization (WHO) announced they will hold the '2023 World Bio Summit' from the 20th to the 21st at the Conrad Seoul Hotel in Yeongdeungpo-gu, Seoul, under the theme of 'Strengthening capacities for pandemic response: Preparation of Workforce, Business, and Civil Society.’ The World Bio Summit is an annual global event where the Korean government invites leaders in the bio field from various countries, companies, and international organizations around the world to discuss measures and seek international cooperation. The event celebrates its 2nd year this year. Last year, 'The Seoul Declaration' was announced, which declares the need to strengthen measures for response to infectious diseases such as vaccines and treatments, and guarantee equal access to such responses. This year, international organizations that played a pivotal role in responding to the COVID-19 pandemic in the vaccine and bio fields, namely, the Asian Development Bank (ADB), Coalition for Epidemic Preparedness Innovation (CEPI), the International Vaccine Institute (IVI), and the Foundation for Innovative New Diagnostics (FIND) and the Korea Disease Control and Prevention Agency are contributing as partner organizations and cooperating in organizing detailed programs, garnering high anticipation for more professional presentations and discussions. Also, Kenya's Minister of Health, India's Minister of Livestock and Dairy Affairs, and the Acting Director-General of the EU Health Emergency Preparedness and Response Authority will visit Korea to attend the event. After the opening ceremony, the regulatory session will be held. The session will focus on discussing ways to bolster vaccine, pharmaceutical, and diagnostics manufacturing and regulatory oversight to strengthen capacities for pandemic response under the theme of 'Regulatory Oversight to Build Capacities for Pandemic Response', with the participation of the Asian Development Bank (ADB) as a partner organization. The session is expected to provide implications for low- and middle-income countries striving to institute a pharmaceutical regulatory system or to make major strides toward an advanced regulatory system, and serve as an opportunity to explore the development of safe and effective local vaccines. In the therapeutics session that is held the next day, under the theme of 'Accelerating Pandemic Therapeutics Development and Manufacturing to Respond to Future Pandemics', the Korea Disease Control and Prevention Agency will serve as a partner organization to share global strategies for therapeutics development and find ways to maximize each country's capacities through regional cooperation at each stage of therapeutics research and development. In the diagnostics session, under the theme of ‘Accelerating pandemic diagnostics development and manufacturing’, the Foundation for Innovative New Diagnostics (FIND) will partake as a partner to discuss future diagnostic market trends, ways to strengthen future diagnostic capacities, and collaborative approaches in South Korea to enhance diagnostic accessibility in low- and middle-income countries. Lastly, at the vaccine session, the Coalition for Epidemic Preparedness Innovations (CEPI) will introduce CEPI’s 100-day mission, CEPI’s collaboration with International Vaccine Institute(IVI), collaboration with industry and engagement with academia, and the achievements of the KDCA’s National Institute of Health Center for Vaccine Research (KVRC) under the theme, ‘Strengthening Global Preparedness Through CEPI’s 100-Day Mission.’ Various other side events such as luncheons, seminars, and business networking will also be held during the event. In addition to workshops and seminars sponsored and partnered by CEPI and ADB each, a ‘Global Business Lounge’ will be set up to foster and facilitate cooperation between international organizations, global companies, overseas health ministries, and domestic bio companies In addition, the 2nd Vice-Minister of Health and Welfare Minsoo Park will have a meeting with WHO Assistant Director-General of External Relations and Governance Catharina Boehme and resume the Korea-WHO policy dialogue, which had been on hold since it was first held in 2019 and discuss various measures to strengthen cooperation between the Korean government and WHO. Also, the MOHW will have a meeting with Saudi Arabia’s Vice Minister of Health for Planning and Development Abdulaziz bin Hamad bin Saleh Al-Ramaih, Ukraine’s Deputy Minister of Health Ihor Kuzin, Laos’ Vice Minister of Health Phayvanh Keopaseuth, and Chair of CEPI Board Jane Halton to proceed with discussions on key issues of mutual interest of both sides.
- Company
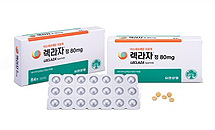
- Yuhan Leclaza clinical phase 3 cost exceeds 100 billion
- by Chon, Seung-Hyun Nov 20, 2023 05:50am
- The development cost invested by Yuhan's new anti-cancer drug Leclaza in the Phase 3 clinical trial has exceeded 100 billion won. Three years after the start of the clinical trial, the cost of development is also increasing. Yuhan was approved as a primary treatment based on the efficacy and safety of Leclaza confirmed in the clinical trial, and the salary expansion was also in the countdown. According to the Financial Supervisory Service on the 16th, as of the end of the third quarter, the total development cost accounting for as intangible assets by Yuhan Corporation was 119 billion won. It increased by 3.9 billion won in three months from 1151 billion won at the end of the first half of the year. In 2019, the Financial Supervisory Service set a standard that accounting assets can only be processed if there is a technical feasibility for R&D tasks such as new drugs. The Financial Supervisory Service proposed the asset-enabled phase of R&D costs, with phase 3 clinical trials for new drugs and phase 1 clinical approval for biosimilars. Generics can be assetized after the biometric test plan is approved. The intangible assets reflected by Yuhan in its development expenses are the largest proportion of Lexha. At the end of the third quarter, Leclaza development cost of intangible assets was 101.1 billion won. It means that more than 100 billion won has been invested in aleclaza clinical phase 3 expenses. Leclaza is a treatment for non-small cell lung cancer that was approved as the 31st new drug developed in Korea in January 2021. Patients with locallyprogressive or metaplastic non-small cell lung cancer who are resistant to T790M after administration of the first and 2nd generation epithelial growth factor (EGFR) tyrosinase suppressant (TKI) are subject to administration. It interferes with the transmission of signals involved in the growth of lung cancer cells and acts as a mechanism to inhibit the proliferation and growth of lung cancer cells. Leclaza recognized 32.6 billion won in development costs as intangible assets for the first time in the fourth quarter of 2020. As the clinical phase 3 trial started in earnest, the development cost was reflected as an intangible asset. Leclaza development cost of intangible assets increased to 61.4 billion won at the end of 2021 and rose to 88 billion won, an additional 26.6 billion won last year. This year, the development cost of Leclaza’s intangible assets increased by a total of 13.3 billion won. 5 billion won and 4.7 billion won in clinical phase 3 expenses were invested in the first and second quarters, respectively. In the third quarter, 3.4 billion won was added, exceeding 100 billion won for the first time. Leclaza is challenging the primary treatment health insurance benefits. In June, Lexar received additional permission as a primary treatment, two years and five months after receiving domestic approval. Reclaza identified statistically significant progression-free survival (PFS) improvements in a multi-country clinical phase 3 trial. A Phase III trial (LASER301) conducted on 393 active EGFR mutant-positive topical advanced or metaplastic lung cancers that had never been treated demonstrated superior safety and effectiveness over conventional therapies. The clinical results were unveiled at the Asian General Conference of the European Society of Oncology in Singapore last year. Yuhan has recently applied to health authorities to apply Leclaza primary treatment. Yuhan plans to provide patients with free drugs for clinical trials for primary therapeutic purposes through the Expanded Access Program (EAP). The price of Leclaza’s monthly medicine is about 6 million won. According to IQVIA, a pharmaceutical research institute, Leclaza’s sales in the first half of last year were 10.3 billion won, up 49.5% from the same period last year. Sales in the first quarter were 5.1 billion won, up 57.4% from the previous year, and in the second quarter, it recorded 5.2 billion won, up 42.5% from the previous year. Cumulative sales since the release of Leclaza have been 30.2 billion won. Cumulative sales exceeded 30 billion won within two years of domestic launch.
- Company
- ‘1+3 will also impact Korea’s patent linkage system'
- by Kim, Jin-Gu Nov 20, 2023 05:50am
- The so-called ‘1+3 restriction on bioequivalence testing’ system is expected to impact the Drug Approval-Patent Linkage System as well. If applied, the 1+3 system is expected to eradicate the practice of multiple generic companies jointly filing patent challenges and improve the practicality of the first generic exclusivity right (first generic exclusivity). Also, the ‘Bill for the Amendment to the Drug Patent Term Extension System’ that is pending at the National Assembly is expected to have a positive effect on generic companies that challenge the original drug’s patent. However, there is also a possibility that this bill will not be passed due to the nearing end of the 21st National Assembly session. "1+3 restriction will improve the effect of first generic exclusivity system… but be disadvantageous for generic companies" Myung Hee Lee, Senior Researcher at the Korea Institute of Intellectual Property presented the prospects mentioned above at the ‘2023 Medical Intellectual Property Policy Forum’ that was held at the Korea Intellectual Property Service Center on the 16th. During her presentation, Lee pointed to 2 changes that will significantly affect the patent linkage system in the future. One is the Bioequivalence Testing 1+3 Restriction System, and the other is the Drug Patent Term Extension System.’ The 1+3 system limits the participating generic makers to 3 consignees per consignor when conducting joint biological equivalence tests or clinical tests. This means that the number of times the clinical data owned by the pharmaceutical company that directly conducted the trial can be shared is limited to 3. Regarding this, Lee predicted that there would be a change in the existing practice of multiple pharmaceutical companies challenging one patent at the same time. At the same time, it was predicted that the effectiveness of the generic exclusivity system would improve. Lee added, “We are currently analyzing the impact of the implementation of the systems. Because the system was not applied to existing applications at the time, the impact of the new system will begin to be felt in earnest from 2022.” “In the past, many companies challenged patents at the same time. In this process, the criticism was that the original purpose of the first generic exclusivity system had faded somewhat. With more than 20 pharmaceutical companies receiving exclusivity at the same time, the significance of the ‘exclusivity’ offered by the system had faded somewhat.” Lee added, “Simultaneous filing of patent challenges will also decrease. We are also observing situations where consignees who have not been granted data from the consignor companies that have directly conducted the bioequivalence tests are unable to join in the early release of generics. This will lead to many small pharmaceutical companies giving up development of generic drugs.” In the discussion session that followed the presentation, In-Bum Kim, an expert consultant at Kimg & Chang added, “From the generic companies’ part, it is now time to seriously consider selecting and focusing on a specific subject. If the companies had all challenged a single patent together, it is now the time for them to each select and pursue a field they can do well at.” "Patent term extension system is beneficial for generic companies…but will it be able to pass NA remains a question" On the other hand, the amendment to the drug patent term extension system was expected to have a positive effect on generic companies. The amendment, which is currently pending at the National Assembly, was presented to limit cases of excessive patent term extensions by original companies. The drug patent term extension system extends the patent term of original companies by the time the company spent on clinical trials or approval, by up to 5 years. However, criticism arose that the system rather works in favor of the original companies in Korea than in the US and Europe. The amendment limits the 'effective patent term’ to 14 years from the time of application for term extension to its expiration, and allows the company to only apply for term extension of one patent among the many that are registered for a single drug. Lee said, “The amendment will be detrimental to original companies and beneficial to generic companies. It is expected to serve in favor of generic companies on the other side of the 1+3 system.” However, whether it will pass the National Assembly review remains in question. To be enacted, the bill must be passed within this year’s session ahead of the general election in April next year. The panels who participated in the discussion predicted that the possibility of passing the amendment in the 21st National Assembly was low. Professor Cha-ho Jung from Sungkyunkwan University Law School who chaired the panel discussions, said, “If it was a bill with no objections, it might have passed; but due to strong opposition from original companies, it will not be easy for the bill to pass NA. I believe the chances are low."