- LOGIN
- MemberShip
- 2026-04-04 13:41:29
- Company
- SK bioscience invests KRW 4.1B stake in a U.S. biotech
- by Kim, Jin-Gu Oct 10, 2024 05:51am
- SK bioscience announced on October 8th that the company has signed an agreement to acquire a stake in U.S. biotech Fina Biosolutions by investing USD 3 million (approximately KRW 4.1 billion). SK bioscience became the first and only strategic investor of Fina Biosolutions. According to the contract between both companies, the specific stake acquisition size remains undisclosed. According to SK bioscience, Fina Biosolutions, established in 2006, has the core technology of conjugate vaccines for pneumoniae, meningococcus and typhoid. Fina Biosolutions is known to manufacture 'CRM197 (cross-reacting material),' a protein carrier important for the conjugate vaccine development, and produce it in high yield. CRM197 conjugates to antigens that induce infectious disease prevention and enhance immune responses. Fina Biosolutions has developed 'EcoCRM' to enhance productivity, which is higher than that for CRM197, using its in-house expression system and filtration technology. Additionally, the company is researching to increase immunogenicity and productivity using the next-generation CRM197 technology, which targets an antigen-binding site. Fina Biosolutions provides various carrier proteins, including CRM197, in collaboration with various biotech companies and agencies, including the U.S. Inventprise and companies in China and India. Furthermore, the company is expanding its business by providing antigen·dextran (carbohydrate-derived polysaccharides) conjugate service using its specialty in conjugate technology. SK bioscience, which owns conjugate vaccines for pneumoniae·typhoid, plans to utilize Fina Biosolutions' CRM197 technology. The company aims to increase profitability by securing the high preventative benefits of conjugate vaccines and high-yield production. "We appreciate the confidence shown by SK bioscience," Fina Biosolutions CEO Andrew Lees said. "With the investment agreement, we anticipate acceleration of global commercialization and conjugate vaccine development." "We are pleased to continuously pursue the partnership opportunities with global companies that have next-generation vaccine technologies," Jaeyong Ahn, CEO of SK bioscience. "Thorugh long-term collaboration with Fina Biosolutions, we will enhance the quality of vaccines in development and enhance our competitiveness for global entry." SK bioscience has recently made active investments to create a synergy with global companies with technologies. The company also signed an agreement to acquire the business rights of a German company, 'IDT Biologica,' which ranked among the top 10 global vaccine CMOs in June and wrapped up the acquisition process earlier this month. In June SK bioscience conditionally acquired a stake in a U.S. biotech company, 'Sunflower Therapeutics.' Sunflower Therapeutics has its own 'yeast-based culturing system' required for antigen·antibody development. "We plan to continue securing competitiveness by making investments and M&As in companies with superior technologies to establish steppingstones as a global company," SK bioscience personnel said.
- Company
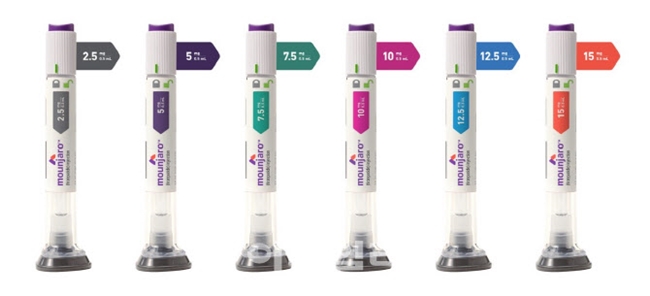
- Who will distribute Mounjaro in Korea?
- by Whang, byung-woo Oct 10, 2024 05:50am
- As Novo Nordisk's obesity drug Wegovy (semaglutide) is set to be released, the industry’s interest is on which domestic distributor Lilly will choose for its Mounjaro (tirzepatide). 마운자로 제품사진. According to industry sources on the 8th, Boryung Pharmaceutical and Chong Kun Dang are in the running to become domestic distributors of Wegovy’s rival obesity drug Mounjaro. Zuellig Pharma Korea, which handled Novo Nordisk's existing obesity drug Saxenda, was announced as the domestic distributor of Wegovy. The supply price is set at KRW 370,000 for 4 weeks’ worth. The reason why the competition amongst domestic distributors for Mounjaro is so interesting is the sales potential of obesity drugs. Due to its global craze, its sales are likely to have a positive impact on the sales of domestic distributors as well. In 2023, global sales of Wegovy reached USD 4.5 billion (about KRW 6 trillion), while Zepbound (the U.S. brand name for the Mounjaro’s obesity indication), which was launched in Q4 last year, generated sales of UDS 176 million (KRW 240 billion). In June last year, Mounjaro received approval as an adjunct for chronic weight management, about a year after it was approved for type 2 diabetes. In the SURMOUNT-1 and SURMOUNT-2 trials which became the basis of approval, all doses showed statistically significant weight loss results compared to placebo, and the rate of achieving weight loss of 5% or more was also higher in the Mounjaro arm than in the placebo arm. Lilly’s decision to add the obesity indication to its existing product rather than file a new drug application is also expected to accelerate its launch. The industry is currently expecting a May launch next year. The first pharmaceutical company being considered as a possible distributor for Mounjaro is Boryung. It has a distribution agreement for Lilly's diabetes drug Trulicity and therefore is expected to have an advantage in Mounjar, which is a similar drug. In addition, Boryung has acquired the domestic rights to the patent-expired cancer drugs Gemzar (gemcitabine) and Alimta (pemetrexed) and the schizophrenia drug Zyprexa (olanzapine) from Eli Lilly in 2020-2022. ‘We have been in charge of Trulicity’s distribution since 2016 after signing a sales agreement with Lilly. However, signing a new drug partnership is a completely different matter, therefore it is difficult to confirm any facts,’ said a Boryung official. Chong Kun Dang, which has experience in distributing obesity drugs, is also in the running. Currently, Chong Kun Dang is the co-promoter and distributor of Alvogen's obesity drug Qsymia. Qsymia was approved in the US in 2012 for the treatment of obesity in adults. It is a combination of phentermine, a short-term appetite suppressant, and topiramate, a neurotherapeutic agent. It was introduced in South Korea in late 2019. “From Lilly's point of view, experience in distributing obesity drugs will be important when choosing a domestic distributor. I think we are being considered because we have experience in distributing the obesity drug Qsymia,” said a Chong Kung Dang official.
- Policy
- "Preferential drug pricing excludes K-made new drug benefits
- by Lee, Jeong-Hwan Oct 10, 2024 05:50am
- The government's revised plan for the drug pricing system has been criticized for reverse discrimination against Korean pharmaceutical companies because it was primarily designed to favor multinational pharmaceutical companies. The criticism concerns the revised drug pricing system plan, announced by the Health Insurance Review and Assessment Service (HIRA) in August, that the plan excludes the core measure in favor of Korean pharmaceutical companies. Baek Jong-heon, a member of the People Power Party, questioned this during the National Assembly's audit of the Ministry of Health and Welfare (MOHW) on October 8th. In December 2023, the MOHW reported to the Health Insurance Policy Review Committee regarding potential improvements to the drug pricing system to reflect the new drug's innovative value and the nation's healthcare security. In February 2024, the MOHW announced the expansion of preferential drug pricing criteria for pharmaceutical companies as part of the 2nd comprehensive National Health Insurance Plan 2024. The MOHW's plan was reviewed by the Drug Reimbursement Evaluation Committee (DREC), and the MHOW announced in August a revision to the specific evaluation criteria for pharmaceuticals during the negotiations process. However, Baek pointed out that the plan excludes 'Preferential drug pricing for new drugs developed by pharmaceutical companies with high R&D contributions' and 'Improved measures to calculate drug pricing to support domestically produced new drug exports.' Because of the announcement of a revised plan for preferential drug pricing, excluding measures to provide benefits to Korea-made new drugs, Jeil Pharm's new drug developed by Onconic Therapeutics, listed in September, suffered reduced export pricing. Document prepared by Baek Jong-heon, a member of the People Power Party. If the plan had been revised according to the Health Insurance Policy Review Committee report, the drug could have been listed under 'essential reimbursement pricing.' However, due to delays in drug pricing system improvement, the company did not receive the benefit. Baek questioned Cho Kyoo-hong, Minister of Health and Welfare, about the rationale for excluding a measure that provides practical benefits to Korean pharmaceutical companies from the revised plan, despite Korean pharmaceutical companies requesting improvements to the current policy related to new drug development and exports. "As part of the 120th national agenda, the Yoon Suk Yeol government has decided to foster bio-health business as the core export business, aiming to leap as a leading global country in the bio·digital health field," Baek said. "The MOHW announced the revised plan without taking into account practical supporting measures." "MOHW's role is to lead the growth and development of the Korean biopharmaceutical industry and enhance international competitiveness, thereby contributing to the nation's health and welfare improvement," Beak emphasized.
- Opinion
- [Reporter's View]Industry expectations rise with new changes
- by Eo, Yun-Ho Oct 10, 2024 05:50am
- The criteria for innovative new drugs that are eligible for the flexible application of the ICER threshold and the measures for the expansion of the risk-sharing agreement (RSA) have been revealed. As always, the industry expressed a mix of expectations and concerns. The disclosed ‘Detailed evaluation criteria for new drugs etc. subject to negotiations’ goes as follows. Firstly, the requirements for ‘innovativeness’ to qualify for the flexible application of the ICER threshold were established. A new drug is regarded as ‘innovative’ when it satisfies all of the following criteria: ▲ there is no substitutable or therapeutically equivalent product or treatment ▲ a significant clinical improvement, such as prolonged survival, is recognized in the final outcome, ▲ the new drug is approved by the MFDS under Article 35(4)(2) of the Pharmaceutical Affairs Act through expedited review or is recognized as equivalent by the committee. The criteria are not very different from what was proposed in the ‘Preferential treatment measures for innovative new drugs’ that was first released last year. The difference is that in that draft, the U.S. FDA Breakthrough Therapy Designation (BTD) and the European EMA Priority Review (PRIME) were required, but this time, only the MFDS’s GIFT (Global Innovative products on Fast Track) designation is required. This is certainly encouraging. Of course, most truly innovative drugs receive BTD and PRIME designations. However, the fact that only MFDS’s GIFT is required as a criterion is, in many ways, removing the hurdle for innovative new drug designations. The most notable changes were made in the RSA eligibility criteria. The amendment expands the scope of the second condition in the RSA criteria, which was ‘drugs eligible for special calculation or equivalent,’ to further specify the ‘or equivalent’ diseases. The ‘or equivalent’ part was specified to severe diseases that do not qualify for the special calculation but are difficult to cure, irreversible disability or organ damage due to the progression of the disease, and hold a significant disease burden. In addition, if the expected additional claim amount for refund-type RSA-type drugs is less than KRW 1.5 billion within the scope of the reimbursement standard expansion, the drug may omit Drug Reimbursement Evaluation Committee evaluations and conduct NHIS negotiations. However, if the drug falls under the second RSA criteria, a new condition was added, stipulating that the expenditure cap must be applied even if it is not a pharmacologic evaluation exemption drug. This raises the potential for a number of issues. Firstly, the removal of the ambiguous phrase ‘special cases or equivalent’ is welcome. The clinical criteria of irreversible disability and organ damage also seem to be specific enough. However, it is unclear how many drugs will be eligible for less than KRW 1.5 billion additional claims criteria. Simplifying the process of expanding the RSA reimbursement criteria has been long desired by the industry, the problem is that while there weren't many drugs that exceeded that 'total amount' in the past, things have changed as we've entered the era of high-priced drugs. Furthermore, the criteria that set all drugs that fall under the second condition to be applied the expenditure cap RSA is a concern. In fact, recently listed drugs are being contracted with combined type RSA that includes the expenditure cap type. The problem is that while there weren't many drugs that exceeded that 'total expenditure cap’ in the past, things have changed as we've entered the era of high-priced drugs. In this situation, it remains to be seen whether the unilateral application of the ‘expenditure cap’ for drugs falling under the second criteria will lead to smooth reimbursement listing.
- Policy
- Yoon Kim ‘40% of the generic drug price is a bubble’
- by Lee, Jeong-Hwan Oct 10, 2024 05:50am
- Kyoo-hong Cho, the Minister of Health and Welfare, positively responded to Democratic Party of Korea Representative Yoon Kim's request to lower the price of generic versions of patent-expired drugs within the year. This refers to a separate generic drug price cut rather than the foreign drug price referencing reevaluations, which the pharmaceutical industry and the MOHW are currently discussing the timing and method of implementation, and has increased the tension amongst domestic pharmaceutical companies on another imminent generic drug price cut. At the National Assembly's MOHW audit on the 8th, Representative Yoon Kim pointed out that there is too much bubble in Korea’s generic price. “There is a significant bubble in the price of generic drugs in Korea,” said Kim, adding, “In a study commissioned by the Welfare Ministry, if you look at how high the price of generic drugs are in Korea is compared to developed countries such as the United States, the United Kingdom, Germany, and France, about 40% is a bubble.” “In monetary terms, about KRW 4 trillion more is being spent by Korea every year on generic drugs compared to developed countries,” he said, asking, “Does the ministry have any plans to adjust generic drug prices?” “A few months ago, I received a report from the Ministry of Health and Welfare’s Division of Health Insurance Benefits that they were planning to implement price cuts this year. When I asked them again recently, I was told that nothing was in progress,” said Kim. “Please reduce the price of generic drugs.” Minister Kyoo-hong Cho responded to Kim's request that the ministry has the intention to reduce the price of generic drugs. ‘We are considering adjusting generic drug prices. The agenda is being reviewed together in linkage with the sustainability of Korea’s national health insurance finances" When asked by Kim to push for a reduction in generic drug prices, Cho briefly replied, “Yes.”
- Company
- Wegovy's release on the 15th…competitor Mounjaro
- by Whang, byung-woo Oct 08, 2024 05:49am
- With the release of Wegovy, the next-generation obesity drug, set for the middle of this month (October), the release of its competitor Mounjaro is also gaining attention. Although Lilly has not yet announced a specific launch date for its Mounjaro, the company has opted to add another indication to its drug rather than apply for new drug approval, which is raising expectations on the company’s early launch scenario. Pic of Mounjaro Novo Nordisk recently announced that it will launch Wegovy (semaglutide) in the Korean market from the 15th of this month. This will be the 9th global launch in a year and a half since the drug was approved by the Ministry of Food and Drug Safety in April last year. With the launch of Wegovy, a representative next-generation obesity treatment, near, attention is also being drawn to the release of Mounjaro (tirzepatide), which was approved by the MFDS in August. As there is a time gap between the global approvals of Wegovy and Mounjaro, the timing of the domestic launch of the two is also set to differ. Even so, the industry is awaiting the release of Mounjaro due to the company’s method of approval. Lilly received approval for its Mounjaro as an adjunct to chronic weight management in June last year, about a year after receiving approval for the type 2 diabetes indication. In the U.S., Lilly received approval for the obesity treatment separately under the brand name Zepbound, just as a separate brand called Ozempic is approved for the diabetes indication of Wegovy. In Europe, as in Korea, Lilly received approval for both the diabetes and obesity indications with one product, Mounjaro. The industry interpreted Lilly's approach as a choice made to accelerate the approval timeline, although it may not necessarily mean an early launch. ‘It's much easier to get multiple indications approved for one product if you don't have to brand the product separately,’ said a pharma industry insider. “Given that the drug showed significant weight loss in the diabetes clinical data already showed, and that the product is already approved in the US and Europe, adding an indication to an existing product is faster.” He added, “Adding an indication only requires safety and efficacy review, but new approvals are allocated a longer time allocated for review, including GMP site assessment, which makes a difference in the speed of (approvals). In general, it is rare for an existing product to receive approval for a new indication as a separate product in Korea.” In other words, it's Lilly's choice whether to add indications to a product and seek new approvals, but in terms of speed of approvals, adding indications is much easier. The company is also expected to have considered the fact that MFDS’s inspection timeline for overseas manufacturing sites is scheduled to be after 2027. In conclusion, there is no doubt that Lilly's decision to add an indication for Mounjaro gave the company an option to ‘choose’ the timing of its launch rather than wait for approval. The industry is now speculating on Mounjaro’s launch in the first half of next year, with a shortlist of potential domestic distributors. The idea is that the company will seek to enter the market before Wegovy has a chance to reap the benefits of first-mover advantage. However, the industry believes that the distributors that are being discussed based on their relationship with Lilly or their experience in distributing obesity drugs need to be watched further. In addition, even if Mounjaro is launched immediately, it is a different matter whether sufficient quantities can be supplied to Korea, which also requires review from various aspects. Lilly Korea is of the view that the launch date should be determined before discussing whether to select a distribution partner. “Both Wegovy and Mounjaro are expected to compete with a greater focus on obesity treatment,’ said another pharmaceutical industry insider. “However, as they have different indications for type 2 diabetes and cardiovascular disease, their impact is likely to vary depending on the patient condition.”
- Company
- Ilaris can be prescribed at general hospitals in KOR
- by Eo, Yun-Ho Oct 08, 2024 05:49am
- Ilaris, which was listed for reimbursement 9 years after receiving marketing authorization in Korea, can now be prescribed at general hospitals in Korea. According to industry sources, Novartis Korea's hereditary recurrent fever syndrome drug Ilaris (canakinumab) has passed the drug committees (DCs) of Seoul National University Hospital, Seoul St Mary's Hospital, and Sinchon Severance Hospital. Ilaris, which was approved in Korea in 2015, has been on the reimbursement list since August. The company quickly accepted the conditional reimbursement decision made by the MFDS’s Drug Reimbursement Evaluation Committee in April and then promptly concluded the difficult drug pricing negotiation process. ILARIS is an interleukin-1 (IL-1) inhibitor recommended for the treatment of CAPS in the 2021 international guidelines from the European College of Rheumatology and the American College of Rheumatology. In a Phase III study, the drug demonstrated significant clinical benefit in remission after a single dose and remission rate at 6 months and can be administered every 8 weeks in patients with CAPS, improving the quality of life for patients and their caregivers. In the CLUSTER study, which included 46 patients with TRAPS and 63 patients with colchicine-resistant FMF, 45% (n=10/22) of TRAPS patients treated with Ilaris 150 mg and 61% (n=19/31) of colchicine-resistant FMF patients achieved a complete response at week 16. "All three of these indications reimbursed for Ilaris are extremely rare diseases, and many patients suffer through a diagnostic odyssey that prevents them from receiving an accurate diagnosis," said Dae-Chul Jeong, professor of Pediatrics at Seoul St. Mary's Hospital, "and even after diagnosis, their treatment options were very limited, which was frustrating for them and us as medical professionals. Patients and us healthcare providers alike have been waiting for the reimbursement coverage of Ilaris for a long time, so the reimbursement approval came as encouraging news.” Ilrais is indicated for the following diseases in Korea: ▲Periodic fever syndromes (PFS), cryopyrin-associated periodic syndromes (CAPS), tumor necrosis factor receptor-associated periodic syndrome (TRAPS), hyperimmunoglobulin D syndrome (HIDS)/mevalonate kinase deficiency (MKD), and familial mediterranean fever (FMF) ▲Active systemic juvenile idiopathic arthritis (Systemic JIA). For CAPS, the indication can be further categorized into the following symptoms: ▲Familial cold autoinflammatory syndrome (FCAS)/ familial cold urticaria (FCU) ▲Muckle-Wells syndrome (MWS) ▲Neonatal onset multisystem inflammatory disease (NOMID)/chronic infantile neurological, cutaneous and articular syndrome (CINCA). With such a small patient population and complex indications, making progress in the reimbursement discussions for the drug had not been easy. The number of patients eligible for the various indications of ILARIS is extremely small. Some indications for ILARIS do not even have disease codes or have only recently been registered.
- Policy
- Daewoong wins nod for first generic version of Ibrance tab
- by Lee, Tak-Sun Oct 08, 2024 05:49am
- Daewoong's first generic for Pfizer's breast cancer treatment, 'Ibrance Tab,' has been approved. Due to the successful patent nullification, the company received priority marketing authorization. The first generic can be launched in March 23rd, 2027, when the product patent expires. The Ministry of Food and Drug Safety (MFDS) has granted approval of Daewoong's "Ranclib tab" 75 mg, 100 mg, and 125 mg. This drug can be used to treat hormone receptor (HR)-positive or HER2-negative metastatic or advanced breast cancer. It is used in combination with aromatase inhibitor as first-line endocrine therapy in women or in combination with fulvestrant after endocrine therapy in women with advanced diseases. PfizerPfizer's Ibrance containing palbociclib is the original medicine. Ibrance was the first CDK4/6 inhibitor to be developed. It was launched with reimbursement coverage in November 2017 through the risk-sharing agreement (RSA). Unlike capsules, tablets can be taken regardless of food. The film-coating of the tablet allows for co-administration with proton pump inhibitors (PPI) or antacids, which are used to manage gastrointestinal disorders or diarrhea that commonly occur in breast cancer patients. Overall, tablets provide easier administration convenience than capsules. Ibrance's 2023 sales were KRW 50.5 billion, according to IQVIA. Due to the drug's high marketability, Korean pharmaceutical companies are attempting at early market entries through patent challenges. In addition to Daewoong, Kwang Dong Pharmaceutical, Boryung, and Samyang have entered patent challenges. After winning the nullification trial for the ingredient patent in March, Daewoong has established a ground to launch generic once the substance patent expires. In addition to successfully winning the patent challenge, Daewoong also acquired priority marketing authorization with approval as it met the requirement for the first generic. The prior marketing authorization period is from March 23rd, 2027, the day after Ibrance Tab's substance patent expires, to December 22nd, 2027. Last year, Kwang Dong Pharmaceutical acquired the priority marketing authorization of 'Alency Cap' for the same period as Daewoong's product. However, Kwang Dong Pharmaceutical won the priority marketing authorization against Ibrance Cap rather than Ibrance Tab. Additional approvals are expected soon as other companies that have challenged the patent have won. At the launch, Ibrance had no competitors in the market for breast cancer. However, the sales are slightly decreasing as similar CDK4/6 inhibitors Kisqali (Novartis, ribociclib) and Verzenio (Lily, abemaciclib) have launched. The analysis suggests that the market size may be downsized upon generic launch. "Although Ibrance generates the highest sales in the CDK4/6 inhibitors market, the sales have been gradually decreasing," pharmaceutical industry personnel said. "Sales could drop even more when generics launch following the substance patent expiration. Korean pharmaceutical companies could have launched generics earlier."
- Company
- Eisai Korea signs Alzheimer's MOU with the Korean Dementia
- by Whang, byung-woo Oct 08, 2024 05:49am
- (From left) the Korean Dementia Association President Seong Hye Choi, Eisai Korea CEO Ko Hong-Byung Eisai Korea announced on October 7th that it has signed a memorandum of understanding (MOU) with the Korean Dementia Association to utilize and manage 'JOY-ALZ,' a registry program for long-term follow-up survey platform of Alzheimer's disease prognosis. The MOU aims to advance Alzheimer's disease treatment and to establish safe Alzheimer's disease treatment settings in South Korea. According to the MOU, both companies will collaborate on establishing and running JOY-ALZ, thereby securing high-quality data on Alzheimer's disease patients. Based on this platform, they will establish and provide safe and optimized treatment settings for Alzheimer's disease patients in South Korea. JOY-ALZ is a long-term follow-up registry program that collects and registers real-world data (RWD) on the efficacy and safety of new drugs for Alzheimer's disease in South Korea. In the United States, a registry program called 'ALZ-NET' is used to follow, manage, and evaluate the efficacy and safety of RWD on new drugs for Alzheimer's disease. Collected data are utilized to report the latest diagnosis, categorization, and treatment options for Alzheimer's disease. JOY-ALZ will enable the collection of RWD information on the efficacy and safety of drugs by collaborating with overseas global platforms, such as ALZ-NET. This platform is expected to aid in systematic treatment and management. Moreover, its data gathering of Korean patients is expected to help establish treatment backgrounds considering Korean-optimized genetic and environmental factors. Based on RWD collected through JOY-ALZ, Eisai Korea plans to execute post-sales drug monitoring duty on its Leqembi, a new drug used to treat Alzheimer's disease. By submitting the long-term safety and efficacy results of Leqembi to the Ministry of Food and Drug Safety (MFDS). Eisai Korea plans to contribute to the safe use of medicines. "In collaboration with the Korean Dementia Association, we are pleased to fulfill the duty of monitoring Leqembi following the Pharmaceutical Affairs Act, as well as contribute to the advancement of treatments for Alzheimer's disease in South Korea," Eisai Korea CEO Ko Hong-Byung said. "Eisai Korea will strengthen treatment monitoring of Alzheimer's disease patients and adverse cases related to medicines. We will strive to provide safe and effective treatment options." "After years of efforts, a drug has been developed to delay the progression of Alzheimer's disease. An overseas registry has been established and is in operation to evaluate the efficacy and safety of new drug," the Korean Dementia Association President Seong Hye Choi, "If JOY-ALZ in South Korea allows for active research, new drug development, and RWD-based treatments for dementia, a global-level treatment environment for treating dementia can be established."
- Company
- Galderma Korea appoints Jai Hyuk Lee as new GM
- by Whang, byung-woo Oct 08, 2024 05:48am
- Jake (Jai Hyuck) Lee, new General Manager of Galderma Korea, Galderma Korea announced on the 7th that it has appointed Jake (Jai Hyuck) Lee, former head of the Aesthetics Business Unit, as its new General Manager, effective as of October 1. The new GM is a seasoned sales and marketing expert with more than 25 years of experience in the global pharmaceutical and aesthetics industries. Lee will succeed Younhee Kim, who led Galderma Korea for 4 years from 2020. Lee started his pharmaceutical career at GSK Korea. He has since held positions at MSD Korea, Sanofi-Aventis Korea, Novartis Korea, and Merz Aesthetics Korea before joining Galderma Korea in 2019. Over the past 5 years, Lee has successfully led the launch of new Restylane brands such as Restylane Kysse and Restylane Eyelight, as well as the renewal of the Sculptra and Dysport brands, strengthening the product portfolio and driving rapid business growth. He has also served as the interim head of the pharmaceuticals BU and the derma-cosmetics BU, accumulating a wealth of hands-on experience and expertise in non-esthetic skin diseases and skincare across all divisions of Galderma Korea. “I am very pleased to be able to represent Galderma Korea, which has grown into a leading company in the skin field under the slogan of advancing dermatology for every skin story,” said the new GM. ’Going forward, I will do my best to support the company’s growth both internally and externally by advancing the company’s organizational capabilities and expertise, while presenting innovative solutions to solve skin problems that are becoming more diverse and fragmented based on the dermatological know-how that Galderma Korea has already accumulated.’