- LOGIN
- MemberShip
- 2026-04-09 01:29:33
- Company
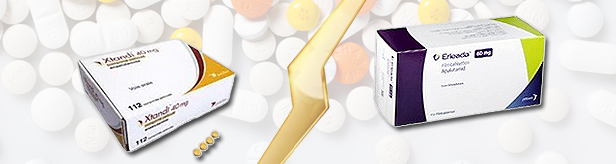
- Erleada chases leader Xtandi in prostate cancer mkt
- by Kim, Jin-Gu Dec 06, 2023 06:00am
- Pic of Astella Astellas Pharm Korea ‘Xtandi (enzalutamide)’ took the lead among oral androgen receptor targeted agents (ARTAs) used to treat prostate cancer. In comparison to declining sales of Janssen's ‘Zytiga (Abiraterone),’ sales of Janssen's other prostate cancer drug ‘Erleada (Apalutamide)’ has been rapidly increasing since being released just 6 months ago, chasing Xtandi in the market. Xtandi's Q3 sales KRW 11.7 billion…top among ARTAs for prostate cancer The ARTA-type prostate cancer treatment, Xtandi, posted KRW 11.7 billion in sales in Q3. According to IQVIA, a pharmaceutical industry research institute, this represents a 53% increase from the KRW 7.6 billion it had raised in the same quarter last year. Xtandi is approved for ▲the treatment of asymptomatic or mildly symptomatic metastatic castration-resistant prostate cancer (mCRPC), ▲mCRPC previously treated with docetaxel, ▲high-risk non-metastatic castration-resistant prostate cancer, and ▲hormone-sensitive metastatic prostate cancer (mHSPC) in combination with androgen deprivation therapy (ADT). It was approved in Korea in June 2013 and received reimbursement in November following year. Recently, Xtandi's sales have increased sharply, especially since Q3 last year. Previously, its sales ranged around KRW 6 billion, but this significantly increased to KRW 7.6 billion in Q3 last year, then to KRW 8.7 billion in tQ4, KRW 10.5 billion in Q1 this year, and KRW 11 billion in Q2. The increase in sales last year is attributed to the expanded reimbursement. In August, selective reimbursement was granted for the use of ADT as combination therapy for patients with progressive prostate cancer accompanied by distant metastasis. Starting this November, reimbursement for ADTs were extended and is applied regardless of the use of other androgen-production-suppressing drugs. Despite a 3% reduction in the insurance price due to its increased usage, its increase in prescriptions is said to have offset this decrease. Quarterly sales of major ARTA prostate cancer treatments (Unit: KRW 100 mil, Data: IQVIA) Zytiga sales 'slow down'...Erleada sales reach KRW 4.8 billion in only half a year ' Zytiga, a previous competitor of Xtandi, has recently shown a decline in sales. Zytiga's sales in the last quarter were KRW 4.8 billion, a 14% decrease from KRW 5.6 billion of the previous year. Since 2021, Zytiga had closely chased Xtandi in terms of sales. In Q1 2021, sales of Xtandi and Zytiga were KRW 7.6 billion and KRW 2.9 billion, respectively, which is a 2.7 times difference. By Q1 2022, this gap narrowed to 1.2 times, with sales reaching KRW 6.4 billion for Xtandi and KRW 5.2 billion for Zytiga. The sales difference between the 2 drugs further decreased to less than KRW 1 billion in Q2 that year. However, Zytiga's sales have continued to remain in the range of KRW 5 billion since then. Meanwhile, Xtandi's sales increased rapidly. From Q2 his year, the sales difference between the two drugs doubled. Pic of JanssenJanssen's new ATRA-type prostate cancer treatment, Erleada, has rapidly increased its sales since its introduction. In December 2020, Erleada was approved as Janssen's third ATRA prostate cancer therapy. It was added to the reimbursement listing in April this year. Following the listing, Erleada generated sales of KRW 2.7 billion in the second quarter and increased to KRW 4.8 billion in the third quarter, marginally surpassing the sales of Zytiga. Therefore, Janssen plans to focus on Erleada as a latecomer follow-on drugs to Zytiga, aiming to challenge the market leader Xtandi. The market stirrer would be the change in the patient co-payment rates for Xtandi and Zytiga, and the generics development by domestic pharmaceutical companies. Last month, the co-payment rates for Xtandi and Zytiga were adjusted from 30% to 5%. This change occurred following the listing of Erleada, the third ARTA prostate cancer therapy, at a 5% co-payment rate. The maximum reimbursement amount (listed price) for Xtandi was adjusted in the process. Xtandi was registered as a subject to essential reimbursement through a risk-sharing agreement (RSA). With expanded reimbursement, the listed price per tablet was reduced from KRW 28,820 to KRW 14,170. This will be reflected after the fourth quarter of this year. The release of a Zytiga generic by a domestic pharmaceutical company is also anticipated to significantly influence the competitive ground. Hanmi Pharm launched Abiraterone, a generic of Zytiga, under reimbursement in October. As a result, the price of Zytiga was reduced by 30%, from KRW 16,780 to KRW 11,746 per tablet. This price reduction is expected to be reflected in sales after the fourth quarter of this year, potentially resulting steeper decline in Zytiga's sales.
- Opinion
- [Reporter's View] Covid-19, endemic is not 'The End'
- by Eo, Yun-Ho Dec 06, 2023 06:00am
- ‘Endemic’ doesn’t mean THE END. Covid-19 has transitioned to an endemic after a long-fight against the virus for three years. Accordingly, each country is making an adjustment to the health protocol in a bid to meet domestic needs. The Korean government had announced that it will work to list Covid-19 treatments for reimbursement within the first half of 2024 through the Covid-19 risk level management roadmap. And the public’s sense of crisis has dropped with the eased restrictions. Covid-19 transitioned from pandemic to endemic. Yet, the continued introduction of new Covid-19 variant make high-risk patients, particularly those with underlying diseases and senior patients, all the more vulnerable. Up until now, excess deaths associated with Covid-19 is continuing, according to the KDCA report citing KOSTAT data submitted to the office of Rep. Jung Chounsook, a member of the Democratic Party of Korea and a member of NA's Health and Welfare Committee. The Covid-19 associated excess deaths toll over 65,000 people, and in this case, ‘excess deaths’ are typically defined as the difference between the observed numbers of deaths in Covid-19 periods and expected numbers of deaths in the same time periods. Be prepared for battle yet again. We should not forget the times when all people worn masks and had restricted gatherings. Continued supply of vaccines and patient management would be the most important part of the preparation. However, a sense of crisis is hard to achieve once lowered. A lowered crisis-level would result in the transition of reimbursement of Covid-19 treatment to non-reimbursement. After initiating phase 2 of the COVID-19 risk level management roadmap, Covid-19 will be treated as level 4 infectious diseases similar to influenza (flu). In other words, drugs such as Gilead Sciences Korea's ‘Veklury’ and Pfizer's ‘Paxlovid’ will now only be available through the regular reimbursement scheme. Although the government promised to continue providing oral Covid-19 drugs without charge, it can just as well abruptly discontinue provision. As a solution to the problem, pharmaceutical companies have submitted insurance reimbursement applications for drugs in October and are in discussion with HIRA. Yet, no progress has been made. Time is ticking for the upcoming crisis-level adjustment deadline. If we were to treat people with non-reimbursed drugs, copayment rate would be skyrocketed. The problem is that people would start to think, ‘Why should we pay this much for the treatment?’ due to a lowered sense of crisis. We are running out of time. The government should promptly proceed with the debate, and the pharmaceutical companies should consider proposing reasonable drug pricing in preparation for a long fight against COVID-19.
- Policy
- New price adj guidelines set for drugs with unstable supply
- by Lee, Tak-Sun Dec 06, 2023 06:00am
- On the 5th, the National Health Insurance Service established the ‘Pricing adjustment negotiation guidelines for insurance ceiling price of drugs.’ The guideline gained attention for specifying the negotiation content for drugs that have an unstable supply. The guideline also includes the submission of pre-negotiation materials for drugs with unstable supply and premium pricing measures. The NHIS explained that the purpose of the Pricing adjustment negotiation guidelines is to stabilize the supply and demand of drugs that are essential for patient care but do not have smooth supply due to low profitability. The pricing adjustment negotiations are conducted for drugs that have received a negotiation order from the Ministry of Health and Welfare as deemed necessary based on the NHIS Drug Reimbursement Evaluation Committee’s evaluation results of submitted pricing adjustment applications. After receiving the negotiation order, subject companies need to submit evidence of drug cost for the drug subject to negotiation and data on the contract production (import) volume that would be produced (imported) after adjusting the upper price limit. The new guideline requires drugs that need prior discussion to speed up the negotiation period by submitting the abovementioned data prior to negotiations. Therefore, if the MOHW requests prior consultation, the company is required to submit evidence of drug cost for the drug subject to negotiation and data on the contract production (import) volume that would be produced (imported) after adjusting the upper price limit. In addition, if there is an urgent request from the public-private consultative body on the need for a specific drug with unstable supply, the estimated contract production (import) volume data after adjusting the upper price limit must be submitted within 5 days of request. The guidelines set for drugs with unstable supply and demand show the government’s plan to first discuss the contracted production volume amount that will be produced after pricing adjustments to speed up production. Also, drugs with unstable supply and demand will be granted a premium when raising the upper price limit. One percent of the price will be additionally raised if the drug satisfies 1 of the 10 items in the list of policy-related premium pricing conditions, which include cases evaluated as ‘exceptional cases such as infectious disease crises or urgent supply shortages, for which central administrative agencies request cooperation related to drug supply.’ In other words, pricing adjustments requested by the public-private consultative body for drugs with unstable supply and demand will be granted a premium. However, the policy-added benefit is not to exceed a maximum of 7%. The guideline also specified a follow-up plan conditional on production expansion. The NHIS and the company can contract a certain amount of mandatory production (import) volume as needed for a certain period of time, which the company must produce (import) and supply faithfully according to the contract. It also prohibits companies from applying pricing adjustments again for 3 years after receiving pricing adjustments but leaves the door open for cases of medical necessity for the public, such as the spread of infectious diseases. Specifically, negotiations and pricing readjustments are allowed for drugs deemed necessary by the Drug Reimbursement Evaluation Committee. According to the formula, only up to 22% of Selling, General and Administrative Expenses (SGA) will be recognized, and non-operating profits and losses will not be recognized. In addition, a maximum of 17% of the total cost is recognized as reasonable profit, and the distribution margin of 3.44% will be applied to high-priced drugs and 5.15% to low-priced drugs. Hae-Min Jung, Deputy Minister of NHIS Pharmaceutical Management Department, said, “We created the new guideline because it took a long time for pharmaceutical companies to prepare data required for the previous adjustment negotiations, and issues such as sales and administrative costs had hindered prompt negotiations. The NHIS will conduct prompt negotiations per the new guidelines and actively sign and manage supply volume contracts to ensure that essential drugs are supplied to the front line." Meanwhile, the government is actively reviewing drug price increases for drugs with unstable supply and demand and is considering including them eligible for adjustment applications. In line with the government's policy, the guidelines for pricing adjustment negotiations also include exceptional negotiation clauses for drugs with unstable supply and demand. Only this year, prices of magnesium hydroxide, pseudoephedrine budesonide, suspension for nebulizer were granted price hikes through pricing adjustment negotiations, and acetaminophen fever reducer syrups and cefditoren pivoxil Fine Granule and constipation treatments that contain lactulose are being reviewed for pricing adjustments.
- Opinion
- [Desk’s View] On resolving the stock shortage of drugs
- by Lee, Tak-Sun Dec 05, 2023 05:49am
- As a parent, I feel like I've been visiting hospitals after the endemic than I did during the COVID-19 pandemic. At the height of the pandemic, I only had to worry about COVID-19, but after the pandemic has passed, children with weak immunity are suffering from various viral diseases including colds, flu, and adenovirus. When my older daughter got sick, my younger one also got sick, and us parents were no exception. After going through a series of these vicious cycles, I pulled out my mask again and wondered whether I had removed my mask too soon. With this increase in hospital visits, drug shortages are quite understandable. However, as a healthcare consumer, I don't understand the seriousness of it. It's not as bad as when I had to go from pharmacy to pharmacy seeking acetaminophen fever reducers for my COVID-19 vaccination. I never had to go to another pharmacy because the prescriptions written by my local pediatrician or ENT were not available at one site. However, when I look at the prescribed drugs, I see the ingredients of the “drug with unstable supply" that are often mentioned in recent articles. Although these are not the items that are being discussed for price hikes, they contain the same active ingredients. This shows that there may be differences in demand for different products even within those that contain the same ingredients. Also, in one corner of the pharmacy, I saw a lot of over-the-counter fever reducers. Although the supply of prescribed fever reducers is running low, the over-the-counter ones still seem to be available. The government's response to the ‘unstable supply of medicines’ seems to have been successful to a certain extent, as consumers are not feeling any inconvenience yet. If the consumers had felt and experienced the drug shortage firsthand, the anger against the government would have been extreme, as people become most emotional when they are sick. However, I am disappointed that the government's solution seems to be focused on 'increasing production.’ Even the most effective card the government has played, the drug price increase, is aimed at increasing production. If only certain products are out of stock and other drugs with the same ingredient are available, efforts should be made to diversify prescriptions. It would be best if the system allows full utilization of the substitution system, but it is unlikely to have an effect under the current system, as the pharmacies need to bear the inconvenience. In this case, why not amend the enforcement decree to temporarily exempt pharmacies from notifying the medical institutions about the substitutions for drugs that are in unstable supply? In the long run, we can also expect to alleviate the concentrated prescription of certain ingredients by removing reimbursement of general medicines used for mild illnesses such as the common cold. However, as such options are not even being considered as a way to solve the supply and demand instability of drugs, this further narrow the options that the government can take. The government should seriously monitor the market first, and consider measures to diversify prescription if other drugs with the same ingredient are left for immediate utilization.
- Company
- AbbVie Korea strengthens its autoimmune disease pipeline
- by Eo, Yun-Ho Dec 05, 2023 05:48am
- AbbVie Korea is actively working to advance its position in the field of autoimmune diseases. According to industry sources, AbbVie Korea successfully expanded the indications for its Interleukin-23(IL-23) inhibitor 'SKYRIZI (Risankizumab)’ and JAK inhibitor ‘RINVOQ (Upadacitinib).’ On November 15th, an indication was added for SKYRIZI to treat adult patients with moderately to severely active Crohn's disease who had responded inadequately or were intolerant to biologic therapy and/or existing therapies. The approval was based on a multicenter, randomized, double-blind, placebo-controlled Phase III trial that demonstrated the efficacy, safety, and tolerability of SKYRIZI in adult patients with moderate-to-severe active Crohn’s disease. In the ADVANCE and MOTIVATE induction trials, 1,419 patients were randomly assigned to receive 6000 mg intravenously, 1,200 mg intravenously, or placebo at week 0, week 4, and week 8. Clinical remissions and endoscopic responses were measured by stool frequency (SF) and abdominal pain score (APS) at 12 weeks. The FORTIFY trial evaluated 462 patients who showed clinical response to 12 weeks of intravenous induction therapy in the ADVANCE and MOTIVATE trials. Patients were randomly assigned to receive either subcutaneous risankizumab 180 mg, subcutaneous risankizumab 360 mg, or withdrawal from risankizumab to receive subcutaneous placebo every 8 weeks for up to 52 weeks. The primary endpoints were clinical remission and endoscopic response at week 52. SKYRIZI is currently approved and reimbursed in Korea to treat moderate-to-severe plaque psoriasis in adults who are candidates for phototherapy and systemic therapy (including biologic treatment), as well as to treat active psoriatic arthritis in adults who are non-responsive to DMARDs (disease-modifying anti-rheumatic drug). As for RINVOQ, its reimbursement has been expanded to the treatment of severe active ankylosing spondylitis from December. Accordingly, patients with severe active ankylosing spondylitis who have stopped receiving treatment due to poor response to one or more types of tumor necrosis factor-alpha inhibitor (TNF-α inhibitor) or IL-17A inhibitor will be able to receive reimbursement for once daily treatment of 15mg of RINVOQ. The reimbursed price of RINVOQ is KRW 19,831 for once-daily dose of 15 mg, and patients covered by special exemption of calculations provisions only have to pay 10% of the drug price. RINVOQ proved efficacy in patients with severe active ankylosing spondylitis who were enrolled in the Phase 2/3 SELECT-AXIS1 and the Phase 3 SELECT-AXIS2 clinical trials. In both SELECT-AXIS 1 and 2 clinical studies, the ASAS40 (Assessment of Spondyloarthritis International Society response standard improvement of 40% or more) response rate for patients treated with RINVOQ 15 mg was 52.0% and 45.0%, respectively, at 14 weeks. This was significantly higher than that of the placebo group (26.0% and 18.0%, respectively.) In addition, the ASAS40 response was observed from Week 2 in SELECT-AXIS1 and from @eek 4 in SELECT-AXIS 2, confirming quicker effect of the drug. In the SELECT-AXIS 2 clinical trial, the average reduction in back pain in the Rinvoq 15mg treatment group was -3.00, which is a significant decrease compared to the placebo group (-1.47), and the intensity of morning stiffness also decreased by -3.08 compared to the baseline value compared to the placebo group (-1.67). Lee Sin Suk, Chairman of Korean College of Rheumatology, said, “RINVOQ has significantly improved the pain, inflammation, and joint damage in patients with severe active ankylosing spondylitis, and also helped improve the quality of life in patients as well as reaching treatment goals. The reimbursement expansion is likely to address the unmet need and alleviate the financial burden of patients who had been unable to receive benefits, and is expected to offer an effective oral treatment option for the patients."
- Company
- Companies in KOR and abroad compete in Eylea biosimilar mkt
- by Son, Hyung-Min Dec 05, 2023 05:48am
- Intense challenges are being made against Bayer's Eylea, the current leader in the macular degeneration market. Not only domestic biosimilar companies like Celltrion and Samsung Bioepis but also traditional pharmaceutical companies such as Sam Chun Dang Pharm have thrown down the gauntlet. Bayer and Regeneron, the original developers of Eylea, thus face fierce market competition not only with biosimilar developers but also with Roche's next-generation macular degeneration treatment, Vabysmo. According to industry sources on the 3rd, among domestic companies, Sam Chun Dang Pharm, Celltrion, Samsung Bioepis, and Alteogen are currently developing Eylea (aflibercept) biosimilars. All four companies have set their sights on the global market, preparing to receive approvals in the U.S. and Europe in addition to the Korean market. In the U.S., Celltrion, Amgen, and Sandoz are close to commercializing their biosimilars in the U.S. These companies have now completed global Phase III clinical trials and applied for marketing authorization in the U.S. and Europe. Eylea’s U.S. new drug exclusivity expires next May, and its European substance patent expires in November 2025. In Korea, Sam Chung Dang Pharm is nearing approval. The company applied for the marketing authorization of its Eylea biosimilar candidate ‘SCD411’ in two formulations - vial and prefilled syringe formulation – in Korea last month. Sam Chung Dang Pharm is the first company to apply for the approval of a prefilled syringe formulation of Eylea. The application was filed based on SCD411’s Phase III trial results. The trial, which had been conducted on 576 patients, demonstrated SCD411’s bioequivalence to Eylea. Based on the results, Sam Chung Dang Pharm is also preparing to apply for marketing approval of SCD411 in the United States and Europe as well. Eylea to inevitably compete with biosimilars and Vabysmo Pharmaceutical and bio companies in Korea and overseas have jumped into developing biosimilars due to the high marketability of Eylea. Eylea’s global sales were approximately USD 1.4 billion (KRW 1.9 trillion) in Q1 this year and USD 1.5 billion (KRW 2.373 trillion) in Q2 this year. Eylea had posted the highest sales in the domestic macular degeneration market. According to the pharmaceutical market research institution IQVIA, Eylea’s sales, which had been around KRW 46.8 billion in 2019, exceeded KRW 60 billion in 2020. Sales continued to grow, and Eylea posted KRW 80.4 billion in sales last year. However, sales generated by Eylea’s competitors last year, Novartis’ Lucentis (ranibizumab) and Beovu (brolucizumab) were only KRW 29.4 and KRW 14.5 billion, respectively. In addition to the challenges made by the biosimilar developers, Bayer’s Eylea is starting competition in earnest with Roche's macular degeneration drug Vabysmo (faricimab). Unlike existing VEGF inhibitors like Lucentis, and Eylea, the drug’s dual mechanism of action allows for it to also block the angiopoietin-2 (Ang-2) pathway to inhibit neovascularization. In particular, in clinical trials, Vabysmo improved visual acuity at a level non-inferior to that of Eylea, which is administered at 8-week intervals, when administered at intervals of up to 16 weeks. Vabysmo was granted reimbursement and released in earnest in October last month. Roche is also known to be planning to introduce a prefilled syringe formulation of Vabysmo next year.
- Policy
- MOHW will expand drugs subject to price adjustments
- by Lee, Tak-Sun Dec 05, 2023 05:48am
- To ensure higher drug prices for medicines in short supply, the Ministry of Health and Welfare (MOHW) announced its plan to revise the evaluation standard for drugs eligible to apply for pricing adjustments to increase the upper limit price (list price). Accordingly, national essential drugs and those selected by public-private consultative bodies as having unstable supply will be eligible to apply for price increase adjustments. According to industry sources on December 4th, the MOHW is reviewing a plan to include national essential drugs and drugs suggested to be in short supply by its public-private consultative body as subjects in its evaluation standard to adjust the upper limit price. The current evaluation standard allows drug pricing adjustments to be made for drugs ▲with no alternative drugs are available; ▲absolutely necessary for treatment; ▲necessary for treatment, and the cost of medication is cheaper than its alternative but the administration and ingredients are the same, and there is only 1 manufacturer produces the drug. A drug essential for treatment should meet the following criteria, ▲have no available alternative therapies (including medicines) ▲is used for serious diseases with low survival rates ▲is used for rare diseases that affect a small group of patients ▲has demonstrated clinically significant improvement such as a substantial extension of survival time, and other cases where the committee evaluates it absolutely necessary for the patient’s treatment. The drugs that have recently had supply shortages and raised concern in pharmacies, often do not meet the current evaluation criteria. To resolve the current issue, the government is considering revising the evaluation criteria to include national essential drugs and drugs deemed necessary for price increase by the public-private consultative body to resolve the supply instability issue. The Ministry of Food and Drug Safety recently added 6 types of 7 pediatric medicines, including acetaminophen syrup and tulobuterol transdermal patches to the list of national essential drugs. The industry views this as a step towards adding the qualification for drug price increase adjustment requests for national essential drugs. The industry views the proposed revision is a measure to quell potential disputes over criteria that may arise in the future, as the government is considering the drug price increase as part of measures to address the supply shortage of particular drugs. Consequently, there is speculation that drugs that do not qualify for adjustment requests will still go through the price increase process, with their legitimacy being granted through subsequent revisions of the criteria. On December 7th, HIRA's Drug Reimbursement Evaluation Committee will review pricing adjustment requests for 5 pharmaceutical products that have supply shortages. The candidate products include Sama Pharm's 'Setophen Suspension', Johnson & Johnson Korea’s 'Children's Tylenol Suspension', Boryung's ' Meiact Fine Granule Boryung', Kukje Pharm's 'Ditoren Fine Granules', and JW Pharmaceutical's 'Dulackhan Easy Syrup', among others.
- Company
- Celltrion applies for US approval of its Prolia biosimilar
- by Chon, Seung-Hyun Dec 04, 2023 05:13am
- Celltrion announced today that it has filed an application for the marketing authorization of its, ‘CT-P41,’ to the US Food and Drug Administration (FDA). CT-P41 is a biosimilar of ‘Prolia,’ a treatment for osteoporosis. Based on the results of a global Phase III clinical trial on CT-P41, Celltrion filed for marketing authorization of its substance for all of Prolia’s indications, including osteoporosis, bone loss, prevention of skeletal complications from multiple myeloma or solid cancer bone metastases, and giant cell tumor of bone. Prolia is a biological osteoporosis treatment that targets the RANKL protein essential for the formation, activation, and survival of osteoclasts that destroy the bone. The same active ingredient is also licensed under the brand name ‘Xgeva’ for the prevention and treatment of bone metastasis complications in cancer patients. Based on IQVIA, the drug generated USD 5.83 billion in sales last year (KRW 7.54 trillion), and its patent is set to expire in the U.S. and Europe in February 2025 and November 2025, respectively. In October, Celltrion conducted a global Phase III trial on 477 patients in postmenopausal women with osteoporosis in 4 countries including Poland and Estonia, to confirm the bioequivalence and pharmacodynamic similarity to the original drug. Celltrion has worked continuously with the FDA and the European Medicines Agency (EMA) throughout the development process to design a global development program that will enable CT-P41 to be approved simultaneously as a Prolia and Xgeva biosimilar, based on the results of the global Phase III study. The company explained that CT-P41 will be allowed to be prescribed interchangeably with the original product once it is approved in the U.S. because it will receive interchangeable biosimilar status. Following the U.S. approval, Celltrion plans to file for approval in other major global markets, including Europe. A Celltrion official said, "After confirming the bioequivalence and similarity of CT-P41 in the Phase III trial, we have submitted our application for its marketing authorization in the U.S. as an interchangeable biosimilar. will continue to work with regulatory authorities to complete the remaining approval processes."
- Policy
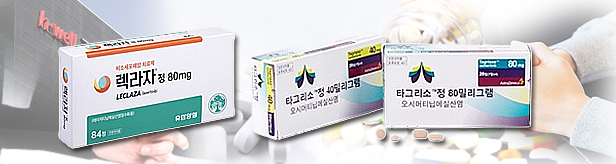
- Tagrisso and Leclaza complete drug pricing negotiations
- by Lee, Tak-Sun Dec 04, 2023 05:13am
- The National Health Insurance Serivce was found to have completed pricing negotiations with the companies for the reimbursement of Tagrisso (AZ, osimertinib) and Leclaza (Yuhan, lasertinib) as first-line treatments for non-small cell lung cancer through the risk-sharing agreement (RSA) system. However, as the two were applied different types of RSA, the actual reimbursed prices of the two drugs are expected to differ somewhat. According to industry sources on the 1st, AstraZeneca, which owns Tagrisso, and Yuhan Corp, which owns Leclaza, recently completed drug price negotiations with the NHIS. The pricing negotiations had been ongoing with the NHIS since late September for Tagrisso, and since late October for Leclaza, but the authorities had reportedly treated the two drugs as a single set for reimbursement deliberations. Yuhan Corp has been providing Leclaza as a first-line treatment to patients free of charge through an Early Access Program (EAP) until its reimbursement, without limiting the number of patients. This was why the initial treatment refund type RSA was not applied during its pricing negotiations., Initial treatment refund-type RSA is a risk-sharing approach that takes into account uncertainties such as the effectiveness of the drug during the initial treatment period. Instead, Leclaza was applied 2 types of RSA during negotiations – Refund type and Expenditure Cap type RSA. On the other hand, Tagrisso was applied initial treatment refund type, refund type, and expenditure cap type RSA. Due to differences in the type of RSA applied, the NHIS seemingly has balanced the prices of the two drugs by setting different refund rates. As such, the refund rate for Leclaza, which was not applied the initial treatment refund-type RSA, will be higher than that of Tagrisso. As such, the actual price of Leclaza is also likely to be somewhat lower than that of Tagrisso The list price is also expected to decrease slightly with the increase in the reimbursement rate. Currently, the insurance price ceiling (list price) is set at KRW 68,964 per tablet for Leclaza, while Tagrisso (80 mg) is set at KRW 212,148 per tablet. As Leclaza is taken three times a day, and Tagrisso once a day, the total drug cost is slightly cheaper for Leclaza. However, depending on the refund rate, the actual difference in price may increase further.
- Opinion
- [Reporter's View] Non-face-to-face treatment pilot project
- by Lee, Jeong-Hwan Dec 04, 2023 05:12am
- Starting December 15th, ’24-hour non-face-to-face treatment’ will be available to the public following the legalization of revised non-face-to-face treatment pilot project by the Ministry of Health and Welfare. The project will extend the non-fact-to-face treatment benefit that had been previously limited to returning patients to new patients. Patients will be able to request non-face-to-face treatment for all types of diseases, whether acute or chronic, at a clinic where they have made an in-person visit within 6 months. A dramatic increase in the number of non-face-to-face treatment is expected due to the updated policy, which has allocated the responsibility of sorting the non-face-to-face treatment eligibility at the sole discretion of the physicians, regardless of severity of disease. Physicians and pharmacists registered with the Korean Medical Association (KMA) and the Korean Pharmaceutical Association (KPA) have voiced concerns against the newly updated non-face-to-face treatment policy. Their primary view regarding the policy is that the government has forcefully implemented the pilot project without a thorough assessment of its side effects. Shortly after the MOHW's announcement, physicians and pharmacists' associations small and large have all issued statements opposing the revised pilot project. On November 30th at 8:00 a.m., a day before the MOHW released the revised plan, a conference had been held between the advisory panel regarding the non-face-to-face treatment pilot project. Although the KMA, KPA, and the Korea Alliance of Patients Organization have stated their opposing views on the extension of the policy at the meeting, their opinions have not been taken into consideration. Therefore, the panels expressed that the advisory panel for the pilot project was held for formality, wherein the government had not planned to regard any of the opinions proposed at the meeting and the extension of the plan was already a done deal. The Yoon Suk-Yeol administration has been putting an emphasis on running the governmental operation based on ‘social consensus and scientific reasoning,’ especially regarding the major issues of national concern. However, no such consensus or reasoning was made in the process of the non-face-to-face treatment pilot project advisory panel meeting and the revision announcement. Neither scientific statistics nor research analysis data have been suggested for concluding extending the potential patient pool of the ongoing pilot project. It is truly questionable whether MOHW has ruled President Yoon Seok Yul’s comment that “parents of children have a hard time seeking non-face-to-face treatment late at night and on holidays” made in the Cabinet meeting a scientifically backed argument. In contrast, all the opposing voices of physicians, pharmacists, and patient organizations were left unheard. As for the revision, the scientific reasoning-based administration is non-existent and the societal agreement-based policy-making principle has been neglected. Because the non-face-to-face-treatment pilot project is a revision to the public health law and not to the medical law, there seems to be no regulation of MOHW's way of conducting an extension. Despite of opposing views by medical professionals, concerns by patient organizations, and major criticisms by the political party, the pilot project which allows ’24-hour non-face-to-face treatment’ will be accelerated. December 15th will be remembered as the date on which the omnipotent pilot project, which extends the scope of non-face-to-face treatment, overpowers the medical law.