- LOGIN
- MemberShip
- 2026-04-04 11:57:39
- Policy
- Third vitamin-based acne treatment approved in KOR
- by Lee, Tak-Sun Jan 02, 2026 07:43am
- Dongwha Pharmaceutical’s nicotinamide-based acne treatment, “Sevita B Gel”Since the first vitamin-based acne treatment was launched in Korea in 2022, other pharmaceutical companies have begun turning their attention to the market. Following Dongwha Pharmaceutical, Chong Kun Dang, and Dong-A Pharmaceutical have now also secured product approvals.On the 29th, the Ministry of Food and Drug Safety (MFDS) approved Dong-A Pharmaceutical’s “Acvita Gel” (over-the-counter drug). Acvita Gel contains nicotinamide and is used for the topical treatment of mild to moderate inflammatory acne.The recommended dosage is to apply a sufficient amount to the affected area twice daily, morning and evening. Each tube contains 30 grams.Nicotinamide is known as the active form of vitamin B3. It helps improve acne symptoms through its anti-inflammatory action.The acne-fighting effects of nicotinamide have already been confirmed in numerous studies and papers. Consequently, products containing nicotinamide, such as Nicomed Cream and Prederma Gel, have long sold overseas.In Korea, the ingredient was first introduced in June 2022, when Dongwha Pharmaceutical launched “Sevita B Gel,” a product containing the same active ingredient.Subsequently, in October last year, Chong Kun Dang received approval for ‘Dermagram Gel’, a pharmaceutical product with the same ingredient. Now, with Dong-A Pharmaceutical's ‘Acvita Gel’ added, three products have been listed.Dong-A Pharmaceutical expects Acvita Gel to generate synergies with its steady seller, “Noscarna Gel.” Noscarna Gel is used for the treatment of hypertrophic and keloid scars, acne scars, and post-surgical scars, and has established itself as a leading acne scar treatment with annual sales exceeding KRW 10 billion.The nicotinamide-based acne treatment also offers advantages over existing prescription drugs, having the advantage of not inducing resistance and being conveniently available as an over-the-counter drug. Dongwha Pharmaceutical's Sevita B Gel recently recorded high growth rates among the company’s OTC products, further boosting expectations for other products containing the same ingredient.
- Company
- Contracted generics 94%↓ over 5 years…entries suppressed
- by Chon, Seung-Hyun Jan 02, 2026 07:43am
- New entries of generic drugs have significantly decreased, with the number of items with certified bioequivalence reduced by 86% compared to five years ago. The number of generics approved through the entrustment method without conducting independent bioequivalence testing has dropped by more than 90%. Since the implementation of the tiered drug pricing system and the joint development regulation, new entries of contracted generics have been suppressed.According to the '2025 Food and Drug Statistical Annual Summary' published by the Ministry of Food and Drug Safety on the 27th, there were 333 bioequivalence-certified items last year, a 6.1% decrease from the previous year.Bioequivalence-certified items are products recognized as equivalent to the original drug, and they consist mostly of newly approved generics. Since reaching 2,358 items in 2019, bioequivalence certifications have shown a continuous downward trend, shrinking by 85.9% over the five-year period.The number of bioequivalence testing and contracted generics with certified bioequivalence by year (unit: number, source: MFDS). BLUE: in-house GREEN: contractedThe number of entries for contracted generics approved has dropped. Contracted generics refer to those approved by outsourcing the entire manufacturing process to another company without performing an independent bioequivalence study.Last year, the number of bioequivalence certifications for contracted generics was 139. While this is a slight increase from 136 in 2023, it represents a 93.9% contraction compared to the 2,277 items recorded in 2019.The reduction in contracted generics due to the reorganization of the drug pricing system and the strengthening of approval regulations led to a significant decrease in new entries, which in turn resulted in a decline in the total number of generic approvals.Under the reformed drug pricing system implemented in 2020, a tiered pricing model was introduced where the ceiling price becomes lower the later a drug is listed for reimbursement. If more than 20 generics are already registered for a specific ingredient, the ceiling price for a newly listed item is set at 85% of the existing lowest price. It is analyzed that the motivation for new approvals has been significantly dampened as the drug prices for generics entering the market late have dropped substantially.Starting from July 2021, the revised Pharmaceutical Affairs Act limited the number of incremental transition drugs and generics that can be approved using a single clinical trial. In cases where a product is manufactured using the same site, prescription, and manufacturing method as a pharmaceutical company that conducted its own bioequivalence study, the use of that bioequivalence data is limited to three times. This means that only four generics can be approved per single bioequivalence study.Previously, there were no regulations on the number of contracted generics approved per bioequivalence study, which led to an excessive number of approvals. However, as approvals were limited to a maximum of three contracted generics per study, a reduction in the number of new approvals became inevitable.Contracted generic approvals reached 515 cases in 2017 but surged more than fourfold within two years before turning downward to 1,405 cases in 2020 and 573 cases in 2021. Since 2022, the figure has remained below 200 for three consecutive years.The number of generics approved per bioequivalence study has also decreased significantly. In 2019, the number of generics approved through direct bioequivalence testing was 81, accounting for less than 4% of all bioequivalence-certified items. At that time, approximately 29 generics were approved per single bioequivalence study. However, as the number of contracted generics decreased, the number of generics approved through direct testing exceeded the number of contracted generic approvals in 2023 for the first time in 23 years since 2011.In 2011, the 543 products with completed bioequivalence studies outnumbered the 366 contracted products, but the proportion of contracted products became much larger afterward. Last year as well, the number of generics recognized through direct bioequivalence testing exceeded contracted generics by 55 cases. As of last year, 1.7 generics were approved per bioequivalence study, a 94.1% decrease compared to 2019.The industry anticipates that if the reduction of the generic drug price calculation standard is implemented, new generic entries will be further restricted.The drug pricing system improvement plan reported by the Ministry of Health and Welfare to the Health Insurance Policy Review Committee last month includes lowering the price calculation rate for generics and patent-expired drugs from 53.55% to the 40% range.Under the current drug pricing system, effective since 2012, generics receive a premium of up to 59.5% of the original drug price before patent expiration upon initial listing, which then drops to 53.55% after one year. Patent-expired original drugs are also reduced to 53.55% of their pre-expiration price. The new calculation standard for generic drugs is highly likely to be set between 40% and 45%.As the drug pricing standard was set at 45% from 53.55%, the maximum price for generics is calculated to decrease by 16.0%. If the standard is finalized at 40%, the maximum price reduction rate for generics will increase to 25.3% as the previous KRW 53.55 would drop to KRW 40.
- Company
- US and Japan ease new drug approval reviews
- by Cha, Ji-Hyun Dec 31, 2025 07:49am
- As the global biopharmaceutical market grows rapidly, competition among major countries' regulatory authorities over expedited drug reviews has intensified. As the success or failure of new drug development hinges critically on launch timing, nations are engaging in a ‘speed race’ by shortening review timelines and expanding conditional approvals. While Korea has also moved to streamline approval procedures, experts say additional measures are needed to improve the effectiveness and real-world impact of the system.According to the 2025 Biopharmaceutical Industry Trends Report published by the Korea Biomedicine Industry Association(KoBIA) on the 30th, the global biopharmaceutical market has been growing at a double-digit annual rate and has become the central pillar of the pharmaceutical industry. In 2024, the global biopharmaceutical market reached USD 632.3 billion, representing an annual growth rate of 13.6%, and is projected to grow to USD 974.2 billion by 2028.As the market expands, competition among regulatory authorities worldwide to accelerate their approval timeline has also intensified. Countries are strategically leveraging fast-track review and conditional approval pathways to expedite new drug launches and gain an edge in the global approval race.The United States and Japan are currently at the forefront of expedited review frameworks.First, the U.S. Food and Drug Administration (FDA) is establishing a multi-layered expedited review system, including ▲Fast Track, ▲Breakthrough Therapy, ▲Accelerated Approval, and ▲Priority Review.The Fast Track designation applies to drugs intended to treat serious or life-threatening conditions that address unmet medical needs. Under this program, the FDA provides more frequent communication and guidance from the development stage and allows rolling submission and review of data, minimizing bottlenecks throughout development and review.The Breakthrough Therapy designation is a more advanced program than Fast Track and is granted when a drug demonstrates or is expected to demonstrate a significant clinical improvement over existing treatments in early clinical trials. When this program is applied, the FDA assigns a multidisciplinary review team to provide intensive support across clinical, statistical, and manufacturing aspects, helping applicants to finalize development strategies at an early stage.Accelerated Approval allows early authorization based on surrogate endpoints or interim clinical indicators, without waiting for final clinical results. However, post-marketing confirmatory trials are mandatory upon approval, and the FDA may modify or revoke approval if clinical benefit is not demonstrated in these trials. This clear delineation of post-approval responsibility alongside expedited review is central to the U.S. accelerated approval system.Priority Review that shortens the review period itself after an application is submitted. It directly accelerates the launch timeline of new drugs compared to standard review. It applies to treatments for serious diseases or those expected to yield significant public health benefits.Japan adopts a strategy that directly guarantees corporate profitability. The Pharmaceuticals and Medical Devices Agency (PMDA) supports early approval of innovative drugs primarily through Priority Review and the Sakigake program, while also fostering a regulatory environment that considers the potential for market success post-approval.The Sakigake program is Japan's flagship fast-track review initiative for innovative new drugs for severe diseases that are expected to demonstrate superior clinical benefit over existing treatments. If a drug designated under Sakigake is the first in the world to apply for approval in Japan, the re-examination period can be extended up to 10 years. This effectively guarantees a long-term monopoly status equivalent to a patent, serving as a powerful incentive for global pharmaceutical companies to prioritize Japan as an initial launch market.Rather than focusing solely on speed, Europe emphasizes regulatory differentiation based on the level of uncertainty. The European Medicines Agency (EMA) concurrently operates systems like Conditional Marketing Authorisation, Marketing Authorisation under Exceptional Circumstances, Accelerated Assessment, and the PRIority Medicines (PRIME) program, applying different approval pathways based on a drug's development stage and level of clinical evidence. The Marketing Authorisation under Exceptional Circumstances system acknowledges situations such as rare diseases where confirmatory trials may be impractical even after market entry, distinguishing Europe’s approach from other regions.China is pursuing aggressive regulatory innovation centered on speed, accelerating its rise as a global biopharma powerhouse. The National Medical Products Administration (NMPA) operates multiple fast-track review systems, including Breakthrough Therapy Designation, Conditional Approval, Priority Review, and Special Approval.Among these, the Special Approval system for public health emergencies is designed to initiate acceptance and initial review procedures within 24 hours, making it one of the fastest response systems globally. Priority review timelines have also been shortened significantly, from the standard 200 days to 130 days, reflecting China’s aggressive time-reduction strategy.Korea has introduced expedited review and conditional approval systems in line with global regulatory trends. Through the GIFT (Global Innovative product on Fast Track) program, the Ministry of Food and Drug Safety (MFDS) shortens review timelines for innovative drugs and treatments for serious or rare diseases, while allowing conditional approval before full clinical evidence is accumulated.Under GIFT, the review period is reduced by approximately 25%, from 120 days to 90 days. Additionally, the system has institutionalized rolling review, where approval data is submitted and reviewed in stages, to enhance review efficiency.However, critics argue that Korea’s system still lags behind major countries in practical implementation. To achieve genuine global competitiveness, Korea’s GIFT program may require bold enhancements on par with those in leading jurisdictions. Just as Japan’s Sakigake program offers strong exclusivity incentives for first-in-world applications, Korea also needs to strengthen commercial incentives for companies by guaranteeing exclusivity for a certain period after approval or implementing exceptional drug pricing preferential policies.Expanding the scientific flexibility of regulations and strengthening the expertise and authority of regulatory agencies are also identified as key tasks to enhance the competitiveness of the GIFT system. In areas with urgent unmet medical needs, such as cancer and rare diseases, there is an argument for more active acceptance of surrogate endpoints while shifting the regulatory paradigm toward strengthened post-marketing surveillance and post-approval verification. Analysis also suggests that strengthening the MFDS's bio-specialist workforce and institutionally guaranteeing the discretion and responsibility of reviewers must precede such changes.An industry official commented, “We are now in an era where the quality of regulation directly determines a country’s biopharmaceutical competitiveness. Beyond simply accelerating approval timelines, the GIFT system's effectiveness will only truly increase when a clear commercial reward structure is established that allows for the recovery of the massive R&D costs invested in new drug development.”
- Company
- Boehringer Ingelheim takes legal action over similar trademark to Ofev
- by Kim, Jin-Gu Dec 31, 2025 07:49am
- Boehringer Ingelheim has filed a trademark invalidation trial against Daewoong Pharmaceutical, challenging a trademark it deems similar to its nintedanib-based pulmonary fibrosis treatment ‘Ofev (nintedanib)’.Within the pharmaceutical industry, the move is being interpreted as a strategic effort to protect the original brand and deter follow-on generics, particularly given that the legal action was initiated after the generic product name had already been changed.According to the Intellectual Property Trial and Appeal Board on the 30th, Boehringer Ingelheim filed a petition on the 29th to invalidate the registration of the trademark ‘Ofevia’ held by Daewoong Pharmaceutical.Daewoong applied for the Ofevia trademark in October last year and obtained marketing authorization for the generic in January this year. The trademark was officially registered in November. Following the registration, Boehringer initiated legal action, citing the similarity in pronunciation and spelling to its original product, Ofev.However, Daewoong had already changed the product name to ‘Ofild’ in June of this year. While no product is currently marketed under the Ofevia name, the trademark itself remains registered.Industry observers believe Boehringer’s move reflects a broader strategy to reinforce brand protection and curb potential generic competition. The analysis suggests an intent to invalidate the active trademark, regardless of actual usage, to solidify the original brand's distinctiveness.Furthermore, it is also analyzed that even if Daewoong Pharmaceutical does not use the Ofevia name, the fact that the trademark remains registered could influence the naming choices of future generic entrants. This is interpreted as an aim to prevent the reappearance of similar trademarks at the source.This interpretation is further supported by the fact that Boehringer has not pursued invalidation actions against other approved Ofev generics. Currently, approved Ofev generics include Daewoong Pharmaceutical's Ofild and ▲Yungjin Pharm’s ‘Nintebro,’ ▲Kolon Pharmaceutical's ‘Effidanib.’ ▲Ildong Pharmaceutical's ‘Cuninta.,’ all of which are considered sufficiently distinct from the Ofev brand.Ofev is indicated for ▲ the treatment of idiopathic pulmonary fibrosis, ▲ slowing the decline of lung function in patients with systemic sclerosis–associated interstitial lung disease, and ▲ progressive fibrotic interstitial lung diseases with a progressive phenotype. However, reimbursement coverage in Korea applies only to the latter two indications, excluding idiopathic pulmonary fibrosis. According to the Ministry of Food and Drug Safety, Ofev’s import volume last year was USD 3.57 million (approximately KRW 5.3 billion).Generic companies obtained marketing authorizations around January this year, coinciding with the expiration of Ofev’s substance patent. Following Ofev’s inclusion in the reimbursement list in May this year, generics were launched with reimbursement two months later in July.
- Company
- OTC drugs 8K↓·health functional foods 25K↑ over 10 years
- by Chon, Seung-Hyun Dec 31, 2025 07:49am
- The gap between the number of registered health functional foods and over-the-counter (OTC) drugs in South Korea has widened significantly. Over the past 10 years, health functional foods have more than doubled, and just nine years after overtaking OTC drugs, the number of health functional foods items has reached five times that of OTC products. OTC drugs have decreased to nearly half their previous level over the past 10 years, indicating that the movement to enter the market has been greatly restricted. It was also found that one out of every two approved OTC products has no production record.According to the '2025 Food and Drug Statistical Annual Publication' released by the Ministry of Food and Drug Safety (MFDS) on the 31st, the number of OTC drug items was calculated at 8,630 last year. This is a decrease of 490 items from 9,120 in 2023.The downward trend in the number of domestically approved OTC drug items is continuing. From 16,717 items in 2014, the count has shrunk to nearly half over the course of 10 years. The number of OTC items dropped by 5,797, from 14,175 in 2018 to 8,378 in 2019, and although it recovered to the 10,000 level in 2020, the decline has persisted since 2021. The number of OTC drug items rose by 307, from 8,813 in 2022 to 9,120 the following year, but returned to a downward trend within a year.The number of over-the-counter (OTC) drugs and health functional foods registered in South Korea by year (unit: number, source: MFDS)The report indicates that more products have withdrawn from the domestic OTC market than new ones have entered. In the pharmaceutical market, many products disappear due to safety management systems such as the continuous renewal of product licenses. The requirements of the drug product license renewal system is that drugs approved by health authorities must re-verify their efficacy and safety every five years for the license to be maintained. For many products, if marketability is determined to have declined at the time of expiration, the renewal is abandoned and the product is withdrawn from the market.This is a stark contrast to the continuous annual increase in health functional food.Last year, the number of manufactured health functional food items was 41,896, an increase of 4,622 from the previous year. Over the 10 years since 2014, when there were 16,632 items, the count has increased by 25,264, expanding by approximately 2.5 times.In 2014, health functional foods had 85 fewer items than OTC drugs. In 2015, the number of health functional food items rose to 18,956, overtaking the 14,892 OTC drug items by a margin of 4,064, and the gap has gradually widened since then. Last year, the number of health functional food items was found to be about five times greater than that of OTC drugs.. The gap becomes even larger when considering only those OTC drugs with actual production records. Last year, the number of OTC drugs with production records was 4,631, compared to 4,873 in 2023. This means that about half of the OTC drugs currently maintaining MFDS approval have no production records. The number of OTC items with production records has decreased by 1,444 over the 10 years since 2014, when it was 6,075.Analysis suggests that as the health functional food market size continues to grow and market entry barriers are relatively lower than those for OTC drugs, new entry activities are more active. Last year, the health functional food market was valued at KRW 4.0131 trillion, a 1.9% decrease from the previous year. It has decreased for two consecutive years from KRW 4.1695 trillion in 2022. The industry analyzes that as new entries into the health functional food market have become active and low-price competition has intensified, it has affected the reduction in market size. However, compared to KRW 2.4130 trillion in 2014, the market size has increased by 146.1%, showing rapid growth recently.The health functional food market size and over-the-counter (OTC) drug production size by year (unit: KRW 100 million, source: MFDS)Following the enactment of the Health Functional Food Act in 2003, the health functional food system was implemented. The purpose of introducing the Act was to contribute to the promotion of public health and consumer protection by ensuring the safety and improving the quality of health functional foods and fostering sound distribution and sales. The domestic health functional food market surpassed KRW 1 trillion for the first time in 2010 and exceeded KRW 2 trillion in 2016. The market size grew to the KRW 3 trillion range in 2020 and has remained above KRW 4 trillion since 2021.While the number of OTC drug items continued to decline, production performance showed a brief upward trend.Last year, the production size of OTC drugs was KRW 4.2357 trillion, a 9.9% increase from the previous year. Last year's OTC production amount was the largest in history.The production size of OTC drugs decreased from KRW 3.1779 trillion in 2020 to KRW 3.0692 trillion in 2021 but returned to an upward trend in 2022. In 2022, the OTC production record was KRW 3.5848 trillion, a 16.8% increase from the previous year, and in 2023, it recorded KRW 3.8554 trillion, a 7.5% increase year-on-year. Last year's OTC production scale increased by 38.0% in three years compared to 2021, surpassing KRW 4 trillion for the first time.Analysis suggests that the recent expansion of OTC production records was most heavily influenced by the COVID-19 pandemic and endemic.Since the end of 2021, when hundreds of thousands of COVID-19 cases were reported daily, sales of antipyretics, analgesics, and cold medicines used for symptom relief significantly increased. It is analyzed that the boom in the OTC market has continued as the number of flu and cold patients surged since the transition to the COVID-19 endemic phase in 2023.
- Company
- Export of plasma-derived products 50%↑…Alyglo effect
- by Cha, Ji-Hyun Dec 31, 2025 07:49am
- Despite a contraction in the domestic plasma-derived products market last year, export performance surged sharply. Industry analysis suggests that GC Biopharma’s immunoglobulin product Alyglo played a pivotal role in driving export growth as it expanded its presence in global markets.The Korea Biomedicine Industry Association(KoBIA) announced on the 29th that it had published the ‘2025 Biopharmaceutical Industry Trends Report, which collects and analyzes industry information and trends in the domestic and international biopharmaceutical industry.According to the report, the size of Korea’s plasma-derived products market in 2024 totaled KRW 354.3 billion, down 12.7% from KRW 405.8 billion the previous year. Stagnant domestic demand for major products such as immunoglobulins and albumin contributed to the overall market contraction.In contrast, production and export indicators showed clear improvement. Production of plasma-derived products reached KRW 548.6 billion last year, up 7.0% from KRW 512.9 billion the previous year. Exports surged 50.3% year-on-year to KRW 248.8 billion, leading overall growth. During the same period, imports declined 6.6% year-on-year to KRW 54.6 billion.Exports were largely driven by GC Pharma’s plasma-derived product Alyglo. In fact, among the top exported plasma-derived products in 2024, IV-Globulin SN Inj 10% and IV-Globulin SN Injection 5% ranked first and third, respectively.Top Plasma-derived products by export value in 2024 (Source: KoBIA)Alyglo is a liquid immunoglobulin product purified from plasma. It is the first plasma-derived therapeutic developed by a Korean company to enter the US. market, having received approval from the U.S. Food and Drug Administration (FDA) in December 2023. It is indicated for the treatment of primary immunodeficiency diseases, including congenital immunodeficiency and immune thrombocytopenia. In Korea, it is marketed under the brand name IV-Globulin SN I.According to the Ministry of Food and Drug Safety (MFDS), production of IV-Globulin SN Inj 10% increased 71.5%, from KRW 26.0 billion in 2023 to KRW 44.5 billion in 2024. Production of the 5% formulation totaled KRW 60.5 billion in 2024, down from KRW 85.8 billion the previous year.In particular, GC Biopharma’s exports of plasma-derived products—including Alyglo —nearly tripled year-on-year, rising from KRW 103.0 billion to KRW 303.6 billion. Much of this growth was attributed to expanding US sales of Alyglo. GC Biopharma USA, responsible for sales in the US, reported cumulative sales of KRW 79.0 billion through the third quarter of this year.Meanwhile, domestic production of plasma-derived products continued to be led by albumin formulations. SK Plasma’s SK Albumin 20% Inj and GC Biopharma’s Green Cross Albumin 20% Inj ranked first and second in production volume, respectively, confirming an albumin-centered production structure. IV-Globulin SN Inj 5% ranked third, while IV-Globulin SN Inj 10% placed sixth.
- Company
- K-medical devices sweep CES2026…'Physical AI'
- by Hwang, byoung woo Dec 31, 2025 07:49am
- Korea's medical device companies will participate in world's largest IT and consumer electronics show 'CES2026,' scheduled to kick off on January 6 (local time) next year.Source: CES websiteAnalysis suggests that the emergence of 'Physical AI', where AI integrates with physical bodies and medical procedures rather than remaining a mere hardware display, is evaluated to have elevated the status of K-medical devices to a new level.According to the 'CES 2026 Preview' report published by the Korea Trade-Investment Promotion Agency (KOTRA), the core shift in this year's CES is the evolution of AI toward combining with physical environments beyond the virtual world. The digital health and medical device industries are at the forefront of this trend.Digital health, Ranked No. 1 in CES trends... The transformation of a 'Consumer Electronics Show'CES is the world's largest tech exhibition, visited by over 180,000 people from 170 countries annually. It serves as a global stage that concentrates the flow of future technologies, including AI, digital healthcare, mobility, and smart homes.In particular, this year's CES is expected to feature medical AI with high utility in clinical settings, automation-based diagnostic technologies, and innovations in women's health as major themes.According to KOTRA's analysis of industry trends for CES 2026, the digital health sector recorded the highest growth rate among all industry groups, with the number of participating companies increasing by approximately 7.4% year-over-year. This is a signal that CES, which once centered on smartphones and TVs, is shifting its center of gravity toward the medical and healthcare sectors.Furthermore, 'Clinical Decision Support Systems (CDSS)', where AI assists in diagnosis and treatment decision-making beyond simple monitoring, and precision medicine have emerged as key keywords. Analysis suggests that the expansion of data infrastructure and the advancement of AI have brought personalized medical solutions into the reality.Korea accounts for 60% of Innovation Award Winners, with the second-largest number of participating companiesThe progress of Korean companies is particularly outstanding in CES 2026.Among the 284 companies receiving Innovation Awards, 168 are Korean companies, accounting for approximately 60% of the total. After maintaining the status as the country with the most awards for three consecutive years, Korea has further expanded its market share.At CES 2026, KOTRA will operate a unified Korea Pavilion comprising 470 companies in cooperation with 38 related organizations and local governments.This is highly regarded as a 'practical export platform' aimed at connecting with global medical institutions, insurers, and partners, moving beyond a simple exhibition.'Non-invasive·Personalized' treatment areas..focus on the expansion of diagnostic technologyFirst, in the field of treatment, non-invasive and personalized were presented as core trends.Gbrain, a two-time Innovation Award winner, highlighted its competitiveness in the invasive Brain-Computer Interface (BCI) field with a fully implantable brain stimulation system for treating brain diseases such as Parkinson's.The device, in which AI analyzes brainwaves in real-time to adjust stimulation, is cited as an example demonstrating the clinical applicability of BCI technology.The keywords for this year's CES are artificial intelligence (AI), robotics, and digital health.EverEx, a digital rehabilitation solution company, drew attention with its 'MORA' platform, which supports rehabilitation based on smartphone cameras without requiring separate equipment. Currently, predictive AI-based rehabilitation is being evaluated as a potential alternative to address medical personnel shortages and cost issues.In the diagnostic field, liquid biopsy and on-device AI technology have emerged.Exopert will showcase an automated early-stage multi-cancer detection platform based on exosome analysis, while Noul plans to present the possibility of automating field diagnostics through its cervical cancer Point-of-Care (PoC) diagnostic solution.By participating in CES, Noul expects to accelerate business development in North America and Latin America, its current areas of focus.Additionally, technologies such as SevenPointOne's voice-based brain health check 'AlzWIN' and WIRobotics' walking assist robot 'WIM S' will showcase technologies that accelerate 'digital transformation' by extending the scope of diagnosis from hospitals to daily life.Wontech presented a total healthcare strategy combining non-contact vital sign measurement and pain treatment devices, while An&Luck proposed an aging response model through a community-integrated care platform. This demonstrates that strategies combining services and platforms are becoming full-scale, moving beyond individual hardware products.The industry views CES 2026 as a turning point where Korean medical devices transition from manufacturing competition to a software- and solution-centered industry.A medical device industry official stated, "The growth indicators for digital health identified at CES are a signal of the competition for global leadership. The next task is to translate these achievements into results through North American regulatory approvals and entry into insurance reimbursement."
- Company
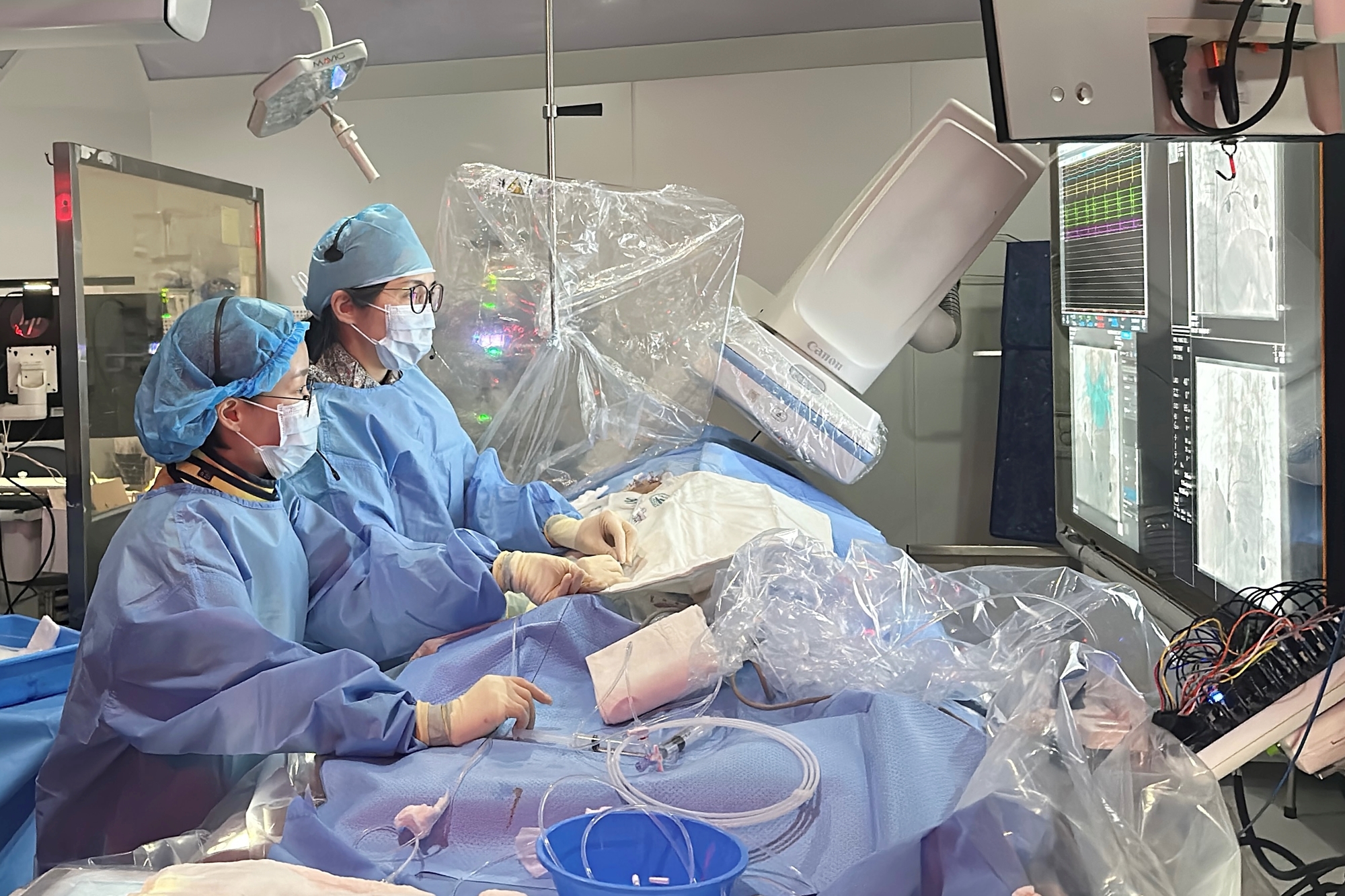
- Adoption of pulsed field ablation gains momentum in Korea
- by Hwang, byoung woo Dec 30, 2025 07:22am
- PFA at Asan Medical CenterThe treatment paradigm for atrial fibrillation (AF) is rapidly shifting from conventional thermal ablation techniques to the non-thermal approach of pulsed field ablation (PFA).Global medical device companies are sequentially entering the Korean market, signaling the beginning of an intense competition for market share.According to a report released by the Korea Health Industry Development Institute (KHIDI) on the 29th, the global PFA market size is projected to grow from approximately USD 1.5 billion (KRW 2.15 trillion) in 2024 to USD 9.2 billion (KRW 13.20 trillion) by 2029, representing a compound annual growth rate (CAGR) of around 41.1%.PFA emerges as the game changer... strengths in safety and procedure efficiencyPFA utilizes the principle of ‘irreversible electroporation (IRE)’, which uses a high-voltage electric field to create microscopic pores in cell membranes.Unlike radiofrequency ablation (RFA) or cryoballoon ablation (CBA), which rely on thermal energy and carry risks such as esophageal injury and phrenic nerve palsy, PFA is evaluated as reducing major complications associated with conventional ablation, such as esophageal injury, phrenic nerve damage, and pulmonary vein stenosis.Clinical data also support this. Results from the ADVENT clinical trial involving 607 patients demonstrated PFA's 12-month treatment success rate of 73.3%, demonstrating non-inferiority compared with conventional thermal ablation techniques (71.3%).Notably, the incidence of pulmonary vein stenosis was just 0.9% in the PFA group, compared with 12% in the conventional ablation group, and no cases of esophageal damage were reported.Building on these strengths, market penetration in Korea is expected to accelerate. The domestic PFA market is projected to grow at an average annual rate of 11.1%, expanding from approximately USD 31 million (KRW 42 billion) in 2024 to USD 53 million (KRW 72 billion) by 2029.Market size and growth rate of PFA in KoreaGlobal “Big 3” secure Korean approvals… adoption accelerates at tertiary hospitalsThe Korean PFA market is currently shaping up as a three-way competition among Boston Scientific, Medtronic, and Johnson & Johnson (J&J).Boston Scientific took the lead with its FARAPULSE platform, obtaining catheter approval in April 2024 and generator approval in September.Following Korea’s first successful PFA procedure at Severance Hospital in December last year, the number of adopting hospitals is rapidly increasing.J&J’s VARIPULSE system received catheter approval in August 2024 and emphasizes its fully integrated workflow with its proprietary 3D mapping system, CARTO 3, as a key competitive advantage.Medtronic's ‘PulseSelect’ followed suit, entering the fray in January 2025 with Class III medical device approval for its integrated catheter and generator system.The company has further strengthened its lineup with the Sphere-9 catheter, capable of high-density mapping and dual RF/PF ablation.Major global medical device companies' PFA approval status“Clear clinical value”…but reimbursement remains the key to wider adoptionClinicians have expressed positive views on PFA adoption.Professor Boyoung Joung of the Cardiology Department at Severance Hospital stated, “PFA cuts procedure time by nearly half compared to conventional methods while significantly reducing complication risks, leading to high satisfaction among both physicians and patients. Currently, about 35% of atrial fibrillation ablation procedures at Severance Hospital are performed using PFA, and this proportion is expected to continue rising.”According to an analysis of real-world data (RWD)-based cost outcomes collected from hospitals in Belgium, Germany, and the Netherlands, the direct costs per patient during the initial procedure (including personnel, time, anesthesia, tests, hospitalization, etc.) were 22% lower than CBS and 35% lower than RFA.From the patient perspective, while immediate cost savings were modest, cumulative reductions were observed over time due to fewer repeat procedures and lower complication management costs.However, regulatory hurdles remain. Although PFA passed the New Health Technology Assessment in December last year, it has yet to be listed for reimbursement, pending inclusion in the National Health Insurance reimbursement list. Currently, whether a patient has indemnity health insurance significantly influences their choice.An industry insider commented, “In markets such as the U.S., PFA already accounts for over 70% of all ablation procedures and has effectively become the standard of care. Given its proven efficiency and safety, reimbursement and other institutional support in Korea would significantly accelerate the pace of market transition.”
- Company
- Bimzelx indication expanded to inflammatory diseases
- by Son, Hyung Min Dec 30, 2025 07:22am
- The inflammatory disease treatment market, which had long been dominated by biologics such as Humira (adalimumab) and Cosentyx (secukinumab), is showing signs of significant change. Competition is intensifying as not only biologics but also Janus kinase (JAK) inhibitors and next-generation targeted therapies are successively expanding their indications.UCB Pharma's ‘Bimzelx’According to industry sources on the 30th, the Ministry of Food and Drug Safety (MFDS) has approved Bimzelx (bimekizumab) for 3 additional indications: ▲psoriatic arthritis, ▲axial spondyloarthritis, and ▲hidradenitis suppurativa.Bimzelx is the first and only biologic that simultaneously inhibits interleukin-17A and interleukin-17F. It received its initial domestic approval in August last year for the treatment of plaque psoriasis.This approval for psoriatic arthritis is based on results from two Phase III clinical trials: the BE OPTIMAL study in biologic-naive patients and the BE COMPLETE study in patients who had inadequate response or intolerance to TNF-α inhibitors.In the BE OPTIMAL study, 44% of the Bimzelx group achieved the primary endpoint of ACR50 at week 16, showing a significant improvement compared to the placebo group (10%). In the BE COMPLETE study, 43% of the Bimzelx group achieved ACR50, and 27% achieved ACR70. No new safety signals were observed in either study.The axial spondyloarthritis indication was approved based on the results of two parallel Phase III clinical trials: BE MOBILE 1 in non-radiographic patients and BE MOBILE 2 in radiographic patients.In the studies, the 16-week ASAS40 response rates in the Bimzelx group were 48% and 45%, respectively, significantly higher than in the placebo group (21% and 23%). Responses were observed within 1 to 2 weeks after the first dose, and the response rate continued to increase through week 24. Patients who switched from placebo to Bimzelx also showed similar response rates at week 24. No new adverse reactions were identified in terms of safety.The hidradenitis suppurativa indication was approved based on two Phase III clinical trials, BE HEARD 1 and 2, conducted in moderate-to-severe patients. The proportion achieving HiSCR50 at week 16 was 48% and 52% in the Bimzelx groups, respectively, significantly higher than the placebo groups (29%, 32%).The more stringent HiSCR75 responses were also higher in the Bimzelx group at 33% and 36%, respectively, compared to placebo. These benefits were maintained or further improved through 48 weeks, with meaningful improvements observed in quality-of-life measures such as DLQI. An integrated long-term extension analysis confirmed sustained efficacy and safety over two years.New competitors enter the field… Potential of TYK2-targeted novel drugs draws attentionWith the expansion of Bimzelx’s indications, competition with existing biologics and JAK inhibitors has become inevitable. Current competitors with overlapping indications include major biologics and JAK inhibitors like Novartis’s Cosentyx, AbbVie’s Humira and Rinvoq (upadacitinib), and Johnson&Johnson’s Stelara (ustekinumab).BMS’s ‘Sotyktu’The rapid advancement of new TYK2 inhibitors is also noteworthy. Currently, BMS's ‘Sotyktu (deucravacitinib)’ has only secured approval for plaque psoriasis in Korea. BMS has completed clinical trials for psoriatic arthritis and is awaiting regulatory review in the US.Unlike JAK inhibitors, TYK2 inhibitors selectively suppress the TYK2 protein, blocking only signaling in the IL-12, IL-23, and Type I interferon pathways. This allows for safer treatment while regulating immune responses, making them a point of interest.Among existing biologics, IL-17 inhibitors have faced concerns about exacerbating inflammatory bowel diseases like ulcerative colitis or Crohn's disease and causing oral candidiasis. While IL-23 inhibitors showed no prominent adverse reactions, concerns exist regarding injection site pain or discomfort and upper respiratory tract infections. This is one reason new treatment options are gaining attention.Competition among latecomers is also inevitable.Takeda’s zasocitinib is regarded as one of the most advanced TYK2 inhibitors following Sotyktu. Its commercial potential was confirmed in a Phase IIb clinical trial published last year in the medical journal JAMA.This study randomly assigned 259 participants to receive one of four daily oral doses of zasocitinib (2mg, 5mg, 15mg, or 30mg) or placebo for 12 weeks.Clinical results showed a dose-dependent response to zasocitinib. The proportion of patients achieving a Psoriasis Area and Severity Index (PASI) 75 (75% or greater improvement in skin condition) was 18%, 44%, 68%, and 67% in the respective dose groups, significantly higher than the 6% in the placebo group. Furthermore, no serious safety concerns were reported in this study, indicating that zasocitinib has a favorable safety profile.Takeda is actively developing zasocitinib for indications beyond psoriasis and is also pursuing approval for psoriatic arthritis. Furthermore, Takeda is conducting Phase II studies for moderate-to-severe active Crohn's disease and ulcerative colitis, and is planning studies for systemic lupus erythematosus, exploring the potential of zasocitinib in other immune-mediated diseases.
- Company
- K-made new drug production reached KRW 800B
- by Chon, Seung-Hyun Dec 30, 2025 07:01am
- Last year, the production amount of domestically developed new drugs in South Korea was the highest in history. With a variety of hit products having been introduced to the prescription market, the production growth rate has neared 50% over the past two years. Annual production for HK inno.N's K-CAB, Daewoong Pharmaceutical’s Fexuclue, and Yuhan Corporation’s Leclaza each surpassed KRW 100 billion. Additionally, three domestically developed new drugs passed commercialization hurdles this year. This year has seen the highest output of new drugs in four years since 2021.Korea-made new drug production amounted to KRW 794.6B, up 47% in 2 years...K-CAB first to surpass KRW 200BAccording to the Ministry of Food and Drug Safety (MFDS) on December 30, domestically developed new drugs combined for a total production value of KRW 794.6 billion last year. This represents a 16.1% increase from the KRW 684.6 billion recorded in 2023, the highested in history. Compared to the 2022 production figure of KRW 540.5 billion, the sector has expanded by 47.0% in just two years.2024 Production Amount of Domestically Developed New Drugs in South Korea(unit: number of items, sources: MOHW, MFDS): (from top) K-CAB, Fexuclue, Leclaza, Pelubi, Kanarb, Zemiglo, Noltec, Duvie, Suganon, Envlo, Supect, Rolontis, Zydena, Pyramax, Mvix, Jaqbo, Besivo, Factive, Easyef SOLN, RG-Q, RevanexK-CAB, Fexuclue, and Leclaza represent key commercial successes among Korea-made drugs, each recording annual production exceeding KRW 100 billion.HK inno.N's K-CAB recorded the highest production last year at KRW 231.0 billion. K-CAB is the first potassium-competitive acid blocker (P-CAB) developed by a Korean company for the treatment of gastroesophageal reflux disease (GERD). K-CAB recorded production performance of KRW 171.9 billion in 2022 and KRW 173.9 billion in 2023. Last year, its production increased by 32.8% year-on-year, surpassing the KRW 200 billion mark. K-CAB is the first domestically developed new drug to exceed KRW 200 billion in annual production.K-CAB continues its high growth by highlighting advantages such as a faster onset of action compared to conventional proton pump inhibitor (PPI) products and the ability to be taken regardless of mealtime. In addition to the tablet form, two types of orally disintegrating tablets (ODT) have also been approved. KRW 28.2 billion-worth ODT formulation was produced last year.The production of Daewoong Pharmaceutical's GERD treatment, Fexuclue, reached KRW 118.1 billion last year, a 66.0% increase from the previous year. Fexuclue is a P-CAB class treatment similar to K-CAB. It received marketing authorization in December 2021 and began full-scale sales in July 2022 upon being listed on the national health insurance (NHI) reimbursement list.Fexuclue rapidly penetrated the market by demonstrated excellence in areas such as ▲rapid onset of action ▲fast and superior symptom improvement ▲ effective nighttime symptom relie ▲dosing convenience ▲low drug-drug interactions and consistent efficacy. Fexuclue's production was KRW 27.4 billion in its launch year (2022), then rose sharply to KRW 71.2 billion in 2023, and surpassed KRW 100 billion last year.Yuhan Corporation's Leclaza recorded a production value of KRW 105.8 billion last year. Leclaza is a treatment for non-small cell lung cancer (NSCLC) approved in January 2021 as Korea's 31st domestically developed new drug. Leclaza entered the prescription market following its listing on the NHI reimbursement list in July 2021.Leclaza's initial production in 2021 was KRW 9.8 billion, which expanded to KRW 39.3 billion in 2022. It surpassed the KRW 100 billion in 2023 with KRW 112.2 billion in production, only three years after launch, and has maintained production levels above KRW 100 billion for two consecutive years.Analysis suggests that the surge in demand is due to the expanded scope of NHI reimbursement for Leclaza as a first-line treatment. Leclaza was initially approved as a second-line treatment for locally advanced or metastatic NSCLC with specific gene (T790M) resistance following the administration of 1st or 2nd generation EGFR Tyrosine Kinase Inhibitors (TKIs). In June 2023, the MFDS approved changes of approval for Leclaza to include the 'first-line treatment of NSCLC'. Since January of last year, scope of reimbursement was expanded to include the 'first-line treatment of locally advanced or metastatic NSCLC with specific genetic mutations'.These three new drugs, K-CAB, Fexuclue, and Leclaza, combined for a total production of KRW 454.9 billion last year. They accounted for 57.3% of the total production value of all domestically developed new drugs, leading the commercial success of the sector.The production of Daewon Pharmaceutical's analgesic drug Pelubi reached KRW 76.9 billion last year, a 58.3% increase year-on-year. Approved in 2007 as Korea's 15th new drug, Pelubi is a non-steroidal anti-inflammatory drug (NSAID) with indications for osteoarthritis, rheumatoid arthritis, low back pain, and fever reduction in acute upper respiratory infections. Pelubi’s production surged as its prescription volume expanded significantly through the COVID-19 pandemic and endemic phases.Boryung's hypertension treatment Kanarb, LG Chem's diabetes treatment Zemiglo, and Il-Yang Pharm's anti-ulcer agent Noltec all recorded production values exceeding KRW 50 billion last year.Products recording over KRW 10 billion in production last year included Chong Kun Dang's diabetes treatment Duvie, Dong-A ST's diabetes treatment Suganon, Daewoong Pharmaceutical's diabetes treatment Envlo, Il-Yang Pharm's leukemia treatment Supect, and Hanmi Pharmaceutical's neutropenia treatment Rolontis. Of the 38 new drugs approved through last year, 21 products recorded production figures. Approximately half of the new drugs approved by the MFDS were found to have no production.Three Korea-made new drugs approved this year…highest since 2021This year, three domestically developed new drugs, GC Biopharma's Barythrax, Medytox's Nuviju, and Dong-A ST's Xcopri, received marketing authorization.List of Newly Approved Domestically Developed Drugs: 1. Sunpla Inj (SK Chemicals) 2. Easyef SOLN (Daewoong Pharmaceuticals) 3. Milican inj (Dong Wha) 4. Q-roxin Tab (JW Pharmaceutical) 5. Factive Tab (LG Chem) 6. Apitoxin Inj (BCWORLD PHARM) 7. Pseudobaccine (CJ CheilJedang) 8. Camtobell (Chong Kun Dang Pharm) 9. Revanex (Yuhan Corp) 10. Zydena (DONG-A ST) 11. Levovir (Bukwang Pharm) 12. Pelubi (Daewon Pharm) 13. Mvix (SK Chemicals) 14. Noltec Tab (IL-YANG PHARM) 15. Kanarb Tab (Boryung) 16. Pyramax Tab (SHIN POONG PHARM) 17. Zepeed Tab (JW Pharmaceutical) 18. Supect Capsule (IL-YANG PHARM) 19. Zemiglo (LG Chem) 20. Duvie Tab (Chong Kun Dang Pharm) 21. RIAVAX (GemVax & KAEL) 22. Acelex Cap (CrystalGenomics) 23. Zaborlante Tab (Dongwha Pharm) 26. Suganon tab (DONG-A ST) 27. Olita Tab (Hanmi Pharm) 28. Besivo Tab (Ildong Pharmaceutical)29. Alzavue Inj (Future Chem) 30. K-CAB (HK inno.N) 31. Leclaza (YUHAN Corp) 32. Regkirona Inj (CELLTRION) 33. Rolontis (Hanmi Pharm) 34. Fexuclue Tab (Daewoong Pharmaceuticals)35. SkyCovione Multi Injection (SK bioscience) 36. Envlo (Daewoong Pharmaceuticals)37. JAQBO Tab (Onconic Therapeutics) 38. Unafra Inj (VIVOZON PHARMACEUTICAL)39. Barythrax (Green Cross) 40. NUVIJU Inj (Medytox) 41. Xcopri tab (DONG-A ST).In April, GC Biopharma's recombinant anthrax vaccine, Barythrax, was approved as Korea's 39th new drug. Barythrax is a vaccine designed for the pre-exposure prophylaxis of anthrax in adults. It uses recombinant technology to manufacture the protective antigen (PA) protein, a major component of the anthrax toxin, to induce antibodies that prevent infection. Barythrax was co-developed by GC Biopharma and the Korea Disease Control and Prevention Agency (KDCA) and was approved 18 months after the application was filed in October 2023.In September, Medytox's Nuviju was approved as the 40th domestically developed new drug. Nuviju is a treatment that reduces submental subcutaneous fat by inducing adipocyte lysis through the disruption of the cell membrane's lipid bilayer via surfactant action. It is approved for adult patients seeking to improve moderate to severe submental fullness or excessive fat. Nuviju is Medytox's first chemical drug and the world's first next-generation fat-dissolving injectable developed with cholic acid as the main ingredient. Cholic acid is a type of bile acid synthesized in the liver and secreted into the bile.This year, the highest number of domestically developed new drugs reached the commercialization stage in four years, following the four drugs approved in 2021. In 2021, Yuhan Corporation's oncology drug Leclaza, Celltrion's COVID-19 treatment Regkirona, Hanmi Pharmaceutical's neutropenia treatment Rolontis, and Daewoong Pharmaceutical's GERD treatment Fexuclue obtained new drug approval.