- LOGIN
- MemberShip
- 2026-04-04 01:51:27
- Policy
- 29 new drugs with 45 ingredients reimb this year
- by Lee, Tak-Sun Dec 31, 2024 05:55am
- This year, 45 new drugs with 29 ingredients were listed for reimbursement benefits under Korea’s health insurance. Among them, the only homegrown new drug was 'Zastaprazan citrate', a P-CAB-based gastroesophageal reflux disease treatment. As of the 30th, 45 new drugs were listed for reimbursement in Korea’s National health insurance system from January to December 2024, improving patient access. In January alone, five 5 drugs were reimbursed, including the anti-cancer drugs ‘Braftovi Cap,’ and ‘Bosulif Tab,’ ulcerative colitis treatment ‘Zeposia Cap,’ asthma and COPD treatment ‘Trimbow Inhaler,’ and neurofibromatosis treatment ‘Koselugo Cap.’ In February, two new drugs, ‘Kerendia Tab’ for chronic kidney disease and ‘Luxturna Inj’ for inherited retinal dystrophy, were approved, followed by ‘Trelegy 200 Elllipta Inhaler’ for asthma in March. In April, four new drugs were covered by health insurance, including cytomegalovirus treatment ‘Livtencity Tab,’ plaque psoriasis treatment ‘Sotyktu Tab,’ and anticancer drugs ‘Poteligeo Inj,’ and ‘Enhertu Inj.’ In May, the atopic dermatitis treatment ‘Adtralza Prefilled Syringe’ and the iron deficiency treatment ‘Ferrinject Inj’ were covered, and in June, morning sickness treatment ‘Diclectin Enteric-coated Tab’ were covered for the first time under the government's health insurance program. New Drugs reimbursed in 2024 in Korea, (number of items) New drug approvals continued into the second half of the year. In July, 3 new drugs were introduced, including the eosinophilic asthma treatment ‘Fasenra Prefilled Syringe,’ the hemophilia treatment ‘Idelvion Inj,’ and the anti-cancer drug ‘Xpovio Tab.’ In August, the CAPS treatment ‘Ilaris Inj’ and schizophrenia and bipolar disorder treatment ‘Latuda Tab’ were listed. No new drugs were listed in September, but five came out at once in October. The glaucoma treatment ‘Rhopressa Eye Drops,’ secondary hyperparathyroidism treatment ‘Orkedia Tab,’ the domestic new drug GERD treatment ‘Jaqbo Tab,’ COVID-19 treatment ‘Paxlovid Tab’ and ‘Veklury Inj’ were covered by health insurance, reducing the financial burden on the patients. In November, ‘Empaveli Inj,’ a treatment for paroxysmal nocturnal hemoglobinuria, was added to the list, and in December, new drugs such as ‘Vyxeos Liposome Inj,’, a treatment for acute myeloid leukemia, ‘Qarziba,’ a treatment for neuroblastoma, and ‘Camzyos Cap,’ a treatment for obstructive hypertrophic cardiomyopathy, were added to the reimbursement list. Of the new drugs on the list, 12 were covered by risk-sharing agreements, which accounts for more than 40% of the total listed items. Ten drugs accepted the average price of alternative drugs and skipped insurance price cap negotiations and only negotiated on the estimated claim amount. Next January, apremilast, an ingredient used for psoriatic arthritis and psoriasis, will be reimbursed for the first time. The five drugs that will be reimbursed are all generics. The original apremilast-based drug was Amgen's Otezla Tab, but it failed to gain reimbursement and was withdrawn from the Korean market. This is the first time that a group of domestic generic companies have entered Korea’s health insurance reimbursement market.
- Policy
- Yungjin Pharm wins the 1st trial against Ofev
- by Kim, Jin-Gu Dec 31, 2024 05:44am
- Yungjin Pharm has won the usage patent dispute for Ofev (nintedanib), a treatment for idiopathic pulmonary fibrosis (IPF). Based on the victory, Yungjin is one step closer to the early launch of its generic version of Ofev. The pharmaceutical industry suggests this decision is significant beyond Yungjin Pharmaceutical's avoidance of Ofev's usage patent. It may serve as a patent-avoidance strategy for generics. Yungjin Pharm wins the first trial for Ofev's usage patent…brings closer to the early launch of generic According to the pharmaceutical industry on December 20, the Korean Intellectual Property Trial and Appeal Board (IPTAB) recently issued a decision of 'claim upheld' in a passive rights scope confirmation trial filed by Yungjin Pharm against Boehringer Ingelheim regarding the usage patent of 'Ofev Soft Cap.' In September, Yungjin Pharm filed claims for a passive rights scope confirmation trial against the usage patent of Ofev. Following that, Cosmax Bio and Daewoong Pharmaceutical also filed claims for the same avoidance trials. Among these companies, Yungjin Pharm was the first to win.. Product photo of OfevOfev is protected by at least three patents: a substance patent expiring in January 2025, a usage patent expiring in September 2026, and a formulation patent expiring in June 2029. Only the substance patent is listed in the Ministry of Food and Drug Safety's (MFDS) patent registry, while the usage·formulation patents are unlisted patents. Having won the first trial for the usage patent, Yungjin Pharm is now closer to the early launching its generic version of Ofev. On December 12, Yungjin Pharm received approval for its generic version of Ofev as 'Nintebro.' The generic can be launched after the substance patent expires on January 25. The unlisted formulation patent remains to be solved but nullifying the substance patent is easier than the substance patent or usage patent. Yungjin Pharm is expected to enter the KRW 5.7 billion (USD 4.3 million) nintedanib market with the sole generic version of Ofeb if it launches its product. According to pharmaceutical market research firm IQVIA, Ofev's sales in 2023 totaled KRW 5.7 billion. Despite not being covered by national health insurance, Ofev has steadily increased its sales, reporting KRW 1.8 billion in 2020, KRW 3.3 billion in 2021, KRW 5.6 billion in 2022, and KRW 5.7 billion last year. A single clause in the generic's indication was the key to determining the patent dispute outcome The pharmaceutical industry closely examines the reasons behind Yungjin Pharm's victory in the usage-patent dispute. This success is due to the company's innovative strategy to address the usage-patent conflict, setting it apart from traditional methods. The key to the victory was the MFDS' acknowledgment of the 'exclusion clause.' Nintebro approval was the first time the MFDS has acknowledged 'excluding X.' Ofev is approved for the following indications: ▲Treatment of IPF ▲Delay of pulmonary function decline in patients with systemic sclerosis-associated interstitial lung disease (SSc-ILD) ▲Treatment of chronic fibrosing interstitial lung diseases with a progressive phenotype (excluding IPF). For Nintebro, it is approved for the following indications: ▲Delay of pulmonary function decline in patients with SSc-ILD ▲Treatment of chronic fibrosing interstitial lung diseases with a progressive phenotype, excluding IPF. Boehringer Ingelheim Yungjin Pharm The analysis suggests a key element in the trial outcome in this usage-patent dispute was the 'excluding IPF' clause in Nintebro's second indication. Yungjin Pharm strategically focused on the remaining indications of Ofev, deliberately excluding IPF to circumvent the drug's usage patent. If the MFDS had not acknowledged the 'exclusion clause,' Yungjin Pharm's strategy may not have been successful. Even if the company removed Ofev's first indication, IPF could be interpreted as being included within Ofev's third indication. In other words, if Yungjin Pharm had not included the exclusion clause in the Nintebro indication, the company would have won the avoidance only if it had deleted Ofev's first and third indications. However, the MFDS recognized the wording 'excluding IPF' for Nintebro's second indication, which Yungjin Pharm strategically emphasized during the patent dispute. Ultimately, the Korean IPTAB accepted Yungjin Pharm's argument, ruling that Nintebro did not infringe on Ofev's usage patent. MFDS has acknowledged the 'excluding X' clause for the first time…will this become the new patent avoidance strategy? Until now, the MFDS has not acknowledged phrases such as 'excluding X' in the indication section during the drug approval process. Generic manufacturers have repeatedly requested MFDS to acknowledge such exclusion clause to circumvent originator companies' specific use patents. However, until now, only the deletion of certain indications was permitted, which was a more passive approach. In the Nintebro case, the MFDS broke from its traditional practices, approving an active approach that recognizes indications excluding certain conditions for the first time. The pharmaceutical industry positively views this precedent as a new strategy for patent avoidance. If generic manufacturers focus on obtaining approval for exclusionary language such as 'excluding X' during the regulatory process, they could secure a significant advantage in subsequent usage patent challenges. "Previously, avoiding patent infringement was difficult if the 'active exclusion clause' was not included in the efficacy and effectiveness section of a generic," Park Jong Hyuk, Yungjin Pharm's patent attorney, said. "The company received the ruling by including the exclusion clause in the efficacy and effectiveness section of the generic. It will now open a new door for avoiding patient group-specific discovery," Park said. Industry experts view that the recent ruling is expected to expand access to nintedanib for patients with rare pulmonary diseases in South Korea. Ofev has not yet been listed for reimbursement under the national health insurance system, leaving patients to pay approximately KRW 3 million monthly for the medication. The high cost has prompted a public petition for its inclusion in the reimbursement list. Now, the generic version of Ofev can be launched earlier. Patients will be able to access medication at a lower price.
- Policy
- K-CAB, Enteresto, Olumiant, Rinvoq signs PVA agreements
- by Lee, Tak-Sun Dec 30, 2024 05:57am
- HK.InnoN The domestic P-CAB-based gastroesophageal reflux disease treatment 'K-CAB Tab (tegoprazan, HK Inno.N) and heart failure treatment 'Entresto Film Coated Tab (sacubitril-valsartan-sodium salt complex) the oral JAK inhibitors Olumiant Tab (baricitinib, Lilly), and Rinvoq ER Tab (Upadacitinib hemihydrate, AbbVie) have agreed on a price-volume agreement (PVA) upon negotiations with the National Health Insurance Service. As a result, the refund rate for K-CAB was adjusted, while the price of Entresto and Olumiant will be reduced next month. According to industry sources on the 27th, 3 products recently agreed to a ‘PVA B type’ negotiations with the NHIS to adjust reimbursement and the price ceiling amounts. Since 2021, K-CAB has been reimbursed under a refund-type PVA agreement with the NHIS. Under the agreement, the pharmaceutical company refunds the NHIS for the increase in usage amount instead of reducing the upper limit amount. The new agreement is likely to have adjusted the refund amount for K-CAB. Since the reimbursement agreement, K-CAB has undergone 2 PVA negotiations with the NHIS and the company refunded an amount to make up for its increased usage. By adjusting the refund rate, it has maintained its price ceiling of KRW 1,300 per tablet since its listing in 2019. The price of the heart failure drug Entresto will be reduced to KRW 1,599 per tablet from KRW 1,683 per tablet for 3 products through the PVA. Entresto’s sales had been KRW 59.4 billion in 2023 according to IQVIA, up 29% YoY. Lilly's Olumiant was also unable to escape PVA negotiations due to increased usage this year. Olumiant also generated KRW 17.9 billion in sales last year, up 16% YoY. The deal will lower the price of the 4mg tablet from KRW 20,635 to KRW 20,172 and the 2mg tablet from KRW 13,757 to KRW 13,448. Another JAK inhibitor, Rinvoq XR tablets, will also see its price ceiling reduced through PVA type B negotiations. The price of the 15mg product will go from KRW 19,831 to KRW 18,740, and the 30mg product will go from KRW 31,628 to KRW 29,850. Rinvoq XR tablets generated KRW 20.7 billion in sales last year, up 81% YoY, according to IQVIA. Type B negotiations will be conducted when the insurance price ceiling has been adjusted through Type A negotiations, or 4 years have passed since the ceiling amount has been adjusted by negotiation without Type A negotiations and the claims of the same product group have increased by 60% or more than 10% from the previous year's claims more than four years after the date of first listing or the date the ceiling amount was adjusted by negotiation, and the increased amount exceeds KRW 5 billion. Two other products, including Janssen's autoimmune disease treatment Tremfya prefilled syringe Inj (guselkumab), will also receive a price cut through PVA Type B negotiations. This is the second year the price reductions were made in a row. For the second year in a row, the insurance price ceiling for Tremfya Prefilled Syringe and Tremfya One-Press Auto-Injector will be adjusted from KRW 1.5493 million to KRW 1.524511 million. Five products of Sanofi's hemophilia B treatment Alprolix will also have their insurance price ceiling reduced from KRW 1,081 to KRW 1,088 through PVA type B negotiations. The price adjustments will be made effective January 1, 2025.
- Policy
- 7 pharmas newly win 'Innovative Pharmaceutical
- by Lee, Tak-Sun Dec 30, 2024 05:57am
- Seven pharmaceutical companies, including Dong-A ST, have newly acquired the 'Innovative Pharmaceutical Companies Certification.' Five companies, including Ildong Pharmaceutical, received extensions on their certifications. The Ministry of Health and Welfare (MOHW) announced the result of reviewing the 'Innovative Pharmaceutical Companies Certification' candidates for 2024 through the Pharmaceutical Industry Development and Support Committee meeting held on December 27 (Friday). The result of reviewing the According to the 'Special Act on Pharmaceutical Industry Development and Support,' the MOHW has been issuing companies that have invested substantially in R&D and those demonstrating superior R&D outcomes for new drugs the 'Innovative Pharmaceutical Companies Certification' since 2012. For instance, the standards for companies based on their pharmaceutical sales are as follows: 7% spending in R&D investment for companies with sales of KRW 100 billion or below, 5% spending in R&D investment for companies with sales of KRW 100 billion or more, and 3% spending in R&D investment in companies that have obtained the U.S·Europe GMP (good manufacturing practice). The 'Innovative Pharmaceutical Companies Certification' for new types is conducted every two years, and for extension cases, it is conducted every three years. When certified, companies can maintain the status for three years. Those certified receive benefits, including added points for participating in government-funded R&D projects, drug price advantages, tax benefits, and regulatory approval support. This round of review for the 'Innovative Pharmaceutical Companies Certification' included both new certification and extension of certification. Twenty-five companies were candidates for new certifications. Seven pharmaceutical companies have passed the certification review, including Dong-A ST, Amgen Korea, Onconic Therapeutics, Curocell, Hanall Biopharma, SK Bioscience, and SK Biopharm. For Dong-A ST, the company won the title again five years after failing the cut due to rebate issues in 2019. List of companies certified as innovative pharmaceutical companies (49 companies as of December 28, 2024). 33 general companies, 12 biotech venture companies, and 4 foreign pharmaceutical companies received the title. For extension reviews, five companies were considered for the review, including Alteozen, ABL Bio, Ildong Pharmaceutical, AstraZeneca Korea, and Janssen Korea, and they all passed. As a result, a total of 49 companies received notifications for obtaining the title of innovative pharmaceutical companies. "Innovative pharmaceutical companies have played a crucial role in advancing the Korean pharmaceutical industry by investing more in R&D than other pharmaceutical companies," Director General Jung Eun-young of the Bureau of Health Industry, MOHW, said. "We will continue to establish support systems and improve policies to encourage these companies to continue to expand their R&D investments, enhance innovativeness, and create values."
- Company
- Yuhan Corp will distribute Canesten·Bepanthen
- by Nho, Byung Chul Dec 30, 2024 05:57am
- Yuhan Corp has been selected as the new distribution partner for Bayer’s Canesten and Bepanthen. According to industry sources, Bayer Korea recently terminated its 10-year co-promotion agreement with Ildong Pharmaceutical and signed a new distribution agreement with Yuhan Corp. Under the new agreement, Yuhan Corp will take over the domestic distribution of Bayer's flagship products ▲ Canesten Cream (antifungal), Vaginal Tablets (vaginitis treatment), Powder (infant antifungal), and Bepanthen Ointment (diaper rash treatment). Based on drug distribution performance, Bepanthen and Canesten recorded sales of KRW 8.9 billion and KRW 3.7 billion, respectively, last year. This is a simple wholesale distribution standard data, and the actual company-based performance is expected to be higher than this, so Yuhan Corp’s OTC drug performance is expected to further increase with the addition of this new lineup. The Canesten lineup consists of Canesten 1 vaginal tablet, Canesten cream, and Canesten powder, which are used to treat vaginal candida infections, athlete's foot, and ringworm. The dexpanthenol-based Bepanthen cream is effective for burns, bedsores, dermatitis, eczema, diaper rash, and sunburn, and its competitors include Hanpoong’s Jaungo. Yuhan Corp is a traditional OTC pharmaceutical powerhouse alongside DongKook Pharmaceutical, Donghwa Pharmaceutical, Ildong Pharmaceutical, Daewoong Pharmaceutical, and GC Biopharma, and has recently pursued a sustained and aggressive consumer product marketing strategy. In particular, Yuhan Corp secured the exclusive rights to fatigue recovery drug Lalaola from Vivozon Pharmaceutical in May 2023, and is making considerable efforts to secure a pipeline of OTC drugs, and has been conducting aggressive marketing such as TV-CFs and the appointment of broadcaster Dong-Yup Shin as its exclusive model. In addition, to secure a foothold in the OTC painkiller market that is worth KRW 150 billion, the company launched liquid soft capsule painkillers Yuhan Vfend and Yuhan Acetaminophen soft capsules last year to expand sales. It is also noteworthy that the company has secured more than 15,000 direct-dealer pharmacies for its multinutrient Beecom-Megatrue, which has improved performance through continuous lineup-strengthening activities.
- Company
- Companies show mixed responses to patent term limitation law
- by Kim, Jin-Gu Dec 30, 2024 05:57am
- A bill to amend the Patent Act to limit the patent period for new drugs has passed the plenary session of the National Assembly. The amendment will limit the upper limit of the remaining patent term to a maximum of 14 years from the time a new drug is approved and allow only one of several patents registered for a single drug to be extended. The pharmaceutical industry has expressed both excitement and concern. Multinational pharmaceutical companies that own a large number of original drugs are worried that their patent life will be shortened. Domestic pharmaceutical companies, on the other hand, are generally excited about the prospect of being able to launch generics sooner. The amendment to the Patent Act raises concerns among multinational pharmaceutical companies...“will rather reduce access to drugs” According to industry sources on the 30th, the National Assembly held a plenary session on the 27th and passed the bill to amend the Patent Law. The amendments to the Patent Law are implemented through the government's promulgation process. Its effective date will be 6 months from the date of promulgation. The amendments will apply to patent applications registered after the amendment is enacted. Assuming that the revised law is promulgated early next year, this means that the new patent term system will be implemented as early as the second half of next year. The revision is expected to result in a shorter patent term for original drugs in Korea. Conversely, this means that generics will be available in Korea sooner. The advantages and disadvantages that the amendment will bring to original and generic companies will be sharply divided. Multinational pharmaceutical companies with a large number of original drugs are expected to be disadvantaged by the revised law. The shorter patent period will allow generics to be launched sooner, which will result in lower drug prices. It is also expected to partially neutralize the original companies’ strategy of delaying generic entry by extending multiple drug patents. When the actual amendment was being discussed by the National Assembly, the multinational pharmaceutical companies submitted a statement opposing the amendment. Korean Research-based Pharmaceutical Industry Association (KRPIA) criticized the amendments, saying, “Adopting foreign practices for only some elements, rather than revising the entire regulation on patent term extension, undermines the public's access to medicines and undermines international harmonization.” A representative of a multinational pharmaceutical company said, “Patent challenges by generic companies have been frequent in Korea due to the first generic prior authorization (first generic) system, and the probability of winning patent lawsuits has been higher for the generic companies than in the US and Europe.” “Under these circumstances, shortening the effective patent term will just further damage the companies that own the original drugs.” Expenctations rise among domestic companies on “launching generics faster”…some play ‘caution’ On the other hand, domestic pharmaceutical companies generally welcome the passage of the Patent Act amendment bill during the National Assembly’s plenary session. As the patent term of original drugs will be shortened, the domestic pharmaceutical companies expect this will accelerate the early release of generics, which already own a fair share of the market. “Compared to advanced countries such as the U.S. and Europe, Korea has been overprotective of the patent term of original drugs,” said an official from a Korean pharmaceutical company. ”I think it's good that the law has been revised so that we can keep pace with the global pharma and biotech industry.” “The extended patent life was virtually impossible to overcome,” the official said, adding, ”The revised law is expected to play a positive role in the national health insurance finances by accelerating the time to market generics.” However, some domestic pharmaceutical companies that own original products were not so quick to welcome the change. “Compared to the past, more domestic pharmaceutical companies have succeeded in developing original drugs. In the future, there will be more companies that own originals,” he said, adding, ”In such a situation, competition with other domestic pharmaceutical companies will inevitably intensify if the patent term is shortened.” 14-year upper limit wet on patent term after marketing authorization...will shorten the total patent term The Patent Act passed by the National Assembly has two main objectives. One is to place a cap of 14 years on the remaining term from the date of drug approval. Previously, the limit for extending the patent term was stipulated to 5 years, but there was no provision for a cap on the total term, including post-authorization patent extensions. On the other hand, the United States and China limit the effective patent term to 14 years and Europe to 15 years. The Korea Intellectual Property Office promoted the introduction of a cap on the effective patent term for equity and international harmonization with other countries. Even if a particular pharmaceutical company is granted a patent term of 20+5 years (normal patent term + extension period), the cap is set at 'up to 14 years from the time of marketing authorization, thus shortening the overall patent term. For example, Pfizer's ALK-targeted anti-cancer drug Xalkori (crizotinib) has a 14-year patent term in the U.S. after FDA approval. Due to this cap, Xalkori was only granted a patent extension of 1 year and 6 months (547 days) in the US. In Korea, however, there is no such cap, so Pfizer was granted a full patent extension. The patent extension period for Xalkori in Korea is 2 years and 10 months (1,034 days), which is about 1 year and 4 months (16 months) longer than in the US. 1One patent extension granted per item...favorable for generic companies The law was also amended to allow generic companies to choose only one patent to extend out of the many patents registered for a single drug. Generally, the original company registers as many patents as possible when developing a drug. A single drug may have more than 10 patents, including product patents, use patents, dosage forms patents, method and dosage patents, formulation patents, and crystalline form patents. The more patents a company has, the better it is to defend its product against generic challenges. This is why only one of the patents registered for a drug can be extended in the U.S. and Europe. For example, for the rheumatoid arthritis drug Xeljanz (tofacitinib), Pfizer applied for a patent extension in the U.S. for its product patent from among several patents. As a result, the patent term for Xeljanz became 20+5 years. In Korea, on the other hand, if multiple patents are registered for a drug, each patent can be extended. In fact, Pfizer applied for an extension (up to five years) for 2 product patents and 1 formulation patent in Korea. An overlap occurred when all 3 patent extensions were granted simultaneously. As a result, the patent term for Xeljanz in Korea is approximately 2 years (732 days) longer than in the US and Europe. The benefits to Pfizer of having 2 more years of Xeljanz patent term in South Korea are significant. For one thing, the company can avoid the price reductions that would come with the introduction of a generic version of Xeljanz. The price of the original drug was reduced by 70% in the first year and 53.55% in the second year upon the release of the generic, but Pfizer delayed this price cut by 2 years. By delaying the launch of the generic, the company can maintain its market monopoly for 2 more years. Considering how the annual sales of Xeljanz are around KRW 15 billion, Pfizer avoided a loss of KRW 12.8 billion + α for 2 years.
- Company
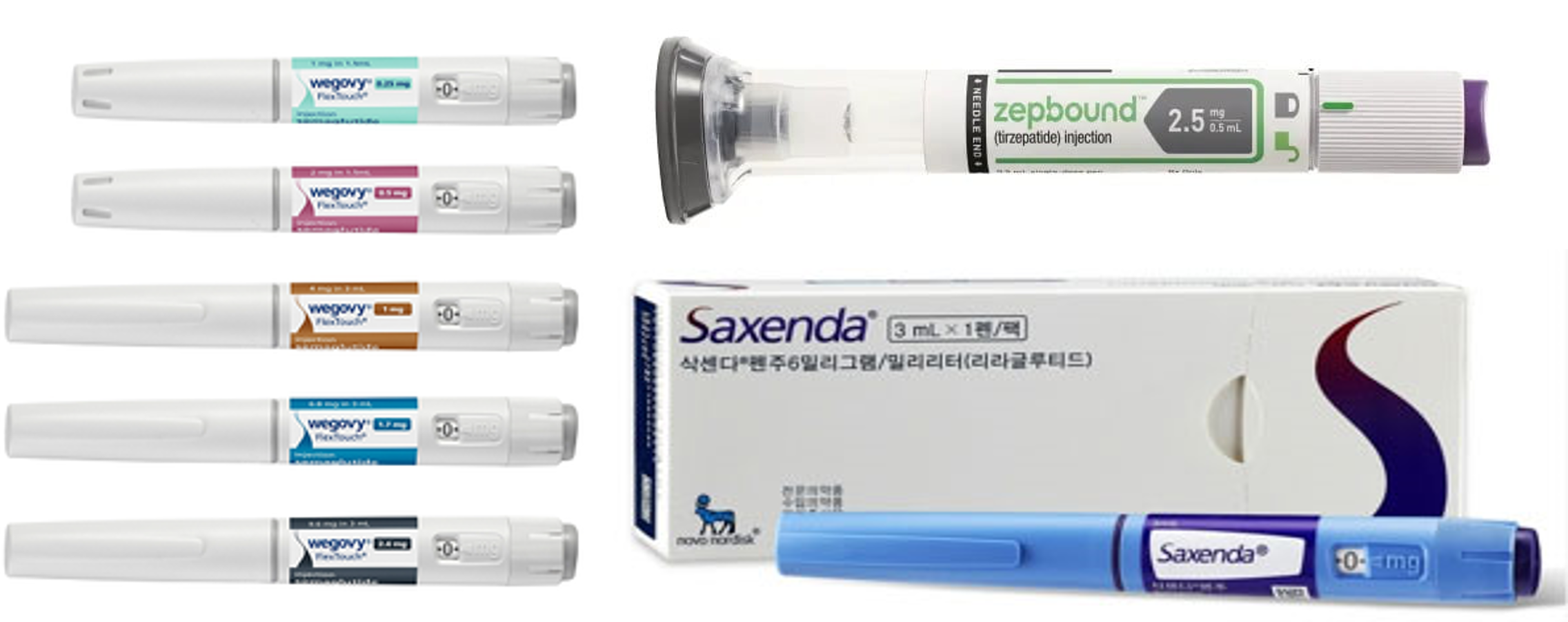
- Generic drugs mkt heat up as GLP-1 obesity meds demand soar
- by Son, Hyung Min Dec 30, 2024 05:57am
- It has been reported that not only new drugs but also generics have joined the competition in the market for glucagon-like peptide-1 (GLP-1)- containing treatment for diabetes and obesity. In the United States, generic versions of liraglutide, which is an active ingredient of the diabetes obesity·treatments Victoza·Saxenda that are GLP-1 receptor agonists. In addition to the development of generic versions of liraglutide, generics of dulaglutide, an active ingredient of Trulicity, are under development. Due to the benefits of effective weight loss with GLP-1 agonists, obesity treatment usage is significantly increasing. Several GLP-1 agonists for treating obesity, including semaglutide and liraglutide, are in shortage worldwide. The shortage in supply is due to a surge in demand for the drug. Consequently, regulatory authorities expect that generic versions of diabetes·obesity drugs that are GLP-1 agonists will likely to resolve drug shortages. The second diabetes drug containing liraglutide has been approved in the United States Novo NordiskAccording to industry sources on December 30, the U.S. Food and Drug Administration (FDA) has granted approval to a Victoza generic developed by a British pharmaceutical company, Hikma Pharmaceuticals. This approval is the second for a generic GLP-1 agonist following Teva's generic for Victoza. Teva Pharmaceuticals launched an authorized generic of Victoza after signing an agreement with Novo Nordisk. Victoza is a GLP-1 agonist for diabetes treatment developed by Novo Nordisk. It was first introduced to the market after obtaining approval in the United States in 2010. After discovering the drug's weight loss effect during a clinical trial, Novo Nordisk introduced obesity drug containing the same active ingredient to the market in 2014. Currently, GLP-1 agonist drugs, including liraglutide and semaglutide, are in global shortage. Regulatory authorities, such as the FDA, have announced that they will prioritize the review of approval applications for generic versions of originals, which will resolve the issue of limited patient access. The latecomers to the market anticipate gaining a market presence due to a surge in GLP-agonist demands. These developers plan to challenge the market by introducing the drugs at lower prices than the originals. Pre-launched Teva's Victoza generic is sold at a 15% lower price than the original drug. Analysis suggests that not only latecomer new drugs but generics will continue to compete fiercely in the market, as GLP-1 agonists are demonstrating effectiveness in treating metabolism dysfunction-related MASH and brain diseases, in addition to diabetes and obesity. Currently, GLP-1 agonist drugs are leading the market for diabetes and obesity treatments. Lily's Mounjaro, a GLP-1 agonist for diabetes, recorded sales of KRW 6.8482 last year, up 971.2% from 2022. The sales of Mounjaro, which was launched in May 2022 and generated the sales of KRW 640 billion in the same year, soared last year. Novo Nordisk's GLP-1 agonist drugs for diabetes also showed growth last year. Ozempic recorded sales of KRW 5.048 trillion last year, up 52% from the previous year. The sales of Rybelsus, containing the same active ingredient, amounted to KRW 1.44 trillion, up 140% from 2022. Saxenda generated sales of KRW 1.2252 trillion last year, up 9.8% from the previous year. Despite the shortage, Wegovy exceeded KRW 300 billion in sales last year. Pharmaceutical companies are closely watching the market for generic versions of GLP-1 agonists Korean companies are also closely watching the market for GLP-1 agonist generics. Handok has successfully launched a generic version of Saxenda, developed by the Indian biotech company Biocon, in South Korea. Both company signed an exclusive domestic sales·distribution agreements for Saxen generic in May. Biocon obtained approval for its first generic of Saxenda in the UK in March and launched the drug this month. Along with Biocon, the Indian companies Dr. Reddy's and Cipla and the Swiss company Sandoz are developing a generic version of liraglutide for diabetes and obesity treatments. Korean and overseas pharmaceutical companies are also focusing on semaglutide and dulaglutide, in addition to liraglutide. Semaglutide is an active ingredient of Novo Nordisk's diabetes treatment, Ozempic, and obesity treatment Saxena. Dulaglutide is an active ingredient of Lily's diabetes treatment Trulicity. Wegovy·Zepbound·Saxenda, GLP-1 agonists for obesity In June, the Chinese company Boan Biotech applied to the Chinese regulatory authority for approval of its generic version of Trulicity. This represents the first approval application for a generic version of Trulicity. Currently, generic versions of GLP-1 agonists for diabetes and obesity are continuing to be developed. Last year, a Chinese company successfully received approval for its Saxenda generic. Sam Chun Dang Pharm is targeting semaglutide generic. In June, the company signed an exclusive sales agreement with a Japanese pharmaceutical company for an oral generic version of semaglutide. The deal entails sales condition after the substance patent of semaglutide expires. In Japan, semaglutide's substance patent expires in 2031. Since 2018, Sam Chun Dang Pharm has been developing a generic version of liraglutide using its formulation-changing technology, called 'S-PASS.' Sam Chun Dang Pharm states that the company can avoid the formulation patent because it has been developing biosimilars and generics that avoid the patents of original formulations. The company recently confirmed the drug equivalence through a pharmaceutical equivalence test.
- Policy
- Movizolo Tab subject to drug shortage prevention program
- by Lee, Tak-Sun Dec 27, 2024 05:56am
- Pharmbio Korea’s Movizolo Tab, a prucalopride succinate-based drug used to treat chronic constipation, will be designated as a drug shortage prevention drug, resulting in an increase in drug price. Until now, prucalopride-based drugs have faced supply challenges due to low drug prices. According to industry sources on January 26, Pharmbio Korea’s Movizolo Tab will be designated as a drug shortage prevention drug as of January next year. The first formulation among the same ingredient drugs to be commercialized was YooYoung Pharm’s Rucalo Tab. The original drug is Korea Janssen's Resolor Tab, however, the drug has been supplied without reimbursement as of now. YooYoung became the first to become reimbursed amount a prucalopride succinate-based drugs in 2020. The problem arose afterward. The price of the drug was directly reduced ex officio by 53.55% of the original price when a generic drug was approved. YooYoung has been challenging the enforcement of the price cut through lawsuits, but the reduction in the reimbursement price was unavoidable. Since then, the company has repeatedly stopped supplying Rucalo Tab due to rising costs. The same goes for generic drugs that are covered under Korea’s reimbursement system. Generic drugs have also struggled to keep up with the rising costs. From 6 reimbursed drugs available for the ingredient, only YooYoung Rucalo Tab and Pharmbio Korea’s Movizolo remain. Most of the licensed drugs are currently distributed without reimbursement. In this situation, Pharmbio has applied for designation as a drug shortage prevention drug for its Movizolo Tab. If accepted, the company will be able to preserve Movizolo Tab’s manufacturing costs. However, it is reported that YooYoung Pharmaceutical, the manufacturer of Rucalo Tab, did not apply separately for its designation as a drug shortage prevention drug. As a result, the price of Pharmbio’s Movizolo Tab 1mg will increase from KRW 70 to KRW 114 and 2 mg will increase from KRW 102 to KRW 202 starting in January. It will be interesting to see if this will address the chronic shortage of reimbursed prucalopride succinate drugs.
- Product
- Price of Pulmican, Pulmicort, Tantum Sol to be raised in Jan
- by Kang Hye-Kyung Dec 27, 2024 05:56am
- As of January 1 next year, items including Pulmican, Pulmicort, and Tantum Sol which are set to receive price hikes, will demand the pharmacists' attention. According to a notice from the Ministry of Health and Welfare on the 24th, the price of ▲ Whanin Imipramine HCl Tab 25mg ▲ Lithan Tab ▲ Myungin Lithium Carbonate Tab ▲ Myungin Lithium Carbonate Tab 150mg ▲ Whanin Trazodone HCl Cap ▲Spiracton Tab 50mg ▲ Pulmican Suspension for Nebulizer ▲ Pulmicort Respule 0.5mg/2mL ▲ Sama Tantum Sol. (100mL) ▲ Movizolo Tab 1mg ▲ Movizolo Tab 2mg ▲ Lavopa Inj ▲ Pine Inj 1000IU/mL ▲ Greencross Heparin Sodium Inj ▲Pine Inj 5000IU/mL ▲ Alkeran Tab will be increased. The price of Pulmican Suspension for Nebulizer will be increased by KRW 126 from 1,121 won to 'KRW 1,247,’ and Pulmicort Respule Nebulized Suspension will be increased by KRW 255 to 'KRW 1,380.’ Sama Tantum Sol's price will increase by KRW 311 from KRW 1,000 to 1,311. There are also items whose drug prices will be reduced. ▲ Epyztek PFS, ▲ Epyztek IV Inj. ▲ Olumiant Tab 2mg, ▲ Olumiant Tab 4mg, ▲ Tremfya Prefilled Syringe Inj, ▲ Tremfya One Press Auto-Injector Inj, ▲ Rinvoq ER Tab 15mg, ▲ Rinvoq ER Tab 30mg, ▲ Entresto Film Coated Tab 100mg, ▲ Entresto Film Coated Tab 200mg, ▲ Entresto Film Coated Tab 50mg, ▲ Alprolix Injection, ▲ Sugar Tree XR Tab, ▲ Vegzelma In, ▲ Pomalyst Cap 1mg, ▲ Pomalyst Cap 1mg 2mg, ▲ Pomalyst Cap 3mg, ▲ Pomalyst Cap 4mg, etc. are included for price cuts. Meanwhile, the prices of Pulmicort Respule Nebulized Suspension and Pulmican will be raised in just one year, as the public-private council has recently requested a preemptive drug price increase to secure domestic imports as Pulmicort supply and demand in global markets such as Japan and Canada.
- Company
- First RSV vaccine Arexvy Inj lands in Korea
- by Whang, byung-woo Dec 27, 2024 05:56am
- Arexvy, GSK's global sales driver as a No. 1 adult RSV vaccine, is set to launch in Korea. Pic of Arexvy GSK Korea's Respiratory Syncytial Virus (RSV) vaccine Arexvy was recently approved by the Ministry of Food and Drug Safety (MFDS) for the prevention of lower respiratory tract disease (LRTD) caused by RSV in adults aged 60 years and older. After setting the milestone as the first RSV-LRTD vaccine, the vaccine was approved by the U.S. Food and Drug Administration (FDA) in May last year and then extended to those 50-59 years of age in June. The approval was based on results from two Phase III studies, RSV OA=ADJ-006 and RSV OA=ADJ-004, in adults aged 60 years and older. Study results showed, Arexvy significantly reduced the risk of RSV-LRTD by 82.6% and the risk of severe RSV-LRTD by 94.1% compared to placebo in subjects 60 years of age and older. In addition, vaccine efficacy for RSV-A-associated LRTD events and RSV-B-associated LRTD events was 84.6% and 80.9%, respectively. In addition, in 2 RSV seasons (from 15 days after the first dose in the Northern Hemisphere to the end of the second season), the mean follow-up period was 17.8 months, with 67.2% efficacy for RSV-LRTD and 78.8% efficacy for severe RSV-LRTD in patients aged 60 years and older. “RSV infections can be severe and even fatal in high-risk groups, such as the elderly, and bring a major social burden,” said Dr. Won Seok Choi, professor of infectious diseases at Korea University Ansan Hospital. ”Global health authorities in the U.S., U.K., and elsewhere are continuing efforts to prevent simultaneous outbreaks of RSV, influenza, and COVID-19.” “Older adults are at greater risk of infection due to age-related immune decline and a greater likelihood of complications,” said Professor Choi. ”Cross-infection can occur between young children and elderly grandparents, so practicing good personal hygiene and getting vaccinated early can help keep you, your family, and society safe.” Arexvy generated KRW 2 trillion in global sales last year...Records No. 1 in global share Currently, Arexvy’s global sales are on an upward trajectory, driven by its title as the first RSV vaccine. According to GSK's earnings release, Arexvy’s global sales in the second quarter were GBP 62 million (KRW 108 billion). Given that the RSV season is in the third and fourth quarters, sales are expected to become greater. Last year, the company's total revenue was GBP1.15205 billion (approximately KRW 2 trillion) GSK has raised its revenue forecast for 2024 based on the growth of its RSV vaccine and expects revenue to increase by 7-9% over last year. In particular, the company’s share is expected to grow despite the arrival of competing vaccines from Pfizer and Moderna. In the short term, the company plans to increase its global presence outside of the U.S. and European markets, including Japan. This includes South Korea, where it recently received approval from the Ministry of Food and Drug Safety. Arexvy is available in a vial containing the antigen (powder) in pre-fusion form of the RSV F-protein and a vial containing the immunostimulant (AS01E, suspension), where 0.5 mL of the reconstituted vaccine is administered intramuscularly using a needle. It can be administered in combination with the non-immune-boosting inactivated seasonal influenza vaccine, and the CDC recommends RSV vaccination for adults aged 75 and older or high-risk groups between 60 to 74 years of age. “RSV infections are a significant physical and economic burden for high-risk groups, including the elderly, and GSK is committed to the successful launch of Arexvy to not only prevent infection in adults but also to reduce the burden of disease for patients in Korea,” said Hyunji Kwon, Head of the Vaccines Business Unit at GSK Korea.