- LOGIN
- MemberShip
- 2026-04-04 11:57:38
- Policy
- MFDS establishes dedicated division to accelerate biosimilar reviews
- by Lee, Tak-Sun Jan 05, 2026 10:39am
- The establishment of a dedicated division for biopharmaceutical approvals is expected to accelerate the review process for domestically developed biosimilars.The Biopharmaceutical Approval Division has been formally incorporated into the Biopharmaceuticals and Herbal Medicine Bureau. With the Biologics Approval Task Force (TF), which began operations in 2024, having been elevated to a permanent division, the review of related products is expected to gain momentum, alongside last year's increase in biosimilar fees.According to the Ministry of Food and Drug Safety (MFDS) on the 1st, the Biopharmaceutical Approval Division was newly established as of the 30th of last month. Hyun-jung Park, a senior civil servant who previously led the TF, has been appointed as division director.Notably, the Biopharmaceutical Approval Division is expected to focus on shortening biosimilar review periods by organizing dedicated teams to review each product.The biosimilar approval review fee was raised last September from the previous KRW 8.03 million to KRW 310 million. The MFDS has stated that it aims to significantly reduce approval timelines using the additional resources secured through the fee increase.Hyun-jung Park, Director of the Biopharmaceutical Approval DivisionDuring a recent presidential policy briefing, MFDS Minister Yu-kyoung Oh announced plans to reduce the average review period for new drugs and biosimilars to 240 days. The current average review timelines are 420 days for new drugs and 406 days for biosimilars. If achieved, this would effectively cut review periods by nearly half.The key to shortening review timelines lies in operating dedicated review teams. By concentrating reviews on a per-product basis, the MFDS aims to eliminate unnecessary delays. The Biopharmaceutical Approval Division is expected to play a central role in this effort.The Ministry of Food and Drug Safety (MFDS) introduced a new expedited review system last year alongside increased fees for new drugs. This system involves forming dedicated product teams (18 members) incorporating specialized personnel for new drug approvals, prioritizing the review of Good Manufacturing Practice (GMP) reviews, and providing customized face-to-face meetings both before and after marketing authorization applications.The establishment of the Biopharmaceutical Approval Division is widely interpreted as an effort to extend this expedited review framework to biopharmaceuticals. All 13 members of the former Biologics Approval TF have been transferred to the new division.Since 2024, the Biologics Approval TF has been responsible for handling approval-related petitions, including preliminary reviews, coordination requests (covering GMP, review processes, risk management plans, and patent issues), issuance of requests for additional data, and approval of extensions for response periods when necessary, and coordination of responses from relevant departments.
- Company
- Chinese API importers outnumber domestic manufacturers in Korea
- by Chon, Seung-Hyun Jan 05, 2026 10:39am
- The number of Chinese active pharmaceutical ingredient (API) suppliers importing APIs into Korea continues to rise. Compared to six years ago, nearly 100 more Chinese companies are now supplying APIs to the Korean market, significantly outnumbering domestic producers. The scale of Chinese API imports has also steadily grown, indicating an expanding influence of Chinese products in the domestic market. In addition, as pharmaceutical companies face price pressure and seek lower-cost APIs, the number of Indian suppliers has also grown substantially.According to the Ministry of Food and Drug Safety on the 3rd, the total number of Chinese API importers in 2024 was recorded at 350. This represents an increase of 19 companies from the 331 recorded in 2023, within just one year.Number of Chinese and Indian API importers and domestic API manufacturers by year (Unit: No. of companies; Source: MFDS)The number of Chinese API importers has shown an upward trend year after year. In 2018, APIs were imported from a total of 254 Chinese companies, meaning 96 new importers have been added over the past six years. Although the number declined from 342 in 2021 to 333 in 2022 and 331 in 2023, it rose again in 2024.As the scale of Chinese API imports continues to grow, the number of newly identified importers is also increasing.The value of Chinese API imports rose from USD 678.09 million in 2018 to USD 816.32 million in 2024, representing a 20.4% increase over six years.Domestic pharmaceutical companies prefer inexpensive imported APIs to reduce costs, leading to a sustained increase in imports from China and diversification of import sources. In 2014, China ranked 6th among domestic drug import sources, but by 2024, it had jumped to 3rd place.The number of Chinese importers now exceeds that of domestic API manufacturers. In 2024, there were 315 domestic API producers, 35 fewer than Chinese importers. While domestic producers were 12 fewer than Chinese importers in 2018 (242 domestic vs. 256 Chinese), the gap widened to 23 in 2024.The volume of Chinese APIs used in the domestic market is comparable to that of domestically produced APIs.In 2024, total API production amounted to KRW 4.4007 trillion. Excluding exports worth USD 2.17314 billion, APIs valued at KRW 1.43 trillion were used in the domestic market, calculated using an average 2024 exchange rate of KRW 1,367 per USD.This calculation indicates that Chinese API used domestically last year amounted to KRW 1.1159 trillion. Considering that Chinese API is cheaper than domestic products, this suggests that domestic companies actually use more Chinese API than domestic API.The number of companies importing active pharmaceutical ingredients from India is also steadily increasing. In 2018, 192 companies imported Indian API, but by 2024, this number had risen to 241, an increase of 49 companies over six years. In 2024, the gap between the number of companies importing Indian API and those producing domestically was only 74.Concerns are spreading that if generic drug prices fall further, the avoidance of relatively expensive domestic APIs will intensify.Under the revised drug pricing system scheduled to take effect this July, the pricing benchmark for generics will drop from 53.55% of the price of the original drug before patent expiration to the 40% range. A setting between 40% and 45% is regarded as most likely. Arithmetically, lowering the maximum generic price from 53.55% to 40% implies a 25% deterioration in profitability.An industry insider stated, “Ongoing drug price-cut policies have significantly expanded efforts to source low-cost APIs from import suppliers in China and India. If the price of generics, a core revenue source for domestic pharmaceutical companies, drops sharply, they will inevitably have to further reduce API costs to cut expenses.”
- Policy
- Rebate penalty in innovative pharma certification reform
- by Lee, Jeong-Hwan Jan 05, 2026 10:39am
- The pharmaceutical industry's attention is drawn to the Ministry of Health and Welfare's (MOHW) proposed reform of the Innovative Pharmaceutical Company certification system.The MOHW is considering a transition from the current 'cancellation of innovativeness certification' rule for illegal rebates to a points-based scoring system.While the MOHW initially announced a shift toward a scoring system, which would give incentives towards new drug development from pharmaceutical companies rather than a penalty disqualifying the certification, it has faced significant pushback from those expressing concerns.On January 2, the pharmaceutical industry is currently anticipating an administrative measure related to rebates concerning innovative pharmaceutical company certification system reform that can sufficiently encourage R&D investment.Even if the current disqualification rules for illegal rebates are maintained without a transition to a scoring system, the industry's demand is for a reasonable reform that prevents certification cancellation for cases that occurred long ago.The industry argues that for R&D plans to be maintained, regulations should be established to ensure that certification is not revoked for extremely old rebate cases or for instances where court rulings determined the company did not actively participate in the illegal activity.The MOHW plans to issue a legislative and administrative notice within January after receiving feedback from both domestic and international pharmaceutical companies. The MOHW noted that the original schedule for the notice was delayed due to the process of gathering diverse opinions from stakeholders.An official from a domestic pharmaceutical company explained, "The MOHW seems to be considering various administrative directions regarding the transition to a scoring system," and "It is crucial that the unreasonable cancellation rules be updated rather than a focus on transitioning to a score system. The goal of the certification is to provide preferential treatment to companies that are dedicated to new drug R&D."
- Company
- Upcoming JPM 2026…K-bio focuses on 'global big deals'
- by Hwang, byoung woo Jan 05, 2026 10:39am
- The '44th Annual J.P. Morgan Healthcare Conference (JPMHC 2026)', the world's largest investment event, will be held from January 12 to 15 (local time), with major domestic pharmaceutical and biotech companies participating to target the global market.This year's event is expected to focus on the reorganization of global supply chains following the implementation of the U.S. Biosecure Act and innovation in drug development based on Artificial Intelligence (AI).Korea's domestic companies plan to strengthen their status through main track presentations while aiming to derive future technology export results through business meetings.JPMHC 2026 is a place where approximately 500 major global pharmaceutical companies, as well as domestic firms, share new R&D achievements and discuss the future direction of the pharmaceutical and biotech industries. For domestic companies, JPMHC 2026 serves as an opportunity to achieve the major goals of 'technology export' and 'partnership formation'.Samsung Biologics and Celltrion to give main track presentations…focus on CDMO strategiesThe most notable participant is Samsung Biologics. The company has received an official invitation for the 10th consecutive year since 2017 and will take the stage in the Grand Ballroom, where only 25 top-tier companies selected by the hosts are featured.CEO John Rim, serving as the speaker, is expected to officially declare the new CMO brand 'ExellenS'.This branding represents Samsung Biologics' core value '4E (Excellence)' and will emphasize company's outstanding competitiveness in the contract manufacturing (CMO) field.Notably, the presentation is likely to highlight the achievement of exceeding $20 billion (approx. KRW 26 trillion) in cumulative orders last year, the expansion of North American bases through the acquisition of the Rockville, Maryland plant, and the 'Super-Gap' strategy to absorb demand shifting away from Chinese CDMOs following the Biosecure Act.Celltrion will also give a main track presentation to demonstrate the synergies of the integrated entity.CEO Jinseok Seo, the eldest son of Chairman Jungjin Seo, is expected to step forward to announce a digital transformation roadmap that introduces AI platforms into areas of drug development to clinical trials and sales.Furthermore, the company is expected to specify plans for a full-scale entry into the new CDMO business based on its recently secured production facilities in the U.S., declaring its leap from a biosimilar firm to a 'Total Healthcare Company'.[Stock Photos] Samsung Biologics CEO John Rim delivers presentation at the 2025 Annual J.P. Morgan Healthcare Conference.APAC track official invitations…scheduled to participate in JPM week meetingsOfficial invitations for presentations on the Asia-Pacific (APAC) track will include presentations of domestic firms.Alteogen, invited as a presenting company, plans to explore additional export possibilities for its human hyaluronidase (ALT-B4) technology, which converts intravenous (IV) injections into subcutaneous (SC) formulations.D&D Pharmatech also announced its selection as an APAC track presenter, unveiling its goal to expand contact with global investors and pharmaceutical companies.D&D Pharmatech is expected to broaden its reach with multinational firms by emphasizing differentiated convenience during the 'New Obesity Drug' rush, presenting clinical progress and oral formulation technology for GLP-1 class obesity and metabolic disease treatments.Hugel is also known to be planning a presentation on its strategy to expand North American market share for its FDA-approved botulinum toxin 'Letybo' and the synergy of its filler business.Beyond the presentation tracks, the activities of biotechs seeking substantive licensing-out (L/O) contracts through unofficial meetings and showcases during JPM Week are also specified.Onconic Therapeutics stated it will participate after receiving an official invitation for the second consecutive year and plans to hold serial meetings with global pharmaceutical companies, biotechs, and investment institutions.ABL Bio will engage in follow-up discussions regarding its blood-brain barrier (BBB) shuttle platform 'Grabody-B', which previously led to large-scale contracts with GSK (approx. KRW 4.1 trillion) and Eli Lilly (approx. KRW 3.8 trillion). Notably, the company plans to share plans for entering Phase 1 clinical trials for its bispecific antibody ADC pipelines (ABL206, ABL209) currently under development through its independent U.S. firm, 'NEOK Bio'.STCube will put forward encouraging clinical data for its immune checkpoint inhibitor 'Nelmastobart'. Based on data proving the biomarker potential of the target protein BTN1A1, STCube is reportedly planning to discuss detailed business structures with global big pharma predicated on actual adoption.ROKIT Healthcare plans to participate in the 'Biotech Showcase 2026' held during JPM Week to present its AI hyper-personalized organ regeneration platform.STCube CSO Seung-han Yu stated, "Nelmastobart has great potential to become a new immune axis that supplements the limitations of existing immune checkpoint inhibitors," and added, "STCube will continue discussions with global companies centered on the mechanism and clinical data at this year's J.P. Morgan Healthcare Conference."An anonymous domestic industry official stated, "J.P. Morgan Healthcare Conference has become an event for South Korean companies to move beyond simple participation to becoming key partners for business discussions on innovative new drugs. As it is a major event at the start of the year, we expect achievements through active meetings during the event."
- Company
- Reimb talks for GVHD drug Rezurock extend into the New Year
- by Eo, Yun-Ho Jan 05, 2026 10:39am
- The graft-versus-host disease (GVHD) treatment Rezurock has stalled at the final stage on its path toward reimbursement listing in Korea.Sanofi Korea and the National Health Insurance Service (NHIS) failed to conclude price negotiations for the ROCK2 inhibitor Rezurock (belumosudil) by the end-of-year deadline and have entered extended negotiations. As a result, reimbursement discussions have carried over into the beginning of this year.Accordingly, it remains to be seen whether Sanofi can complete the listing process in 2026.Rezurock, which was granted accelerated approval from the U.S. Food and Drug Administration (FDA), was approved in Korea in August 2024 and launched as a non-reimbursed product in November. Its key feature is the selective inhibition of ROCK2, a novel mechanism targeting the inflammatory response and fibrosis process in chronic graft-versus-host disease (cGVHD).Chronic GVHD is a complication that occurs in approximately half of patients who receive allogeneic hematopoietic stem cell transplantation. While the patient population may be small due to the disease's nature, it affects half of transplant recipients and is a severe, life-threatening condition requiring essential treatment.Graft-versus-host disease is the leading cause of death in hematologic malignancy patients, accounting for 37.8% of deaths excluding relapse. The problem is that as hematopoietic stem cell transplants increase annually in Korea (1,794 cases in 2023), treating chronic GVHD is becoming increasingly important. Among transplant patients, 42% experience chronic GVHD within an average of 3 years, and 66% have already experienced acute GVHD.However, a significant treatment gap remains. Steroids, recommended as first-line therapy in both domestic and international treatment guidelines, are not suitable for long-term use. Prolonged steroid therapy can cause Cushing’s syndrome, which causes various systemic side effects when used for extended periods, including osteoporosis, joint necrosis, organ failure, hyperlipidemia, gastrointestinal disorders, and growth retardation.While 96% of patients with chronic GVHD receive steroids as first-line therapy, 70% require second-line treatment, and as many as 50% ultimately need third-line therapy. When second-line therapy fails, patients are left to be treated with a combination of steroids and immunomodulators due to a lack of effective third-line options.Furthermore, 97% of GVHD patients treated with steroids experience at least one complication, with infection (79.5%) being the most common. Systemic, multiple symptoms significantly impair patients' quality of life, and host reactions occurring in the lungs or liver are particularly fatal.Against this backdrop, attention is focused on whether Rezurock, if granted reimbursement, can establish itself as a new treatment option.Meanwhile, in clinical trials involving patients who had failed two or more lines of systemic therapy, Rezurock demonstrated a high overall response rate (ORR) of 75%, confirming superior efficacy over existing treatments. Notably, it showed response rates of 71%, 39%, and 26% in the joints, liver, and lungs, respectively—areas where improvement is difficult with conventional therapies.Professor Hee Je Kim, Head of the Department of Hematology at Seoul St. Mary’s Hospital, said, “In 42% of patients with chronic graft-versus-host disease, symptoms occur at multiple sites throughout the body, significantly lowering quality of life. Host reactions occurring in the lungs and liver, in particular, can have a fatal impact on blood cancer patients, making effective management therapies urgently needed.”
- Policy
- 'Platform wholesaler ban law' forum faces backlash over bias
- by Lee, Jeong-Hwan Jan 02, 2026 08:00am
- Rep. Hankyu Kim of the Democratic Party of Korea (center) hosted an emergency forum titled 'Asking the Venture Industry's Opinions on the Pharmaceutical Affairs Act Amendment' on December 16.Rep. Hankyu Kim of the Democratic Party of Korea received criticism from the healthcare and medical communities for openly opposing a bill that would prohibit platforms from also serving as pharmaceutical wholesalers. Rep. Kim opposed this bill, which passed the Health and Welfare Committee and theLegislation and Judiciary Committee with agreements reached by both parties, during an emergency National Assembly forum that he hosted.During the forum, Rep. Kim reportedly voiced strong opposition to the statement from a division head from the Ministry of Health and Welfare (MOHW), the primary department responsible for the proposed amendment to the Pharmaceutical Affairs Act. Some in the medical community expressed concern that the "host lawmaker appeared to be targeting the official by demanding an immediate change in stance or a revision to the legislative proposal". Objections have been raised that the forum was operated with a coercive manner, essentially ordering the ministry to draft an amendment in favor of platform industries and startups for a bill that is already awaiting a final plenary session vote.During the emergency forum on December 16, Rep. Kim stated, "Today’s seminar was co-hosted by 10 members of 'Unicorn Farm', but if the MOHW does not listen to the opinions [of startups and VCs], I will gather 20 members next time for a seminar. If that is still insufficient, I will gather 30 members for discussion."On December 17, representatives from the medical community and bipartisan officials from the National Assembly’s Health and Welfare Committee evaluated Rep. Kim's remarks as biased. They noted that holding a division head responsible for follow-up measures regarding a bipartisan agreement that has already cleared the Welfare and Judiciary committees is likely to draw criticisms of partiality.Medical industry insiders and committee officials argued that the operational direction of the forum, titled "Asking the Venture Industry's Opinion on the Doctor Now Prevention Act," could be perceived as an attempt to intimidate the MOHW administration, which is working to protect public health and the drug distribution ecosystem.Jun-Hyuk Kang, Director of the Pharmaceutical Policy Division at the MOHW, stated during the event, "The Pharmaceutical Affairs Act amendment is different from the 'Tada Ban Law.' The expression 'Doctor Now Prevention Act' is also incorrect, and it is not a law that hinders innovation." He explained, "If a platform operates a wholesaler, it cannot be free from the incentive to drive the prescription and dispensing of its own distributed medications. This law is designed to prevent such conflicts of interest."Kang's statement implied that the delay in passing the bill, which has stalled despite clearing committees, must be resolved to prevent telemedicine platforms from abusing the system for private profit.Immediately after Kang's statement, Rep. Kim continued with remarks that appeared to demand an immediate revision of the bill.Rep. Kim said, "Just because the MOHW says I shouldn't call it a 'Tada Ban Law' doesn't mean I can't. Because many people see it that way, I call it the second 'Tada Ban Law,' and I believe 'Doctor Now Prevention Act' is an accurate term." He added, "The MOHW seems to question why companies enter the platform wholesale business, but what business a company chooses to conduct should be a matter of corporate freedom."Rep. Kim continued, "It is unreasonable for the MOHW to judge whether something constitutes innovation. While the ministry wants to separate the dispensing and prescription of drugs from wholesaling, the correct legislative approach would be to regulate specific conduct, such as penalizing a wholesaler or platform that negatively influences a pharmacist’s prescription or forces generic substitution, rather than banning the wholesale business itself." He argued, "It is strange to insist on this amendment just to block a wholesaler that distributes 90 types of drugs, as Doctor Now does."Rep. Kim further challenged the official, saying, "I could offer a rebuttal to every single point the MOHW makes. I am questioning the procedural legitimacy. I believe the ministry should state its willingness to seek alternatives to the venture industry's opinions. What is most disappointing is that, based on the division head's words today, the MOHW seems to have no intention of changing its position or engaging in further discussion. Is it the official position of the department that no further discussion is possible?"As these remarks became public, the healthcare community questioned whether the emergency forum was intended solely to pressure the MOHW.A pharmaceutical industry official noted, "I heard that Rep. Kim openly pressured the ministry for a revision. Why demand a modification from the MOHW for a bill that secured bipartisan agreement and is awaiting a plenary vote? Remarks about gathering 20 or 30 lawmakers if the ministry doesn't comply sound like a threat."The official added, "The delay in the Pharmaceutical Affairs Act amendment is a matter that could shake the domestic healthcare system and the public health safety net," and added, "Dismissing doctors and pharmacists as focused on protecting their own turf, while framing platforms and startups as the 'weak' party to block the bill, is an act that ignores people’s right to health and life."The official said, "Despite numerous explanations for the legislation and reports of illegal platform activities during the pilot project, Rep. Kim’s insistence on protecting specific corporate interests through a distorted frame is difficult to understand," and concluded, "This attempt to modify the bill will only increase the burden on the Democratic Party, the government, and the President."
- Company
- Korean Pharma wins first-instance ruling in patent challenge over Sirturo
- by Kim, Jin-Gu Jan 02, 2026 08:00am
- Generic drugmakers have secured a first-instance victory in a patent dispute surrounding Sirturo (bedaquiline), a treatment for tuberculosis.According to industry sources on the 19th, the Intellectual Property Trial and Appeal Board (IPTAB) ruled in favor of BC World Pharmaceutical and Yungjin Pharm in a passive scope confirmation (non-infringement) trial concerning the composition patent for Sirturo Tab (Patent No. 10-1514700), filed against Janssen. The two companies had submitted the petition in September.With the favorable ruling in the avoidance trial, analysts say BCWorld and Yungjin have moved one step closer to early market entry for generic versions of Sirturo.Three Sirturo Tab patents are listed in the Ministry of Food and Drug Safety's patent directory. The substance patent expired this June. Excluding this, a use patent expiring in December 2026 and a composition patent expiring in December 2027 remain.BC World and Yungjin’s strategy is to avoid the composition patent expiring in 2027 and strategically launch generics early, promptly upon the expiration of the use patent next year.Sirturo is Janssen’s tuberculosis treatment that was approved in 2014. It features a novel mechanism of action that inhibits the energy metabolism of Mycobacterium tuberculosis and is used in the treatment of multidrug-resistant tuberculosis (MDR-TB). The World Health Organization (WHO) recommends Sirturo as one of the standard treatments for MDR-TB.According to the MFDS, Sirturo recorded import sales of USD 4.1 million (approximately KRW 5.7 billion) in 2023. Although the overall market size is relatively small, the drug has become a key target for generic manufacturers due to the lack of suitable alternatives in MDR-TB treatment. Furthermore, given that tuberculosis treatment regimens last nearly a year, the launch of a generic version is expected to enable stable revenue generation.
- Company
- Roche accelerates drug reformulation strategy
- by Son, Hyung Min Jan 02, 2026 07:45am
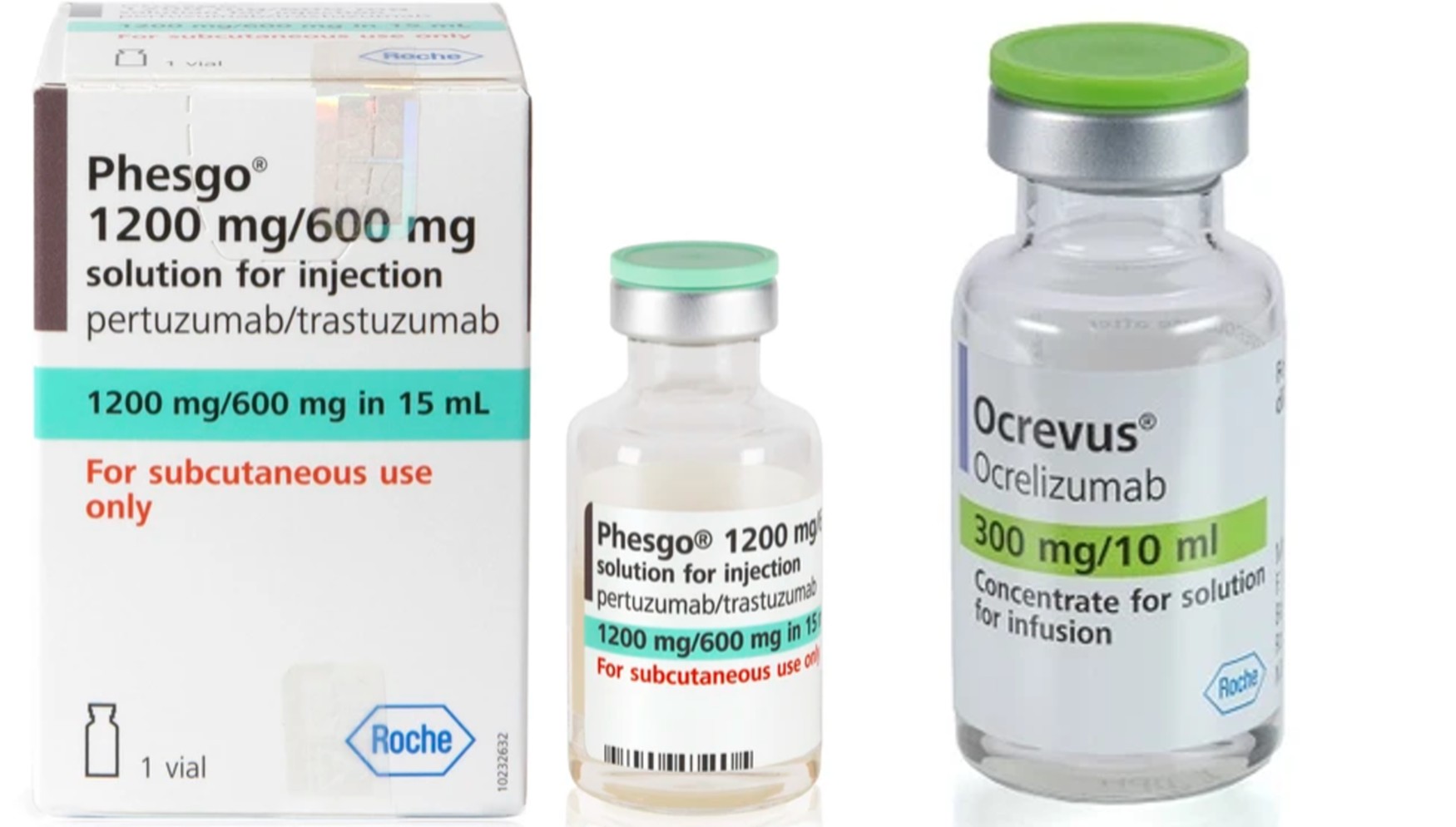
- Roche is driving a paradigm shift across the oncology field by leading with subcutaneous (SC) formulations.The company is actively pursuing formulation changes for its new drugs, positioning ease of administration as a core competitive advantage. Its R&D competitiveness has been confirmed through multiple SC formulation development achievements across both solid tumors and hematologic malignancies.FDA approves Lunsumio Velo…1-minute administration of bispecific antibodies possibleHematologic cancer therapy Lunsumio2According to industry sources on the 2nd, Roche recently received approval from the U.S. Food and Drug Administration (FDA) for Lunsumio Velo, the SC formulation of the bispecific antibody Lunsumio (mosunetuzumab). The drug is indicated for adult patients with relapsed or refractory follicular lymphoma (FL) who have received at least two prior systemic therapies.Lunsumio is the first CD20/CD3 T-cell-engaging bispecific antibody approved for relapsed or refractory follicular lymphoma.The approval was granted under the accelerated approval pathway based on results from the Phase I/II GO29781 study. Full approval will be contingent upon confirmation of clinical benefit in confirmatory trials.With the approval of Lunsumio Velo, convenience in administration has improved markedly. While the intravenous (IV) formulation requires 2–4 hours of infusion, the SC formulation can be administered in approximately 1 minute. Roche explained that this dramatically reduces the time patients spend in the hospital, enabling treatment tailored to individual clinical needs and preferences.In the Phase 1/2 GO29781 study, which formed the basis for this FDA approval, Lunsumio Velo demonstrated meaningful anticancer effects even in patients with third-line or later follicular lymphoma, a group with limited treatment options.Clinical results showed an objective response rate (ORR) of 75% in the Lunsumio Velo treatment group, with 59% achieving complete response (CR). The median duration of response (DOR) among responding patients was 22.4 months.Safety was considered manageable. The most common adverse events included injection-site reactions, fatigue, rash, cytokine release syndrome (CRS), musculoskeletal pain, and diarrhea. CRS occurred in 30% of patients, with most events being Grade 1–2, typically occurring during Cycle 1 and resolving within a median of two days. Grade 3 CRS was reported in only 2.1% of patients.Like the IV formulation, Lunsumio Velo can be administered in the outpatient setting and features a fixed-duration treatment approach. Treatment duration may be as short as six months, differentiating it from therapies that require indefinite administration until disease progression.The IV formulation of Lunsumio has already established itself as the first approved bispecific antibody for third-line or later follicular lymphoma. Roche presented long-term follow-up data for both SC and IV formulations at the American Society of Hematology (ASH) Annual Meeting last year and has submitted these data to regulatory authorities globally, including in Europe. Recently, the European Commission (EC) granted conditional marketing authorization for the Lunsumio SC formulation.Roche is also conducting studies to move Lunsumio Velo into earlier lines of therapy, including combination with Polivy (polatuzumab vedotin) in second-line or later diffuse large B-cell lymphoma (SUNMO study) and combination with lenalidomide in first-line follicular lymphoma (MorningLyte study).From Ocrevus to Tecentriq and Phesgo: Roche expands its SC portfolioRoche has systematically built its SC portfolio by developing and expanding SC formulations for multiple core products.Recently, the SC formulation of the multiple sclerosis treatment ‘Ocrevus (ocrelizumab)’ also entered the domestic market.Roche obtained domestic regulatory approval for Ocrevus SC last month. Ocrevus is a humanized monoclonal antibody that selectively targets CD20-expressing B cells and is classified as a high-efficacy therapy that reduces disease activity and delays long-term disability progression in patients with multiple sclerosis.Compared with the IV formulation, Ocrevus SC reduces administration time to approximately 10 minutes and can be used even in healthcare settings with limited IV infrastructure. The dosing schedule remains unchanged at once every six months, allowing treatment with just two administrations per year—another factor enhancing patient convenience.Roche’s Phesgo, Ocrevus, and TecentriqThe shift to SC formulation is also gaining momentum in the solid tumor field. Roche has already secured SC versions of key oncology products, including the immunotherapy Tecentriq (atezolizumab) and the HER2-positive breast cancer therapy Phesgo (pertuzumab/trastuzumab). In particular, Phesgo has been recognized as a successful case where switching from the existing IV combination therapy to a single SC formulation simultaneously improved administration convenience and operational efficiency in clinical practice.Underpinning this SC transition is Halozyme Therapeutics' ENHANZE drug delivery technology. Roche’s subsidiary Chugai Pharmaceutical secured global rights to use the ENHANZE technology through a partnership with Halozyme in 2022.ENHANZE is a drug-delivery platform that uses recombinant human hyaluronidase (rHuPH20) to convert IV drugs into SC formulations.By temporarily injecting hyaluronic acid within subcutaneous tissue, the technology facilitates drug dispersion, enabling high-dose biologics, previously only available via IV, to be administered as a single SC dose. This allows SC administration within 5 minutes compared with IV formulations, significantly reducing treatment burden on patients while greatly enhancing the efficiency of healthcare providers and the entire hospital system.
- Company
- Celltrion secures '4T sales'…generating profit after merger
- by Hwang, byoung woo Jan 02, 2026 07:43am
- In 2025, Celltrion posted 'record sales' by surpassing KRW 4 trillion in annual revenue and 1 trillion KRW in operating profit.The company successfully eliminated concerns regarding declining profitability after merger with Celltrion Healthcare late-2023. Last year, Celltrion achieved an operating margin of 36%.In 2026, growth is expected to continue based on the expanding sales of new pipelines like Zymfentra and the growth of the CDMO (Contract Development and Manufacturing Organization) business.Steady sales growth, 'Profitability quantum jump' in 2025Reviewing Celltrion's performance over the last two years, 2024 can be viewed as an 'adjustment phase' during which the company absorbed accounting costs related to the merger. 2025 can be seen a 'recovery phase', characterized by sales growth and cost reduction.During an online briefing last November, Chairman Seo Jung-jin expressed confidence, stating, "We have emerged from the long tunnel of merger-related adjustments through the third and fourth quarters."Such statement was validated by the 2025 results, which showed profitability improving at an accelerating pace toward the end of the year.Celltrion's sales and operating profits over 4 years (source: FSS, unit: KRW 100 million). GREEN: sales, ORANGE: operating profitsAccording to preliminary announcement results disclosed on December 31, 2025, Celltrion's Q4 revenue reached KRW 1.2839 trillion with an operating profit of KRW 472.2 billion, yielding an operating margin of 36.8%. This represents a significant year-over-year surge in operating profit.At the time of the merger, Celltrion Healthcare's existing inventory was recorded at market value. In 2024, as this high-cost inventory was recognized as revenue, profit margins were suppressed to single digits.However, as this inventory was depleted during the first half of 2025 and replaced with products manufactured directly by Celltrion at lower costs, the Cost of Goods Sold (COGS) ratio plummeted.Celltrion official said, "With the depletion of high-cost pre-merger inventory and the completion of development cost amortization, which were factors that exerted inevitable pressure on operating profit, combined with titer improvement, operating profit is expected to grow steeply."By Q4 2025, the COGS ratio fell to a tentative 36.1%, a 3 percentage point decrease from the 39% recorded in Q3. Q4 EBITDA is projected to reach KRW 538.9 billion, a record quarterly high.Celltrion analyzes its success to the global stabilization of high-margin new products alongside steady growth in its core portfolio.Despite conservative accounting for market volatility, new products, including Remsima SC (U.S. product name: Zymfentra), Yuflyma, Vegzelma, and Steqeyma, all recorded double-digit growth in Q4. Celltrion's quarterly sales and operating profit over two years (source: FSS, unit: KRW 100 million). PURPLE: sales, ORANGE: operating profitsThese new products are expected to show stark growth, accounting for over 60% of total revenue.Zymfentra is expected to generate even higher revenue in 2026, as the impact of PBM listings and initial marketing investments in early 2025 transitions into full-scale prescription expansion in the second half of the year.Celltrion official said, "For some new products, the timing of their launch was delayed due to factors such as patent settlements for stable product supply, resulting in a somewhat limited effect on annual performance improvement," and added, "From next year, as we expand market share and enter a full-scale profitability enhancement track, we expect a high growth trend in 2026."2026 revenue target of KRW 5.3 trillion…CDMO business in full-scaleCelltrion proposed 2026 revenue target of KRW 5.3 trillion.While Celltrion's 2026 revenue target of 5.3 trillion KRW is slightly lower than previous long-term projections , which once aimed for KRW 7 trillion. The company is showing commitment to crossing the KRW 5 trillion threshold.In 2026, the effects of Zymfentra's inclusion in major U.S. PBM listings will be reflected for a full year. As the company enters a phase where increased prescription volume leads directly to profit, significant revenue growth is expected from Zymfentra as a single item alone.In addition, the company plans to pursue an aggressive bidding 'tender strategy' focused on new products with high net profit, concentrating on substantive growth centered on high-margin product groups rather than expansion of external size through increased supply volume.Furthermore, with a portfolio of 11 biosimilar products completed in major global countries, Celltrion revealed a strategy to accelerate product launches in each country, focusing on new products, while simultaneously reducing the proportion of high-cost products and maximizing the profitability of high-margin product groups.Product photo of ZymfentraCelltrion has a total of 11 products commercialized in the U.S. and Europe. Celltrion aims to expand the number of commercialized products to more than 22 by 2030.Along with this, the company is emphasizing the full-scale launch of the CDMO business as a key to revenue expansion.Previously, to specify its mid-to-long-term CDMO business strategy, Celltrion established Celltrion BioSolutions, a specialized CDMO subsidiary, in December 2024, and in August 2025, its subsidiary Celltrion USA decided to acquire a local company with raw material medicine production facilities in the United States.This is a strategic measure to maintain the framework of the existing CDMO business roadmap while flexibly responding to changes in the external environment, such as U.S. tariff policies, and strengthening responsiveness to global customers.In addition, the company plans to secure multiple new drug product (DP) and drug substance (DS) production facilities in South Korea.Celltrion official said, "In this quarter, to enhance investor predictability and timeliness, we announced our forecast performance for the first time before the end of the quarter by applying conservative assumptions considering market volatility," and added. "From 2026, we will focus on steady growth based on high-margin product groups."
- Policy
- Gvn’t to invest over ₩1T in Medical AI, Pharma, and Biohealth in 2026
- by Lee, Jeong-Hwan Jan 02, 2026 07:43am
- The Ministry of Health and Welfare (MOHW) will invest more than KRW 1 trillion in healthcare research and development (R&D) in the new year (2026).On the 31st, the ministry announced its integrated implementation plan for next year's R&D projects and simultaneously issued the first integrated call for research proposals for 19 healthcare R&D projects. The budget allocated for new projects is KRW 171.5 billion, while KRW 62.5 billion will be invested across the 19 projects in the first call.The MOHW’s total R&D budget for 2026 amounts to KRW 1.0652 trillion, representing a 12.6% increase year on year.Of the 83 total R&D programs, KRW 1.0014 trillion has been allocated to 69 ongoing programs, while KRW 63.8 billion will be invested in 14 new programs. Over the past five years, the ministry’s R&D budget has grown at an average annual rate of 11.1%.The Ministry will pursue this R&D investment centered on four key strategic pillars: technological innovation for public health, securing future growth engines in biohealth, AI-based digital and medical innovation, and establishing a foundation for biohealth innovation.The total scale of new projects for 2026 is KRW 171.5 billion. A first integrated call for proposals will be conducted for projects scheduled to begin in April (19 projects, KRW 62.5 billion).The application period runs from December 31, 2025, to 2:00 PM on January 30, 2026. Research institutions will be selected in April to commence research. Projects scheduled to begin in July (6 projects, 15.9 billion won) will be recruited through a second consolidated call in April 2026.Key new programs included in the first integrated call are dementia medical technology R&D, patient safety technology development, technology development to address suicide-related social issues, translational clinical research in anti-aging and reverse-aging regenerative medicine, establishment of an AI-based surgical robot innovation lab, structure-based AI drug discovery, advanced bio convergence talent cultivation, recruitment of top-tier overseas talent, K-MediST support, and the Health and Medical R&D Core Technology Early Boost Program.In addition, new projects will be selected and supported under nine ongoing programs, including global physician-scientist training, translational research linked to clinical field demand, and technology development to address low birth rates. Starting with new projects in 2026, researchers must submit a research data management plan when applying for a project, and data obtained during the research process must be managed according to a standardized format.Eun-young Jung, Director General of the Bureau of the Health Industry Policy at the Ministry of Health and Welfare, stated, “The role of healthcare R&D is critical for the development of sustainable medical and care technologies and the expansion of biohealth exports. We look forward to active participation from researchers in the new projects for 2026.”Soon-do Cha, President of the Korea Health Industry Development Institute (KHIDI), said, “We will do our utmost to support researchers so that R&D outcomes translate into tangible benefits for the public.”