- LOGIN
- MemberShip
- 2026-04-04 11:57:39
- Policy
- PNH drug 'Piasky Inj' for adults·children wins nod
- by Lee, Tak-Sun Dec 26, 2025 08:39am
- The Ministry of Food and Drug Safety (MFDS), led by Minister Yu-Kyoung Oh, announced that it granted approval of 'Piasky (crovalimab)', a new orphan drug from Roche used for the treatment of paroxysmal nocturnal hemoglobinuria (PNH) in adults and pediatric patients aged 12 and older who weigh 40 kg or more, on December 24.PNH is an acquired hematopoietic stem cell disorder characterized by hemolytic phenomena (hemoglobin escapes from red blood cells) that causes release of dark-colored urine during the night.'Piasky Inj' is an orphan drug that inhibits complement-mediated intravascular hemolysis in patients with PNH. Complement proteins assist the immune response by attacking pathogens such as bacteria and viruses.As a complement protein C5 inhibitor, this drug prevents the binding of C5 to abnormal red blood cells produced by hematopoietic stem cell disorders, thereby protecting the cells from lysis.Piasky is the first treatment approved for pediatric use in South Korea, and it is expected to significantly broaden the therapeutic landscape for both adult and adolescent patients (12 years and older).The MFDS stated that it will make every effort to ensure that new treatments for rare diseases are rapidly made available based on its regulatory science expertise, thereby expanding clinical opportunities for patients.
- Company
- New cardiomyopathy drug joins the fray… Camzyos’s competitor
- by Son, Hyung Min Dec 24, 2025 08:09am
- A new option has entered the U.S. market for obstructive hypertrophic cardiomyopathy (oHCM). Attention is focused on the potential shift in treatment paradigms and market dynamics as a competitor emerges in the cardiac myosin inhibitor class, a space largely dominated by Bristol Myers Squibb's (BMS) ‘Camzyos (mavacamten)’.On the 22nd, U.S. biotech company Cytokinetics announced that its drug ‘Myqorzo (aficamten)’ received approval from the U.S. Food and Drug Administration (FDA) for improving exercise capacity and symptoms in adults with symptomatic oHCM. This marks the first FDA approval in Cytokinetics’ history.Myqorzo is an allosteric modulator that reversibly inhibits the contractile activity of cardiac myosin that works by reducing excessive myocardial contraction and left ventricular outflow tract (LVOT) obstruction, placing it in the same class as the already marketed drug Camzyos.Ironically, this competitive landscape has historical ties. Cytokinetics was involved in the development of Camzyos through its collaboration with MyoKardia in 2012. MyoKardia was later acquired by BMS for USD 13.1 billion. As a result, Camzyos received FDA approval in 2022, becoming the first myosin inhibitor for oHCM.This approval is based on results from the Phase III SEQUOIA-HCM clinical trial. After 24 weeks of treatment, the Myqorzo group showed a significant improvement in peak oxygen consumption (pVO₂) of 1.8 mL/kg/min compared to baseline, versus 0.0 mL/kg/min in the placebo group. Consistent efficacy was observed across key subgroups, including age, gender, and beta-blocker use.Regarding safety, no treatment discontinuations due to worsening severe heart failure or low left ventricular ejection fraction (LVEF) were reported during therapy. However, due to its mechanism of inhibiting myocardial contraction, the warning about the risk of heart failure remains in place.Like Camzyos, Myqorzo carries a boxed warning regarding heart failure risk and is only available through a Risk Evaluation and Mitigation Strategy (REMS) program. It also requires LVEF monitoring via echocardiography before and during treatment.“Will the monopoly break?”... Competition intensifies in the targeted therapy marketCurrently, the targeted therapy market for oHCM is effectively monopolized by Camzyos. Approved by the FDA in 2022, Camzyos is the world's first cardiac myosin inhibitor and is widely credited with changing the treatment paradigm for oHCM, where drug options were previously limited.Until now, the only drug options available for HCM treatment were chronic disease medications like beta-blockers and calcium channel blockers. While these drugs could indirectly manage HCM symptoms, if symptoms did not improve, there were no treatment options other than surgery. The arrival of Camzyos opened a new path for targeted HCM therapy.Camzyos has also demonstrated strong commercial performance. According to BMS, the drug surpassed USD 600 million in annual sales in 2024 and continues to grow this year. Its status as the only FDA-approved myosin inhibitor for oHCM to date has underpinned its market dominance.Despite this, attention is now turning to the potential differentiation between the two drugs. Myqorzo highlights several advantages, including ▲ simple and flexible dose adjustment ▲ no need for drug-drug interaction monitoring ▲ predictable pharmacokinetic properties. Some investment analysts suggest these features could make Myqorzo a more user-friendly option, particularly for newly diagnosed or treatment-naïve patients.Market observers believe that while Myqorzo is unlikely to rapidly overturn Camzyos’ dominance, gradual competition is expected, especially in newly diagnosed and early-treatment populations. Given that oHCM is a chronic condition requiring long-term therapy, dosing convenience and monitoring burden are likely to be key factors influencing real-world prescribing decisions.Myqorzo is scheduled for commercial launch in the U.S. in January 2026. Pricing has not yet been disclosed, but its strategy relative to Camzyos—currently priced at approximately USD 100,000 per year—is expected to play a crucial role in determining the pace of market penetration.Attention is focused on whether the oHCM treatment market can shift from a Camzyos monopoly to a competitive landscape among myosin inhibitors, and on whether Myqorzo can successfully establish itself in actual clinical practice.
- Policy
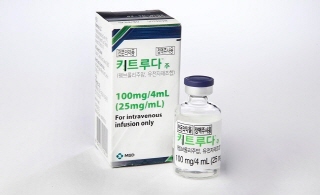
- Expanded reimb for Keytruda·Dupixent
- by Jung, Heung-Jun Dec 24, 2025 08:08am
- As the review of expanded reimbursement for Keytruda (pembrolizumab) and Dupixent (dupilumab) has passed the Health Insurance Policy Review Committee (hereafter referred to as the committee) today (DEC 23), the out-of-pocket cost will drop by up to 95% starting next year.The committee also finalized a restructuring of the compensation system for clinical laboratory testing CDMOs, the formulation of an adjustment system for relative value units, and the launch of an 'innovative pilot project for community-based primary care', set for the second half of next year.The Ministry of Health and Welfare (MOHW) approved expanding health insurance coverage for Keytruda and Dupixent during the committee. While Keytruda was previously covered for four cancer types, including non-small cell lung cancer (NSCLC), coverage will now extend to 17 therapies across nine additional cancer types.Specifically, the expanded coverage includes head and neck cancer, gastric cancer, esophageal cancer, endometrial cancer, small bowel cancer, biliary tract cancer, colorectal cancer, triple-negative breast cancer (TNBC), and cervical cancer.For patients meeting the eligibility criteria for these expanded indications, the annual out-of-pocket cost per patient is expected to drop significantly, from approximately KRW 73.02 million to KRW 3.65 million (with a 5% co-payment for monotherapy).Dupixent, which was previously covered for chronic severe atopic dermatitis, will also be covered for severe Type 2 inflammatory asthma starting in January. The annual cost for severe asthma patients is expected to decrease from KRW 15.88 million to approximately KRW 4.76 million (based on a 30% co-payment).The committee also concluded this year's reimbursement re-evaluation for eight therapeutic ingredients. The reimbursement for spherical adsorptive carbon and artemisia herb extract will be maintained due to price reductions.2025 Reevaluation Results for Pharmaceutical Reimbursement Appropriateness: Reimbursement maintained for 1. olopatadine hydrochloride 2. Clematidis Radix-Trichosanthes Root-Prunella Spike 3. Bepotastine…Drug price reduced and reimbursement maintained for 7. spherical adsorptive carbon and artemisia herb extract (upon voluntary requests for drug price reduction by pharmaceutical companies).The oral formulation of L-aspartate-L-ornithine will remain covered, though its reimbursement criteria will be restricted to hepatic encephalopathy. For three other ingredients, including sulglicotide, the evaluation has been deferred on the condition that a portion of the reimbursement will be recouped if clinical trials fail to prove efficacy.Abolition of clinical lab management fees... new institution-specific fees establishedThe compensation structure for CDMO clinical laboratory testing will be reformed. The consignment fee, which overlaps with existing testing fees, will be abolished, and new fees specific to the CDMO institutions will be established. Furthermore, the billing and payment system will be improved to prevent diagnostic fee discounting.The CDMO fee criteria will be determined based on ▲the current management fee ▲the respective roles of the institutions ▲the financial impact during the regular RVU adjustment process. The KRW 240 billion saved from the abolition of the management fee will be reallocated to increase reimbursement for under-compensated areas, such as consultation fees.Examples of Lab Fee Adjustments: Examples of over-compensated areas, and actual adjustments will depend on finalized cost data. Under-compensated areas within clinical lab testing will not face fee cuts but will be prioritized for balance adjustments.The MOHW plans to revise relevant CDMO regulations in the first half of next year and implement the changes in alignment with the regular relative value unit adjustment cycle. Certification standards for clinical laboratory testing will also be updated.Regular adjustment of the relative value unit (previously updated every 5-7 years)To rationalize the fee compensation system, the government will formulate a regular adjustment model for the relative value unit. Previously, relative value unit restructuring occurred every 5 to 7 years, which critics argued failed to reflect rapid changes in medical practice.The government will review under-compensated and over-compensated services based on medical cost analysis and adjust them to balanced fees.In particular, funds from adjusting for over-compensation in areas such as clinical laboratory tests and imaging (CT and MRI) will be redistributed to under-compensated basic medical services, such as consultation and hospitalization fees at clinics and hospitals. Funds will also be directed toward strengthening compensation for essential healthcare, including surgeries and care that are severe·emergency and pediatrics·deliveries.Medical cost analysis results for relative adjustment...Low margins for drug administration and dispensingThe committee discussed the 2023 medical cost analysis results calculated by the Medical Cost Analysis Committee. These results will serve as the foundation for the 2026 regular RVU adjustments. The '2023 Fiscal Year Cost Analysis Report', which for the first time includes cost-to-revenue ratios by healthcare institution type and specific fee items, is scheduled for publication in the first quarter of next year.The analysis was expanded to include tertiary hospitals and clinics in addition to general hospitals. The report calculated the cost-to-revenue of reimbursements following the establishment of standardized medical cost calculation guidelines.Key findings regarding the cost-to-revenue ratio for covered services (based on tertiary hospitals) showed that clinical laboratory fees (192%), special radiologic imaging fees (169%), and radiation therapy fees (274%) had relatively high profit margins compared to costs.Conversely, drug administration and dispensing fees (11%), basic physical therapy (33%), and basic consultation fees (63%) generated significantly lower revenue than costs.'Innovative Pilot Project for Community-based Primary Care' to launch in JulyThe pilot project will launch next year, initially targeting patients aged 50 and older who require integrated management, with plans for gradual expansion.Under this project, registered patients can receive personalized preventive care, disease and medication management, and lifestyle coaching at their designated clinics, linked to their health check-up results. When necessary, patients can be referred to appropriate medical institutions or receive home-based primary care.Clinics that complete the required training are eligible to participate. Institutions capable of providing multi-professional, multidisciplinary team support can join as hub institutions.The government will introduce the 'Primary Care Functional Enhancement Integrated Fee', which compensates for 'patient registration and continuous management efforts' rather than traditional fee-for-service. The MOHW also plans to pilot multidisciplinary team-based service support and performance-based rewards.The pilot program is scheduled to run for three years, from July 2025 to 2028, with plans to expand its scope to additional regions and institutions starting in 2029.
- Policy
- Gov't to enforce non-face-to-face care Medical Service Act
- by Lee, Jeong-Hwan Dec 24, 2025 08:07am
- Following a Cabinet meeting today (December 23), the South Korean government is set to announce an amendment to the Medical Service Act that formally institutionalizes non-face-to-face care. The Ministry of Health and Welfare (MOHW), led by Minister Jeong Eun Kyeong, is the primary department overseeing this transition.Currently operating as a pilot program, non-face-to-face care will officially take effect one year from the date of announcement, in accordance with the supplementary provisions of the Act. This means that December 24, 2026, will mark the official date when non-face-to-face care becomes a permanent, legalized system in South Korea.This legislative reform comes 15 years after the first amendment proposal was submitted during the 18th National Assembly in 2010.The core principle of the Act is that non-face-to-face care will be centered on returning patients and primary care clinics.First-time patients are also eligible for non-face-to-face care, but with specific caveats. Based on MOHW standards, they can only apply for services within their residential area and will be subject to restrictions on the types of drugs prescribed and the total number of prescription days allowed.For the first time, a provision for the delivery of pharmaceuticals outside of pharmacies will be legalized. This applies to specific groups, including residents of islands or remote areas, long-term care recipients, registered persons with disabilities, patients confirmed with Class 1 or 2 infectious diseases, and patients with rare diseases.Prescribing narcotics or psychotropic substances via non-face-to-face care is strictly prohibited. Additionally, the use of electronic prescriptions will be officially institutionalized specifically for non-face-to-face care services.The National Assembly maintained that while the principle of in-person treatment remains paramount, this legislation provides the legal grounds to supplement care via non-face-to-face care. It also establishes the Minister of Health and Welfare’s authority to manage and supervise non-face-to-face care services via online platforms.Those intending to provide or operate a non-face-to-face care platform must report to the MOHW. Furthermore, if the number of platform subscribers exceeds a certain threshold, the operator must apply for official certification from the MOHW Minister.The MOHW Minister reserves the power to revoke certification if a platform obtains it through fraudulent means or fails to meet the required certification standards.The platforms are prohibited from interfering with a physician’s professional medical judgment, encouraging the misuse or abuse of medical services or pharmaceuticals, brokering, inducing, or instigating collusion in violation of the Pharmaceutical Affairs Act.Furthermore, the platforms are prohibited from demanding or receiving financial incentives, goods, or other economic benefits from medical professionals or pharmacies in exchange for patient or prescription referrals.Prohibited provisions also include recommending or inducing patients to choose specific medical institutions, pharmacies, or medical devices.Both doctors and pharmacists are now mandatorily required to use and verify the Drug Utilization Review (DUR) system when prescribing or dispensing narcotics and psychotropic substances during both in-person and non-face-to-face care consultations. Failure to comply will result in a fine of up to KRW 1 million. This provision was institutionalized as a safeguard during the legalization of non-face-to-face care.Meanwhile, an amendment to the Pharmaceutical Affairs Act aimed at prohibiting non-face-to-face care platforms from concurrently operating as pharmaceutical wholesalers (which was expected to pass alongside the telemedicine bill) remains pending in the National Assembly. The timeline for its final approval remains uncertain.
- Policy
- The implications of Keytruda’s reimb expansion
- by Jung, Heung-Jun Dec 24, 2025 08:06am
- MSD Korea’s immune checkpoint inhibitor Keytruda (pembrolizumab) is expected to complete the process of expanding its reimbursed indications at the Health Insurance Policy Deliberation Committee (HIPDC) meeting scheduled for today (the 23rd).Following approval by the committee, Keytruda’s reimbursement will be significantly expanded from January next year, increasing from the current 7 indications across 4 cancer types to 18 indications spanning 13 cancer types.This marks the culmination of a process that began with the reimbursement application in 2023—nearly three years in the making. Beyond the numerical addition of reimbursement for 9 additional cancer types and 11 new indications at once, the decision signifies enhanced treatment accessibility for previously underserved cancer types.It also stands as a significant precedent, having found a breakthrough for expanding reimbursement indications under the existing RSA framework before the government’s formal introduction of its indication-specific drug pricing system.According to industry sources on the 23rd, the National Health Insurance Service's Health Insurance Review and Assessment Service (HIRA) will vote on expanding Keytruda's reimbursement indications at its meeting today at 2 PM.Starting next January, Keytruda will be reimbursed for gastric cancer, esophageal cancer, endometrial cancer, colorectal cancer, squamous cell carcinoma, cervical cancer, breast cancer, small cell lung cancer, and bile duct cancer. Previously, reimbursement was applied to 7 indications across 4 cancer types: non-small cell lung cancer, Hodgkin lymphoma, melanoma, and urothelial carcinoma.While coverage was previously concentrated on non-small cell lung cancer, the expansion will now allow insurance coverage for a broader range of patients, including those with women’s cancers such as endometrial, breast, and cervical cancer.Furthermore, for several cancer types, including endometrial cancer, small cell lung cancer, and bile duct cancer, reimbursement will be extended to patients with MSI-H (microsatellite instability-high) tumors, significantly improving access for patient groups that had previously been excluded from treatment options.A milestone in multi-indication coverage ahead of indication-based pricingKeytruda, which received a multi-indication approval, has long been at the center of discussions surrounding the potential introduction of an indication-based pricing (IBP) system in Korea.While the government recently announced plans to introduce IBP through a drug pricing system reform, concrete measures have yet to be finalized. In this context, Keytruda's case can be seen as an example where the Health Insurance Review and Assessment Service (HIRA), the National Health Insurance Service (NHIS), and the pharmaceutical company found a broad solution for expanding coverage within the current Reimbursement Strategy Agreement (RSA) framework.With several other multi-indication therapies awaiting reimbursement review, this case is expected to serve as a reference model for future reimbursement decisions, regardless of whether IBP is formally adopted.Keytruda currently holds approvals for 18 cancer types and 35 indications, so further attempts to expand reimbursement coverage are anticipated going forward.Of course, this expansion of Keytruda's indications does not eliminate the need for IBP. How the government's drug pricing system will accommodate blockbuster drugs with multiple indications remains an unresolved policy challenge.However, the significance of Keytruda's recent coverage expansion lies in its active use of RSA to achieve results effectively approaching an indication-based pricing system. The case is meaningful not only for enhancing patient access to treatment but also for clarifying the extent to which existing reimbursement mechanisms can be leveraged to address complex multi-indication therapies.
- Company
- Samsung Bioepis wins JPN approval for autoimmune disease biosimilar
- by Choi Da Eun Dec 24, 2025 08:06am
- Samsung Bioepis has received marketing authorization from Japan’s Ministry of Health, Labour and Welfare (MHLW) for its biosimilar version of Stelara (ustekinumab), a treatment for autoimmune diseases. The approved product, developed under the project name SB17 (ustekinumab), will be marketed in Japan under the product name “ウステキヌマブBS皮下注45mgシリンジ「ニプロ」”.Stelara is an autoimmune disease treatment that works by inhibiting the activity of interleukin (IL)-12 and IL-23. It holds various indications, including plaque psoriasis, psoriatic arthritis, and ulcerative colitis. It is a leading blockbuster biologic, generating annual sales of approximately KRW 15 trillion (USD 10.361 billion) in the global market.Samsung Bioepis headquarters./ Photo=Samsung BioepisSamsung Bioepis plans to launch the product in Japan in May 2026 through its local commercial partner, Nipro Corporation. The two companies signed a partnership agreement in June to commercialize the product in Japan. This marks Samsung Bioepis’ first collaboration with a local company for entry into the Japanese market.Byoungin Jung, Vice President of Regulatory Affairs at Samsung Bioepis, said, “Through this approval, we expect to improve treatment access for Japanese patients with autoimmune diseases while also establishing an important foothold for further global expansion as a leading biosimilar company.”Meanwhile, Samsung Bioepis conducted a global Phase III clinical trial of SB17 from July 2021 to November 2022, involving 503 patients with plaque psoriasis across eight countries. This trial confirmed the clinical equivalence of SB17 to the original product in terms of efficacy and safety.Subsequently, Samsung Bioepis launched this biosimilar in Europe and the United States under the brand name Pyzchiva® through its marketing partner Sandoz. In Korea, it is sold directly by Samsung Bioepis under the brand name Epyztek®.
- Company
- Hemlibra improves joint health and physical activity in hemophilia patients
- by Lee, Seok-Jun Dec 23, 2025 08:00am
- JW Pharmaceutical announced on the 22nd that patients with hemophilia A who switched to prophylactic treatment with Hemlibra (emicizumab) showed improvements in joint health indicators as well as increased levels of physical activity.Hemlibra is an innovative new drug that mimics the function of coagulation factor VIII, which is deficient in patients with hemophilia. It is the only treatment for hemophilia A that can be used in both patients with antibodies resistant to existing treatments (Factor VIII products) and non-antibody patients. Another key feature is its sustained preventive effect with subcutaneous administration as infrequently as once every four weeks. In May 2023, health insurance reimbursement in Korea was expanded to include non-antibody severe hemophilia A patients aged one year and older. In October 2025, Hemlibra was also added to the World Health Organization’s Essential Medicines List (EML) and the Essential Medicines List for Children (EMLc).A research team led by Professor Rebecca Kruse-Jarres of the Department of Hematology and Oncology at the University of Washington in the United States is conducting the ‘BEYOND ABR Study’ to evaluate changes in joint health and physical activity when patients with hemophilia A switch to Hemlibra.The team presented interim analysis results in poster format at the 67th Annual Meeting of the American Society of Hematology (ASH 2025), held in Orlando, USA, over four days starting on the 6th (local time). Unlike previous studies that primarily evaluated bleeding reduction effects, the BEYOND ABR study uniquely observed joint function and physical activity.The study enrolled 136 patients with moderate-to-severe hemophilia A who did not possess antibodies against conventional Factor VIII products.A total of 88 patients were included in the analysis of joint health using the Hemophilia Joint Health Score (HJHS). HJHS is a clinician-assessed measure evaluating the function and mobility of major joints such as the knees, ankles, and elbows. Scores range up to 120 points, with lower scores indicating better joint health. The analysis showed that the mean HJHS improved from 10.1 points at baseline—reflecting generally mild joint damage—to 2.8 points at 12 months after switching to Hemlibra. Among all patients analyzed, 23 patients (26.1%) experienced an improvement of 4 points or moreFurthermore, the 27 ‘target joints’ (joints prone to recurrent bleeding) identified in 15 patients before the study initiation at baseline were no longer observed at the 12-month mark after switching to Hemlibra, with no recurrence of repeated bleeding.Physical activity levels also showed improvement. The research team comprehensively assessed patients' walking and physical activity at various intensities using the International Physical Activity Questionnaire (IPAQ). As a result, the proportion of patients classified as ‘low physical activity’ according to IPAQ criteria decreased from 30.8% (32 out of 104) to 23.4% (22 out of 94) at 12 months post-switch. Conversely, the proportion of patients in the ‘high physical activity’ category increased from 44.2% (46 out of 104) to 52.4% (54 out of 103) at 3 months post-switch and remained at 50.0% (47 out of 94) at 12 months.The proportion of patients experiencing no bleeding also remained stable. Between weeks 25 and 48 after Hemlibra administration, 105 out of 134 patients (78.4%) reported no bleeding requiring treatment. Furthermore, at the 6-month follow-up, 125 out of 130 patients (96.2%) stated they preferred Hemlibra over their previous Factor VIII prophylaxis.JW Pharmaceutical plans to accumulate additional long-term observational data through follow-up and expand the evidence base that can be utilized in establishing treatment strategies in actual clinical practice.A JW Pharmaceutical representative stated, “This study presents interim analysis results confirming the bleeding prevention efficacy of Hemlibra in patients switching from conventional Factor VIII prophylaxis, along with changes in joint health and activity indicators. It provides meaningful clinical data to address key concerns, joint status, and exercise performance, key considerations when deciding whether to switch therapies.”
- InterView
- [Reporter’s View| Global trials turn eyes toward Asia
- by Son, Hyung Min Dec 23, 2025 08:00am
- The key theme at the recent European Society for Medical Oncology Asia Congress ‘ESMO ASIA 2025’ held in Singapore went beyond simply global pharmaceutical companies presenting data on Asian subgroups.What became clear at the meeting was a deeper shift: the center of gravity for where clinical outcomes are generated, and where investment and development strategies follow those outcomes, is moving toward Asia.One of the most illustrative examples of this trend was AstraZeneca’s presentation at ESMO Asia. According to the company, 60–70% of patients enrolled in gastrointestinal cancer clinical trials are now being recruited in Asia. Trial designs are increasingly built around Asian data, with approximately 50 active clinical trial sites operating across the region.This signifies that, beyond mere participation rates, the core evidence determining the reliability of clinical outcomes is being generated in Asia. This is why projections indicate that incorporating Asia-centric data will become inevitable in future discussions regarding new drug approvals and reimbursement.The message global pharmaceutical companies are sending is now clear. Asia is no longer merely a site for secondary analysis in global clinical trials; it is becoming the point where clinical outcomes are first generated and where new drug development strategies originate. This signals that the day when the weight of primary endpoints shifts to Asia, rather than being relegated to subgroup analyses, is not far off.Within this wave of change, Korea's role becomes increasingly crucial. As long as Asia remains a diverse region rather than a monolithic entity, which country and which company will be chosen as a strategic partner ultimately depends on each company's level of preparedness. The question of what position Korea will occupy within this Asia-centered restructuring of clinical trials and investment has now become an unavoidable challenge.The shift in clinical outcomes naturally leads to changes in global pharmaceutical companies' investment strategies. From early development to late-stage trials, combination strategies, and follow-up studies, there is a clear move to place Asia at the center. In effect, clinical development and capital are relocating together. Essentially, a structure is forming where clinical trials and investment move simultaneously.A particularly noticeable change in this process amid China's growing presence. China is no longer merely a country with a large patient population. Building on national-level expansion of clinical infrastructure, regulatory innovation, and capital investment, it is rapidly accumulating the capability to independently drive development from early-stage clinical trials through to late-stage Phase III trials. Cases are also increasing where global pharmaceutical companies design clinical strategies by partnering with Chinese biotech firms for development.China's strengths extend beyond speed and scale. In specific mechanisms of action and indications, scenarios are emerging where China becomes the starting point for global development. This suggests that clinical trials and investments shifting to Asia could converge back to China.Korea has long established itself as a reliable clinical trial execution country in global trials. Rapid patient recruitment and excellent medical infrastructure are clear strengths. However, as China strengthens its design and leadership capabilities, maintaining a strategic presence through execution capabilities alone becomes difficult.What global pharmaceutical companies seek in Asia is not merely a large patient pool, but partners capable of jointly discussing development strategies. Engagement at the clinical design stage, interpretive capabilities for specific patient populations, and a research ecosystem capable of leading follow-up studies are becoming increasingly important.Korea possesses the conditions to move beyond being a mere participant and take on roles in clinical design, interpretation, and expansion. Its specialized clinical capabilities centered around large tertiary hospitals, accumulated experience with specific cancers and patient groups, and rapid data production speed are competitive even within Asia. The problem is that this capability remains confined to individual researchers or institutions and is not consolidated into a national-level strategy.The shift to Asia does not offer equal opportunities to all countries. While many nations conduct clinical trials, only a limited number accumulate results and sustain investment. As China rapidly moves to become a leading developer, what Korea needs is not a question of how much to participate, but a choice of how deeply to engage.
- Policy
- 3 supply shortage drugs set to receive a price premium
- by Jung, Heung-Jun Dec 23, 2025 08:00am
- Hanmi Pharm's Vildagle Tab and Kyongbo Pharm's Vilda TabWhile drug prices for approximately 4,000 items will be reduced next January, including those affected by the Actual Transaction Price adjustment, prices for certain products at risk of supply shortage will be increased.These adjustments apply to drugs for which the government is either extending price premiums due to low profitability or raising prices to offset production costs for 'discontinuation-prevention drugs'.According to industry sources on December 22, the price premium period for two salt-modified vildagliptin products, scheduled to expire in January, will be extended for another year.Hanmi Pharmaceutical's Vildagle Tab 50mg (vildagliptin hydrochloride) and Kyongbo Pharm's Vilda Tab 50mg (vildagliptin nitrate), both DPP-4 inhibitor diabetes treatments, were initially set to lose their four-year price premiums next month.However, in line with the Drug Reimbursement Evaluation Committee (DREC)'s opinion on stable supply, the premium period will be extended by 1 year.Currently, Vildagle Tab receives a premium of KRW 300 from KRW 240, and Vilda Tab receives KRW 314 from KRW 240, a premium of 25%-30% at the ceiling price.These two products entered the market in 2022 through salt modifications, before the expiration of the Novartis patent on its original drug, Galvus (vildagliptin).While prices typically drop following patent expiration and the entry of generics, these salt-modified products successfully defended their pricing. By continuously raising their price premiums, they are now priced above the KRW 240 ceiling price of the original Galvus.Additionally, the ceiling price for Bukwang Pharmaceutical's Antiroid Tab (propylthiouracil), an antithyroid agent designated as a 'discontinuation-prevention drug', will increase by approximately 11% next month, from KRW 34 to KRW 38.Price compensation for 'discontinuation-prevention drug' is granted to medicines that are essential for clinical treatment but are at risk of production stoppage due to low profitability.As an ingredient designated as a 'National Essential Drug', it has faced concerns about supply instability amid rising raw material costs, given its low drug pricing. The 11% price hike for Antiroid Tab will take effect on the 1st of next month.Meanwhile, the products undergoing price reductions next month total around 4,000 items, including 3,940 drugs whose prices are being adjusted following the government's Actual Transaction Price investigation.
- Company
- K-Bios acquire US plants to mitigate tariff risks
- by Chon, Seung-Hyun Dec 23, 2025 08:00am
- Korea’s flagship biotech companies, Samsung Biologics and Celltrion, are moving in tandem to acquire manufacturing plants in the United States. Following Celltrion, Samsung Biologics has made a decisive move by acquiring a multinational pharmaceutical manufacturing facility with an investment exceeding KRW 400 billion. These two companies—Korea’s largest biotech exporters to the U.S.—are leveraging strong profitability to pursue large-scale mergers and acquisitions (M&A) as a preemptive strategy to mitigate tariff risks.Samsung Biologics acquires GSK plant in the U.S. for KRW 410 billion...first overseas investmentAccording to industry sources on the 22nd, Samsung Biologics signed an agreement with GSK to acquire a biologics manufacturing facility formerly operated by Human Genome Sciences (HGS), located in Rockville, Maryland. Samsung Biologics America, the U.S. subsidiary of Samsung Biologics, will invest $280 million (approximately 410 billion won) to acquire the facility. The asset acquisition process is expected to be completed within the first quarter of 2026.Human Genome Sciences' biopharmaceutical production facility in Rockville, Maryland, USAThe Rockville production facility is a 60,000-liter API manufacturing plant located in the heart of Maryland's biocluster. It consists of two manufacturing buildings. The facility is equipped with infrastructure capable of supporting antibody drug production at various scales, from clinical development to commercial manufacturing.This marks Samsung Biologics' first overseas plant acquisition. Samsung Biologics currently operates five plants in Songdo, Incheon. All five plants were constructed using internally sourced funds.Since its inception, Samsung Biologics has sequentially built Plant 1 (30,000 liters), Plant 2 (155,000 liters), and Plant 3 (180,000 liters). In October 2022, just 23 months after groundbreaking, it launched Plant 4, which boasts the world's largest manufacturing capacity (240,000 liters) for a single plant. With the start of operations at the 180,000-liter Plant 5 last April, Samsung Biologics' total manufacturing capacity expanded to 785,000 liters. Samsung Biologics invested KRW 5.9089 trillion in the construction of Plant 5.The GSK plant Samsung Biologics is acquiring this time is not large in scale compared to the domestic plants it built itself or the investment amounts involved. Samsung Biologics invested over KRW 2 trillion each in the construction of Plants 4 and 5.Samsung Biologics explained, “This acquisition establishes a dual-source production system connecting Songdo, Korea, and Rockville, USA, providing flexible and stable production options to global customers.” The company plans to expand its collaboration base with North American customers and enhance its ability to respond to regional supply environment changes, thereby further elevating its CDMO competitiveness.Samsung Biologics' acquisition of the US plant is analyzed as a strategy to preemptively mitigate tariff risks.South Korea and the U.S. agreed at the APEC summit in Gyeongju last October to apply Most-Favored-Nation (MFN) treatment in the pharmaceutical sector. This means domestically manufactured drugs in the U.S. will receive MFN treatment, similar to Japan and the EU, with tariffs capped at a maximum rate of 15%.According to the detailed agreement released last month, the White House announced a Joint Fact Sheet (JFS) from the Korea-U.S. summit stating that tariffs on Korean pharmaceuticals would not exceed 15%. It was confirmed that any tariffs imposed on pharmaceuticals would not exceed a 15% tariff rate. Generic drugs would be subject to zero tariffs. Nevertheless, industry players note that future tariff uncertainties remain.Celltrion acquires Lilly plant for KRW 460 billion... resolves tariff risk for top two U.S. exportersFrom the perspective of domestic pharmaceutical and biotech companies, building factories locally in the U.S. is the most realistic and optimal strategy to eliminate tariff risk.Celltrion has taken the most proactive steps in preparation for U.S. tariffs. In September, Celltrion USA, a subsidiary of Celltrion, signed a definitive agreement to acquire a biopharmaceutical manufacturing facility located in Branchburg, New Jersey, from ImClone Systems Holdings, a subsidiary of Eli Lilly. The acquisition price is approximately USD 330 million (about KRW 460 billion). In addition to the plant acquisition cost, Celltrion plans to invest a total of KRW 700 billion, including initial operating expenses.Celltrion received approval from Ireland's competition authorities last October and completed the final review by the U.S. Federal Trade Commission (FTC) in November. These two reviews are key procedures where regulatory agencies assess whether combining corporate assets could harm market competition, representing the final hurdle determining the deal's success.With these acquisitions, Korea’s leading biotech exporters have invested over KRW 1 trillion to secure U.S. production bases, effectively insulating themselves from tariff risks.According to the Ministry of Food and Drug Safety (MFDS), Korean pharmaceutical exports to the U.S. reached USD 1.49 billion (approx. KRW 2 trillion) last year, accounting for 16.1% of Korea’s total USD 9.28987 billion pharmaceutical exports. Finished pharmaceutical products accounted for USD 1,298.99 million (87.1%), while active pharmaceutical ingredients (APIs) amounted to just USD 192.19 million (16.9%. Samsung Biologics and Celltrion dominate this export volume.Samsung Biologics recorded US regional sales of KRW 1.1741 trillion out of its total sales of KRW 4.5473 trillion last year, representing 25.8%. The proportion of Samsung Biologics' sales to the US was 28.5% in 2022 and 26.3% in 2023. Samsung Biologics calculates regional sales based on the location of its CDMO clients. Its cumulative third-quarter U.S. sales reached KRW 1.6482 trillion, already surpassing last year's total exports. U.S. sales accounted for 38.8% of Samsung Biologics' cumulative third-quarter revenue of KRW 4.2484 trillion.Celltrion Regional Sales (Unit: KRW 1 billion, Source: Celltrion)Celltrion has secured 11 FDA approvals in the U.S. Its North American biologics revenue reached KRW 1.045 trillion last year. While Celltrion's North American sales decreased by 11.3% from KRW 709.5 billion in 2022 to KRW 629.2 billion in 2023, they surged 66.1% year-on-year last year, surpassing KRW 1 trillion for the first time. In Q3 alone this year, North American exports surged to KRW 265 billion, more than three times the figure from the same period last year, driven by biosimilar expansion and preemptive shipments ahead of tariff risks.Securing new revenue streams through U.S. manufacturing bases... Generous investments with cash accumulated from high-purity performanceBoth Samsung Biologics and Celltrion avoided tariff risks while securing new revenue streams by establishing U.S. manufacturing bases.Samsung Biologics acquired the Rockville production facility, inheriting contracts for existing products manufactured there and securing a stable supply of large-scale contract manufacturing (CMO) volumes. The company explained that by retaining all 500+ local employees with operational experience and expertise, it established a system that preserves manufacturing continuity and operational stability post-acquisition.Celltrion secured a stable revenue base from the outset by acquiring Lilly's plant and taking over the existing CMO volume of active pharmaceutical ingredients (APIs) previously produced by Lilly. Celltrion anticipates that once the manufacturing line conversion is completed, following post-acquisition validation and re-approval procedures, full-scale production of its own products and CMO volume supply to Lilly will commence next year. This is expected to yield tangible results, structurally mitigating U.S. tariff risks while improving manufacturing efficiency and expanding profitability.Samsung Biologics and Celltrion were able to flexibly secure U.S. manufacturing bases thanks to cash reserves accumulated through high performance.Samsung Biologics recorded cumulative sales of KRW 4.2484 trillion and operating profit of KRW 1.6911 trillion during the first three quarters of this year. Its operating profit margin reached 39.8% relative to sales. Samsung Biologics held KRW 922.1 billion in cash and cash equivalents as of the end of the third quarter.Celltrion posted cumulative sales of KRW 2.8323 trillion and operating profit of KRW 693.3 billion for the first three quarters of this year. This represents an operating profit margin of 24.5%. Celltrion's cash and cash equivalents totaled KRW 810 billion as of the end of the third quarter.