- LOGIN
- MemberShip
- 2026-04-04 13:41:29
- InterView
- "Recruiting a company to co-develop pitavastatin+ezetimibe"
- by Kim, Jin-Gu Oct 30, 2024 05:54am
- Seung Hee Baek, Shinil Pharmaceutical Shinil Pharmaceutical will collaborate with other biopharmaceutical company to co-develop a generic 'pitavastatin+ezetimibe' combination drug for treating hyperlipidemia. Through joint-collaboration, Shinil Pharmaceutical plans to improve not only the contract manufacturing of medicines but also cost savings and productivity. In the meantime, the company plans to teach its construction technology for active ingredients and production know-how to companies participating in joint development in addition to out-licensing. "The pitavastatin+ezetimibe combination drug is set to enter the generics market in 2027," Seung Hee Baek (52), Shinil Pharmaceutical's Executive Director who is directing the co-development·out licensing project, said. "We started to prepare for the current project amid searching for a new sales approach following the implementation of 1+3 bioequivalence regulation." 'pitavastatin+ezetimibe' combination drug sales are skyrocketing…Sinil has completed the bioequivalence test for its generic drug, set to enter the market The company's pitavastatin+ezetimibe combination drug for treating hyperlipidemia received approval under the name Livalozet (JW Pharmaceutical). Livalozet sales are skyrocketing in the prescription market. According to the pharmaceutical market research firm UBIST, Livalozet generated sales of KRW 70.4 billion in prescription sales, more than twice the previous year's growth. Up to September 2024, it generated KRW 66.8 billion in sales. It is expected to surpass last year's prescription record. It seems that Korean pharmaceutical companies have started to show interest in these generics, as Livalozet has shown strong sales in the prescription market. During 2020-2021, many generics containing pitavastatin+ezetimibe received approvals. Livalozet generics are currently subjected to reconsideration. When the reconsideration period ends after 2027, product launch is possible. Sinil Pharmaceutical focused on pitavastatin's low statin side effects and significant treatment effects. The company has already completed a bioequivalence test and is set to enter the market. "The advantage of pitavastatin is that it lowers the risk of diabetes than other statin agents," Baek said. "Sinil Pharmaceutical had selected the pitavastatin+ezetimibe combination as an in-house project. In April 2024, the company demonstrated the bioequivalence of the drug to the control drug through a bioequivalence test." "Searching for a company to co-develop…we will make a technology transfer, including active agent designing·production know-how" Sinil Pharmaceutical will begin technology marketing based on its completed bioequivalence demonstration. The company plans to secure a co-development company and make a technology transfer to the partner, including active agent designing·production know-how·how-to prepare documents for approval. Baek anticipated that such a co-development·technology transfer strategy would be a win-win for both Sinil Pharmaceutical and its partnering company. Baek explained that Sinil Pharmaceutical could benefit from improving its profitability by sharing the development cost. A co-development company can learn various know-how during the technology transfer process. In addition, a co-development company can appoint a CMO company. "Sinil Pharmaceutical conducts the process of product development·approval·production in-house. We can make a technology transfer of necessary technology for each stage up to launching a product," Baek emphasized. "Partnering company can secure various know-how, including the production technology needed in a plant and CTD documentation for applying for approval, in addition to active ingredient designing that made Sinil Pharmaceutical successfully complete the bioequivalence test." Baek emphasized that 'timing is a key to generics development.' Baek explained that the timely development of drugs secures high drug pricing and establishment in the market. Additionally, a company has to consider the cost-effectiveness of the development for an agent that is difficult to demonstrate bioequivalence. "Sinil Pharmaceutical has already demonstrated a bioequivalence of its pitavastatin+ezetimibe combination," Baek emphasized. "Considering cost-effectiveness, joining one of the three co-development companies will provide benefits." Changes occurred following the 1+3 bioequivalence regulatory policy…"It's our new sales strategy" Sinil Pharmaceutical strategized this approach following the 1+3 bioequivalence regulatory policy, which the government implemented in July 2021. The policy requires that a bioequivalence document can be used up to three times when a manufacturing plant produces a pharmaceutical that is equivalent to one for which a pharmaceutical company conducted a bioequivalence test. Only four generics can receive approval based on a single bioequivalence test. After the implementation of the policy, CMO companies faced significant changes, Sinil Pharmaceutical likewise. Concerns have risen in the company that profitability could be lowered because the company can appoint only three CMOs companies. "Sinil Pharmaceutical has been actively developing first-generics," Baek said. "After the implementation of the 1+3 policy, the company had to find a different sales approach for its CMO business." Baek explained, "Recruiting only three CMO companies can hinder development, but on the bright side, expanding to technology sales can be a win-win strategy for both CMO and consignment companies." Sinil Pharmaceutical plans to expand its business field based on the current joint development·technology transfer experience. "We have tried technology transfer of previously manufactured products after the implementation of the 1+3 policy. However, we had difficulty starting a business because too much information had already been shared," Baek said. "For a new product, it is different. We plan to pursue technology transfer sales for a new product actively."
- Company
- Difficulty expanding indications for antibody-drug conjugate
- by Moon, sung-ho Oct 30, 2024 05:54am
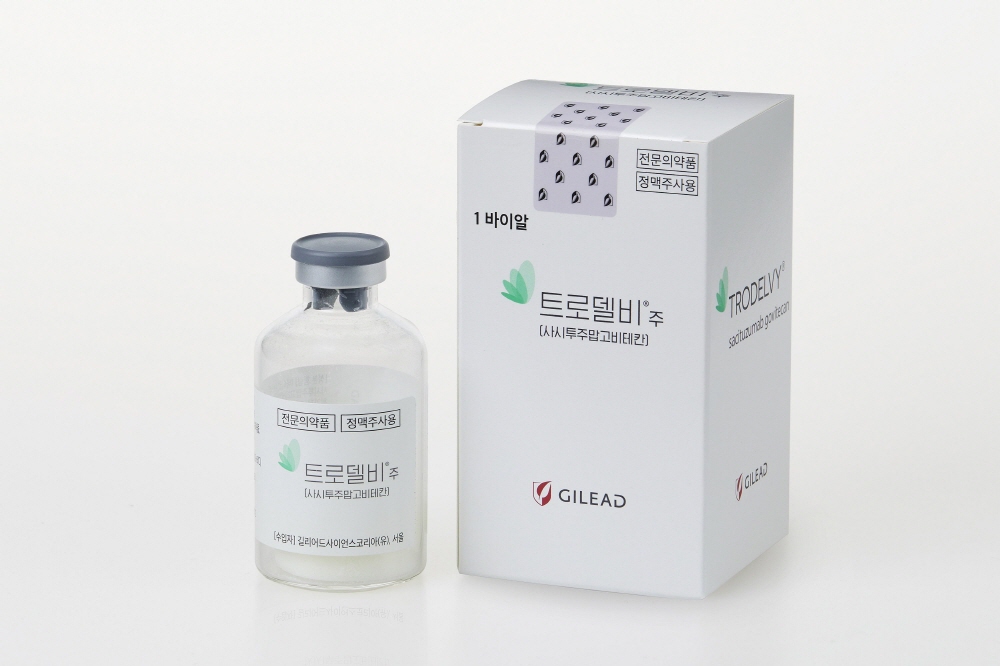
- Drug candidates that received much expectation as next-generation antibody-drug conjugates (ADCs) based on the success of Enhertu are on a struggling path to reimbursement in Korea. Although the companies aimed to expand the scope of coverage of their drugs based on their effectiveness in specific cancer types, the expectations damped somewhat due to the disappointing clinical research results. Nevertheless, the domestic and international pharmaceutical and bio industries are attempting to develop new drugs by starting clinical research on the combined use of ADCs and immuno-oncology drugs. The TROP-2 class of drugs has been receiving the most attention as the next generation of ADCs in the global pharmaceutical and biotechnology industries and clinical sites. The leading products in the market include TRODELVY® (sacituzumab govitecan-hziy, Gilead), and datopotamab deruxtecan (AstraZeneca, Daiichi Sankyo). However, these candidates have recently struggled to expand their indications. According to industry sources on the 25th, Gilead recently formalized the ‘abandonment’ of its application to expand Trodelvy’s indication to urothelial cancer in consultation with the US Food and Drug Administration (FDA). Troldelvy binds to the Trop-2 protein, which is highly expressed in a variety of cancers including breast cancer, and releases the drug inside the tumor cells, destroying not only the tumor cells but also the tumor microenvironment, with minimal impact on healthy cells. Currently, it has demonstrated efficacy in advanced triple-negative breast cancer (TNBC) and HR+/HER- breast cancer. However, the company abandoned its other promising indication - urothelial carcinoma. The company failed to demonstrate overall survival (OS) in the TROPiCS-04 study, a Phase III confirmatory trial that was part of the conditional approval. The Keytruda (pembrolizumab, MSD) and Padcev (enfortumab vedotin, Astellas) combination therapy, which has been introduced in Korea as a first-line treatment option for urothelial carcinoma, stood out in the trial, demonstrating its relative effect. Another TROP-2 class ADC, datopotamab deruxtecan, Dato-DXd, is also facing difficulties. The final OS analysis HR+/HER2 low- or negative-expressing breast cancer did not show a statistically significant improvement over chemotherapy. At the same time, it failed to achieve statistical significance in OS compared to docetaxel in advanced/metastatic NSCLC. Following the failure of Trodelvy in demonstrating survival benefits in metastatic stage IV NSCLC, datopotamab deruxtecan is also struggling to gain traction in the lung cancer space. However, AstraZeneca and Daiichi Sankyo have applied to the FDA for the approval of datopotamab deruxtecan for advanced/metastatic non-squamous NSCLC, as they believe it could be a new treatment option. As such, the FDA is expected to make a decision on whether to approve datopotamab deruxtecan for NSCLC by December 20th. Dr. Min-Hee Hong, professor of Medical Oncology at Severance Hospital, said, “If you think about the role of ADCs in NSCLC, nothing has been approved so far. “However, if you look at Trop-2-targeted ADC studies, there is some talk that some subgroup analysis results were good. But whether the company can get FDA approval with the data remains in question, as it didn't meet its primary endpoint.” “I think it shows what we call “cherry picking” in the medical community, where they're really cherry-picking the data that they want to show, so I think it's fair to say that there's no approved drug and no role for the drug at this point. But we'll have to wait and see because the FDA’s decision hasn't been announced yet.” While global pharmaceutical companies' next-generation ADC candidates are struggling to expand their scope, the domestic pharmaceutical and biotech industry's presence in ADC development has been growing. The Korean pharmaceutical and biotechnology industries are attempting various clinical trials with the combination of ADCs and immuno-oncology drugs, which are being actively conducted around the world and are leading to actual technology export results. Gilead, AstraZeneca, and Daiichi Sankyo are already attempting clinical studies with immuno-oncology drugs such as Keytruda or Imfinzi (durvalumab) in combination with Trodelvy and datopotamab deruxtecan. One representative domestic company is LigaChem Biosciences. LigaChem Biosciences is a company specializing in ADC development that has signed a total of 10 technology transfer agreements. Starting with the technology transfer to China's Fosun Pharma in 2015, the company successfully exported its LCB84 technology to Janssen in December last year. LCB84 is an ADC candidate that can target various solid cancers, including triple-negative breast cancer and non-small cell lung cancer. LigaChem Biosciences also recently announced that it has signed 2 technology transfer agreements with Japan's Ono Pharmaceutical Industries, including LCB97, which targets the L1CAM protein. The total value of the two agreements is worth over USD 700 million (KRW 943.5 billion), with the specific upfront payment amount undisclosed as per the parties' agreement. LCB97 targets L1CAM, a protein expressed in several solid tumors, including lung, pancreatic, and colorectal cancers. In February of last year, LigaChem Biosciences entered into a research and development agreement with Elthera AG in Switzerland for the L1CAM antibody, securing worldwide rights, including for ADC use. In addition, various domestic biotech companies such as NeoImmuneTech and TXINNO Bioscience are attempting clinical research with the concept of ADC and immuno-oncology combination therapy. A domestic pharmaceutical and biotech industry official said, “The success of the Padcev+Keytruda combination attracted much attention to the combination of ADCs and immuno-oncology drugs. And the combination’s reimbursement is gaining great interest in Korea. Although it has no commercialized items yet, LigaChem Biosciences gained attention for signing licensing agreements with global big pharma companies such as Ono Pharmaceutical, Janssen, and Amgen. This is because ADCs are being regarded as the next cash cow in the global market.”
- Policy
- Gvnt agrees on the need for dispensing substitute drugs
- by Lee, Jeong-Hwan Oct 30, 2024 05:54am
- In this year's NA Audit, the Minister of Health and Welfare Kyoo-Hong Cho said that he would prioritize the ‘dispensing of substitute drugs’ as a solution to the problem of unstable supply of medicines such as cold medicines that are often out of stock. Two bills related to alternative dispensing were submitted to the National Assembly on the 28th, including a bill to amend the Pharmaceutical Affairs Act, which was introduced as representative by Rep. Byung-Duk Kim and Rep. Soo-Jin Lee of the Democratic Party of Korea. The bills aim to enable pharmacy pharmacists to substitute prescribed drugs with other generic versions that have undergone bioequivalence tests certified by the head of the Ministry of Food and Drug Safety as having the same ingredients, formulation, and dosage as the drug listed in the doctor's prescription. By clarifying whether or not a pharmacist should notify the doctor of the substitution, the bill is intended to promote information sharing among pharmacists and improve convenience in prescribing and dispensing medicines. Specifically, Rep. Byung-Duk Kim's bill expands the post-notification subject for the pharmacists’ dispensing of substitute drugs to the Health Insurance Review and Assessment Service, while Rep. Soo-Jin’s bill expands the notification to HIRA and changes to the term from “dispensing of substitute drugs” to “international nonproprietary name prescriptions.” The 22nd National Assembly is regarded as being more proactive in addressing the issue of the unstable supply of drugs by changing the substitute dispensing method, as it has introduced 2 bills to activate the practice early in its term, compared to the 1 bill submitted during the 21st National Assembly. In addition, Rep. Young-Seok Seo and Rep. Yoon Kim of the Democratic Party of Korea are also considering legislation to streamline the dispensing of substitute drugs and international nonproprietary name prescriptions, so there is a possibility that more related legislation will be introduced in the future. In addition, Minister Cho is expected to actively participate in the parliamentary review of relevant legislation, as he had cited the promotion of dispensing substitute drugs as a solution to the unstable supply and demand of drugs after the COVID-19 pandemic. However, opposition from the medical community is one barrier that must be overcome. Doctors argue that increasing substitute dispensing could adversely affect patient health, as different products with the same ingredients, whether original or generic, can have different clinical effects when taken by patients. Nevertheless, lawmakers who agree with the need to promote dispensing substitute drugs say that the legislation is essential to overcome the 5-year-long problem of unstable drug supply. Some doctors find it difficult to understand why the government would consider promoting dispensing substitute drugs, which can hinder public health when it is already operating an incentive system that provides incentives to pharmacies for dispensing low-cost substitute drugs. “We shouldn't have another case of stalled legislation due to inter-functional differences among professionals about dispensing substitute drugs,’ said an official from the Health and Welfare Committee, adding, “The problem of unstable drug supply and demand has been infringing on the public's access to medicines for several years now, deteriorating public health.” “Many Health and Welfare committee members are in agreement with the need for legislation, and some are considering expanding the scope of INN prescriptions,” said the official, “As Minister Cho answered in the NA Audit that he will prioritize INN prescriptions, we plan to focus on passing the legislation together with the MOHW.”
- Company
- Dupixent sales skyrocket with 'expanded indications'
- by Son, Hyung Min Oct 30, 2024 05:54am
- Sales for 'Dupixent,' a biological agent developed by Sanofi, continue to skyrocket. Net sales for Dupixent from Q1 to Q3 of 2024 reached 10 billion euros. The analysis suggests that Dupixent's added indications to treat various immune diseases, including atopic dermatitis, asthma, esophagitis, and chronic obstructive pulmonary disease (COPD), have contributed to its continued sales increase. According to Sanofi's performance report on October 29, Dupixent's Q3 global sales amounted to 3.476 billion euros (about KRW 5.2 trillion), up 18.1% year-over-year (YoY). Dupixent's sales for 2024 were 9.614 billion euros (about KRW 14.4 trillion), up 24.5% compared to Q3 of last year. Dupixent After recording 2.835 billion euros in Q1 2024, it surpassed 3 billion euros in Q2. Dupixent generated 3.476 billion euros in Q3, up 115.4% compared to 1.614 billion euros in Q1 2022. Dupixent is the first biological agent to target the signaling transmission of interleukin (IL)-4 and IL-13, which are the main causes of type 2 inflammation. Dupixent was developed from a global collaboration agreement between Sanofi and Regeneron Pharmaceuticals. The drug received marketing authorization as a treatment for atopic dermatitis in various countries, including South Korea, the United States, Canada, Europe, Japan, and Australia. Launched in the global market in 2018, Dupixent continues to generate sales growth. After recording 107 million euros in Q1 2018, it generated more than 1 billion euros (about KRW 1.5 trillion) in Q1 2022. Since then, Dupixent has grown into a blockbuster drug with annual sales of more than 10 billion euros (about KRW 15 trillion). Sanofi expects Dupixent's yearly sales to reach around 13 billion euros this year. Confirmed safety and efficacy…added indications contributing to sales hike Dupixent, SanofiDupixent is regarded as a biological agent that has proven extended efficacy and safety. Unlike conventional immune inhibitors, this drug reduced the number of side effects in clinical and real-world data. It has the efficacy in targeting only the inflammatory responses of Th2 cells. Biologics are medicines that are manufactured based on pathogenic microorganisms such as proteins, antibodies, nucleotides, and cells. This agent effectively targets proteins and cells to treat diseases, and it has the advantage of lowering side effects compared to chemical agents. Dupixent's other advantage is its continued addition of indications. In 2017, the U.S. Food and Drug Administration (FDA) granted approval to Dupixent for its indication to treat atopic dermatitis. The following year, it expanded prescription areas to treat patients with moderate or higher symptoms. In the United States, Dupixent is the only asthma drug to target all ages. Since then, Dupixent has expanded the scope of approval to various immune diseases, including chronic sinusitis with nasal polyps, nodular rashes, and eosinophilic esophagitis. Dupixent has recently added an indication to treat COPT. On October 27, the FDA approved Dupixent for the treatment of COPD. Dupixent is the first biological agent to secure a COPD indication. Dupixent has shown strength in diseases triggered by type 2 inflammation, from atopic dermatitis to asthma, chronic sinusitis with nasal polyps, and eosinophilic esophagitis. It is expected to add additional indications for diseases caused by similar mechanisms. Currently, Sanofi is conducting a Phase 3 clinical study of Dupixent, aiming to secure indications to treat chronic idiopathic urticaria (CSU), chronic pruritus (CPUO), and bullous pemphigus (BP).
- Product
- KMA ‘non-face-to-face Wegovy prescriptions cause harm'
- by Kang, Shin-Kook Oct 30, 2024 05:53am
- A Korean doctor's association has called for an immediate halt to the government's plan to allow non-face-to-face medical treatment. The Korean Medical Association (Chairman Hyun-Taek Lim) issued a statement on the 28th claiming, “The government should immediately stop allowing all non-face-to-face medical treatment as its pilot project and ensure that the non-face-to-face treatment pilot project is operated only as a supplementary means for returning patients, under the principle of face-to-face treatment.” The Ministry of Health and Welfare implemented a pilot project for non-face-to-face treatment in June last year, focusing on clinic-level medical institutions and returning patients who have received face-to-face medical treatment, and then allowed overall non-face-to-face treatment as a pilot project in February this year. In response, the Korean Medical Association pointed out, “With the full permit of non-face-to-face treatment, online platforms are encouraging patients to use non-face-to-face treatment platforms through various advertisements, SNS, influencers, and other promotional activities. Rather than providing essential medical treatment services for returning patients that have received face-to-face treatment, patients are being seriously induced to receive non-reimbursed initial medical treatment related to beauty such as hair loss, diet, and acne through online medical platforms.” “In the case of Wegovy, an injectable obesity drug that has been controversially overprescribed through non-face-to-face online platforms, the manufacturer suggests gallstones, hair loss, and indigestion as common side effects, and in rare cases, fatal side effects such as pancreatitis can occur, so it is a specialty drug that should be prescribed through BMI criteria. However, due to the full allowance of non-face-to-face medical treatment, consumers, not patients, can easily acquire and abuse specialty drugs, and we would like to question whether this is in line to allow non-face-to-face treatment,’ said KMA. It added, “To prevent the distortion of the medical market and indiscriminate prescriptions caused by non-face-to-face treatment and the damage to public health, we need to restrict prescription of non-face-to-face treatment for drugs that should be prescribed after strictly identifying the patient's condition. As such, the government should strengthen the monitoring system for online platform companies and prepare measures against factors that may disrupt the medical market.”
- Opinion
- [Reporter's View] Super precision medicine
- by Eo, Yun-Ho Oct 30, 2024 05:53am
- Super Precision medicine. New drugs have been developed to selectively target with more significant efficacy than before. Lately, news articles related to anticancer agents have frequently covered keywords related to genetic mutations, including MET, RET, ALK, EGFR, and ROS1. Customized treatments are becoming available, where effective treatments can be provided depending on the patient's particular genetic mutations. Pharmaceuticals targeting HER2, ALK, and EGFR have been developed, and they show superior effects in patients with particular conditions. Lately, pharmaceuticals have been developed selectively targeting difficult-to-develop gene mutations, such as ROS, NTRK, and RET. Such development of super precision medicines suggests that prescription criteria will likely change from 'disease' to 'genes'. A customizable healthcare era has arrived. However, it is still unfamiliar. Will the South Korea successfully incorporate these drugs that show efficacy regardless of cancer type once gene mutations in patients are confirmed? Previously listed targeted anticancer drugs and immune checkpoint inhibitors have faced difficulties in expanding reimbursement. One such difficulty is the high costs. However, when a drug's new use is discovered, the values and the volume of usage must be re-assessed. The process is required by the National Health Insurance system in South Korea. However, recently developed new drugs target very few patients; in other words, the number of identified gene mutations cases is few. This means that not many patients are likely to be prescribed for these new drugs. In South Korea, patients with rare diseases comprise below 1% of overall solid cancer cases. Less than 200 patients are diagnosed. Experts explain that these types of patients do not respond well to standard therapies (conventional drugs). The problem is reimbursement. Anticancer agents targeting gene mutations mentioned earlier have already been approved domestically. However, most of these drugs are still non-reimbursable. Pharmaceutical companies have continued to apply for coverage, but their attempts 'failed.' It is time to consider a reimbursement track for super precision medicine. Rather than going against the current system, we must establish different reimbursement review criteria for drugs that can be used in patients regardless of their cancer types.
- Company
- "Early intervention" needed for treating multiple myeloma
- by Whang, byung-woo Oct 29, 2024 05:49am
- The survival rate of patients with multiple myeloma increased following new drug development. However, concerns have been raised that South Korea's survival rate is still far from that of advanced countries. Expert opinions indicate that the Korean medical treatment field changes with new drug approvals and reimbursement listings, yet patient access remains restricted. Dr. Chang Ki Min, a Professor of Hematology at Seoul St. MaryDuring the conference commenced by Janssen on October 28th, Dr. Chang Ki Min, a Professor of Hematology at Seoul St. Mary's Hospital, emphasized the need for early treatments customized to the nature of multiple myeloma. According to the Health Insurance Review and Assessment Service (HIRA), the number of Korean patients with multiple myeloma is on the rise due to the aging population. Multiple myeloma patients were 7,063 in 2017, and the number increased to 11,219 patients in 2023. "The average multiple myeloma onset is 60 years or higher. As the aging population continues to increase, the number of multiple myeloma patients will likely increase as well," Dr. Min explained. The problem is that remission of multiple myeloma is difficult to achieve. Despite undergoing treatments, patients are likely to experience relapse. Therefore, treatment focuses on selecting effective treatments in each stage and extending the progression-free survival period. Typically, treatment duration shortens as the number of treatments increases. For example, 95% of patients undergoing first-line treatment relapse, 15% of patients undergoing fourth-line treatment relapse, and the percentage drops to 1% by the time patients receive fifth-line treatment. "When multiple myelomas relapse after several years following treatment at an early stage, the disease becomes chronic. It is important to undergo effective treatment at early stage so that the disease does not advance to secondary, tertiary, etc," said Dr. Min. As new drugs are being developed for treating multiple myeloma, a variety of treatment options are available. Originally starting from simple chemotherapy, the treatment evolved to combination therapies, including proteasome inhibitors, immune modulators, and high-dosage steroids. Recently, new treatments, such as anti-CD38 monoclonal antibodies and BCMA-targeted immune therapy, became available. The survival rate following the development of treatments for multiple myeloma (based on a document presented by Dr. Chang Ki Min) In South Korea, new treatment options are emerging as new drugs for treating multiple myeloma receive the Ministry of Food and Drug Safety (MFDS) approvals: elranatamab in May and Talvey in June. However, the limitation to utilizing such treatments is that the National Health Insurance reimbursement is limited. A 'DVTd combination therapy (Darzalex+bortezomib+thalidomide+dexamethasone),' containing Darzalex (daratumumab), recently received an appropriateness decision from the Drug Reimbursement Evaluation Committee (DREC) for expanding reimbursement as the first-line treatment of multiple myeloma. However, analysis suggests that the medical field still needs overall improvement. "Within the limited government fund, many pharmaceuticals are covered by reimbursement. Yet, there still needs improvement based on the global standard," Dr. Min said. "Despite having good treatment options, including existing medications and new drugs, patient access is limited. Thus, it requires improvement." The survival rate of patients with multiple myeloma in South Korea and several advanced countries (based on a document presented by Dr. Chang Ki Min) The survival rate of Korean patients with multiple myeloma is 51%, showing a 10% difference compared to that of advanced countries, 61% in the United States and 62% in Germany. Consequently, the Korean medical field is changing due to new drug approvals and reimbursement listings. Various approaches may be needed to improve treatment outcomes, such as considering treating the disease at an early stage and optimizing the order of treatments. Dr. Min has suggested an improvement plan as ▲Early use of the monoclonal antibody combination therapy ▲Triple combination drug that can be used in patients who are non-responsive to Lenalidomide ▲A new class treatment for patients relapse or refractory to over third-line therapies. It suggests that administering a new drug that would significantly improve the survival rate at an early stage could achieve both the treatment effects and the National Health Insurance finance. "We understand the problem of limited National Health Insurance finance, but we expect that early administration of an effective treatment method would reduce relapse rate and increase the survival rate, thereby potentially reducing the treatment cost," Dr. Min said. "Most patients currently undergo 6th and 7th treatments. Using effective treatments at an early stage will, in turn, be effective for the National Health Insurance." Lastly, Dr. Min added, "It would be difficult for patients to fully experience the treatment effects when effective medications are used in patients with poor conditions after the 3rd and 4th treatments. By administering treatments early, we expect to see the global standard therapy results."
- Company
- Will the third time be the charm for Mylotarg?
- by Eo, Yun-Ho Oct 29, 2024 05:49am
- The industry’s attention is focused on whether the insurance reimbursement discussions for the acute myeloid leukemia drug ‘Mylotarg’ will make progress this time. According to the industry sources, Pfizer Korea’s acute myeloid leukemia (AML) drug ‘Mylotarg (gemtuzumab ozogamicin)’ is set to be submitted to the Health Insurance Review and Assessment Service’s Drug Reimbursement Evaluation Committee. However, it is not yet confirmed whether the agenda it will be presented in November. Mylotarg’s reimbursement agenda was deliberated by HIRA’s Cancer Disease Review Committee in May 2022 but was unable to set reimbursement standards then. It passed the CDDC review in October last year, but subsequent discussions had collapsed. Pfizer resubmitted the application for its reimbursement in June, which passed the Pharmacoeconomic Evaluation Subcommittee. Mylotarg is an antibody-drug conjugate (ADC) indicated for the first-line treatment of newly diagnosed acute myeloid leukemia whose tumors express the CD33 antigen (CD33-positive AML) . The drug, which received marketing authorization in Korea in December 2021, is an ADC composed of a CD33-targeting monoclonal antibody linked to calicheamicin, a potent cytotoxic agent. The drug works on cells that express the CD33 antigen, which is expressed in more than 90% of AML patients. This blocks cancer cell growth and induces apoptosis. Mylotarg’s approval was based on a clinical trial (ALFA-0701) conducted on 271 patients aged between 50 to 70 with newly diagnosed AML with no prior treatment experience. The ALFA-0701 trial was an open-label, randomly assigned, multicenter Phase III study that compared the existing standard front-line chemotherapy, daunorubicin+cytarabine combination therapy, with Mylotarg+ daunorubicin+ cytarabine combination therapy. Results showed that the median event-free survival (EFS) in the Mylotarg+daunorubicin+cytarabine combination therapy group was 17.3 months, a 7.8 month extension compared to the 9.5 months in the daunorubicin+cytarabine combination therapy group. Also, the Mylotarg combination therapy reduced the risk of induction failure, relapse, or death by 44% compared to chemotherapy alone. The median relapse-free survival (RFS) was 28.0 months in the Mylotarg+daunorubicin+cytarabine combination therapy group and 11.4 months in the daunorubicin+cytarabine combination therapy group, showing a significant 16.6-month difference with the addition ofMylotarg. In the case of median overall survival (OS), the median OS was 27.5 months in the Mylotarg+daunorubicin+cytarabine combination therapy group and 21.8 months in the daunorubicin+cytarabine combination therapy group, but there was no statistically significant difference between treatment arms in OS. Meanwhile, Mylotarg is administered intravenously under close monitoring over 2 hours at the recommended dose with dilution depending on the treatment regimen, including remission induction therapy and remission maintenance therapy. Mylotax was approved by the U.S. FDA in September 2017 and by the European Medicines Agency (EMA) in April 2018.
- Company
- ABL Bio receives ₩7B as milestone payment from Sanofi
- by Kim, Jin-Gu Oct 29, 2024 05:49am
- ABL Bio has received additional milestone payments from Sanofi for its degenerative brain disease treatment candidate ‘ABL301’. Including the upfront payment., ABL Bio will receive a total of KRW 170 billion, which includes the upfront payment. On 28 February, ABL Bio announced the completion of the manufacturing technology transfer of ABL301 to Sanofi and the receipt of USD 5 million (KRW 7 billion) as a milestone payment. ABL Bio signed a licensing-out agreement with Genzyme for ABL301, a bispecific antibody candidate for the treatment of Parkinson's disease and other neurodegenerative diseases. Genzyme is a fully-owned subsidiary of Sanofi. The total value of the agreement was estimated to be approximately $1.06 billion. As part of the agreement, ABL Bio received a non-refundable upfront payment of USD 75 million (KRW 100 billion). In September of the same year, the company received an additional USD 20 million of the USD 45 million in milestone payments for completing preclinical studies. In January last year, the company received an additional USD 25 million as a milestone (KRW 35 billion) for administering the first dose in the Phase I clinical trial. This, coupled with an additional USD 5 million (KRW 7 billion) in milestone payments for the transfer of manufacturing technology, brings the cumulative amount received from Sanofi to more than $125 million (KRW 170 billion). ABL Bio is eligible to receive up to an additional USD 940 million in other milestone payments based on further clinical, regulatory, and commercial achievements. In addition, upon successful commercialization of ABL301, the company may receive an agreed-upon percentage of net sales as an ongoing technology fee (annual fee). ABL301 is currently in a Phase I clinical trial in the U.S. led by ABL Bio. Upon completion of Phase 1 trials, Sanofi will be responsible for initiating Phase II trials. Sanofi retains global development and commercialization rights from Phase II trials. ABL301 is a bispecific antibody that uses ABL Bio’s Grabody-B platform technology to effectively deliver antibodies that inhibit the build-up of alpha-synuclein, the protein that causes Parkinson's disease, into the brain for improved treatment effect. The company's Grabody-B platform leverages the IGF1R (Insulin-like Growth Factor 1 Receptor) to maximize the blood-brain barrier (BBB) penetration of therapeutic candidates for various central nervous system diseases.
- Policy
- Low reimb approval rate hinders Soliris’s use for aHUS
- by Lee, Tak-Sun Oct 29, 2024 05:49am
- The industry’s eyes are on whether the preliminary reimbursement review process for Soliris, a treatment for the rare disease aHUS (atypical hemolytic uremic syndrome), will be eased in Korea. Until now, patients wishing to use Soliris for aHUS with reimbursement had to pass a preliminary review process. However, the problem is its low approval rate. With an approval rate in the 30-40% range every year, it has been a regular topic of discussion in the medical field and at the NA audit. This year, the issue was again raised during the NA audit, and attention has been drawn to the fact that HIRA expressed its intention to conduct a prospective review of the process. According to industry reports on the 27th, Rep Yoon Kim, a member of the Democratic Party of Korea, said to HIRA, “Soliri Inj, a treatment for atypical hemolytic uremic syndrome, has a very low approval rate at the preliminary reimbursement review committee process. If it cannot be used because of too strict standards, the standards need to be improved.” “As it is an acute disease, its use should not be bound by the preliminary review system, but should be started first, and the decision on whether to continue the treatment should be made afterwards.” aHUS is a severe, rare, hereditary disease where 79% of patients die within 3 years of onset, require dialysis, and suffer permanent renal failure. Without Soliris, it can be fatal. However, there are many cases of pre-approval rejections, rendering it difficult to treat patients in the field. However, HIRA has been citing the lack of required application data from long-term care institutions as the reason for the low approval rate, and that there was no problem with the reimbursement standards and review process. However, whether Rep. Yoon Kim’s criticism will improve the system is gaining attention. In its response to the NA’s written inquiry, HIRA said, ‘We will collect opinions from relevant societies on whether first allow the initial treatment with “Soliris Inj,” a drug for atypical hemolytic uremic syndrome, and then conduct preliminary review for further administration of the drug.” However, it seems that the authorities will keep the preliminary review system will be maintained. NA Rep. Jong-Heon Baek pointed out, “It seems inappropriate to operate the pre-review system for aHUS, as it is an acute rare disease. In other countries, considering the characteristics of acute rare diseases, they do not place them in the pre-review system or process the review very quickly. Korea is the only country that does not consider such characteristics of the disease.” To this, HIRA only responded, ‘Considering the nature of the disease, the drug can be administered immediately after submission of the pre-review application if it is an emergency case, and if approval follows, It is retroactively applied.” This suggests that the authorities are not considering plans to convert the preliminary review process to regular review. The implication is that they will consider exempting the initial dose only. In November, the government decided to switch the preliminary review process for the reimbursement of Soliris and Ultomiris for PNH (Paroxysmal nocturnal hemoglobinuria) to regular review. This is because the existing approval rate has remained stable at over 90%. Upon the change in their reimbursement process, there have been growing calls in the medical community for the removal of prior authorization aHUS in addition to PNH.